Trimethylolpropane: A Cornerstone in Modern Chemical Manufacturing
Looking Back: How Trimethylolpropane Came About
Trimethylolpropane, often called TMP in the industry, stands as one of those materials that quietly changed the landscape of coatings, plastics, and lubricants production. In the early 20th century, organic chemists developed methods to convert basic organic building blocks into substances with more reactive centers. TMP came into the spotlight during the rise of synthetic resins and polymers, around the post-war manufacturing boom. Chemists discovered that a simple molecule—trimethylolpropane, marked by its three alcohol groups attached to a central propane backbone—provides a robust, customizable starting point for a host of chemical products. Laboratories began tapping into its function as a crosslinker and branching agent, driving forward innovations in everything from automotive paints to resilient industrial foams.
What Trimethylolpropane Really Is
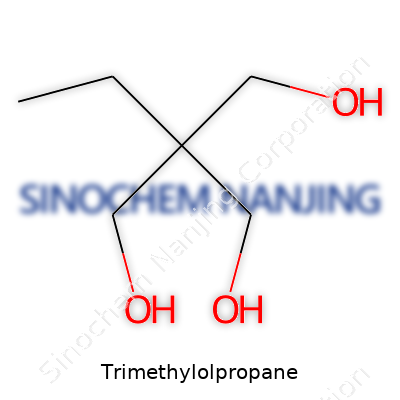
In clear liquid or crystalline form, TMP appears colorless, thick, and carries a mild odor—a far cry from most caustic or hazardous raw chemicals. Its molecular skeleton features a propane base with three hydroxyl groups, earning the name “triol.” This basic structure sets TMP apart from simpler alcohols: the multiple reactive ends allow it to link with acids, isocyanates, and other building blocks, increasing structural complexity in finished products. Synonyms like 2-ethyl-2-(hydroxymethyl)-1,3-propanediol pop up in technical papers, yet the functions always circle back to this layout: a centerpiece for flexibility, durability, and stability in materials science. TMP lurks behind a range of names, which points to its adoption beyond English-speaking markets and within tightly regulated industrial channels.
Technical Details and Physical Behavior
At room temperature, TMP typically forms a white crystalline powder or a viscous, clear liquid depending on purity and storage. It mixes easily in water and polar solvents, tipping the scales at a molecular weight near 134.2 g/mol. The melting point ranges between 56-61°C, and the boiling point hits around 295°C, supporting use in high-heat industrial processes. The chemical’s flash point clears most safety thresholds for mid-scale manufacturing, though storage still demands sensible containment to prevent contact with strong acids or bases.
Making TMP: Synthesis on an Industrial Scale
Industry mainly produces TMP by condensing n-butyraldehyde and formaldehyde under base-catalyzed conditions, followed by hydrogenation. This approach, refined over decades, takes advantage of accessible raw materials and mature chemical processes like aldol condensation. Production scales reliably, and manufacturers can consistently churn out metric tons with tight control over impurity levels. That consistency translates right into material performance on the factory floor and helps finished products conform to international standards for everything from paint hardness to thermal stability in polyurethane foams.
Chemical Reactivity and Route to Modification
TMP’s triol nature sets the table for plenty of downstream chemistry. The hydroxyl groups form ester linkages with acids, react with isocyanates to create urethanes, and connect with epoxides for specialized resins. Additives, flame retardants, and even flexible plastics take shape around reactions that start with TMP’s reactive backbone. This means research teams can “tune” the resulting polymers by playing with TMP’s ratios or by introducing other functional groups at those hydroxyl centers. The versatility here explains why it remains a staple for polymer scientists pursuing custom-fit materials.
Labeling and Compliance in Real-World Settings
TMP may look benign on paper, but labeling falls in line with chemical safety practices. Packaging details show the substance’s CAS number (77-99-6) and hazard statements touching on mild irritation risks. In my role overseeing process safety, I learned firsthand how these compliance details travel alongside bulk shipments and small samples alike, keeping transporters, handlers, and product developers all speaking the same “safety language.” Industry standards and the push for green chemistry led to tighter tracking, reliable traceability, and demands from downstream users for rigorous reporting.
Staying Safe: Handling and Operational Guidelines
No one wants to see spills or inhalation events in a plant environment, even with low-to-moderate-risk chemicals like TMP. My experience in on-site chemical management stressed the importance of good airflow, personal protective gear, and written guidance for anyone transferring or mixing TMP. Standard procedures stress keeping the substance sealed, off the skin, and away from reactive chemical storage. Data from toxicity studies raise minimal alarm compared to more dangerous alcohols, but long-term, repeated exposure stories drive plant supervisors to train up on first aid and emergency response anyway—complacency never pays off.
Why TMP Found a Place in Every Corner of Industry
Walk down any assembly line or peek into a specialty paint shop, and the influence of TMP becomes clear. It acts as a core building block for alkyd resins found in durable outdoor paints, PU foams cushioning car seats, adhesives holding complex electronics together, and lubricants powering engines under tough conditions. Its high reactivity, combined with chemical stability, opens doors for blending into surfactants, printed circuitry coatings, and more. OEMs stick with TMP because trials—both in lab and in real-world stress conditions—keep proving its worth year after year.
Cutting-Edge Research and New Frontiers
In research circles, TMP shows up almost every month: scientists are spinning out new urethane foams for light-weight aerospace components, water-repellent coatings for wind turbine blades, and bio-based transformations looking to wean the industry off fossil-fuel derived ingredients. Sustainable reformulations ask tough questions. Can TMP be produced from renewable resources? Can its reactivity help trap other pollutants or strengthen bioplastics? Studies are underway looking at extending TMP’s reach through catalysis tweaks, new reaction solvents, and greener manufacturing at scale. These incremental changes don’t make front-page headlines, but the combined global impact—reduced carbon footprint, longer-lasting consumer goods, safer industrial practices—shouldn’t be underestimated.
Weighing Toxicity and Long-Term Health
Safety data today reflects extensive toxicity reviews from regulatory agencies and decades-long use. Acute hazard remains low, with skin and eye irritation as the most common complaints from lab settings and factory floors. Chronic effects have received attention too, with animal studies pointing to little bioaccumulation or major organ risk. That said, regulatory bodies keep a close eye on loading thresholds, waste handling routines, and emission controls—which is how industry keeps confidence high among workers, neighbors, and end users. Consistent monitoring and honest reporting run hand-in-hand with keeping TMP’s reputation strong.
Outlook: What’s Next for Trimethylolpropane
Even with digital chemistry and emerging bioplastics nipping at its heels, TMP holds onto relevance in high-value segments. I’ve watched development labs pivot to greener initiators and bio-derived feedstocks, hoping to lessen the environmental footprint without losing out on performance. Antimicrobial coatings, flexible electronics, high-efficiency solar cells—all now depend on clever tweaks to classic trimethylolpropane chemistry. As new regulations kick in, especially in the EU and Asia, industry’s pressure to recycle, extend product lifetime, and lower toxicity may open up next-generation TMP derivatives we haven’t seen yet. That evolution, built on one molecule’s flexibility, promises to keep material science on its toes for years to come.
What is Trimethylolpropane used for?
Shaping Daily Materials Behind The Scenes
Most people never hear about trimethylolpropane, though it shapes plenty of products we count on every day. From the seat of a city bus to the frame of a pair of glasses, TMP shows up as a kind of silent partner. I’ve seen firsthand how much manufacturers lean on TMP when aiming for toughness, elasticity, or resistance to the wear-and-tear of daily life.
What TMP Brings To Industry
Trimethylolpropane pops up often in coatings, especially paints and varnishes. Manufacturers mix it with other chemicals to create polyester and alkyd resins. These resins coat doorframes that face rain, sleet, or sunlight every season. When walking through a city, I know street signs and railings owe some of their durability to TMP-based finishes. It plays a big part in making surfaces last longer and look better.
TMP also finds a home in plastics and polyurethanes, from foam mattress cores to skateboard wheels. Chemists favor it because of the way it links up with other molecules, weaving stronger, more resilient chains. Furniture makers choose foams made with TMP since they balance springiness with the ability to bounce back after months or years of daily use.
Why It Matters For Performance And Sustainability
Durability remains important, but so does environmental responsibility. TMP-based resins help cut down the need for frequent repainting, saving money and reducing waste. I remember talking with building contractors who track savings made possible by coatings that hold up season after season. Every time a surface needs attention, more water, energy, and raw material gets used. Better coatings can delay that cycle.
TMP doesn’t just serve the paint or plastics world. Lubricant manufacturers also count on it. By creating esters from TMP, they make oils that stick around inside engines and machines without breaking down or gumming up. Car owners and industrial workers feel the impact over time, as these oils keep equipment running longer, cleaner, and with less downtime.
Worker Safety And Handling Concerns
I have seen plenty of factory safety posters and personal protective equipment—TMP shouldn’t be handled carelessly. While not highly toxic, it can cause irritation with repeated or direct contact. By training workers and using proper ventilation, companies keep risks down. Manufacturers who treat worker safety seriously protect not just those in the factory but also everyone who depends on a reliable supply chain.
Challenges And The Push For Better Alternatives
Despite many strengths, TMP-based products run into some hurdles. As demand for biodegradable and eco-friendly materials rises, researchers keep searching for something that matches TMP’s toughness with a lighter touch on the planet. Bio-based feedstocks and greener resins show promise, though they haven’t matched TMP’s scale or price yet.
From what I’ve witnessed, real progress comes from partnerships—chemists linking up with manufacturers and consumer advocates to steer innovation in a healthy direction. The companies that stay nimble, swapping in safer or more sustainable ingredients, often earn the trust of both employees and the public.
Where TMP Fits Into Tomorrow’s Materials
Innovation moves quickly, but for now, trimethylolpropane keeps doing the unseen work of holding together floors, frames, and finishes. Every push for something stronger, more flexible, or longer lasting brings new questions about what goes into these materials and how the choices we make ripple outward. For those of us who care about what products are made of, TMP remains a good reminder that even small molecules can leave a big impact.
What are the main properties of Trimethylolpropane?
Getting to Know Trimethylolpropane
Trimethylolpropane, or TMP, shows up in a surprising number of products. Anyone working in coatings, plastics, or resins probably deals with TMP, even if it’s behind the scenes. It’s a small molecule, but it packs a punch when it comes to the features it brings to the table. The heart of its promise lies in the fact that it has three hydroxyl groups sticking out from a central carbon skeleton. Picture a tripod, each leg grabbing onto a chemical reaction and holding on tight. This structure sets it apart from simpler alcohols and explains why industries keep coming back to it.
Physical and Chemical Traits
TMP looks a bit like table sugar—white, chunky, and crystalline. Its melting point lands around 56-58°C, which means it’s solid at room temperature but pretty easy to melt down for mixing into formulas. Like many alcohols, TMP dissolves in water, though not freely. It does much better with solvents like acetone or ether.
A draw of TMP is its stability under most storage conditions. It won’t break down or change character just sitting on the shelf. Plus, it doesn’t stink—unlike some chemicals with industrial reputations. TMP doesn’t easily evaporate, so you don’t have to worry about losing it to thin air. Handling it feels about as safe as things get in a chemical lab. There’s always a need for common sense with any industrial chemical, but TMP doesn’t carry the acute toxicity worries you’d find with some other raw materials.
The Role TMP Plays in Manufacturing
People reach for TMP mostly because of how those three hydroxyl groups behave. Each one creates a spot to react with acids, turning TMP into the backbone of many synthetic resins. Alkyd and polyester resins depend on TMP to boost flexibility and keep their structure tough over time. In coatings, that translates to paint that weathers years of sunlight and still holds its color and finish. In the lab, these reactions aren’t just textbook chemistry—they make the difference between a coating that cracks and one that lasts through a decade on a busy school hallway wall.
TMP also lets manufacturers control the thickness and flow of resins by changing just how much of it they mix in. There’s a direct link between molecule structure and product performance. I’ve seen production lines where flipping between TMP grades let them speed up cure times or tweak gloss without introducing risky additives.
Possible Downsides and Where Things Go Next
No story about industrial chemicals feels complete without talking about trade-offs. TMP comes from a process that relies on formaldehyde and n-butyraldehyde, so the environmental cost sits upstream unless producers use modern, cleaner synthesis routes. Also, over-reliance on TMP can lead to materials that are too hard or brittle unless formulations get balanced with softer polyols. Anyone aiming for sustainability has to think about where the TMP comes from and what happens when the final products reach their end of life.
Cleaner processes have started surfacing, and some producers recycle or recover byproducts, trimming back on waste. This kind of attention will only grow in importance. Customers ask more questions now about every stage of a product’s journey, from raw material to disposal. TMP’s strong performance will keep it in the conversation as sustainability expectations rise.
Is Trimethylolpropane hazardous or toxic?
Looking at Trimethylolpropane in Everyday Terms
Trimethylolpropane, known in industry circles as TMP, shows up in more places than you expect—coatings, polyester resins, even lubricants that keep machines running. Most folks don’t spray it on their hands or pour it in their coffee. People often bump into TMP not through direct touch, but by coming in contact with products that rely on its chemistry.
Factories deal with loads of TMP each year. The stuff plays a big role in the performance of durable paints, adhesives, and even certain plastics. But the moment science stirs up a name like “Trimethylolpropane,” caution sirens blare for many. Folks want to know if it causes trouble for health or the planet.
Health Concerns and Exposure
TMP doesn’t drift through city air by itself. Those working at chemical plants sit closest to risk through skin contact or breathing in dust during manufacturing. Studies suggest that breathing in high amounts may irritate the nose, throat, or lungs. Getting TMP on bare skin may cause redness or itching, especially if someone skips gloves or the substance splashes without warning.
The US National Library of Medicine gives TMP a pretty clear profile: not classified as cancer-causing, not mutagenic by current research, not likely to poison workers through trace exposure. Acute toxicity sits low—meaning short and accidental contact hardly ever leads to hospital visits. That said, rubbing it into your skin daily or swallowing a tablespoon comes loaded with risk just like most chemical agents.
TMP does not explode in sunshine or pollute rivers in blob form. Waterways don’t often fill with big TMP spills, but wastewater treatment setups keep prepared just in case. It’s not pleasant to taste or safe to sprinkle on food. It should stay sealed and far from children and pets, same as cleaning sprays or antifreeze.
Safety in Industrial Use
The Material Safety Data Sheet (MSDS) rules the day in plants handling TMP. Workers wear safety specs and gloves to skip skin troubles. Proper fans run to sweep away any loose powder. Companies do not just toss TMP barrels beside nearby creeks. Risk management teams work to avoid leaks and keep workers healthy with periodic health checks.
From experience working around factories with similar chemicals, training makes the difference. Reading a safety label never replaces giving people the chance to pause and double check labels or equipment before pouring a batch of resin or sweeping up powder. That’s the line where most injuries start—too many shortcuts or not enough attention.
Environmental Impact and Responsible Handling
TMP scores lower on long-term environmental harm when compared to heavy-duty solvents or oil-based chemicals. Still, a spill could kill fish or plants in a stream by taking up too much oxygen. Making a habit of leak checks and storing the chemical away from flood-prone areas keeps accidental releases in check.
Solutions to minimize risk come straight from action—strong training programs, strict housekeeping, locking up TMP in labeled drums, and keeping emergency cleanup gear ready. Innovation can also play a role. Alternatives show up over time that use renewable sources or create less waste. Green chemistry models point the way toward safer substitutes, though finding one that truly matches TMP’s technical magic remains a tall order.
Why Oversight Remains Key
No need to panic over the presence of TMP in a plastic bench. Still, the small print stays important—every workplace or community near a chemical facility stands to gain from transparent reporting, fast cleanup drills, and a culture where questions get answered right away. Laws calling for disclosure and safety reporting keep everyone honest.
At the end of the day, respect for this colorless solid comes through real-world routines—handling it with care, storing it right, and never forgetting that every chemical brings costs as well as benefits.
How should Trimethylolpropane be stored and handled?
Understanding the Substance
Trimethylolpropane shows up in many workplaces, especially in resin production, paints, and coatings. It takes the form of a white, crystalline solid. If left unchecked, dust from this chemical can float around and cause irritation to skin or eyes. Inhaling it isn’t safe either. Folks who have worked in chemical plants know a slip in basic precautions turns an easy day into an accident report.
Personal Experience on the Shop Floor
I remember shadowing an old-timer in a midwestern paint facility. He told me stories about the “bad old days” when dust masks were a suggestion, not standard gear. His hands, always a bit rough, reminded me what happens without simple gloves or eye protection. For us, gearing up with goggles, gloves, and solid shoes became as automatic as clocking in. These steps go a long way in keeping splashes and dust out of places they don’t belong.
Storage Matters
One lesson stood out early: don’t stack trouble. Trimethylolpropane should stay in cool, dry areas, away from heat or open flames. The containers need thick, tight-fitting lids. Keeping things dry keeps clumps and spills down, plus it cuts down the chance of slipping on stray powder. Clear labeling keeps everybody on the same page, especially during shift changes. Fire codes usually call for good ventilation. That’s not just a rule for inspectors; dumping vapors out of the room means one less thing to worry about during a long shift.
Good Habits Make a Difference
Leaving a mess is an invitation to trouble. Using a vacuum built for fine powders beats pushing dust into the air with a broom. Everyone in the crew, from new hires to veterans, cleans as they go. The old “leave it better than you found it” motto never wears out.
I once watched a mix-up when two very similar barrels got swapped. Nobody got hurt, but it caused a scramble and wasted a good afternoon figuring out what went wrong. The label on top saved the day. Now? Red tags for potential hazards and a sign-out system for containers keeps the team honest and organized. It sounds simple, but it works.
Handling Emergencies
Bad things happen fast. The local safety data sheets stay posted close by, but nobody wants to be reading those in a panic. Fire extinguishers sit by every entrance, and spills get covered with dry absorbents, wiped up, and put in the waste bin immediately. Eyes need washing stations even if it feels old-fashioned. I’ve seen experienced folks skip safety drills, then regret it during real spills. Running drills with the actual team, pretending it’s the worst day possible, means everyone knows their role, even if nerves take over.
Training Builds Confidence
I used to think all those refresher classes were overkill. Turns out, most accidents come from small mistakes—like hauling containers without checking lids, or plugging gaps with the wrong label. Good training tightens up those loose ends. Ongoing talks—real conversations, not checkbox meetings—help everyone look out for each other. Nobody wants to see their buddy get hurt on the shift.
No Shortcut Replaces Care
Safe storage and mindful handling of trimethylolpropane protect both people and property. Each smart habit passed from one worker to another makes it harder for accidents to sneak in. After years around chemicals, keeping things organized, labeled, and ventilated isn’t just routine—it’s a way to make sure everyone goes home safe at the end of the day.
What industries commonly use Trimethylolpropane?
The Backbone of Paints and Coatings
Coatings keep cars shiny and protect industrial equipment from rust. The secret sauce for many of these hard-working paints comes from trimethylolpropane (TMP). This chemical brings flexibility, gloss, and toughness, letting paints endure weather and wear. Paint manufacturers often look to TMP when they need to boost the durability of surfaces that take a beating. Floor coatings and exterior paints owe a lot of their toughness to this one ingredient. In my own work renovating homes, I’ve seen coatings with TMP hold up season after season under sun and rain.
Plastics and Polymers: Holding Strong
TMP plays a vital part in making polyurethanes. These appear everywhere—from skateboard wheels and phone cases to insulation foam. Manufacturers blend TMP into polyols, and these eventually turn into polyurethane plastics. By tweaking the formula, companies adjust flexibility or rigidity, letting products last longer or resist cracking. The result shows up in car seats that resist flattening and shoe soles that stay bouncy year after year. A recent report from Grand View Research pegged the polyurethane market at $81.3 billion in 2022, and demand keeps climbing—partly buoyed by ingredients like TMP.
Lubricants: Keeping Machines On The Move
Lubricant makers call on TMP for its ability to make stable, long-lasting oils. Synthetic lubricants built with TMP esters hold up under high temperatures and heavy loads. They show less breakdown over time, giving machines a longer and more reliable life. I’ve worked with a small engine repair shop, and they swear by these oils in everything from lawnmowers to motorcycles. Even commercial airlines and wind turbines count on lubricants featuring TMP, since failures in those settings cost big money and risk safety.
Adhesives and Sealants
Strong adhesives keep furniture joints locked tight, hold shoes together, and play a part in everything from bookbinding to construction. TMP strengthens the chemical backbone in certain glue formulas. Formulators can count on reliable curing times and bond strengths, cutting down product failures. In the building industry, a good adhesive means fewer callbacks for repairs and less waste from thrown-out materials. Even in basic woodworking, I noticed joints with TMP-based glue tend to resist peeling and splitting much better.
Flexible Solutions in the Chemical Industry
TMP gives chemical engineers room to experiment. It acts as a starter molecule for producing a range of ethers, esters, and resins. These form the raw materials for specialty inks, textile treatments, and even some cosmetics. I chatted with a few chemists who mentioned TMP-based resins offering resistance to moisture in specialty paper products—think shipping boxes that don’t fall apart during humid summers.
Getting the Details Right—And Safer
Safety matters, both for workers and the environment. Chemical makers have spent decades refining how TMP gets produced, stored, and used to limit emissions and reduce waste. Regulatory bodies and industry groups keep a watchful eye on handling practices. My time covering industrial safety taught me that proper training and equipment go a long way toward keeping these innovations both productive and safe. Investing in closed-loop systems and proper ventilation remains a priority for the facilities that handle TMP in bulk.
Putting It All Together
Trimethylolpropane might not be a household name, but it’s mixed into so many daily products that its absence would be noticeable. Industry groups, material scientists, and regulation all play their part to keep its benefits rolling into paints, plastics, lubricants, adhesives, and more. Its story underscores an idea I see over and over: essential chemicals, handled right, bring comfort and function to modern life, often behind the scenes.
| Names | |
| Preferred IUPAC name | 2-ethyl-2-(hydroxymethyl)propane-1,3-diol |
| Other names |
2-Ethyl-2-(hydroxymethyl)-1,3-propanediol Tris(hydroxymethyl)propane Trimethylolpropyl alcohol TMP |
| Pronunciation | /traɪˌmɛθɪloʊlˈproʊpeɪn/ |
| Identifiers | |
| CAS Number | 77-99-6 |
| Beilstein Reference | 1910526 |
| ChEBI | CHEBI:35582 |
| ChEMBL | CHEMBL62908 |
| ChemSpider | 5461 |
| DrugBank | DB14024 |
| ECHA InfoCard | ECHA InfoCard: 012119457661-43-0000 |
| EC Number | 202-254-3 |
| Gmelin Reference | 8419 |
| KEGG | C06233 |
| MeSH | D014260 |
| PubChem CID | 8675 |
| RTECS number | UZ2990000 |
| UNII | 7BRR5G8K77 |
| UN number | UN2323 |
| Properties | |
| Chemical formula | C6H14O3 |
| Molar mass | 134.18 g/mol |
| Appearance | Clear, colorless liquid or white crystalline solid |
| Odor | Odorless |
| Density | 0.966 g/cm³ |
| Solubility in water | miscible |
| log P | 0.28 |
| Vapor pressure | 0.01 mmHg (20 °C) |
| Acidity (pKa) | 14.7 |
| Basicity (pKb) | 8.06 |
| Refractive index (nD) | 1.446 |
| Viscosity | 75 mPa·s (25 °C) |
| Dipole moment | 2.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -603.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3706 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319 |
| Precautionary statements | P280: Wear protective gloves/protective clothing/eye protection/face protection. |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 199°C |
| Autoignition temperature | 400 °C |
| Explosive limits | Explosive limits: 1.1–7.0% |
| Lethal dose or concentration | LD50 (Oral, Rat): 2769 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Trimethylolpropane: 5,000 mg/kg (oral, rat) |
| NIOSH | NIOSH = "XD5250000 |
| PEL (Permissible) | 100 mg/m3 |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Glycerol Pentaerythritol Trimethylolethane Neopentyl glycol Ethylene glycol |

