Understanding Trimethylamine Hydrochloride: A Practical View on Its Role and Future
Historical Development
Trimethylamine hydrochloride has a background that tracks the path of both curiosity and necessity in chemical science. Chemists observed how the manipulation of amines in the late 19th and early 20th centuries opened up novel pathways for new compounds. Trimethylamine emerged as a compound of interest because of its basic structure—three methyl groups bonded to a single nitrogen atom. The hydrochloride salt revealed a way to stabilize and handle an otherwise highly volatile substance. Over decades, researchers moved from small-scale isolations in analytical labs to industrial applications, reflecting a broader story of how chemicals with pungent smells and reactive personalities often build fundamental connections across disciplines like agriculture, pharmaceuticals, and synthetic chemistry.
Product Overview
It’s easy to overlook trimethylamine hydrochloride in the collection of specialty chemicals unless you’ve worked with it directly. This compound shows up as a white, crystalline solid, presenting much less of an olfactory challenge than its parent, trimethylamine, which is notorious for its sharp, fishy odor. That conversion to a stable salt doesn’t just make storage and handling easier, it creates a launching pad for reactions that call for a reliable source of a methylating agent. I remember keeping a vial tucked away in a desiccator because exposure to the air could make it sticky, given its hygroscopic nature. The product typically lands in labs looking for predictable behavior in both synthesis and research.
Physical & Chemical Properties
In my personal experience and across standard chemical catalogs, trimethylamine hydrochloride reveals itself as a fine crystalline powder that dissolves quickly in water. The hydrochloride form eliminates the strong ammonia-like vapor, and the substance remains solid at room temperature. Heat sticks to it, leading to decomposition rather than melting in the way other organic salts do. It behaves as a quaternary ammonium compound, marked by its strong ionic pairing—so it stands up well to regular lab conditions but shouldn’t cross paths with strong bases, which will swing it back toward the intensely smelly amine form. Its chemical formula, C3H9N·HCl, makes it direct enough for calculation in stoichiometry-driven syntheses.
Technical Specifications & Labeling
Every reputable supplier pays close attention to purity, moisture content, and visual appearance. Labels may include CAS numbers, formula weight, and best-by dates, but real confidence comes from suppliers willing to provide Certificates of Analysis and third-party verification. Laboratories doing fine work demand consistent melting points and low levels of residual impurities—it’s frustrating to find batch-to-batch differences during a project. While these specs don’t matter much in casual conversations, they can make or break things in pharmaceutical intermediates or analytical standards.
Preparation Method
Chemists typically build trimethylamine hydrochloride by reacting the parent amine vapor with concentrated hydrochloric acid. It’s a straightforward acid-base reaction; the hydrochloride salt drops out as a solid almost immediately. This synthesis calls for well-ventilated hoods and acid-resistant containers. Anyone who’s carried out this prep will recall handling trimethylamine as a pressurized gas, then watching a crystalline mass deposit as the acid flows in—a certain satisfaction comes from seeing chemistry work so reliably, even if it’s not glamorous. On a larger scale, the simplicity of making this compound gives it an edge, and process engineers mostly focus on managing vapors and minimizing byproducts.
Chemical Reactions & Modifications
This salt opens the door to a range of synthetic reactions, including quaternization and alkylation steps central to making surfactants or pharmaceuticals. The hydrochloride form doesn’t engage in as many direct reactions as the free amine, but in the right solvent or with the right base, you can ‘free’ the amine and use it as a nucleophile or base. For anyone pursuing organic synthesis, trimethylamine hydrochloride often serves as a stepping-stone—simple, stable, and always ready when you need a methyl group source or a buffer against stronger acids.
Synonyms & Product Names
Anyone researching or ordering this chemical meets alternate labels. Chemists call it trimethylammonium chloride or TMA-HCl in shorthand. Different suppliers brand it according to their catalogues, but those looking for it in literature should keep all common synonyms on hand. The point isn’t to confuse, but rather to align with the conventions set by generations of researchers before us—whether reading Russian journals or supplier certificates.
Safety & Operational Standards
Real-world chemistry always brings safety to the foreground. Trimethylamine hydrochloride, unlike the free amine, presents fewer acute hazards, but care stays essential. Moisture absorption can change weight calculations if containers aren’t closed tight. In any lab or industrial setting, operators wear gloves and avoid breathing dust; the mild acidity of the hydrochloride doesn’t carry the same punch as the parent gas, but chemical hygiene stays non-negotiable. Storage looks like any other hydroscopic chemical: tightly closed, labeled, and away from strong bases. The most common headaches in labs relate not to acute toxicity but to improper storage, which shows the difference between following the letter of the MSDS and working day-after-day toward a safe environment.
Application Area
Trimethylamine hydrochloride moves quietly into a surprising range of applications. It supports the synthesis of pharmaceuticals, especially as a building block for quaternary ammonium compounds used in everything from disinfectants to neurologically active drugs. Its role in organic synthesis spans dye manufacture, ion-exchange resin creation, and even serves in analytical chemistry as a calibration standard. The versatility found in labs and plants traces back to its reliable supply of methyl groups without bringing in much synthetic baggage.
Research & Development
Chemistry research leans heavily on reliable intermediates, and trimethylamine hydrochloride has proven to be a dependable partner. Studies push its limits in catalysis and drug development, with researchers testing new derivatives for both improved physical properties and novel biological effects. Anyone who’s spent time optimizing a synthetic sequence knows that having a stable, easy-to-handle precursor goes a long way toward building more complex molecules. Some work even investigates modifications that might lead to more environmentally friendly or less toxic analogs, although these always take years to translate from bench to market.
Toxicity Research
Research into toxicity focuses on both acute and chronic exposure. Compared to its parent compound, the hydrochloride salt doesn’t stir up the same level of concern for accidental release, but oral, inhalational, or dermal exposure can still irritate mucous membranes and may cause headaches or gastrointestinal symptoms in large doses. Long-term studies remain limited, likely because industrial volumes rarely lead to large-scale exposures—unlike solvents or pesticides that saturate certain industries. Regulators keep it on their watchlists, though, and review its safety as part of broader evaluations of the amine family. Real lab experience—whether at the bench or in regulatory departments—teaches that routine respect for this compound sidesteps the majority of potential problems.
Future Prospects
As industries shift toward greener and safer chemicals, attention to simple, well-characterized compounds like trimethylamine hydrochloride stays high. Ongoing research explores options for using it in environmentally friendlier catalytic systems and turning the focus to derivatives with lower environmental impact. Its direct value in synthesis may never attract headlines, but it builds crucial links in producing new drugs, materials, and analytical tools. Chemists working in sustainability see promise in redesigning these basic chemicals to either biodegrade more easily or to support greener process chemistry. Whether exploring amine-modified sensors or ion-exchange systems for water purification, trimethylamine hydrochloride looks set to stay in the repertoire of working chemists looking to balance reliability, safety, and opportunity.
What is Trimethylamine Hydrochloride used for?
Where You’ll Find Trimethylamine Hydrochloride
Trimethylamine hydrochloride isn’t a name you see plastered across supermarket shelves, but it shows up behind the scenes in many places that touch our daily lives. Chemists rely on it in laboratories, especially in organic synthesis. It serves as a helpful building block in the process of making medicines, dyes, and photographic chemicals. Anyone who has ever taken certain antihistamines or antihypertensive medications has probably benefited from procedures that use this compound.
The Roles Trimethylamine Hydrochloride Plays in Chemistry
In the lab, this chemical isn’t flashy. It appears as a simple, odorless white crystalline powder. What gives it a place of value is its ability to transfer its methyl groups to other molecules. This property makes it helpful for researchers working on new drug candidates or exploring how to build bigger molecules from smaller pieces. Facilities focused on pharmaceutical research turn to this compound time and again because it enables a range of key reactions. The smooth workflow it supports matters more than any fuss over branding or appearance.
Industrial Uses That Reach Further Than You Might Expect
People might never see trimethylamine hydrochloride at work, but industrial chemists do. In the world of polymer manufacturing, it helps fine-tune the conditions needed to make plastics that form the basis for electronics, medical devices, and countless everyday products. Its presence in the production of phase transfer catalysts makes complicated chemical reactions far more practical at scale. Many people depend on plastics and chemicals produced in these processes, though they may not even know the starting ingredients.
Pharmaceutical Importance and Safety Concerns
Pharmaceutical companies often use trimethylamine hydrochloride as an intermediate. It acts as one of several steps in the creation of more complex molecules found in drugs for treating illnesses ranging from high blood pressure to allergies. Because it is used in synthesis, people never take it directly. Instead, it serves as a tool for building safer, finished products.
Handling this compound, though, calls for respect. Exposure to trimethylamine gas, which can be released if the hydrochloride breaks down, may irritate eyes and respiratory tracts. Researchers and workers use protective gear and work in controlled environments to avoid accidents. Safety training and regulations play a big role in making sure that only skilled professionals work with this material.
Environmental Responsibility and Alternatives
Production and disposal of chemicals like trimethylamine hydrochloride can have consequences for the environment. Wastewater from labs or factories sometimes contains traces of this compound, and without proper treatment, it could migrate into soil or waterways. Over the years, regulations from bodies such as the Environmental Protection Agency (EPA) have pushed manufacturing facilities to clean up their waste streams and find less hazardous alternatives where possible.
My experience in a research facility showed me the importance of having clear protocols in place. We kept logs detailing how much chemical entered and left the lab. Strong partnerships with waste disposal companies proved just as critical. Over time, greener chemistry has become more than just a buzzword. Scientists search for catalysts and reagents that lower toxicity and environmental risk, cutting down dependency on substances like trimethylamine hydrochloride.
Looking for Smarter Solutions
As demand for safer and more sustainable chemistry grows, labs and manufacturers can keep raising the bar. Training, investment in proper equipment, and embracing innovative alternatives support people and the planet, without losing sight of the essential tools that help move science forward. Trimethylamine hydrochloride might not draw much attention in the public eye, but it serves as a reminder of the invisible work that goes into chemistry and industry every single day.
Is Trimethylamine Hydrochloride safe to handle?
Why People Care About Chemical Handling
Many of us remember the first time lab safety rules got drilled into our heads in high school science. Safety glasses, gloves, and the odd horror story about what happens if you don't follow instructions. Nobody wants a skin rash or worse from an unknown compound. Trimethylamine hydrochloride doesn’t stand out in everyday conversation, but folks who work with chemicals or in research settings often run into it. Ignoring the basic safety information can lead to a short ride to the nurse’s office – or a much longer trip to the hospital.
What Trimethylamine Hydrochloride Is
Trimethylamine hydrochloride shows up in labs, chemical plants, and sometimes classrooms. It looks like a white crystalline powder. People use it in organic synthesis, drug manufacturing, and sometimes for research. Unlike household cleaners or sugar, it isn’t something you want lying around your kitchen. The chemical brings a strong, fishy odor that you won't soon forget. The smell alone pushes most people to treat it with respect.
Safety Profile Backed by Experience
My first brush with trimethylamine hydrochloride came as an undergraduate in an organic synthesis lab. The safety data sheet sat stapled above the workbench. It laid things out clearly: skin and respiratory irritation, eye damage if splashed, and serious nausea if inhaled in quantity. The stuff soaks into skin much faster than you’d expect. Wearing gloves and working under a fume hood became automatic. A classmate once worked outside that hood to save time, and got a headache that chased him for the rest of the day.
A look at public records and chemical safety databases like PubChem and the European Chemicals Agency shows warnings that echo that experience. Trimethylamine hydrochloride doesn’t easily catch fire, but the dust and vapor can sting your lungs in seconds. The hydrochloride form means it dissolves easily in water, making accidental contact more likely. Even a small spill on the bench, if not cleaned up right away, can make a room smell fishy for hours and bother anyone with sensitive lungs.
Risks and Real Solutions
The Centers for Disease Control and Prevention (CDC) offers straightforward rules for handling chemicals like this: goggles, gloves, full lab coat, never work without ventilation. Accidents have a habit of happening fast, and sometimes complacency sets in after handling dozens of safe or routine compounds. I’ve seen labs skip the last step, letting vapor drift without a fume hood. In the worst cases, someone gets sick or chemical burns develop.
Training remains the biggest hurdle. Many smaller companies don’t run regular safety refreshers. Even major labs get busy, and someone new ends up learning safety procedures from a roommate, not a professional. Real change comes from constant reinforcement – hanging up posters, holding short weekly safety chats, keeping extra gloves and splash goggles stocked.
The Bottom Line
Trimethylamine hydrochloride won’t hurt anyone sitting, sealed in a bottle. The minute the cap comes off, caution needs to kick in. As long as people follow time-tested safety advice — gloves, eye protection, ventilation, never eating or drinking where chemicals are handled — the risks drop dramatically. Treating every chemical with a little respect saves more than time; it protects health and careers.
What are the storage conditions for Trimethylamine Hydrochloride?
Why Proper Storage Really Matters
Trimethylamine hydrochloride often shows up in research labs and chemical facilities. It comes with its own quirks, and storing it the wrong way causes a lot more trouble than people might imagine. The stuff gives off a strong odor, and if you’ve ever cracked open a bottle in a small room, you’ll remember it. Breathing those fumes makes for a bad day, and careless storage raises safety risks for anyone around. Not long ago, I helped set up a shelf in a university storeroom. One leaky container left the staff with headaches and complaints. Those lessons don’t stick until you experience them yourself.
Moisture: The Main Enemy
Trimethylamine hydrochloride, being hygroscopic, grabs hold of water from the air. Even a little extra moisture makes it clump or degrade, which messes with its purity and measurement for experiments. Keeping it in an air-tight container protects it from humidity. Some people swear by glass bottles with sturdy screw caps — I’ve used them, and they really hold up under daily handling.
Store Away From Heat and Light
Heat will shorten this chemical’s shelf life, and direct sunlight can mess with its stability. The safest bet is a cool, dry place, out of sunlight. Most research labs use a dedicated storage cabinet, away from heat sources — not on top of radiators or near a sunny window. On crowded days, people get tempted to set containers wherever there’s space. Give in to that temptation, and it’s not long until quality drops or accidents multiply.
Avoiding Incompatible Substances
Chemicals don’t get along with every neighbor. Trimethylamine hydrochloride especially hates strong oxidizers and acids. If stored nearby, fumes and traces can react, sometimes releasing toxic gases. In most lab safety workshops, instructors press the point: keep incompatible chemicals separated, never mix bottles on quick shelves, use secondary containment if possible. These habits cut the risk of cross-contamination or something much worse.
Handling and Labeling
There’s no excuse for unclear labels. Faded tags or missing hazard signs can put everyone in danger, especially shift workers and new staff. Whenever I restock, I double-check labels and safety sheets. If I mess up, I expect my coworkers to call me out. It saves time and safety issues later on. Wearing gloves and eye protection makes sense, even for short tasks, since the dust and fumes hit hard on exposed skin and eyes.
Safe Disposal Matters Too
Expired or spilled trimethylamine hydrochloride shouldn’t just go in the trash. Most labs follow local hazardous waste guidelines. A sealed waste bin and regular disposal schedule keep the risk low. Years ago, a colleague tried to wash traces down the sink, which ended with an emergency call to the safety officer. One bad move can taint plumbing or hurt cleaning crews.
Finding Solutions
Strong habits in storage are built with real practice. Training new staff, using lockable cabinets, regular inventory checks, and clear signage — all make for fewer mishaps. For facilities handling large amounts, humidity monitors and backup ventilation give peace of mind. At the end of the day, a well-stored trimethylamine hydrochloride means fewer headaches, safer labs, and a much better experience for everyone involved.
What is the molecular formula of Trimethylamine Hydrochloride?
Why the Formula Matters
Trimethylamine hydrochloride carries the molecular formula C3H9N·HCl. This might seem dry at first glance, but behind these letters and numbers lies a substance connecting not only bench-top chemistry, but also real-world problems and opportunities. As someone who spent afternoons in university labs surrounded by vials, I learned that knowing a molecular formula is more than trivia. It helps researchers vouch for purity, avoid accidents, and plan reactions with confidence.
The Building Blocks: What’s Inside
The structure comes down to three methyl groups attached to a nitrogen, with the hydrochloride representing an added chloride ion. Altogether, the compound features three carbons, ten hydrogens, a nitrogen, and a chlorine atom. Breaking it down in plain terms, trimethylamine forms a gas with a strong fishy odor, and bringing hydrochloric acid into the mix turns it into a solid, more manageable for shipping, storage, and measurement. Chemists rely on these forms for a reason: some substances work better as solids, especially in lab environments where control and safety come first.
Importance in Research and Industry
Labs and factories value trimethylamine hydrochloride for its versatility. Pharmaceuticals, agriculture, dyes, and many other fields need reliable sources of nitrogen-based compounds to synthesize medicines, solvents, or even antistatic agents. If someone’s ever looked into antihistamines or local anesthetics, they’ve benefited from chemistry that traces back to simple building blocks like trimethylamine hydrochloride.
I realized in my first research job that memorizing these formulas isn't just good for passing exams. Toxicity, reactivity, and even industrial pricing depend on knowing exactly what’s inside a bottle. Take the hydrochloride version: solid form keeps fumes at bay. Safety data sheets warn about the hazards, like throat or eye irritation from the gas version. Regulations often require chemical handlers to mark and store each compound with exact details, which means nobody can afford to act on guesses or roundabout estimates.
Sourcing and Safety
Reliable sourcing depends on careful documentation. Whether a factory imports bulk or a school orders a few grams, suppliers rely on the precise formula to track shipments and maintain compliance. Mishandling pure trimethylamine isn’t a minor concern—one whiff leaves a lasting impression, and shipping laws mirror those risks. Hydrochloride forms bring reassurance, letting researchers and technologists focus on developing new materials, medicines, and processes instead of worrying about hazardous leaks.
Improving Practice and Knowledge
Greater transparency, quality control, and education go a long way. Rigorous training helps staff recognize labels and understand what formulas mean for storage and usage. Government agencies and public health organizations often suggest workshops and regular reviews based on real case studies—some inspired by lab mistakes I’ve heard about in safety seminars. Frequent audits, clear communication, and a culture of double-checking formulas protect workers, communities, and projects alike.
Looking Forward
New advances mean more demand for chemicals like trimethylamine hydrochloride. Researchers are already using it in greener synthesis methods and more targeted pharmaceuticals. Just as the basics—knowing the formula, respecting the risks, and ensuring proper training—stay central, the opportunities for innovation keep growing as well. Nobody benefits more than the everyday user when industry-wide standards raise the bar, and it all circles back to a solid grasp of what’s inside the jar.
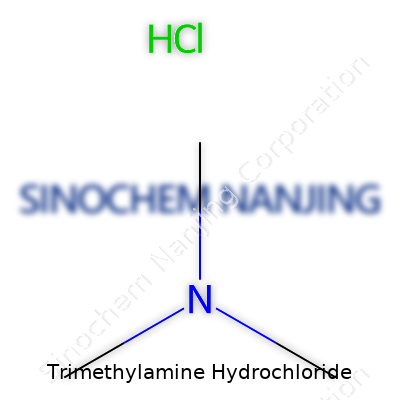
Molecular formula: C3H9N·HCl.
How should Trimethylamine Hydrochloride be disposed of?
Understanding the Problem
Trimethylamine hydrochloride, with its sharp fishy smell, shows up in research labs and some manufacturing sites. Some people even bump into it in niche chemistry projects. For anyone who’s ever dealt with this stuff in person, one thing becomes clear right away—mishandling could mean runaway odors, safety hazards, or even breaking the rules.
Disposing of chemicals isn’t just a checklist item. The choices we make protect others who share the same workspace, the people who drive trash trucks, and anyone living nearby—plus whatever wildlife lives downstream from our sinks and sewers. I remember once having to clean up after a forgotten bottle leaked in an old storeroom. Not only did the work take hours, but the effects stuck around for days. Responsible handling matters, whether you’re in a glass-walled lab or a school science room.
Risks at a Glance
Chemicals like trimethylamine hydrochloride aren’t as simple as tossing table salt. If this compound goes into regular trash or gets poured down a household drain, there’s a real risk to water treatment facilities and anyone exposed to the fumes. This isn’t fear-mongering—municipal wastewater systems across the globe fight against chemical contamination every week. One wrong move in one sink can mean headaches for everyone downstream. Emergency response teams in cities like New York and London log calls from improper chemical disposal every year.
Copyrighted, Regulated Means of Disposal
Federal and local rules, especially under agencies like the Environmental Protection Agency (EPA), address exactly this sort of risk. Labs and factories learn to treat trimethylamine hydrochloride as hazardous. Based on direct experience, the rules aren’t written just to be difficult. They make it clear: never mix this compound with general waste or household drains. In many research settings, every single disposal step gets logged and checked.
Instead, experts recommend segregating trimethylamine hydrochloride into a labeled, sealed container. In every facility where I’ve worked, these containers sit in ventilated holding cabinets until a professional chemical waste contractor picks them up. Trained staff handle the final disposal at licensed facilities. This step is not negotiable—no matter if it’s a full container or a few grams left from an experiment.
Making Things Better
No one wants to feel stuck in a maze of paperwork, especially when cleaning up after a project. But skipping steps creates harm down the line. Setting up clear disposal guidelines keeps everyone on the same page. I’ve seen universities set up annual training sessions, making sure every new person knows the dangers—not just relying on posters or slides. In another lab, QR codes posted by the disposal cabinet sent staff straight to digital manifests and collection forms.
At a broader level, cities could help by hosting regular hazardous waste collection days for individuals, not just companies—especially since home chemistry kits and DIY projects keep popping up in the news. Public outreach goes a long way. Local governments can push out reminders by text or email, so the information reaches everyone using such materials. For those handling larger amounts, consulting with hazardous waste companies before starting a project can save piles of trouble.
Long-Term Outlook
We get one shot at keeping workplaces and communities safe from chemical mishaps. Every step from labeling to pickup day protects real people, not just checkboxes on a form. The more effort people put in before disposal day, the fewer headaches and the greater peace of mind for everyone involved. Knowing where your waste goes is part of taking pride in your work—and respecting the folks next door.
| Names | |
| Preferred IUPAC name | N,N-Dimethylmethanamine hydrochloride |
| Other names |
Trimethylamine hydrochloride Trimethylammonium chloride N,N-Dimethylmethanamine hydrochloride |
| Pronunciation | /traɪˈmɛθɪl.əˌmiːn haɪˌdrɒˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 593-81-7 |
| 3D model (JSmol) | `/lodestone/generic/mol/?id=100-69-6` |
| Beilstein Reference | 1690803 |
| ChEBI | CHEBI:60032 |
| ChEMBL | CHEMBL1232056 |
| ChemSpider | 15116 |
| DrugBank | DB11173 |
| ECHA InfoCard | 100.037.334 |
| EC Number | 214-686-6 |
| Gmelin Reference | 8288 |
| KEGG | C01080 |
| MeSH | D019311 |
| PubChem CID | 11456 |
| RTECS number | XN6476000 |
| UNII | 7U31753GJZ |
| UN number | UN3241 |
| CompTox Dashboard (EPA) | DTXSID9020442 |
| Properties | |
| Chemical formula | (CH₃)₃N·HCl |
| Molar mass | 81.56 g/mol |
| Appearance | White crystalline powder |
| Odor | Fish-like |
| Density | 0.730 g/cm3 |
| Solubility in water | Very soluble |
| log P | -0.296 |
| Acidity (pKa) | 10.05 |
| Basicity (pKb) | 4.19 |
| Magnetic susceptibility (χ) | -49.5e-6 cm³/mol |
| Dipole moment | 1.29 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 117 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −174.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -357.5 kJ/mol |
| Pharmacology | |
| ATC code | A16AA06 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Autoignition temperature | 275 °C |
| Lethal dose or concentration | LD50 oral rat 1000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 415 mg/kg |
| NIOSH | PH7225000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 40 mg/m3 |
| IDLH (Immediate danger) | Not listed. |
| Related compounds | |
| Related compounds |
Methylamine hydrochloride Dimethylamine hydrochloride Tetramethylammonium chloride |

