Looking Closer at Vinyl Neodecanoate: More Than Just a Building Block
Historical Development: A Case of Chemistry Meeting Changing Needs
Years ago, before high-performance coatings took over bridges, stadium roofs, and industrial machinery, chemists grappled with materials that couldn’t handle the roughness of modern life. Vinyl neodecanoate arrived just as industries searched for ways to toughen up paints and resins. While folks were still using simple vinyl esters in the 1960s and 70s, it became clear something more weather-resistant was needed for marine, automotive, and protective uses. Enter this unique ester, developed from neodecanoic acid, which brings a heavy, branched structure to the vinyl group. This tweak didn’t just boost stability; it opened doors for acrylics and vinyl copolymers in ways no one expected. Watching paints hold up on oil rigs or public transit over decades, it’s easy to see why chemists kept digging into what vinyl neodecanoate could offer beyond the basics.
Product Overview: Behind the Name
Vinyl neodecanoate stands out because of its bulky, branched tail—a design that blocks moisture, chemicals, and sunlight from breaking up the polymer. In practice, this means paints won’t peel so fast in coastal air; adhesives stay put even after years of temperature swings on a city parking deck. The compound’s structure allows it to play well with a variety of comonomers, so it gives formulators an edge when trying to balance flexibility, hardness, and chemical toughness in one stroke. I’ve walked through factories where this molecule acts almost like a secret ingredient, bringing together latex binders that would otherwise crumble or chalk up under heavy use.
Physical & Chemical Properties: What Sets It Apart in the Lab and in Use
Sitting down with a bottle of vinyl neodecanoate, you notice two things: a faint, sweet odor and its surprising resistance to water. Boiling points run high, and the stuff shows little interest in mixing with water, but has no trouble dissolving in organic solvents. This is a big plus for paint shops and coating plants facing problems with humidity. Its branched hydrocarbon side chain, bigger than what you find on straight-chain vinyl esters, shields the carbonyl group from attack by alkali and acids. Because molecules like this refuse to fall apart in caustic washes or acid rain, construction crews trust them to hold up insulation panels, water tanks, or road markings. The physical form looks clear or slightly yellow, staying stable even after months at room temperature, which makes storage and transport less of a gamble for chemical suppliers.
Technical Specifications & Labeling: Keeping Things Clear and Honest
Every barrel of vinyl neodecanoate comes with purity above 97 percent if made properly. This compound holds specific gravity right around 0.9, which lines up with the lighter end of organic liquids, so drums weigh less and are easier to move around crowded warehouses. The best suppliers print batch numbers, production dates, and shelf life clearly. Experience shows that labeling with hazard information using the Globally Harmonized System (GHS) makes storage and handling less risky, especially for new hires. Ensuring proper flash point markings and guidance for incompatible materials also gives plant managers peace of mind. These aren’t just legal boxes to check—they keep the crew out of emergency rooms and prevent the sort of accidental mixing that has ruined coatings lines before.
Preparation Method: From Raw Acid to Hard-Working Vinyl Ester
Making vinyl neodecanoate starts with neodecanoic acid, a highly branched carboxylic acid that rarely gets headlines, but underpins the whole process. Chemists use a vinylation reaction—throwing ethylene into the mix with a catalyst under mild pressure. The result? The acid and ethylene react, forming a vinyl ester that’s primed for use in copolymerization. Heading into a pilot plant, the smell of ethylene and esters hangs in the air, and the reactors churn away, recycling any unreacted monomer. The purification steps include careful distillation, since leftover acid or catalyst traces can throw off the performance in paints or adhesives down the line. The payoff for all this care comes at the application stage, where a tiny bit of variation in preparation changes how a product weathers its first year outside.
Chemical Reactions & Modifications: Why Structure Means Everything
Vinyl neodecanoate isn’t just blended into latex and forgotten. Customization starts in the reactor, where it trades places with other monomers like vinyl acetate or acrylics, forming alternating or block copolymers with properties that adjust like a dimmer switch. This adaptability gives coatings chemists freedom—a little more of this, a little less of that, and suddenly paints resist graffiti, pick up gloss, or turn matte as the market demands. Radical polymerization is the norm here, making use of initiators that break the double bond and turn the monomer into a long, repeatable unit. Crosslinking with carbodiimide or isocyanate agents gives finished materials unbeatable stain and weather resistance. I’ve seen polymer labs run test panels as long as a football field to see how slight shifts in formulation affect real-world aging, all starting from choices made at the monomer level.
Synonyms & Product Names: More Than One Face in the Market
Depending on the supplier or region, you might hear this molecule called “VeoVa 10,” a shorthand for vinyl ester of versatic acid, or just vinyl neodecanoate monomer. Industry insiders use these names interchangeably, though the underlying chemistry stays the same. This multiplicity can trip up new buyers searching for data sheets or regulatory files, so it pays to ask for CAS numbers or structural diagrams when sourcing, rather than just going by marketing lingo. Out on the trade show floor, you’ll spot technical brochures with all these names gracing the same booth, reminding everyone that chemistry solves problems, not branding.
Safety & Operational Standards: More Than Just Getting the Job Done
Vinyl neodecanoate doesn’t explode or catch fire at the drop of a hat like some monomers, but the risks aren’t zero. Liquid contact can irritate skin, and vapor builds up can trigger headaches or nausea in closed mixing rooms. Proper ventilation, personal protective equipment, and spill controls matter, as I learned watching a factory near-miss that could have sent half a shift for screening. Operators post hazard signage, and newer facilities use closed-loop handling to cut exposure. Spills clean up best with absorbent materials, not water, which just spreads the risk. Fire training covers chemical extinguishers, not just water hoses, because burning monomer releases nasty fumes. Seeing the impact of even a small accident leaves no doubt: safety standards aren’t just checklists; they’re the shield protecting lives and livelihoods.
Application Area: Why This Molecule Matters Across Industries
Go into a bridge painting project or a marine equipment overhaul, and chances are high you’ll find vinyl neodecanoate at work. Its resistances to moisture, alkalis, and UV rays mean marine and exterior architectural paints actually hold their color and stick to metal or masonry. In the adhesives space, its flexibility stops tiles from debonding in bathroom humidity or freezing winter temperatures. Paper coatings benefit too—no more yellowing along the edges of a quality magazine after a summer on a newsstand. Roof coatings, road marking paints, even toughened wood finishes count on this molecule to avoid breakdown for years. Green building certifiers also watch for low volatile organic compound emissions, and this material typically scores well when formulated thoughtfully, helping building owners keep up with tighter regulations on indoor and outdoor air quality.
Research & Development: Chasing New Answers in Polymers
Scientists keep pushing the limits of vinyl neodecanoate, whether it’s about improving durability in graffiti-resistant paints or tweaking formulations for heat-resistance in automotive treatments. A big push goes toward lowering residual monomer content to hit tighter environmental standards, because regulatory pressure on VOCs and hazardous air pollutants gets stricter by the year. Smart coatings use this ester as a backbone for self-cleaning or self-healing surfaces, adding nanomaterials or functionalized acrylics to bring new effects. I’ve spoken with researchers at university-industry collaborations who see vinyl neodecanoate as the springboard for greener, smarter materials—not just more of the same old paint. Looking at patents, a spike in filings proves industries aren’t done finding new angles for what once seemed a pretty simple vinyl ester.
Toxicity Research: Keeping an Eye on Health and Environment
The story here stays complicated, as it always goes with chemicals that hit so many applications. Toxicity tests on vinyl neodecanoate point toward low acute hazards under normal use conditions—long-term inhalation or skin exposure still bring up concerns. Regulatory agencies flag risks tied to chronic exposure by workers handling large quantities, so stronger ventilation and protective gear remain the norm. Runoff from production or improper disposal sometimes pops up in environmental screens near chemical plants; tighter containment protocols knock down these problems before they reach wildlife or drinking water. Because breakdown byproducts and trace monomer leaks are always possible in older facilities, new approvals often require extensive environmental impact studies. The clock doesn’t stop on toxicology checks, and the drive for non-toxic alternatives influences every new round of research for paints that land in schools, hospitals, or water infrastructure projects.
Future Prospects: Not Just Holding on to Old Strengths
Looking ahead, the uses of vinyl neodecanoate look set to expand along with green chemistry and the drive for decarbonization. Labs focus on ways to make the monomer from renewable feedstocks, cutting petrochemical ties and shrinking carbon footprints. As the world looks for repairable, recyclable coatings and adhesives, tweaks to this vinyl ester’s structure build in better end-of-life options without dropping durability. Building codes call for longer lifespans and tougher surfaces, making this compound a focus for both incremental improvements and big leaps in performance. As a writer who’s spent years talking to chemists and builders alike, I see vinyl neodecanoate less as a finished chapter and more as a toolbox that expands as demands on materials keep shifting. The challenge isn’t just technical—it’s about keeping products safe, effective, and sustainable in a world that asks more from every molecule in circulation.
What is Vinyl Neodecanoate used for?
Everyday Products Start Here
Step into any hardware store. Rows of paint, glue, protective coatings line the aisles. On the surface, they seem pretty basic. The real story sits much deeper. Materials scientists and chemists have put years into tweaking molecules so things stick, shine, and stay strong. In this arena, vinyl neodecanoate is a quiet workhorse. Not many people outside the industry talk about it, but nearly everyone unknowingly benefits from it.
The Backbone of Modern Paint and Adhesives
Factories use vinyl neodecanoate as a building block for special polymers. These polymers end up in paints primed to resist weather, adhesives that keep surfaces together, and floor coatings that handle foot traffic day in and day out. The key lies in how this molecule handles water and sunlight. It gives paints and coatings a shield against fading, peeling, and blistering—even if the sun beats down for years or a wall faces constant cleaning.
My own experience comes from summer work at a hardware supplier in the Midwest. Farmers used to demand new fence paint every spring. Old formulas turned chalky within a year. Eventually, companies started switching to polymers based on vinyl neodecanoate. Complaints dropped sharply. People noticed their barns and fences held a clean, bright finish much longer, even in tough heat and endless rain. Stores wasted less stock. Customers saved money. A direct result of one molecule.
Industrial Applications With Real Human Impact
It doesn’t stop at paint. Manufacturers depend on vinyl neodecanoate polymers to create waterproof sealants for windows and bridges. Think of all the small leaks that can eat away at a home’s insulation or let rainwater corrode steel rails. Adding this chemical keeps sealants hanging tight and fending off the elements.
In another corner, textile mills use it to coat fibers or backings to keep stains from sinking in. Hospitals and schools, where cleaning is constant and surfaces take a beating, look for these coatings to keep things sanitary and fresh. Food processing plants and breweries often want easy-to-wash surfaces. Companies that supply these places keep returning to vinyl neodecanoate because it stands up to harsh cleansers, repeated scrubbing, and plenty of water.
Balancing Performance with Safety
Scientists and regulators hold chemicals like this one to strict standards. Workers who handle it in manufacturing plants wear gloves, masks, and work with proper ventilation. Completed paint or glue products, after curing, release little to nothing harmful. The EPA and EU have dug into its safety record. As long as factories handle it with basic chemical safety in mind, risk stays very low.
Responsible companies take this seriously. Most now invest in continuous training, rigorous audits, and transparent labeling. Some have shifted to water-based systems instead of solvent-heavy ones, which slashes emissions and exposure for everyone involved.
Looking Ahead
No one expects people to memorize chemical ingredients in every product. Still, the presence of something like vinyl neodecanoate means paints last longer, adhesives don’t fail unexpectedly, and buildings stay sealed up tight. These practical wins mean fewer repairs, less wasted material, and a better shot at keeping up with sustainability goals for the next generation.
What are the safety precautions when handling Vinyl Neodecanoate?
Understanding the Risks
Vinyl neodecanoate carries potential dangers that anyone in the paint, coatings, or plastics industries needs to treat seriously. You might notice a fruity odor near open containers, but the real problem lies in its ability to irritate eyes, skin, and lungs with even brief exposure. Chronic contact can worsen allergies or trigger asthma-like reactions for some workers. It’s not a mystery chemical: the facts are clear from the safety data sheets and regulatory agencies alike. Some research flags the risk of organ toxicity after prolonged contact, so thinking ahead makes a real difference.
Personal Experience Shows the Gaps
Walking through a coatings facility last year, I saw the shortcuts workers sometimes take. Gloves tossed aside to save a minute, cracked goggles hanging from hooks. It’s no surprise: hot warehouses get uncomfortable, and gear can feel cumbersome. Yet, in the real world, one splash or fume inhalation can send someone straight to the emergency room. I remember talking with a foreman who regretted his own short-sleeve days; he developed a nasty rash that lasted weeks. That lesson stuck with me.
Practical Protective Steps
Products like vinyl neodecanoate demand strong protocols, not just hope. Splash-proof goggles, not simple safety glasses, form a decent barrier for the eyes. Chemically resistant gloves—think nitrile or neoprene, never basic latex—block skin contact. Lab coats or coveralls keep the chemical off arms and legs. Simple mistakes like wearing street clothes in a mixing room leave skin exposed, and that ramps up the risk.
Respiratory exposure deserves attention, especially in rooms with poor airflow. Cartridge respirators rated for organic vapors create a real shield. Relying only on open doors or small fans doesn't provide much reassurance when fumes are strong, particularly during warm months. The right mask beats improvisation every time, and smart workers never “just risk it” to finish a shift faster.
The Value of Training and Vigilance
Clear labeling and safety data sheets should stay accessible. Teams who know where to find emergency eyewash stations and showers react faster during spills. Regular training isn’t busywork: it saves hands, eyes, and lungs. I’ve seen crews skip fresh glove changes between tasks, sometimes using rags instead. That spreads hazardous residue onto tools and even food break areas, turning a small error into a bigger mess. Keeping chemical storage areas dry, cool, and well-marked also matters. Overloaded shelves or leaky caps waste money and create surprise hazards for the next shift.
Solutions That Work
Spill kits with proper absorbents and disposal bins give teams a fighting chance against small leaks before they become disasters. Ventilation fans and hoods remove vapors from the work zone, making every breath a little safer. Weekly safety meetings, posted rules, and clear reporting routes all keep safety front-of-mind. Supervisors shouldn’t hide behind clipboards—real leadership walks the shop floor, spots bad habits, and encourages better choices. Even simple habits, like washing up before meals and removing gloves correctly, block accidental ingestion and cross-contamination.
Many accidents trace back to overlooked routines. Vinyl neodecanoate doesn’t reward carelessness, and it never cuts a break for old hands or rookies alike. No job speeds up enough to excuse shortcuts with personal protection or sloppy storage. A strong safety culture, backed by real-world experience and smart procedures, gives every worker a fair shot at heading home healthy at the end of the day.
What is the chemical formula of Vinyl Neodecanoate?
Understanding Vinyl Neodecanoate
Vinyl neodecanoate holds a real place in the modern chemical world. Its formula, C12H22O2, tells more than its composition—it shows its potential where coatings and adhesives come alive. Looking at chemical formulas, every atom counts. Here, twelve carbons, twenty-two hydrogens, and two oxygens link together in a branched structure. From every bit of hands-on work I’ve seen in labs and factories, that branching sets it apart from straight-chain monomers. This isn’t some obscure bit of trivia. That form gives different weather resistance and toughness to the polymers based on it.
Why Structure Drives Performance
Putting formulas on paper doesn’t make a story. What’s behind it marks the real value. Vinyl neodecanoate brings a mix of hydrophobic qualities and flexibility not every vinyl monomer shares. Engineers pick vinyl neodecanoate for coatings that outlast sun, rain, and rough handling. Try painting steel in a damp summer, hoping your finish won’t start flaking in a few years—that’s the difference a well-designed monomer formula brings. C12H22O2 isn’t just an answer to a chemistry quiz, it’s the difference between maintenance headaches and long-lasting performance.
Industry Use and Safety
People wonder why some monomers fade out and others stay part of production lines. Vinyl neodecanoate stays because it meets tough regulatory standards and handles well in factories. Its formula gives low vapor pressure, so fewer fumes float into the workspace compared to older monomers like vinyl acetate. Workers notice cleaner air, fewer headaches, and gains in safety. Facts from safety data sheets back that up—less inhalation risk leads to fewer workplace medical incidents. The European Chemicals Agency and the U.S. EPA have both reviewed these kinds of monomers, and vinyl neodecanoate’s record looks strong compared to some past products.
Challenges Facing Vinyl Neodecanoate
Making, transporting, and using any synthetic chemical asks for responsibility. I’ve seen more push for sustainable chemistry each year. Vinyl neodecanoate doesn’t solve every challenge. It comes from fossil raw materials, and end-of-life recycling still runs into barriers—those polymers don’t just break down on their own. On the practical side, managing waste and avoiding environmental release demand good process controls and smart facility design. On a personal level, reading about microplastics keeps the pressure on chemists and manufacturers to chase greener alternatives.
Looking Toward Solutions
Finding cleaner ways to make and use chemicals pushes this industry forward. For vinyl neodecanoate, new biosourced routes have started to show up in research circles, using renewable feedstocks instead of petroleum. Improved catalyst systems can cut unwanted byproducts and energy costs. Over years working with formulators, I’ve seen better recycling procedures and cleaner processing equipment drive down environmental impact. Policy and market choices help—when buyers ask for greener products, companies respond with investment and ingenuity.
Trust and Science Go Together
Vinyl neodecanoate is more than a chemical name or a formula written in a textbook. It illustrates how chemistry, good industry practice, and public trust all weave together. Each move to make it safer and cleaner builds credibility. Solid research, transparent safety data, and open communication matter every step of the way.
How should Vinyl Neodecanoate be stored?
Understanding What You’re Dealing With
Vinyl Neodecanoate isn’t exactly olive oil. Pulled out of the world of industrial chemicals, this colorless liquid comes with a mild odor and a volatility that tells you not to get casual with it. Most folks I know who have worked with it keep a mental list of its quirks—flammable, reactive, susceptible to slow polymerization if you forget it for just a few warm days. These qualities make it crucial to get storage right, not just because of regulations, but because nobody likes dealing with a leaking drum or a fire risk in the warehouse.
Temperature and Ventilation Make All the Difference
Let a drum of Vinyl Neodecanoate sit in the sun for a while, and you’re basically inviting trouble. Heat speeds up chemical reactions, and with this substance, it means polymerization. Once that process gets going, you’re looking at thickening or gelling that ruins product quality and chokes lines in production. I tell everyone who asks—store it in a cool place. Think 15–25°C, away from heaters, direct sunlight, or hot pipes. Keeping things ventilated matters just as much. This liquid gives off vapors that mix easily with air. Without fresh air moving through the storage area, you’re flirting with flammable vapor accumulation. Any spark—maybe just the flick of a switch—can be all it takes if the ventilation isn’t right.
Containers and Compatibility
I remember one story where a barrel developed soft spots because someone tried using a generic plastic drum. That’s a lesson. Use original containers, or if you need to transfer, go with solid, well-sealed metal drums or tanks lined for organic chemicals. Avoid copper or copper alloys since they can react and kick off polymerization, which in the worst cases, heats up the whole batch. Even the best drums won’t save you if you’re careless with sealing. Any time air or moisture sneaks in, the clock starts on chemical degradation—and those same air gaps can let vapors out.
Keeping Out Contamination
One often-overlooked risk is cross-contamination. Mixing with acids, bases, or strong oxidizers can turn an ordinary spill into an emergency. Training warehouse staff to use separate tools for different chemicals isn’t overkill; it’s just wise. In some shops I’ve seen, each class of chemicals gets its own section and color-coded tools. It feels strict, but clean boundaries keep reactions from starting when and where they shouldn’t.
Safety Steps That Matter
Label every drum. I’ve opened boxes where the only way to tell what’s inside was by sniffing the edge—never a safe habit. Clear, durable labels with hazard symbols, product name, and date received or filled prevent mistakes when folks start moving things around. Some teams run regular inventory checks and rotate stock so nothing gets too old. Vinyl Neodecanoate doesn’t last forever; storing it too long runs the risk of losing not just money but safety margin.
Spill kits with absorbent material, goggles, nitrile gloves, and a plan for drumming up leaks help keep small problems from turning into warehouse-wide crises. If a drum tips or a valve drips, you want to be able to deal with it quickly, not scramble to find help.
Why All This Attention Matters
Nobody likes filling out incident reports or calling the fire department. Safe, smart storage of Vinyl Neodecanoate isn’t just about following rules. It’s about keeping people safe, keeping product usable, and keeping everyone’s workday a little less stressful. Good habits beat luck every time.
Is Vinyl Neodecanoate hazardous to health or the environment?
The Chemistry in Plain Sight
Vinyl neodecanoate isn’t a household name, yet it quietly supports the coatings and adhesives industry. This chemical joins hands with other compounds to make paints last longer and surfaces more durable. You’ll find it in construction, automotive, and even some everyday products. The trouble with many modern chemicals, though, is that their effects often stretch far beyond their intended use.
Health Risks: Hidden or Overstated?
Anyone who has spent time in a paint shop—like I did in my younger days—knows how easily you can overlook what’s floating through the air. Vinyl neodecanoate itself is not well studied compared to older, infamous chemicals like lead or benzene. According to the European Chemicals Agency, this chemical doesn’t cause acute toxicity easily at the levels most workers encounter. Still, chronic exposure stories often come out years after new materials hit the market.
Still, breathing in fumes or letting a chemical touch skin isn’t just about the big dramatic poisonings. Workers, especially those in manufacturing and application jobs, can develop irritation in their eyes and airways over long periods. Even without a stack of conclusive lab results, the rule of “keep it off your skin, keep it out of your lungs” still stands strong. Even if something isn’t acutely toxic, repeated low-level exposure can add up, especially for people who are more sensitive or who face multiple chemical exposures at work.
Environmental Fate: Where It Ends Up
It’s not just factory workers or painters at risk. I’ve learned firsthand from years living near industrial areas that when chemicals end up in wastewater or soil, things can go sideways for the local ecosystem. The technical literature suggests vinyl neodecanoate sticks around in the environment longer than some other common chemicals. Its structure resists breaking down quickly. Fish and other aquatic animals are sensitive to many industrial residues—if this compound builds up, it could interfere with their growth or survival.
Once chemicals like these slip into waterways or soil, the cleanup gets expensive and tricky. So, the safest bet is to prevent releases in the first place. That means holding companies accountable—to use tight storage and disposal practices and to follow laws on chemical discharge.
Practical Steps: Reducing Risk in Daily Life
Regulations mean little unless people follow them. On job sites or in hobby spaces, using gloves, masks, and decent ventilation can cut risks fast. Old habits die hard, but swapping open buckets and dirty rags for closed containers and regular hand washing goes a long way. Most importantly, companies owe it to everyone—their workers and their neighbors—to share safety sheets and give real training.
My time volunteering on community safety boards taught me that transparency moves mountains. If you don’t know what’s in the products you use or the air you breathe, you can’t make smart choices. When people speak up about what’s being stored near their homes or schools, policy-makers listen.
Looking Forward
It takes only one oversight to turn a useful chemical into trouble for health and the environment. Until more long-term studies come along, caution and honesty—backed by strong rules and real enforcement—are the right tools for keeping vinyl neodecanoate safe for everyone.
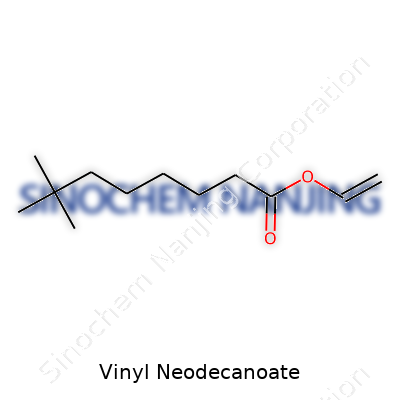
| Names | |
| Preferred IUPAC name | Ethyl 3,5,5-trimethylhex-3-enoate |
| Other names |
Vinyl neodecanoic acid Vinyl esters of neodecanoic acid Vinyl ester neodecanoate Neodecanoic acid, vinyl ester VEOVA 10 |
| Pronunciation | /ˈvaɪ.nɪl niː.oʊˌdɛk.əˈnoʊ.eɪt/ |
| Identifiers | |
| CAS Number | 51000-52-3 |
| Beilstein Reference | 1718731 |
| ChEBI | CHEBI:87447 |
| ChEMBL | CHEMBL1627182 |
| ChemSpider | 11805718 |
| DrugBank | DB14041 |
| ECHA InfoCard | '03e1be22915e-41be-b245-2845c5b5320c' |
| EC Number | 271-721-9 |
| Gmelin Reference | 623586 |
| KEGG | C21110 |
| MeSH | D004313 |
| PubChem CID | 85947 |
| RTECS number | XR1985000 |
| UNII | 3B1B1UC92J |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C12H22O2 |
| Molar mass | 212.33 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Mild |
| Density | 0.91 g/cm3 |
| Solubility in water | insoluble |
| log P | 3.87 |
| Vapor pressure | 0.24 mmHg @ 20°C |
| Acidity (pKa) | ~25 |
| Basicity (pKb) | 13.5 |
| Magnetic susceptibility (χ) | -8.0e-6 cm³/mol |
| Refractive index (nD) | 1.426 |
| Viscosity | 12 mPa·s |
| Dipole moment | 1.45 D |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH⦵298) | -471.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6121 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P261, P271, P280, P304+P340, P312, P403+P233 |
| Flash point | 84°C |
| Autoignition temperature | > 236 °C (457 °F) |
| LD50 (median dose) | LD50 (median dose): >5,000 mg/kg (rat, oral) |
| NIOSH | No established NIOSH value. |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
Vinyl acetate Vinyl laurate Vinyl propionate Vinyl pivalate |

