Trioxane: Looking Beyond the Lab into Real-World Impact
Historical Development and My First Encounter
Stories about trioxane rarely turn up on the evening news, but anyone who’s cracked open an organic chemistry textbook will likely bump into it early on. My own journey with trioxane began in a university lab where someone opened a bottle and pointed out its acrid, medicinal scent. At that moment, I didn’t realize I was dealing with a chemical that has stood at the crossroads of industrial production and field rations for decades. Trioxane’s story began in the late 19th century, showing up in German patents focused on formaldehyde polymerization, then steadily entering both academic texts and industrial catalogs worldwide. Understanding that history helps explain why this substance still matters—industries continue searching for solvents, fuels, and intermediates that thread the needle between performance, price, and safety.
Product Overview: Beyond the White Powder
Off-white and crystalline, trioxane often doesn’t grab much attention visually. At first glance, it looks like many other industrial solids. People in chemical engineering circles, though, see it differently. For decades, factories processed massive quantities of trioxane mainly as a formaldehyde source. Compared to handling pure formaldehyde, trioxane offered easier storage and more predictable behavior. Over time, manufacturers leveraged it to produce everything from resins used in household goods to field fuel tablets favored by military and hiking communities. Some may recognize it as the little blueish square tucked into a portable stove kit—reliable and simple, with a direct, hot burn.
Physical & Chemical Properties That Matter in Practice
Handling trioxane day-to-day, you notice straightaway how moisture-sensitive it is. This chemical dissolves swiftly in water, breaks down under basic conditions, and sublimes if left in a warm room long enough. Its molecular structure—basically a ring made up of three formaldehyde units—gives it strong ties to every chemistry professor’s favorite molecule. Melting occurs at about fifty-eight degrees Celsius, so a hot day in an uncooled warehouse spells trouble. Its volatility and chemical reactivity create both opportunities and headaches: perfect for controlled release of formaldehyde, risky for anyone not storing it tightly sealed.
Technical Specs and Clear Labeling: The Quiet Foundation of Safety
Experience teaches the value of readable labels on chemical containers. Too often, companies treat these as an afterthought, yet a mistake in labeling can provoke confusion or danger on the shop floor. For trioxane, the important figures tend to focus on purity, appearance, and moisture content. Chemists usually want to know how much water and impurities such as formic acid sneak in, since those affect everything from utility as a starting material to shelf life. Transparent technical documentation isn’t just a regulatory hassle—it gives workers and researchers the detail they need to avoid mishaps and ensure consistent product outcomes.
Preparation Method: More Than Just Academic
Few discussions go into the nitty-gritty of manufacturing, but the details behind trioxane production make a real difference. Industrial plants rely on acid-catalyzed polymerization of formaldehyde in concentrated solutions, followed by careful cooling and isolation. This preparation creates a product pure enough for both large-scale plastic synthesis and finicky analytical work. Small mistakes ripple throughout the chain—using the wrong catalyst or failing to control temperature can lead to contaminated batches that upend production schedules or even create dangerous byproducts. Practical experience in manufacturing keeps theoretical chemistry grounded, reducing the gap between the lab bench and the warehouse.
Chemical Reactions & Modifications: Unleashing Versatility
For those in research, trioxane usually acts as a gateway to formaldehyde chemistry. Its slow, controlled breakdown offers an approachable route to polymers and crosslinking reactions. In production environments, it steps in wherever a less volatile formaldehyde source becomes essential. Trioxane also opens doors for making high-strength plastics like polyoxymethylene, which show up in everything from gears to zippers. Beyond its solo act, creative chemists modify trioxane to fine-tune molecular weight or craft specialty intermediates. In applied labs, I’ve watched technicians leverage that modularity, creating tailored products without dealing with the health and handling nightmares of raw formaldehyde.
Synonyms & Product Names Waste No Time
Ask for trioxymethylene, paraformaldehyde trimer, or 1,3,5-trioxane—most chemical suppliers know these names lead to the same substance. Students sometimes stumble on this overlap, thinking they’re reading about different chemicals, when they’re actually revisiting the same molecule through several naming conventions. Accurate language and product designation shouldn’t get lost in translation. Misunderstandings lead to mix-ups, either in classroom experiments or high-stakes industrial supply chains.
Safety & Operational Habits I’ve Grown to Trust
Years working around industrial chemicals reinforce a simple lesson: know the hazards. Trioxane delivers both fire and health concerns in the same package. Its combustibility matters for storage and handling, especially in facilities that already use lots of flammable solvents. Health-wise, inhaling its dust or vapors can lead to eye and respiratory irritation. Outdated habits, like working with open containers or keeping chemicals near ignition sources, invite accidents. Over time, I started following a checklist built on direct experience—tightly closed bottles, isolated storage, well-marked secondary containers. The basics matter most, and trioxane offers no margin for overlooking them.
Where You’ll Find Trioxane Put to Work
Real-world applications surprise people unfamiliar with chemistry’s reach. Military field rations stand apart for their frequent reliance on trioxane-based fuel tablets, since these offer a stable, weather-resistant energy source. In industry, companies withdraw trioxane from warehouse shelves to synthesize plastics that resist wear, solvents, and even acids—critical for consumer and automotive products worldwide. In labs, it acts as a pure, easy-to-measure source of formaldehyde for analytical and synthetic tasks. Hobbyists sometimes use it for emergency stoves or even as a controlled release agent in model-making adhesives. These uses add up to lasting demand and underline the limits of substitutes, especially for field fuel and polymer production.
Research & Development: Chasing Better Paths Forward
Decades of innovation haven’t sapped trioxane’s relevance. Recently, research teams in both the public and private sectors keep pushing for new synthesis routes that give higher yields with fewer unwanted byproducts. Environmental regulations and cost pressures challenge traditional manufacturing, driving a hunt for greener catalysts and reduced waste. In the lab, curiosity keeps sparking fresh applications—from controlled release of biocides to improved composite materials. My time reading through patent filings and research journals proves that the search for better, safer, and more efficient uses continues, reflecting broader trends aimed at sustainability and operational resilience.
Toxicity Insights Learned from Evidence
Many chemicals labeled as “safe” for one use hide toxic edges elsewhere; trioxane lands somewhere in the middle. Animal studies and occupational exposure reports reveal mild to moderate acute effects—enough that proper ventilation and personal protective equipment move from optional to necessary in active environments. Long-term toxicity doesn’t rival the danger of direct formaldehyde, though regulators keep a close watch on cumulative exposure risks and breakdown products. My own protocols now lean heavily on controlling dust, ensuring tight seals, and tracking exposure—even when official guidelines appear less than strict. This cautious approach reflects both practical workplace wisdom and a growing body of toxicological research.
Looking Ahead: Future Roles for Trioxane
Every day, firms and researchers confront growing regulations, tighter supply chains, and new performance demands. Some experts predict trioxane’s role as a source for greener thermoplastics will only grow, provided industry shifts toward catalysts and processes that shrink environmental footprints. Emergency aid organizations seek reliable, safe fuels for remote areas, prompting renewed interest in trioxane tablets that store well and burn cleanly. Synthetic chemists, on the hunt for alternatives to gaseous or liquid formaldehyde, find trioxane’s shelf stability and measured breakdown invaluable for both routine synthesis and advanced polymer engineering. Trioxane may never grace magazine covers, but for those building tomorrow’s materials and getting things done in unpredictable places, it will keep earning a spot in the toolkit.
What is Trioxane used for?
Understanding What Trioxane Does
Trioxane looks like a chemical name you’d only see in a lab, but it’s got much more to offer. I learned about it volunteering at a makerspace with a few survival enthusiasts and later, through reading about advanced plastics. Trioxane steps up both in chemistry and real-world uses.
Fuel in the Field
One of the most practical uses for trioxane shows up in camping and military life. I’ve watched friends prepping for long hikes pull out packets of solid fuel labeled “trioxane bars.” These blue bars light up even when it’s raining. Soldiers use them to heat field rations and boil water with little more than a spark. This chemical packs high energy into a small, lightweight form, hugely valuable where every ounce of fuel counts. Unlike wood, trioxane burns clean with almost no smoke or ash. In survival circles, someone having a few trioxane bars gets hot meals without carrying bulky propane or unreliable fire starters.
Reliable energy matters. Bad storms knock out power. Wildfires or earthquakes leave people stranded. For emergency kits, the solid, stable nature of trioxane means it won’t spill or evaporate. The US military still includes trioxane bars in some rations to this day. That’s telling.
The Lesser-Known Chemical Backbone
Read a label on plastic household storage, and you rarely see the words trioxane, but its fingerprint is there. The industrial world knows trioxane as a building block for polyoxymethylene (POM), a tough polymer used in car parts, gears, and zippers. I once fixed a broken latch on my backpack, not realizing the little plastic cog inside probably started life as trioxane. If you drive a modern car or use a sturdy kitchen tool, you’re sitting one step away from trioxane chemistry.
The manufacturing process needs consistent, high-quality inputs. Trioxane, as an intermediate, offers just that. Factories rely on it to keep supply lines moving. If supply suddenly dried up—factory output goes down, costs go up. In 2020, pandemic disruptions exposed the risks of depending on just-in-time chemical deliveries. Trioxane’s stability and shelf-life give manufacturers more control, which trickles down to daily life—fewer shortages, better products, less waste.
Challenges and Responsible Use
Chemicals like trioxane have risks. Inhaled fumes irritate eyes and lungs. Trioxane’s relatives even show up in the formaldehyde family, which gets flagged for health effects. Here lies the balance: trioxane delivers real, tangible value in fuel and manufacturing, but only with good safety practices. Storing and handling needs tight controls. I’ve seen shops set up fume hoods and require gloves even for small amounts, something every user should take seriously.
Future solutions? Better labeling on consumer products helps people understand what they’re working with. Easier recycling for plastics that started from trioxane sources could reduce environmental costs. More research around alternatives can broaden safer choices, especially if large-scale industrial uses keep growing.
A Chemical With Reach
Trioxane won’t become a household word, but its impacts stretch across industries and day-to-day life. Reliable field fuel and strong plastics both trace back to this one chemical. Knowing what’s behind these hidden tools helps us value both the advances—and the responsibility—they demand.
Is Trioxane safe for human use?
What Is Trioxane, Really?
Trioxane might sound mysterious, but most folks know it as those little white solid fuel tablets you see tossed into camping or survival kits. This chemical—formaldehyde linked in a loop, forming a ring—burns clean and hot. People use it to spark up stoves, purify water, and sometimes in chemistry labs. The focus here turns to a practical worry: does trioxane pose risks when handled or used by people?
Breaking Down the Risks
My time around outdoor gear taught me that convenience can’t excuse danger. Trioxane burns steady and doesn’t give off a ton of smoke, true, but it does release formaldehyde. Anyone who’s spent time in science classes hears that word and thinks of warnings—eye irritation, breathing trouble, possible long-term cancer risk. Those tiny tablets, if burned in a small tent or tight shelter, can spike indoor air with fumes nobody wants to breathe. The American Cancer Society calls formaldehyde a probable carcinogen. I have camped with groups where someone put a trioxane fuel block on a can in a closed space just to avoid cold morning air. Within minutes, headaches or itchy eyes cropped up—body signals that clean air matters.
Facts from the Research
A review of data from trusted medical resources like the Centers for Disease Control and Prevention (CDC) confirms that formaldehyde, even in brief contact, irritates eyes, nose, throat, and skin. Folks with asthma feel it sooner. Extended or repeated exposure links to greater cancer risks, especially for those with weak immune systems. A single weekend of using trioxane in open air probably causes little harm, but folks using it year-round for cooking, heat, or industrial projects run into real health worries. Researchers in Europe and the United States track workplace exposure, setting strict limits for air quality. Military manuals warn soldiers not to use trioxane inside enclosed vehicles or bunkers without strong airflow. No surprise—the health lessons pile up quickly since accidents and overexposure don’t always show up right away.
Safer Ways to Use Trioxane
To lower risks, anyone using trioxane should work outside whenever possible. Good ventilation isn’t just a suggestion, it’s basic survival sense. Kids and people with breathing trouble should stay far from burning tabs. I tell new hikers to avoid touching the fuel with bare hands and never use the residue left after burning as kindling or in cooking. The tablets remain popular because they light fast and work in the rain, but stacking up safety practices doesn’t take extra effort. If anybody forgets, symptoms like stinging eyes serve as quick reminders.
Better Alternatives Exist
Trioxane works in a pinch, but today’s market offers safer options. Solid alcohol stoves, newer synthetic fuel bars, and even pressurized gas canisters give backpackers heat without sketchy chemicals. Cities ban burning some fuels indoors for a reason—air quality drops hard with the wrong products. My own kits swapped trioxane out years ago for bio-ethanol or propane blends, leaving behind problems linked to formaldehyde and adding real peace of mind for family outings.
Health Comes First
Safe habits start with knowing what you’re handling. Trioxane remains handy in emergencies or old-school camping setups, but it never deserved blind trust. Fresh air, gloves, and alternatives turn basic safety into common sense. The bottom line stays simple: if something feels harsh in your nose or chest—step away and rethink the fuel before the harm piles up.
How should Trioxane be stored?
Trioxane Demands Respect in the Storage Room
Trioxane shows up in a lot of places, especially where you need a reliable chemical building block. Most folks working with it quickly notice: this isn’t something you just toss on a shelf with last year’s paint cans. In my own work around chemical stocks and supply rooms, I’ve seen how a casual approach can lead to headaches—or worse.
Why Storage Matters for Trioxane
Nobody likes surprise reactions. Trioxane doesn’t wait around patiently if it gets too warm or damp. It starts breaking down before you know it, and the next thing you notice is a mess or even fumes you don’t want to breathe. Across several workplaces, I’ve found that treating trioxane storage with a bit of extra care pays off every time.
You don’t need a fancy, high-tech vault. Solid shelves, a dedicated spot, and a consistent routine make more difference than you might think. Walk into any well-run chemical store, and you’ll spot the telltale metal or plastic containers, clearly marked, often double-checked by more than one pair of eyes.
Keep Trioxane Cool and Dry
Moisture turns trioxane storage into a problem. Humid air seeps through improper seals or old packaging, causing product loss or even forming dangerous by-products like formaldehyde. In my first job, I watched a new hire set several drums near a swinging door facing the loading dock. Within months, labels began to peel, and the contents no longer matched the records. Keeping trioxane in a cool, dry space makes a world of difference.
Rooms between 15°C and 25°C stay safer year-round. If you can keep the humidity under 60%, your trioxane stays as it should. That sounds basic, but it saves resources and money in the long run—nobody likes disposing of degraded chemicals.
Sealed Containers Remain Non-Negotiable
Trioxane loves to pick up any stray moisture or contaminant, and that ruins its value. Containers with airtight seals work best, and nobody should treat them like everyday buckets or jars from the hardware store. At one site, reusable drums with reliable gaskets delivered peace of mind for years, while carelessly closed bags always wound up costing more in disposal and reordering.
Think about transport, too. Unsealed containers can tip, leak, or spill during any routine move. A proper drum palletized on stable shelving lowers those risks right away.
Labeling and Rotation: Not Just Paperwork
Everyone knows people who just want to move the boxes and get on with their day. Still, labels and stock cards provide a critical backup, especially once multiple staff members get involved. Dates, batch numbers, handling instructions—clear labeling keeps things transparent. No more guessing what’s in the container.
Use older material first. Want to avoid expired or ruined trioxane? Bring the FIFO (first-in, first-out) process into your routine. A warehouse I visited kept a large whiteboard, tracking every trioxane package. Their annual disposal fees for expired stock dropped nearly to zero.
Ventilation and Fire Safety Cannot Slip
Trioxane carries a flammability risk. Storing it away from ignition sources, sunlight, and open flames ranks high on every checklist. Good air movement pushes away any fumes—since even a tiny leak can cause problems over time. Sparking outlets, overloaded power strips, or carelessly parked forklifts don’t belong anywhere near trioxane storage.
A fire extinguisher suited for chemical fires, installed nearby, adds another practical layer of safety. I’ve never seen it used in a well-maintained storage room, but knowing it’s there heads off the worst fears and satisfies the most demanding safety audits.
What are the side effects of Trioxane?
Understanding Trioxane’s Impact on the Body
Trioxane has important medical uses—especially in malaria treatment as an active metabolite of artemisinin. Still, stepping into the world of antimalarial medications, side effects can turn even a small pill into a source of confusion or concern. As someone who pays close attention to medication safety, I know reading long lists of side effects can put anyone on edge. Not every person experiences problems, but those side effects deserve a real look.
Common Side Effects
Trioxane doesn’t go easy on the stomach for many folks. Nausea and vomiting often top the list, disrupting daily routines. Headaches pop up too—nagging and sometimes enough to send you searching for a quiet, dark room. Just like other artemisinin derivatives, dizziness can make walking around risky, especially for older adults. Digestive troubles, such as loose stools or a bitter taste, don’t help either. One patient shared how even after the malaria improved, the stomach upset lingered for days, cutting short family meals.
Uncommon but Serious Reactions
Most people don’t face life-threatening complications. A small group sees their blood counts drop, especially their white cells. This can open the door to other infections. Liver troubles appear in rare cases, sometimes showing up as yellow skin or dark urine—the kind of signs you recognize fast if you watch for them. Allergic responses, like rash, swelling, or breathing difficulties, can seem scary, though they remain rare. For patients with a history of reactions to artemisinin-based drugs, the risk jumps.
Why Side Effects Matter
Ignoring side effects isn’t smart. A tough bout of vomiting or dizziness might mean someone skips pills—and unfinished malaria treatment puts lives at risk. Poor tolerance slows down progress in communities where malaria sweeps through every year. Trioxane’s side effects highlight a real-world challenge: keeping people on medication until the job is done. I’ve seen firsthand how trust in medicine builds one good experience at a time. When that trust breaks, patients drop out of treatment programs fast.
Reducing The Risks
Doctors weigh the pros and cons before handing out Trioxane. Liver and kidney checks help catch issues early. Nurses in malaria zones watch patients for warning signs, stepping in when confusion, fever, or strange bruises show up. Staying hydrated during treatment can soften the blow of digestive side effects. For folks with previous allergic reactions, alternatives like chloroquine or doxycycline might make more sense, though resistance and availability always shape decisions. Making sure people have a support network—family, health workers, local clinics—improves outcomes, especially for those hit hardest by side effects.
Learning from Experience
Every year, new reports from clinics shape how doctors use Trioxane. Long-term monitoring and honest talk between patients and health workers go far. Watching out for warning signs and acting fast keeps small side effects from turning into bigger, lasting harm. By sharing stories, listening to patient concerns, and giving practical advice, the cycle of malaria and its treatments can slowly shift toward safer, more effective care.
Can Trioxane be used as a fuel source?
Understanding Trioxane’s Nature
Trioxane usually lands in the conversation about solid fuels you find in camping gear or military rations. As someone who hikes regularly, solid fuel tabs have saved my dinner plans more than once when wood stayed wet and unreliable. The stuff burns hot and steady—a real asset far from civilization. Trioxane’s role in these tabs isn’t flashy, but it holds the spotlight for compact, portable fuel.
Why Chemists and Tinkerers Get Interested
Trioxane draws eyes in chemistry labs due to its structure: a polymer of formaldehyde. It shows up as a white, crystalline compound that breaks down easily, releasing energy with almost no smoke. Folks who want lightweight, easy-to-carry heat—backpackers, soldiers in the field—respect Trioxane’s simple power. Its ability to burn without making much mess means less cleanup, fewer lingering smells, and a lower fire profile. Those are perks any outdoor enthusiast can respect.
Comparing It To Other Fuels
Gasoline rules the world of transportation for good reasons—energy density, cheap storage, and established supply lines. Propane and butane bring similar strengths, with valves and canisters ready for grills and stoves everywhere. Trioxane can’t push cars down highways or feed factories. Its energy density sits well below liquid hydrocarbons. You can’t load up your gas tank with it and drive cross-country. Even ethanol and methanol, which fall short of gasoline, offer more versatility and fewer risks linked to formaldehyde derivatives.
Risks Hidden in the Details
Trioxane stands out for short, precise tasks. Prolonged or unintended exposure pulls formaldehyde into the picture, which the International Agency for Research on Cancer lists as carcinogenic to humans. Breathing the vapors or touching broken-down fuel over and over might not look dangerous during a quick meal in the woods, but it collects over weeks and months. Working as a safety officer on a wildland fire crew some years back, I learned to check fuel sources for hidden hazards. Nothing ruins a job or an adventure like chemo-absorbed regrets. Some fire tabs ignite easily—too easily if left unattended near kids or pets—so storage and use demand attention.
Scale and Real-World Potential
Trioxane’s industrial production never gained widespread traction for large-scale energy needs. Fossil fuels remain cheap and accessible, with infrastructure built over generations. Trioxane’s creation uses resources better suited for plastics, resins, and construction glue—fields where it finds its real niche. If society pursued a shift to Trioxane for mass fuel, costs, pollution, and health problems would shadow every gain.
Where Trioxane Fits
Not every fuel belongs in every engine or fire pit. For short burns off the grid—melting snow, boiling coffee, keeping a field medic’s hands warm—Trioxane does the job. I’ve relied on it more than once, with water on the boil in no time. As a primary energy source for homes or transportation, though, it falls short against safer, denser, and less hazardous options.
Moving Forward With Smarter Choices
Outdoor communities and emergency planners benefit from understanding the place of specialty fuels like Trioxane. Knowledge, in this case, matters more than hype. Look beyond the marketing and weigh actual risks against real advantages. For anyone planning expeditions off the beaten track, it pays to know what burns clean, what stores easily, and what keeps life comfortable without rolling out new dangers every time supper’s on.
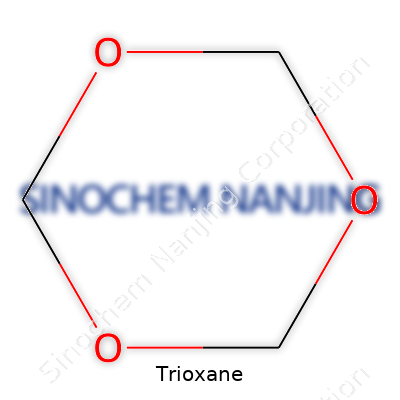
| Names | |
| Preferred IUPAC name | 1,3,5-Trioxane |
| Other names |
1,3,5-Trioxane Paraformaldehyde trimer Trioxymethylene |
| Pronunciation | /traɪˈɒk.seɪn/ |
| Identifiers | |
| CAS Number | 110-88-3 |
| 3D model (JSmol) | `JSmol` "3D model" string for **Trioxane** (1,3,5-Trioxane): ``` C1OCOCO1 ``` |
| Beilstein Reference | 1207022 |
| ChEBI | CHEBI:36256 |
| ChEMBL | CHEMBL1230971 |
| ChemSpider | 53138 |
| DrugBank | DB06436 |
| ECHA InfoCard | 100.003.248 |
| EC Number | 200-963-8 |
| Gmelin Reference | 8277 |
| KEGG | C06042 |
| MeSH | D014353 |
| PubChem CID | 7901 |
| RTECS number | MN3225000 |
| UNII | 2B2B472G6N |
| UN number | UN2814 |
| Properties | |
| Chemical formula | C3H6O3 |
| Molar mass | 90.08 g/mol |
| Appearance | Colorless crystalline solid |
| Odor | odorless |
| Density | 0.9 g/cm³ |
| Solubility in water | 8.3 g/100 mL (20 °C) |
| log P | -0.53 |
| Vapor pressure | 1 mmHg (20 °C) |
| Acidity (pKa) | 5.10 |
| Basicity (pKb) | 6.89 |
| Magnetic susceptibility (χ) | -51.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.128 |
| Viscosity | 1.13 mPa·s (at 20 °C) |
| Dipole moment | 2.30 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.7 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -292 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2054 kJ/mol |
| Pharmacology | |
| ATC code | ``` P01BX01 ``` |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H228, H302 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338 |
| NFPA 704 (fire diamond) | 1-3-1 |
| Flash point | 83°C |
| Autoignition temperature | 427 °C |
| Explosive limits | Explosive limits: 3.5–15.5% |
| Lethal dose or concentration | LD50 oral rat 5200 mg/kg |
| LD50 (median dose) | 1540 mg/kg (rat, oral) |
| NIOSH | CY9275000 |
| PEL (Permissible) | PEL = 5 mg/m3 |
| REL (Recommended) | 0.5 mg |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
1,2-Dioxetane 1,3-Dioxetane Ethylene glycol dioxalate Formaldehyde Paraformaldehyde |

