Triisopropyl Borate: A Closer Look at a Modern Chemical Ingredient
Historical Development
Triisopropyl borate didn’t emerge from the fog of early chemistry but gained real traction in the late twentieth century, as organoboron chemistry zoomed forward. In academic labs experimenting with new ways to build carbon-boron bonds, chemists realized the value of borate esters not just as lab curiosities, but as serious tools for synthesis. Triisopropyl borate, while overshadowed sometimes by its methyl and ethyl cousins, started finding fans because its bulkier isopropyl groups gave it a unique edge in selectivity and reactivity. From the late 1970s onward, this compound showed up in patents, journals, and finally on industrial shopping lists where better ways to introduce boron atoms made a noticeable difference. The story here lies less in brand names and more in relentless tinkering by researchers determined to draw new molecules, faster and cleaner, than what came before.
Product Overview
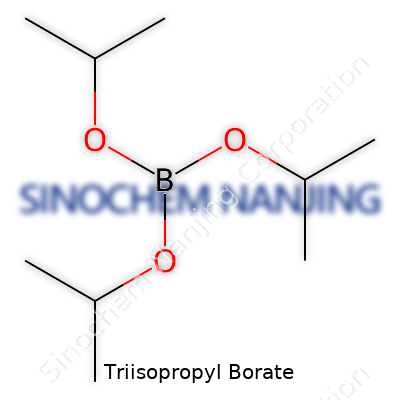
Triisopropyl borate looks the part of an organic liquid, clear and shimmering even in the laboratory glass. Chemists use it mostly as a reagent—its main draw isn’t in how it looks, but in how predictably it delivers boron into other molecules. Its formula, B(OCH(CH₃)₂)₃, tells the tale: a boron atom holding court among three isopropyl arms. Sometimes it’s overshadowed by simpler borates, but the combination of volatility and reactivity sets it apart, especially when fine-tuning reactions in organic synthesis. While it rarely grabs headlines in popular science, many chemists see it as an unsung workhorse—quietly dependable, ready to build new boron-based compounds or serve in technical applications like specialty coatings, electronics, and flame retardants.
Physical & Chemical Properties
This liquid brings a unique blend of volatility and relatively high boiling point into the lab. It boils at around 181°C, a notch above some common esters, and displays a characteristic sweet odor that’s hard to forget if you’ve worked with it. It dissolves nicely in organic solvents like ether or benzene, showing little interest in water thanks to the isopropyl groups. Unlike the simplest borates that sometimes hydrolyze at a single splash, triisopropyl borate’s bulkier arms offer a little extra resistance, which can matter during multi-step syntheses. Lighter than water, it demands careful handling, mostly because it can still break down in the presence of moisture, sending out isopropanol and boric acid.
Technical Specifications & Labeling
If you’ve handled triisopropyl borate in a supply room, you know to expect quality standards set by purity levels, often 98% or higher for research use. Labels should spell out any water content, and the packaging routinely comes sealed under nitrogen or another inert gas to keep atmospheric moisture at bay. The chemical’s physical hazards—mainly flammability and skin irritation—must stay front and center in any documentation, according to regulations that keep labs and industries in check for safe handling. Storage always stays away from oxidizers, acids, or open flames, and container material often shifts to brown glass or steel to resist either chemical attack or light-induced decomposition.
Preparation Method
Triisopropyl borate gives a neat example of classic ester synthesis. Often, producers react boric acid with isopropanol in the presence of an acid catalyst—sometimes sulfuric acid, sometimes p-toluenesulfonic acid. The process drives off water, pushing the equilibrium toward product. Anyone who has performed a Fischer esterification knows the drill: heat, remove the water, distill the product, and purify it further if top-notch quality’s needed. Some commercial operations might shift gears to continuous reactors or tweak reagent ratios, but the heart of the process remains unchanged since those early formative years in the lab.
Chemical Reactions & Modifications
This borate ester finds itself drafted into a number of clever reactions. Synthetic chemists often use it for the formation of boronic acids, handy intermediates for Suzuki-Miyaura cross-couplings, which are mainstays in drug and materials development. In a reaction with Grignard reagents or organolithiums, triisopropyl borate hands off its boron center, then after a little hydrolysis, the boronic acid emerges—an elegant bit of chemistry, precise and reproducible. The compound sometimes recruits modifications through transesterification, giving way to other borate esters, or gets used as a building block for boron-modified polymers. The chemistry world leans on its selective reactivity, making it valuable beyond what a simple structural formula suggests.
Synonyms & Product Names
Names pile up for a compound like this. You might hear it called boric acid, triisopropyl ester, or just triisopropoxyborane in older literature. Some supply catalogs abbreviate it as TPIB or list the CAS number for cross-reference. These varied identities arise from decades of discovery and commercial use, each name reflecting a different slice of research or industrial tradition. For anyone navigating chemical inventories or searching scientific papers, recognizing these synonyms can mean the difference between finding what you need and missing a hidden gem.
Safety & Operational Standards
Nobody who has worked with triisopropyl borate forgets its flammability. It catches fire as easily as many organic solvents, demanding grounded equipment and diligent handling. Splashing it on skin causes irritation, reminding handlers to suit up with gloves, goggles, and solid ventilation. Regulations flowing from agencies worldwide track this compound as a fire risk, assigning hazard symbols and transport restrictions that put safety first. Spills must get handled swiftly using absorbent materials, and storage stays away from open sunlight and incompatible chemicals. Through my years in the lab, treating triisopropyl borate with healthy respect meant fewer accidents and a longer, safer career at the bench.
Application Area
Triisopropyl borate finds itself most at home wherever boron chemistry steps into the spotlight. In pharmaceuticals, it serves as a reagent in the creation of key intermediates, lending a hand to synthetic routes that shape complex organic molecules. Electronics manufacturers use it in thin film deposition, where boron materials are in demand for their electrical properties. Specialty coatings sometimes turn to borate esters for added flame retardancy or chemical resistance. Paints and lubricants sometimes benefit from boron additives, allowing triisopropyl borate to add value where it’s least expected. Looking at industrial landscapes, it’s clear this compound isn’t boxed into one specialty but spreads across several sectors, always lending some of its unique chemical features to the process.
Research & Development
Academic and industrial labs constantly push the boundaries with triisopropyl borate. Research groups run studies to fine-tune new synthetic routes, experiment with alternative catalysts for greener production, or probe the use of borate esters in next-generation polymers and electronic devices. Some projects dive into ligand design for catalysis, where boron-containing moieties shape new organometallic platforms. Others explore sustainable materials, where the control over boron content can help create lightweight, strong composites. My own experience tells me that collaborative effort makes the real difference—chemists sharing techniques and findings to raise the collective knowledge, rather than protecting trade secrets behind closed doors. Conferences and academic papers offer regular reminders that the story of this borate is still being written.
Toxicity Research
Toxicologists have spent years tracking the behavior of borate esters like this one. Though triisopropyl borate doesn’t carry the acute toxicity that grabs headlines, chronic exposure remains a real concern. Boron compounds can irritate eyes and skin, and inhalation of vapors sometimes leads to headaches or nausea. Animal studies suggest that, at high enough doses, effects on reproductive health could emerge, sending a trial balloon to safety agencies to keep exposure limits tight. Industry recommendations call for monitoring air levels in workplaces and maintaining strict engineering controls to keep risks managed. Having spent some late nights reading safety data sheets and wrangling with safety protocols, I’ve learned that respect for chemical hazards starts with good information and clear-headed risk management.
Future Prospects
The future of triisopropyl borate rides closely with new needs in materials science, green chemistry, and electronics. As researchers hunt for better syntheses that avoid heavy metals and minimize waste, boron-based compounds draw more attention—clean, versatile, and effective under mild conditions. In semiconductor fabrication, with every new generation of chips hungry for more precise dopants and coatings, borate esters line up as reliable candidates. Flame-retardant chemistry also has room for smarter, less toxic additives; here, triisopropyl borate’s chemical quirks offer further promise. Watching grant proposals and industrial investments flow, I see a compound ready for reinvention—not just as another reagent, but as a platform for sustainable, advanced technologies. Whether the community finds new applications or pushes for greener methods, triisopropyl borate stays relevant in a world always chasing improvements.
What is Triisopropyl Borate used for?
Why Chemists Reach for Triisopropyl Borate
Stepping into a modern chemistry lab, glass bottles packed with all sorts of compounds line the shelves, and somewhere among them is triisopropyl borate. This colorless liquid might not win any “most famous” awards, but for those doing real synthetic work, it’s something you pick up more than once. I remember working through a tough organic synthesis—there’s almost always a point where introducing a boron atom leads to a world of easier transformations. That’s where this stuff comes into play.
Triisopropyl borate steps up as a reagent when boronic esters or acids become necessary for Suzuki coupling reactions—a staple for making carbon-carbon bonds. Ask any chemist putting together pharmaceuticals or advanced materials, and you’ll hear the same: forming reliable bonds with biaryl structures or fine-tuning electronic properties often circles back to boron chemistry. This liquid helps you get there without a lot of fuss.
How Industry Puts It to Work
Outside the bench, the uses for triisopropyl borate show up in silicone processing and glass manufacturing. I saw this on a site visit to a specialty glassmaker in Germany: borates, including this one, help boost the thermal stability or adjust the refractive index for fiber optics. This isn’t a product that screams from the packaging, but it quietly improves performance everywhere from your phone’s screen to the cabling under city streets.
There’s also a need for it in making flame retardants and specialty paints. Anyone working in coatings technology recognizes how small changes in molecular structure can control how a coating performs under heat. Additives made from borates reduce flammability for paints used in aircraft and public spaces. It isn’t a glamorous story, but keeping people safe in emergencies matters more than the chemicals used to do it.
Sourcing, Handling, and Environmental Responsibility
More facilities rely on triisopropyl borate, so questions about sourcing and safety don’t go ignored. Proper handling comes down to the usual lab best practices: goggles, gloves, and keeping it sealed until use. If you’ve ever spilled even a tiny drop, you learn quickly about its volatility—nobody wants to breathe in the vapors. Most suppliers stick to international guidelines for labeling and transport, which protects not just the buyer, but anyone involved as it moves from plant to lab.
Environmental considerations mean looking past just the immediate hazard. I’ve seen plants develop take-back programs for unused chemicals, and some switch to greener alternatives in lower-stakes applications. Regulators push for reduced emissions and tightly managed waste streams, so disposal never happens without a plan. Workshops on chemical stewardship hammer home how small changes in workflow keep pollutants out of the water table.
Where Challenges Crop Up
The biggest headaches come not in use, but in scaling up or down production. Supply chain disruptions make it hard to guarantee quality or keep costs manageable for specialty borates. COVID-19 made this even more obvious—one missed shipment causes weeks of delay. Companies are starting to diversify sourcing and keep larger stockpiles on hand. Collaboration with academic labs sometimes leads to finding substitutes, but so far, triisopropyl borate keeps its seat at the table for tricky syntheses.
Everyone I’ve met in the industry shares the same view: it’s not about ditching chemicals altogether, but about smarter decisions. Leaning on experience, following safety protocols, and seeking cleaner waste management lighten the environmental load. Daily decisions about these substances, even less celebrated ones like triisopropyl borate, stack up to change the way whole industries work.
Is Triisopropyl Borate hazardous or toxic?
Understanding the Chemical Background
Triisopropyl borate pops up in chemical catalogs and research settings more often than in your average household. Sometimes I see the question floating around: just how risky is this stuff? Since most of us don’t run a small chemistry lab in our basements, it’s fair to wonder if it poses any significant hazard. Personally, the last time I worked with an unfamiliar solvent, I double-checked the label, scanned the safety sheet, and hesitated before cracking the cap. Habit, maybe, but for good reason.
Direct Hazards: What the Safety Data Says
No need to squint at old textbooks—reliable sources like industrial safety databases and the European Chemicals Agency (ECHA) keep the details current. Triisopropyl borate hits a few standard red flags. Get it on your skin, and it may cause irritation. Splash some in your eyes, expect discomfort. Breathing in high concentrations can irritate your airways. Spill enough of it and don’t open a window, and you’ll soon wish you had. This chemical carries a “flammable” reputation, so storing next to the stove runs against common sense.
What about actual toxicity? Animal studies reveal that large exposures can affect major organs, with metabolic byproducts like isopropanol factoring into toxicity. While the data sets aren’t vast, borate compounds overall have drawn concern for potential reproductive effects in high exposures over long periods. The everyday user, rarely in contact with neat triisopropyl borate, usually sidesteps this particular risk, but industry workers shouldn't ignore it.
Real-World Use and Risk Management
Chemical handling involves more than just tossing on a lab coat. Anyone who’s ever poured a solvent without gloves recalls the sting or lingering smell. The right protective gear—nitrile gloves, splash goggles, and a working fume hood—offers reassurance. Factories and schools that keep triisopropyl borate on hand rely on such measures. I’ve learned through experience, in both school labs and commercial settings, that regular reminders about protocols beat fancy posters every day.
Spills happen. Labs prepare absorbent materials and spill kits for moments like these. Quick action—not waiting around or looking for help—matters most. With flammable solvents, grounding equipment and controlling ignition sources becomes part of the daily checklist.
Environmental and Long-Term Concerns
Borates stir debates around ecological safety. High concentrations, if dumped carelessly, risk harming aquatic life. Wastewater treatment can help, but responsible labs avoid pouring leftovers down the sink. Instead, they collect and dispose via licensed professionals. I’ve seen institutions face fines and warnings for missteps; local regulators visit more often than people think.
Inside the body, studies don’t suggest acute danger in typical low-level exposure. Chronic health effects present a wider set of questions. California has considered some borate compounds for the state’s Prop 65 list, driven by animal evidence. Most job sites act with caution, preferring to limit air concentrations and keep skin contact brief.
Clear Steps Forward
Tackling chemical hazards means training, regular audits, and easy access to safety info. Digital MSDS sheets make it easier for workers to check safe handling guidelines on the fly. Regular review of procedures—don’t just leave the binder on a dusty shelf—helps avoid problems before they start. If more folks brought up safety during hiring, orientation, and regular meetings, chemical incidents wouldn’t show up on the news as often.
So, triisopropyl borate deserves respect, not panic. Treat it with the care you’d give any flammable, slightly toxic compound. That usually means staying alert, keeping up with best practices, and relying on real experience—not just reading the warning label and moving on.
What is the chemical formula of Triisopropyl Borate?
Diving Into Triisopropyl Borate
Triisopropyl borate doesn’t often make the headlines, but its chemical formula—B(OCH(CH₃)₂)₃—ends up mattering in quite a few practical ways. To many, that formula looks like a scramble of letters, numbers, and parentheses, but for folks in chemistry, those symbols spell out real-world value. Triisopropyl borate consists of a boron atom bonded to three isopropoxy groups. In simpler terms: one boron, three oxygen atoms, and nine carbon atoms, all backed with twenty-one hydrogens. Putting it together, the molecular formula reads C₉H₂₁BO₃.
Why Accuracy In Chemical Structure Needs Attention
Working in a laboratory setting, I’ve seen firsthand how a single letter or number missing from a chemical formula derails an entire synthesis. This is especially true with borates, since swapping an alkoxy group or confusing it for another ester shifts the reactivity dramatically. Triisopropyl borate reacts differently in organic transformations compared to its cousin, triethyl borate, all due to the structure you spot in the formula.
The right formula saves time and cuts down on dangerous mistakes. If someone grabs the wrong compound for a Suzuki coupling reaction, projects stall, budgets swell, and risks surface. The structure of triisopropyl borate gives it just enough steric bulk to moderate its reactivity, making it a preferred choice in certain syntheses where milder boron reagents end up as safer bets. So every character in C₉H₂₁BO₃ counts.
Beyond The Lab Bench
Triisopropyl borate crops up often in research, industrial organic syntheses, and flame retardant production. Its ability to hand over boron atoms with reasonable ease suits it to cross-coupling chemistry—a backbone of pharmaceutical and materials manufacturing. This kind of cross-coupling turns complex molecules from idea into a physical product, from medicines to smartphone screens.
Getting the structure right helps protect workers and consumers. Accurate formulas make sure environmental health scientists know what they're registering, and hazards get mitigated before anyone's exposed. Triisopropyl borate, because of its particular configuration, can hydrolyze to generate isopropanol and boric acid. Both have their own handling instructions, so clear labeling with the correct formula keeps safety data sheets meaningful.
Supporting Reliability in Science and Industry
Publishing precise chemical formulas means the science remains reproducible. In my early research days, reading a published paper with a wrong borate formula chewed up time and resources. By sharing correct, clear formulae—like C₉H₂₁BO₃ for triisopropyl borate—the community shares not just information, but trust. Students, researchers, and engineers avoid confusion. Industries avoid production loss and liability. It’s not only about what sits in a bottle; it’s about consistency, communication, and safety.
Steps Toward Safer And Smarter Handling
Clear documentation stands as the first line of defense against accidents. Scientists and companies can go further by reinforcing chemical safety training tied to understanding formulas and structures. Digital inventory systems flag unusual orders or dangerous substitutions. Regulatory bodies and peer reviewers keeping an eye out for accurate formulas deepen the reliability of published work. Sharing best practices across institutions makes sure that no one finds themselves guessing at the meaning behind a formula again.
How should Triisopropyl Borate be stored?
Why Storage Matters More Than Folks Think
Ask anyone who’s ever set foot in a chemical stockroom: some bottles demand more respect than others. Triisopropyl borate definitely fits in that category. This colorless liquid finds homes in labs, electronics manufacturing, and organic synthesis. Its job might sound technical, but the stakes get very real if nobody pays attention to how it’s stored. One careless shelf pick or leaky cap can trigger an uncomfortable chain of events, from ruined raw materials all the way to safety nightmares.
Humidity: Hidden Enemy Lurking on Every Shelf
I’ve watched more than one researcher race out of a storeroom cursing the day humidity seeped into their precious chemical. Triisopropyl borate picks up water from the air fast, breaking down into boric acid and isopropanol. Let a container sit open for just a few minutes, and you’re not just losing the chemical—you’re setting up useless, even hazardous byproducts. The smartest move? Air-tight bottles with strong seals. Keep the bottle closed every single time, and save the chemistry for the experiment, not the shelf.
Temperature Swings Cause Trouble
Ignore that “Store in a cool, dry place” advice and troubles pile up quickly. Extra heat makes triisopropyl borate more volatile and bumps up the risk of container pressure build-up. Left in a hot supply room or near sunlight, evaporation speeds up, and even trace leaks can fill the air with flammable vapors. In my own experience, the best practice means keeping bottles in locked flammable liquid cabinets cooled by good ventilation—not above head height, not beside radiators, not under direct lights. Night janitors and weekend shifts rarely check for leaks, but fumes can build up long after everyone heads home.
Keep Away from Accident-Prone Products
Incompatibility isn’t a fancy lab word, it’s real-life messes. Triisopropyl borate reacts with strong oxidizers. Spill bleach or nitric acid nearby, and you’re inviting an unpredictable, uncontrolled reaction. So on stockroom rounds, I always keep these further apart than the minimum guidelines suggest—nobody wants an accidental mix-up during a busy delivery day. Double-check secondary containment trays, just in case of a cracked bottle or loose cap that could let chemicals run wild.
Labels Matter: Stay Ahead of Hazards
Forget placeholder labels or faded marker scribbles. Every chemical bottle deserves a clear, legible label with storage instructions and hazard pictograms. In an emergency or high-stress situation, those few seconds spent reading can make all the difference. I’ve lost count of times I caught a mix-up because a label stood out, preventing a pile of wasted work or an accident nobody wanted.
Compliance Keeps People Safe
OSHA and local safety codes don’t just add paperwork—they make sure nobody cuts corners. Regular safety training reminds workers of risks, and documented inspections mean problems get spotted before turning into news stories. Storing Triisopropyl borate the right way builds trust, from the lab bench up to the loading dock. Skipping steps or getting lazy may save a minute, but it costs much more if something goes wrong.
What are the safety precautions when handling Triisopropyl Borate?
Understanding the Risks
Triisopropyl borate, used in labs and some industrial work, looks unassuming: clear, often sold in bottles that don’t grab much attention. Still, it carries a punch if handled with carelessness. This isn’t the sort of chemical that sits safely on a shelf in your garage. Anyone dealing with it faces risks of breathing trouble, skin and eye irritation, and fire hazards. Too many incidents start from folks brushing off these dangers. It’s easy to cut corners, especially when the compound seems pretty tame compared to the harsher acids or bases nearby, but the consequences hit just as hard.
Personal Experience in the Lab
Early in my career, I watched a colleague pour triisopropyl borate into a flask without gloves. He believed the stuff would wash off and shrugged off the warning. By the end of the day, he had red streaks trailing up his arms. Maybe it didn’t blister, but he scratched for a week and blamed “sensitive skin.” The real culprit? Overconfidence.
Worse, a minor spill on a hotplate once sent up sharp fumes. No one in the room forgot that lesson. Most of us learned to show respect for chemicals that don’t even advertise their danger with bright colors or strong smells.
Everyday Steps Make the Difference
Avoiding harm starts before even uncapping the container. Many forget to check for fresh gloves, goggles, or splash-resistant lab coats. Bypassing these tools adds real risk. Never trust skin exposure just because past experience seemed harmless. Nitrile gloves hold up better than latex. Eye protection beats quick rinses at the sink once vapors start burning. Ensuring good ventilation matters—working under a chemical fume hood keeps the lungs safe.
It doesn’t stop with what lands on your skin. Triisopropyl borate lights up fast if it meets an ignition source. Open flames, hot surfaces, and even static could turn a routine experiment into a fire drill. Fire extinguishers must sit close. Simple things—fixing a loose jacket cuff, putting away spark sources—save time and keep you out of harm’s way.
Storing and Cleaning Up
People sometimes shove bottles just anywhere afterward, hoping the chemical never leaks or reacts with something else. That gamble isn’t worth it. The right answer remains simple: keep containers tightly closed, labeled in plain language, and away from heat or sunlight. Flammable storage cabinets cost money, but so do emergency calls and cleanup crews.
Once finished with the compound, dispose of any waste at collection points designed for hazardous chemicals. Pouring leftovers down the drain sounds easy, but it pollutes the environment and creates lingering risks for others down the line. A single step to separate spill kits for borates ends up saving a lab—and the people in it—a world of regret.
Relying on Facts Over Luck
Triisopropyl borate doesn’t care if its dangers seem less dramatic compared to other chemicals. It reacts to carelessness with the same speed and certainty as its more notorious cousins. Real safety springs from respect: staying informed, gearing up, and staying ready for the unexpected.
Many accidents fade from memory, but routines built around preparation last. Whether you’re a student, researcher, or technician, recognize the hazards each time you reach for that bottle. Health and safety don’t hinge on luck; they grow from good habits and solid information—something every lab and workplace can put into practice.
| Names | |
| Preferred IUPAC name | Tri(propan-2-yl) borate |
| Other names |
Boronic acid, triisopropyl ester Triisopropyl borate Borate(3-), triisopropyl- Isopropyl borate |
| Pronunciation | /traɪˌaɪsəˈprəʊpɪl ˈbɔːreɪt/ |
| Identifiers | |
| CAS Number | 5419-55-6 |
| Beilstein Reference | 822690 |
| ChEBI | CHEBI:52086 |
| ChEMBL | CHEMBL185111 |
| ChemSpider | 71148 |
| DrugBank | DB11154 |
| ECHA InfoCard | ECHA InfoCard: 100.011.739 |
| EC Number | 212-216-7 |
| Gmelin Reference | 7872 |
| KEGG | C18912 |
| MeSH | D017921 |
| PubChem CID | 12265 |
| RTECS number | EY2975000 |
| UNII | 4J63R68EYC |
| UN number | UN1993 |
| Properties | |
| Chemical formula | B(OiPr)3 |
| Molar mass | 190.07 g/mol |
| Appearance | Colorless liquid |
| Odor | Faint alcohol-like odor |
| Density | 0.885 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | 0.93 |
| Vapor pressure | 0.3 mmHg (20 °C) |
| Acidity (pKa) | 20.0 |
| Basicity (pKb) | 2.90 |
| Magnetic susceptibility (χ) | -92.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.369 |
| Viscosity | 2.7 mPa·s (20 °C) |
| Dipole moment | 2.16 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 445.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -479.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2116 kJ/mol |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | Precautionary statements: "P210, P240, P241, P280, P303+P361+P353, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-1-2-W |
| Flash point | 80 °C |
| Autoignition temperature | 410 °C |
| Explosive limits | Explosive limits: 0.9–8.5% (in air) |
| Lethal dose or concentration | LD50 (oral, rat): 10000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 10,000 mg/kg |
| NIOSH | BZ1040000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | Fume Hood |
| Related compounds | |
| Related compounds |
Trimethyl borate Triethyl borate Tripropyl borate |

