Zinc Pivalate: More Than Just a Catalyst for Change
Historical Development
Chemistry never stays still. Zinc pivalate entered the stage well after other basic zinc compounds, and for good reason. In the early stretches of the twentieth century, chemists hunted for materials that could push reactions in new directions, free from the drawbacks of other zinc salts like zinc acetate and zinc chloride. Pivalic acid, a carboxylic acid with a notably bulky profile, gave an intriguing twist when paired with zinc. This compound showed that tweaking the organic portion of the salt can influence how zinc behaves in organic synthesis and even in polymer chemistry. My colleagues in academia recalled old German patent literature from the sixties beginning to highlight zinc pivalate for its solubility and catalytic quirks, especially in polyester research.
Understanding the Material
Mention zinc pivalate to a class of sophomore chemistry students and you probably get blank stares in return. It’s not on most high school curricula, and you won’t catch it on the shelf at the local hardware store. In its pure state, zinc pivalate appears as a white crystalline powder, giving no hint of the unique role it serves in industry. Unlike zinc sulfate or nitrate, it’s not vying for attention as a dietary supplement or fertilizer. Those bulky “pivalate” ligands, derived from pivalic acid (trimethylacetic acid), make a big difference in the way this zinc compound interacts with organic systems.
Physical & Chemical Properties
Anyone familiar with organic salts knows that structure matters. Zinc pivalate stands firm at room temperature and doesn’t invite trouble the way zinc oxide dust does. Its moderate melting point and limited water solubility make it easy to handle, though you’ll still need gloves and a mask in the lab. It dissolves well in certain organic solvents, which helps when blending it into other reagents or polymer mixtures. The hexagonal lattice structure, if you care to look under a microscope, owes much to the steric bulk of its pivalate groups, crowding out water and lending some measure of stability others lack.
Technical Specifications & Labeling
Chemists, regulatory bodies, and even those of us double-checking compliance in the paint industry pay close attention to specifications. Purity above 98%, checked by GC or HPLC, tells a story about quality control, as does the need to keep moisture below 1%. Good labeling matters for safety and traceability, with batch numbers and shelf life information ensuring that what comes out of the bag matches what’s written on the invoice. MSDS documentation focuses on skin and eye irritation, so there’s no excuse for lax handling.
Preparation Method
Building zinc pivalate isn't nuclear science, yet it benefits from care. The classic route pairs zinc oxide, or sometimes zinc acetate, with pivalic acid in a solvent such as ethanol or methanol. Heating stirs up the necessary reaction, releasing water or acetic acid as byproducts. Crystallization from organic solvents cleans up the mess, and vacuum filtration or centrifugation separates out the product. In my own research, I found that using a slight excess of pivalic acid nudges the reaction toward greater yield, especially if the starting zinc oxide comes with minor impurities.
Chemical Reactions & Modifications
Zinc pivalate’s magic happens on the reaction bench. In transesterification, it acts as a nudge for monomers to link up, expediting polyester synthesis. Everyone I know in polymer chemistry appreciates alternative catalysts that don’t leave toxic residues behind. Given its non-coordinating pivalate ligands, reactions involving alkoxylation or metathesis sometimes proceed with cleaner byproducts. Substitution of the pivalate for other carboxylates allows researchers to play with solubility and reactivity, and I recall one clever project swapping out related zinc carboxylates in an attempt to improve epoxy resin hardening.
Synonyms & Product Names
Few people call it “zinc trimethylacetate” outside of scientific journals, though that’s just as valid. “Zinc pivalate” remains the preferred name in most research circles. You might run across alternative designations in overseas markets, but as a rule, chemical suppliers and catalogs recognize the IUPAC name to avoid confusion. Nicknames in the lab rely more on context—“the bulky zinc salt” or “ZnPiv2”—but those never appear on invoices or safety data sheets.
Safety & Operational Standards
Everyone who has handled fine, powdery chemicals in a shared lab understands the value of proper ventilation. Skin and respiratory protection isn’t for show—the slight acerbic tang you notice in the air after tipping a pouch of zinc pivalate is enough to make you cautious. Chronic exposure hasn't been tied to cancers or severe systemic injuries in accessible literature, but standard protocols require gloves and dust masks all the same. Good practice demands storing the powder in sealed containers, away from acid fumes and moisture, to prevent unwanted caking or slow degradation. Cleanup often involves sweeping up spillage and collecting it as hazardous waste rather than rinsing it into the drainage system, an approach reinforced by regional environmental regulations.
Application Area
The weight of zinc pivalate in the industrial world falls most heavily on the shoulders of polymer chemists and coatings developers. In rigid polyesters, it hurries along esterification, saving both time and energy in the plant. Its lesser-known but growing role involves catalyzing cross-linking reactions in some epoxy systems, where bulky pivalate ligands lead to fewer side reactions and cleaner products. I’ve seen research into using zinc pivalate as an additive in anti-corrosion primers. Here, it offers a less environmentally toxic alternative to heavier metal salts, a small but promising shift as more governments levy penalties against hazardous substances in paint. Some pharmaceutical researchers experiment with it too, evaluating its use in constructing organometallic frameworks, though that still sits at the margins.
Research & Development
Universities and industrial R&D teams have long tracked how subtle tweaks to the pivalate group alter the performance of catalyst systems. Over the last decade, attention has shifted toward greener alternatives to traditional organometallic catalysts, especially in polymerization and organic synthesis. There’s interest in how recyclable the catalyst might be after a run, how much residual zinc ends up in the final product, and whether you can recover spent reagents without fouling wastewater. My own experience consulting on polyester research confirms that small savings in processing steps, made possible by switching to zinc pivalate, produce big impacts on profitability and waste reduction. Ongoing studies probe the synergy of zinc pivalate mixed with other metal pivalates, examining if combined systems can cut down reaction times or improve yields for specialty elastomers.
Toxicity Research
The legacy of older metal salts reminds us not to assume anything about safety. Animal testing and carefully measured in vitro studies of zinc pivalate have highlighted mostly mild irritation risks, especially in acute exposures. Delving into long-term toxicity literature turns up few red flags, but gaps remain regarding environmental persistence and biological breakdown. Regulators in Europe and Asia have leaned toward the precautionary principle, listing it among substances requiring careful waste handling and disposal. In labs where I’ve advised, no one dumps spent solutions down the drain, not just for legal compliance but out of a sense of prudence given incomplete toxicity data.
Future Prospects
As polymer science pushes toward high-performance and lightweight materials, pressure builds on researchers and industrial chemists to trim down the ecological footprint of every component. Zinc pivalate earns renewed attention for its role as a cleaner, less reactive alternative to older, more contentious catalysts like tin or lead carboxylates. Policymakers apply stricter rules on what goes into paints, coatings, plastics, and even advanced composites, so finding ways to improve the recyclability and final product safety of zinc pivalate-based systems takes on new urgency. I see hope in pilot projects that cycle catalyst streams or in new, bio-based pivalic acid sources that cut down the petrochemical burden. Investment in green chemistry, combined with transparent reporting of residuals and toxicity data, invites confidence in zinc pivalate’s ongoing relevance—not only for industry, but for the broader push to build safer, less wasteful chemical processes. As research fills in the missing toxicity and environmental impact details, innovators stand poised to transform this humble compound from niche reagent to indispensable tool.
What is Zinc Pivalate used for?
Digging Into What Zinc Pivalate Does
Zinc pivalate doesn’t pop up often in conversations outside labs or manufacturing plants, but it plays a bigger role than you might expect. This compound pulls its weight in science because zinc brings a lot to the table—for health, for industry, and for innovation. The “pivalate” part of the name refers to a specific kind of organic fragment that acts as a chelator or partner for zinc. That sounds technical, so here’s what matters: zinc pivalate works as a strong source of zinc that can travel or dissolve into different systems—something that matters in specific chemical processes, especially where plain zinc salts fall short.
Industrial and Scientific Roles
Chemists lean on zinc pivalate when they need a zinc-based catalyst that resists moisture or harsh conditions. Many factories that produce fine chemicals, polymers, or specialty coatings have experimented with zinc pivalate because it handles reactions that ordinary zinc salts mess up or leave incomplete. Paints and plastics get more durable when zinc additives prevent breakdown and keep materials from going brittle. Research teams found that it gives more reliable results in high-precision reactions, thanks to the unique shape of the pivalate molecule and its tendency to keep zinc “available” rather than locked up as a clump or crystal.
Medical and Nutritional Uses
Zinc is an essential trace element for people, animals, and plants. Most nutrition stores stock basic zinc sulfate or zinc gluconate, but these forms sometimes taste harsh, irritate the gut, or trigger nausea. Zinc pivalate tackles some of those headaches because its structure softens the acidic nature of standard zinc salts. Supplement makers keep looking for zinc forms that absorb well, don’t taste metallic, and are affordable. In some studies, zinc pivalate showed good absorption and kept zinc levels stable in experimental nutrition trials, but more clinical data is needed before doctors make it their first pick.
Environmental and Production Considerations
Making zinc pivalate on a large scale requires careful handling of chemicals, which raises concerns about waste and energy use. Many companies face tight emissions rules and need to keep costs low. The field gets caught between finding greener production methods and staying price competitive. Chemists are tinkering with solvent-free processes, aiming for ways to cut down on waste by reclaiming or reusing pivalic acid during synthesis. Recycling and improved filtration have helped. These changes make a difference over years by shrinking the environmental footprint and building public trust in specialty chemicals.
Looking Forward: More Than Just a Zinc Source
No matter if it's boosting the function of a coating, powering a chemical reaction, or, in the future, ending up in a health supplement, zinc pivalate keeps attracting research. Zinc shortages or absorption problems hit hardest in developing regions, and new forms of zinc that work in both medicine and agriculture can nudge health outcomes upward. Personally, I’ve seen how even small shifts in supplement formulations can improve the lives of people recovering from illness or dealing with poor diets. The challenge lies in making safe, effective forms of nutrients that don’t break the bank or harm the planet. Zinc pivalate represents a step forward, but real progress always depends on connecting what science learns in the lab to what people live through day by day.
What are the benefits of using Zinc Pivalate?
What Zinc Pivalate Brings to the Table
In recent years, more folks have been talking about Zinc Pivalate. There’s a reason health circles, supplement makers, and even some research teams are giving this compound a closer look. At the root, zinc forms part of our daily needs. You’ll barely feel it ticking along in your system, but without enough of it, things slow down or break down, sometimes in ways you can’t even see coming.
Zinc Pivalate isn’t just another form of zinc. By pairing zinc with pivalic acid, people who work in chemistry figured out that you can deliver zinc to the body in a way that sometimes works even when traditional zinc supplements might stumble. As someone who’s seen neighbors struggle with zinc from food alone—especially in places where fresh produce or shellfish are out of reach—there’s a genuine relief in knowing other options exist.
The Health Side—Going Beyond Deficiency
Zinc fuels hundreds of functions in your body. Everything from skin healing to sorting out your taste buds runs a bit better with the right zinc levels. Deficiency creeps up on people—poor appetite, wounds that drag on, or getting sick too often all have ties to low zinc. Zinc Pivalate steps into the game here. This form tends to dissolve better and gets absorbed efficiently. For people with sensitive stomachs, traditional zinc supplements can be brutal. Stomach cramps and nausea knock them off course. Zinc Pivalate, with its milder approach, can make daily use bearable, letting more people stick to their regimens.
Even the immune system gets a boost. In the depth of flu season, I remember handing out zinc tablets at a local shelter. Doctors pointed out that keeping regular zinc intake can knock infection time down. By using a form like Zinc Pivalate that’s gentler on the gut, more folks could actually keep up with daily supplements instead of quitting after a week of belly aches.
Why Absorption Matters
Not all zinc gets used by the body in the same way. Some forms barely get through the gut wall before they end up wasted. Zinc Pivalate brings something else—a higher chance for your gut to actually take in the mineral you swallow. Nutrition studies show that better absorption means you don’t need mega doses, trimming down on the risk of side effects. Fewer side effects, less resistance to giving zinc a try again.
Real World Value and Industry Uses
Zinc isn’t just about people, either. In crop fields, zinc keeps grains and vegetables growing strong. Some industrial setups now look at Zinc Pivalate because of its stability and how it releases zinc smoothly—no sudden spikes, no crashes. Machinery and coatings also benefit, as zinc guards against rust and breakdowns. That steady protection helps keep infrastructure ticking longer, which trickles down to less waste and better reliability.
Finding the Balance
Like every supplement, understanding what you’re taking matters. Some folks will need more zinc; some might actually get too much and run into trouble—nausea, headaches, even immune suppression. It’s always smart to get a doctor or nutritionist’s input before starting any new supplement, even one that seems gentle. Yet in a world that asks more from our bodies and our environments, Zinc Pivalate gives people and industries choices, especially where flexibility and ease of use are needed most.
Is Zinc Pivalate safe for human consumption?
What is Zinc Pivalate?
Zinc has always played a big part in staying healthy. It shows up in supplements that promise to strengthen immunity, improve skin, and keep energy steady. Zinc pivalate comes from mixing zinc with pivalic acid—a compound sometimes used in pharmaceuticals. It’s not one of the standard forms like zinc gluconate or zinc citrate, which you might spot on the back of a multivitamin bottle. Zinc pivalate sounds technical, but the real question circles back to safety.
Existing Safety Research
Looking at the science, research on zinc pivalate’s safety for people stays pretty thin. The bulk of official talk about dietary zinc focuses on the tried-and-true compounds, mainly gluconate, sulfate, and oxide. These forms have decades of data showing what they do for the body and how the body handles them. Available medical literature doesn’t share much about the long-term effects or absorption of zinc pivalate in people, so anyone touting it as safe or risky mostly runs on theory.
Human bodies need around 8-11 mg of zinc each day, according to the National Institutes of Health. Too little, and immune systems sputter. Too much, and side effects roll in—metallic taste in the mouth, stomach cramps, even lower immunity. As for the source, most experts recommend choosing zinc forms that have gone through rigorous testing and approval. The FDA and EFSA have set up clear guidelines for supplement ingredients, looking for proofs of safety and clear benefits.
Concerns with Pivalic Acid
Pivalic acid, the other half of this compound, turned up a few questions in drug research. Some studies flagged that pivalic acid, when consumed regularly as part of drugs, can bind to carnitine—a nutrient muscles rely on for energy. Too much of this binding can drop carnitine levels and, in rare cases, lead to muscle weakness, especially over long periods. Medical reviews stress that short-term exposure doesn’t usually create big problems, but there remains an open question about ongoing, low-level intake from a supplement setting.
For a parent looking to support a teenager’s diet, or for someone balancing multiple prescriptions, tracking this risk becomes necessary. Zinc pivalate’s unique chemical combination creates safety questions that science hasn’t fully settled. Experience suggests caution when a supplement’s main ingredient hasn’t been studied well in humans.
Regulatory Approval and Quality
With supplements showing up everywhere, quality control stays crucial. Agencies like the FDA don’t check every supplement before it hits the shelves; they react to problems as they pop up. Products that list zinc pivalate might not pass more stringent reviews found across Europe or North America. High-quality manufacturers often stick with better-known compounds that have passed international reviews and peer scrutiny.
If someone wants reliable zinc, turning to familiar, established forms helps avoid blind spots. Drugstores usually carry zinc gluconate, acetate, and sulfate for good reason—the safety data is clear.
Smart Choices for Better Health
Supplements fill real nutritional gaps when food alone doesn’t cover all the bases. I’ve talked to doctors who remind patients that more isn’t always better. If a new form of any nutrient lands online, taking time to check the science behind it means taking yourself seriously. Health should never feel like guesswork. Consulting with healthcare professionals ensures a clear, credible plan for nutrition. That simple rule makes the biggest difference, especially with less familiar names like zinc pivalate showing up in the ingredient list.
What is the recommended dosage of Zinc Pivalate?
Understanding Zinc Pivalate
Zinc grabs headlines as an everyday mineral, known best for supporting the immune system, wound healing, and cell growth. Most people have heard about zinc supplements—some may even line up for them during cold and flu season. Zinc pivalate, though, steps a bit outside the mainstream. This form binds zinc with pivalic acid and, just like its cousins zinc picolinate or gluconate, aims to enhance absorption.
Looking for Dosage: Where Evidence Stands
If someone tries to find an exact recommended dose for zinc pivalate, the search ends up short. There’s not the same mountain of robust, easy-to-read studies as with zinc sulfate or zinc gluconate. Most evidence on zinc supplementation focuses on elemental zinc, the part of any zinc compound that actually does the job inside the body.
For adults, health authorities like the National Institutes of Health recommend a daily intake of about 11 mg of elemental zinc for men and 8 mg for women. This guideline doesn’t care about the specific form—it talks strictly about elemental zinc, whether it comes in sulfate, acetate, gluconate, or pivalate form.
Zinc pivalate carries its own molecular weight and contains a specific portion of elemental zinc per dose. For any supplement, it makes sense to check the label carefully. Some products mark the amount of the entire compound, some give a breakdown by elemental zinc. Too much can backfire—excess zinc blocks copper absorption, raises the risk of nausea and, at high levels, becomes toxic.
Practical Experience: Not All Zinc Is Alike
Years working alongside pharmacists and doctors taught me that each zinc salt works a bit differently in the gut. Pivalate hasn’t drawn big headlines, but questions around long-term pivalate use come up. Medicines that release pivalic acid can sometimes trigger carnitine deficiency if used in high doses or for lengthy periods. While nobody shouts about this risk with mineral supplements at typical doses, it still sticks in the mind.
Why Dose Recommendations Matter
People looking for safer supplementation want hard numbers and clarity. Over-the-counter supplement bottles sometimes toss out reassuring claims or serve up high numbers unsuspectingly. Sticking to daily amounts set by reliable organizations serves as protection. Multiple clinical research studies report that too much zinc—often defined as regular doses over 40 mg per day—brings side effects like stomach cramps, lowered immunity, and copper deficiency. The evidence isn’t fuzzy: getting enough zinc guards health, but doubling or tripling the amount backfires.
Solutions and Smarter Choices
Doctors and pharmacists continue to steer folks toward trusted sources for supplement advice, not random websites or social media posts. If the goal is to use zinc pivalate, compare the amount of elemental zinc to the recommended daily intake. For those dealing with unusual health needs or specific deficiencies, professional input protects against guesswork. Laboratory testing can even identify zinc status in the body.
Manufacturers and health shops also have a role. Clearer labels, tested products, and selling by elemental content—not just total compound weight—help everyone make sense of what goes in the bottle. When industry and medicine team up, safe and effective supplementation gets easier.
Takeaway: Zinc pivalate adds another option in the supplement aisle but doesn’t change the guiding principle: aim for daily zinc intake in the range set by science, check labels carefully, and never hesitate to ask an expert.Are there any side effects of Zinc Pivalate?
Zinc Supplements and the Daily Routine
Zinc shows up on many supplement shelves, and for good reason. People recognize the role zinc plays in immunity, skin health, and wound healing. Most folks stick to zinc gluconate, picolinate, or acetate, but there’s a new name popping up: zinc pivalate. I’ve spent years researching dietary supplements, and even though companies market zinc pivalate as a novel source, many people ask about its safety and side effects.
Understanding the Unique Case of Pivalate
The pivalate part makes this compound different from regular zinc blends. Pivalic acid, which comes with pivalate, never naturally occurs in the human body. Doctors have seen it before in antibiotics like pivmecillinam, where repeated exposure drains carnitine stores in the body. Carnitine helps turn fat into energy inside cells. If carnitine falls too low, fatigue or muscle weakness might start creeping in. Evidence on zinc pivalate specifically is limited, but pivalic acid’s track record raises important concerns.
Kidney Health and Carnitine Loss
Pharmacists worry that pivalic acid gets stripped from the compound after you swallow it. The liver sends it to the kidneys, and your body flushes it out, but not before it grabs carnitine along the way. The kidneys then clear that carnitine. This isn’t some tiny detail that comes up every so often in research—it’s a real pattern. In children, repeated doses of pivalate antibiotics led to carnitine drops bad enough to catch pediatricians’ attention. Adults might be less sensitive than kids, but vegans, older adults, or anyone with an inherited carnitine issue could see risks climb if they use zinc pivalate every day.
Stomach and Gut Reactions
Zinc itself, no matter the form, can upset digestion. Nausea, cramping, and even vomiting aren’t rare at doses over 40 mg each day. The addition of pivalate offers no evidence of improvement. Instead, there’s reason to think that, at high doses, more unpredictable reactions could appear. I’ve spoken to nutritionists seeing picky eaters develop loose stools and stomach aches on unconventional zinc blends.
Comparing With Classic Zinc Supplements
Clinicians have long chosen simple forms of zinc, not because they’re perfect, but because years of trials show what they do. Pivalate doesn’t offer the same comfort of decades of monitoring. In Europe, regulatory bodies ask supplement makers to prove that new compounds like these won’t sap people’s carnitine over time. No such long-term studies on zinc pivalate exist today.
Better Pathways for Getting Enough Zinc
Whole foods still supply zinc best—seafood, beef, seeds, and beans never saddle people with odd chemicals or new acid structures the body isn’t used to. Whenever a doctor suggests supplements, they look up risks, side effects, and the body's ability to process all pieces of the compound. Until better data turns up, zinc gluconate, acetate, or even plain old zinc sulfate stay safer picks for most people.
What to Ask at the Pharmacy or Clinic
If you see zinc pivalate on a bottle, start a conversation. What’s known about pivalic acid leaving the body? Ask if your diet or medications already lower carnitine. Let your practitioner know about muscle aches, tiredness, or gut troubles, since these might mean your body’s not taking the new supplement well. For now, the unanswered questions around carnitine loss matter more than the marketing spin about new zinc “innovation.”
References and Good Practice
Trusted organizations like the National Institutes of Health flag carnitine depletion as a risk from pivalate compounds in kids exposed long term. If you want to read more, peer-reviewed journals on pharmacology and clinical nutrition offer reassuring transparency about classic zinc forms, but studies on zinc pivalate are just starting.
| Names | |
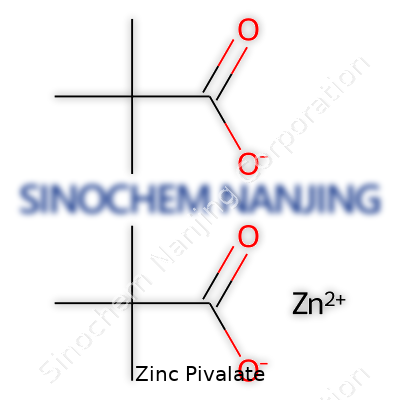
| Preferred IUPAC name | zinc 2,2-dimethylpropanoate |
| Other names |
Zinc trimethylacetate Zinc neopentanoate |
| Pronunciation | /ˈzɪŋk pɪˈveɪlət/ |
| Identifiers | |
| CAS Number | [7448-77-9] |
| Beilstein Reference | 1902226 |
| ChEBI | CHEBI:91144 |
| ChEMBL | CHEMBL4151819 |
| ChemSpider | 21576989 |
| DrugBank | DB14596 |
| ECHA InfoCard | echa.europa.eu/substance-information/-/substanceinfo/100.042.286 |
| EC Number | 237-931-5 |
| Gmelin Reference | 1294886 |
| KEGG | C18615 |
| MeSH | D000402 |
| PubChem CID | 86484 |
| RTECS number | ZH8050000 |
| UNII | X9804K6BQ1 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C10H18O4Zn |
| Molar mass | 297.65 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.15 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.76 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 11.0 |
| Basicity (pKb) | 7.98 |
| Magnetic susceptibility (χ) | -6.2 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.452 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 371.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -935.5 kJ/mol |
| Pharmacology | |
| ATC code | A12CB02 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin and serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, Exclamation Mark |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P362+P364, P501 |
| Autoignition temperature | > 340 °C (644 °F) |
| Lethal dose or concentration | LD50 Oral Rat 2820 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 282 mg/kg |
| NIOSH | Not assigned |
| PEL (Permissible) | 15 mg/m³ |
| REL (Recommended) | 0.5 to 1 mg Zn/kg bw |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Pivalic acid Zinc acetate Zinc propionate Zinc butyrate Zinc isovalerate |

