Trifluoromethanesulfonic Acid: Unpacking the Power, Promise, and Pitfalls
Historical Development
Walking through the history of chemistry, every once in a while, a compound comes along that transforms the way we think about making molecules. Trifluoromethanesulfonic acid, CF3SO3H, always grabs my attention for that reason. Discovered as part of the postwar boom in organofluorine chemistry, it showed up just as laboratories grew braver about working with aggressive chemicals. Its story runs parallel to superacids like fluorosulfuric acid, but triflic acid’s appearance in the late 1950s wasn’t just a matter of stronger acid strength – it changed how organic chemists tackled some of their toughest transformations. Research labs and chemical manufacturers recognized the power in that – an acid even stronger than sulfuric acid, yet surprisingly stable and less oxidizing. Not every chemical gets to say it helped write chapters in textbooks on acidity, catalysis, and functional group activation.
Product Overview
Calling triflic acid a “superacid” may undersell its real-world bite. Chemically, it’s a bronzed liquid with a distinctly sharp, biting smell. Early researchers described its clarity and its destructive power on human tissue in breathless terms. Unlike some hazardous acids that fume up aggressively at room temperature, triflic acid holds together, glass-clear, and straightforward to measure by experienced hands. Chemists who need a strong acid that stays unreactive toward many organic substrates return to it with a kind of professional respect. They know its power doesn’t stop at its ability to protonate alcohols and hydrocarbons – it stretches into tough territory, like activating arenes and stabilizing cations no other acid would touch.
Physical & Chemical Properties
Looking at the molecule itself, CF3SO3H stacks up as a compact, highly polar acid with thermal stability that sets it apart from more volatile mineral acids. It boils over 160°C and holds up in air as long as water and bases stay far away. It outmatches hydrochloric acid and even sulfuric acid on the Hammett acidity scale. Its solution in water releases plenty of heat, making lab work risky for the unprepared. Its dielectric constant allows it to slip through polar media and interact directly with substrates that traditional acids barely touch. That’s why chemists turn to it for tasks that other acids can’t do cleanly or quickly, like cleaving stubborn t-butyl protecting groups or directing Friedel–Crafts alkylations with better selectivity.
Technical Specifications & Labeling
In my experience, working with triflic acid demands more vigilance than many other strong acids. Bottles arrive, labeled with bold hazard warnings and corrosion symbols, in shatterproof containers inside secure shipping boxes. Typical technical-grade triflic acid comes around 99% pure, which sounds high, but even trace amounts of dissolved water or organic impurities can set off unwanted reactivity. Major suppliers specify the minimum hydrogen ion content and screen for iron, chloride, and sulfate contamination. For all the lab’s talk about purity, in practice, storage and handling may introduce problems if bottles grow crusty, turn yellow, or emit fumes. Veterans in the field learn fast to avoid glassware with ground-glass joints – triflic acid welds these shut and can ruin precious glass with pitted, etched surfaces.
Preparation Method
Making triflic acid has always struck me as a recipe that calls for nerve and excellent technique. The earliest commercial runs built triflic acid through direct fluorination of methanesulfonic acid using sulfur tetrafluoride. Modern processes usually start from trifluoromethanesulfonyl chloride, a versatile building block, which can be hydrolyzed safely and efficiently to the acid. This shift from brutal fluorination to more manageable chlorosulfonation techniques made the acid more accessible, safer, and cheaper. Industrial reactors run the conversion under strict temperature and isolation controls, not least because uncontrolled hydrolysis kicks off clouds of corrosive fumes. Large-scale operators take environmental containment seriously — a spill or accidental release causes headaches that linger for months.
Chemical Reactions & Modifications
The reason so many chemists keep triflic acid on the shelf isn’t only its raw acidity. Its chemistry opens channels modern catalysis depends on. It can generate triflate esters by reaction with alcohols, a backbone of modern organic synthesis. These triflates, in turn, flip basic carbon compounds into reactive partners for cross-coupling and other C–C bond-forming reactions. On its own, CF3SO3H activates hydrocarbon skeletons, flips double bonds into carbocations, and pushes electrophilic aromatic substitution past tricky obstacles. The clean, non-nucleophilic nature of the triflate anion means it gets out of the way after doing its catalytic job. For me, that non-interfering aspect sets it apart. Other acids jam up reaction mechanisms with side reactions; triflic acid runs its course and then disappears with a rinse of water, washing away as its salt.
Synonyms & Product Names
Every long-term chemist picks up the jargon. In journals and catalogs, triflic acid often appears as “trifluoromethanesulfonic acid,” “CF3SO3H,” or simply “TfOH.” Some circles call it “triflic acid,” an affectionate shorthand. In the world of pharmaceuticals, it sometimes turns up on labeling as “methanesulfonic acid, trifluoro-” or “trifluoromethylsulfonic acid.” For all these names, none truly convey the careful approach and caution that the bottle deserves.
Safety & Operational Standards
Safety in the lab turns quickly to ritual with this acid. I remember my first time moving a bottle – heavy gloves, full-face shield, and chemical apron, even in a fume hood. Skin contact means instant burn, pain, and risk of deep tissue injury. Splashes on metal bench surfaces leave blackened, pitted scars. Handling guidelines stress slow addition, careful chilling, and never mixing with bases or organics except under controlled conditions. Acid-resistant plastics stand in for glass wherever possible. Waste neutralization, even in small amounts, runs under dilute conditions, since the exotherm with water—think sizzle and popping steam—can surprise newcomers. Standards back up these habits with regular review and mandatory emergency protocols for accidental exposure or spills. Over time, I’ve come to see these procedures less as burdens, more as the mark of respect the acid demands.
Application Area
Triflic acid works behind the scenes in synthetic chemistry, pharmaceuticals, and the world of advanced materials. It gives a crucial push in creating triflate esters, which have become bread and butter in cross-coupling chemistry. Medicinal chemists rely on it to build complex drug scaffolds that would fall apart or sulfonate if treated with other acids. Petrochemical researchers dig into its talent for hydrocarbon activation, tweaking reaction selectivity and yield. In electrochemistry and energy storage, triflic acid and its salts turn up as stable charge carriers and electrolytes, pushing batteries and supercapacitors forward. The electronics industry, always chasing speed, leans on it for special etching, doping, or surface treatments, especially where a clean, aggressive acid with controlled oxidation is necessary. Each of these applications leverages that combination of strength and selectivity, a combination that makes it irreplaceable in some workflows.
Research & Development
Research on triflic acid tracks the shifting needs of the chemical industry. Labs keep finding ways to push its role in green chemistry, looking for safer, less wasteful reaction regimes. For example, clever teams experiment with catalytic cycles where triflic acid can turn over multiple times, reducing waste acid disposal and cost. Polymer chemists chase high-performance plastics and coatings by harnessing its superacidity without needing metal catalysts, sidestepping issues like metal leaching in sensitive products. Academic groups continue diving into structure-activity relationships, figuring out how tiny tweaks in substrates or solvents unlock new reactivity. I’ve seen young chemists fixate on it for late-stage functionalization, where it does what milder acids cannot, letting them tack new groups onto otherwise finished pharmaceuticals. Demand for smarter, safer, and more recyclable uses shows no signs of slowing down, especially as manufacturing keeps tightening regulations on acid waste and emissions.
Toxicity Research
You do not need to be careless with triflic acid to learn about its risks – the data speaks for itself. Exposure means real danger. Inhalation of vapors, even at low levels, causes immediate nose, throat, and lung irritation. Small droplets on skin demand immediate washing to prevent deep, lingering burns. Animal studies reveal severe acute toxicity, though the acid doesn’t seem to act as a classic systemic poison. What stands out is the local destruction: massive protein denaturation, saponification of fats, and cell lysis, features it shares with other strong mineral acids. Standard operating procedures grew up around these facts. Research on chronic exposure stays limited, though environmental consultants note that any triflate run-off acidifies water rapidly and must be kept from drains. Waste managers and safety officers dig into every step of triflic acid disposal, sometimes treating small spills with cold sodium bicarbonate or lime under strict observation, knowing the risk of violent exotherms.
Future Prospects
Looking down the road, the chemistry community has big questions about the future of triflic acid. Its track record in catalysis and activation keeps it relevant as industries chase cleaner, faster, and more selective synthetic routes. Pushes to reduce environmental burden call for microreactor technology or closed-loop systems where acids like these cycle back to the start, slashing costs and hazards. High-value pharmaceuticals and advanced polymers still lean on its power, so demand isn’t dropping, but the focus keeps shifting toward sustainable use, recycling, and waste minimization. As battery and electronics research barrels along, expectations ride high for new applications or safer, functionally similar acids. From where I stand, the legacy of triflic acid comes from that rare blend of explosive strength and precise control – qualities that promise both risk and reward for chemists willing to respect its bite, and who won’t stop looking for the next breakthrough in safe, sustainable synthesis.
What are the main uses of Trifluoromethanesulfonic Acid?
Unpacking Trifluoromethanesulfonic Acid
Trifluoromethanesulfonic acid, often called triflic acid in labs, doesn’t get headlines like sulfuric or hydrochloric acid. Still, chemists pay it a lot of respect. Just spending time in a university chemistry department or hearing stories from process chemists makes it clear: this acid pulls serious weight. It’s tough, direct, and gets reactions done where others might stall out.
Catalysis: Bringing Molecules Together
Organic chemists like to talk about “superacids.” Triflic acid has that badge for a reason. Its main job is kickstarting reactions that build and break bonds in complicated molecules—think pharmaceuticals or fancy plastics. If I’m working on a tricky reaction that refuses to go the final mile, triflic acid often acts as that stubborn, no-nonsense coach pushing things over the finish line. It can take a stalled alkylation or acylation and get it moving without leaving unwanted leftovers in the mix. That can mean higher yields, cleaner products, and less time spent cleaning up the mess afterwards.
Boosting Silicon Chemistry
Modern electronics wouldn’t look the way they do without good silicon chemistry. Here, triflic acid speeds up the creation of organosilicon compounds. These materials show up everywhere from computer chips to waterproof coatings. Triflic acid helps by making the process more efficient compared to traditional mineral acids. I remember seeing how a small tweak—swapping sulfuric acid for triflic acid in the lab—cut the reaction time in half and led to less waste. That kind of improvement matters for both the environment and the budget.
Cleaning Up the Mess: Deprotection
In organic synthesis, chemists often “protect” parts of a molecule to keep them stable through harsh steps, then “deprotect” them at the end. Many protecting groups won’t budge unless you bring in something strong—triflic acid does the job quickly, with less drama. Drug companies in particular value this, since getting rid of protecting groups cleanly can mean the difference between a batch passing or failing quality checks. With tighter regulations and high material costs, that efficiency stands out.
Generating Useful Reagents
Labs often use triflic acid to make special salts or intermediates that don’t come easy with weaker acids. For example, it can create triflate salts, which act as good leaving groups in chemical reactions. This tool gives chemists more options for designing routes to new compounds. Sometimes there’s no replacement for a reagent like this—without it, certain drugs or specialty chemicals would just cost a lot more or take much longer to produce.
Handling the Hazards
Triflic acid works fast, but it doesn’t forgive mistakes. Early in my time at the bench, I saw a cracked flask from careless handling—luckily, the instructor stepped in right away. Direct contact can cause more damage than even sulfuric acid, so protective gear and good ventilation are non-negotiable. Only trained staff should handle it, and companies need strict protocols for storage and disposal. The benefits never justify cutting corners on safety.
Looking Ahead
While some labs push for greener alternatives, triflic acid’s effectiveness sets a high bar. There’s ongoing research into recycling the acid, and companies now supply it in forms that reduce handling risks. Synthetic chemists keep a close eye on waste management and push for better containment, recognizing both the potential and the dangers of this powerful acid.
What safety precautions should be taken when handling Trifluoromethanesulfonic Acid?
The Risks Behind the Name
Trifluoromethanesulfonic acid might not be as famous as sulfuric acid, but it packs a punch for anyone working with it in the lab. Its powerful acidity draws attention, especially when you realize a single drop on the skin starts burning tissue almost right away. I once watched a bottle slip from gloved hands; just the splatter on the countertop was enough to etch marks and force the lab team to clear out for ventilation. The lessons stuck with me since then, because triflic acid means business every time someone opens the bottle.
Personal Protective Equipment: No Corners to Cut
Before even thinking about opening a triflic acid container, a full set of protective gear belongs on your body. Chemical splash goggles don’t offer enough — an actual face shield sits between you and the acid’s vapors or splashes. Nitrile gloves rated for acids give a better grip and add a layer of confidence. A heavy-duty acid-resistant lab coat wraps up arms and torso. Bare forearms or single-layer cotton guarantee burns, so double-check the buttons and cuffs. Thick rubberized aprons make sense for larger operations, especially when pouring or transferring. All skin stays covered.
Ventilation and Safe Handling
This acid fumes up fast, creating an environment loaded with choking odors. Fume hoods can’t wait — always work inside them. Open air benches leave no margin for error. Lab air filters might struggle to clear the mist, so use secondary ventilation when transferring anything over a small flask. Colleagues in the area deserve a heads-up, especially if airflow or extraction runs low. I’ve seen coworkers tempted to keep the fume sash open for easier movement, but reducing the opening slows down exposure risk dramatically.
Storage Practices Matter
Some acids stay stable for years on a shelf. Triflic acid can corrode glass stoppers, metal, and weak plastics. High-density polyethylene and Teflon containers hold up best in my experience. Always seal tightly, but release pressure slow — triflic acid builds up invisible fumes. Store it away from bases, water, and any strong oxidizers. Any water contact throws off instant heat and spattering. Stash only on bottom shelves, inside acid-resistant trays, so leaks run nowhere. Clean up any small spills with copious sodium bicarbonate, but never use water to neutralize — direct water risks splattering.
Emergency Plans Can’t Wait Until Later
I always walk through the lab and check that emergency showers and eyewash stations work, since seconds matter after an accident. Colleagues drill for splashes onto skin or eyes: rinse for at least 15 minutes and call medical services right after. Keep spill kits right in the work space — with proper neutralizers, PPE, and instructions. Find out if your safety data sheet stays up to date and visible. Triflic acid vapors aggravate the lungs, so if someone breathes them in, move them to clean air and seek medical help right away.
Training and Teamwork Build Real Safety Culture
Working solo multiplies the risk. Walk through protocols with teammates before starting. Many labs document procedures and lock up dangerous acids to stop unauthorized access. Respect for the material runs deeper than any warning label. If someone feels uneasy, listen; small hesitations can prevent major injuries. Regular training refreshers keep skills sharp and people confident. Nobody forgets the sting — or the stories — from handling this acid wrong. Learn those lessons before you start, and pass them along.
What is the shelf life and proper storage conditions for Trifluoromethanesulfonic Acid?
Why Shelf Life Matters in the Lab
Trifluoromethanesulfonic acid, often called triflic acid, has a reputation for being both powerful and persistent. Its strong acidity means chemists respect it—one drop too many can wreck a reaction. When I worked in an industrial setting, I learned firsthand how a poorly stored bottle could cause problems, from ruined experiments to safety hazards. Knowing how long you can store this acid without worry, and how to do it right, prevents bigger headaches down the road.
Factors Affecting Shelf Life
The actual shelf life for triflic acid depends on how it’s kept. If stored with care, its chemical structure stays steady for years. According to Merck and Sigma-Aldrich, sealed containers that keep out moisture help triflic acid last at least two to three years, sometimes even longer if humidity and heat stay low. Unopened bottles from reliable suppliers usually offer the freshest stock, but once you crack the seal, the clock starts ticking faster.
Risks of Bad Storage
I remember a time a colleague left a triflic acid bottle uncapped on the bench. Fumes formed, condensation collected, and next week that acid looked more like soup than clean chemical. Contaminated or degraded acid can create unexpected byproducts in organic synthesis, putting months of research at risk. Improper storage can also lead to leaks, pressurization, and dangerous reactions with air or water vapor.
Best Practices for Storage
Treating triflic acid right starts with the container. Always use original packaging—glass with Teflon-lined caps often works best. Keeping it tightly sealed keeps out water vapor, which this acid loves to attack. Store the bottle in a dry, cool spot, away from direct sunlight or heat sources. A temperature range from 15°C to 25°C (59°F to 77°F) usually keeps things safe and stable. Never store with bases, amines, or materials that could react violently with acidity. Even today, I label acids like triflic acid not just with names and dates, but also warning stickers so there’s never any guesswork for others using the fridge or cabinet.
Handling and Safety Matters
Each time you open the bottle, work quickly and in a fume hood. Wear gloves and goggles rated for corrosive substances. Have spill clean-up materials ready before you even pick up the bottle. If you see any sign of fog or droplets collecting on the cap, get rid of that bottle safely, don’t try to salvage it. Trusting my gut and checking for subtle changes in color or viscosity has kept me out of trouble more than once.
How to Make Stock Last Longer
To stretch the shelf life, limit how often bottles get opened. Moving triflic acid into smaller vials reduces air and moisture getting inside. Buying only what the lab expects to use in a year or less often beats the risk of spoiled stock. Digital inventory logs help spot old bottles before they become hazards. Training every new coworker on these steps, not just posting a sign, makes all the difference.
Final Thoughts
With proper storage, careful handling, and a solid respect for its power, trifluoromethanesulfonic acid keeps its punch and doesn’t turn dangerous. Chemistry always rewards those who prep for the long haul, and treating strong acids with a little extra care pays off every time. Learning these routines from seasoned chemists taught me more than any textbook ever could.
Is Trifluoromethanesulfonic Acid compatible with common laboratory materials?
What Triflic Acid Brings to the Table
Trifluoromethanesulfonic acid, or “triflic acid,” claims a spot among the world’s strongest acids. Chemists count on it to do the things sulfuric acid can’t. Its super-strength means it breaks down molecules too stubborn for most other acids. But that strength brings a hidden price: even in small spills, triflic acid eats through lab materials people tend to trust. If someone expects it to play nice with their plastic gear or glassware, they run into trouble.
Battles With Plastics and Metals
I’ve seen more than a few students melt their pipette tips or corrode their benchtop gear just by letting triflic acid sit in the wrong flask for a few minutes. Ordinary plastics like polyethylene, polypropylene, and even familiar Teflon (PTFE) don’t always stand up for long. Teflon tubing offers a bit more protection than most polymers, but leaks still crop up after repeated use. PVC, another lab workhorse, curls up almost instantly.
Metal offers no easy answer either. Stainless steel, aluminum, copper—triflic acid cuts through them. Labs that stick to metal-based stoppers or containers pay for it quickly. Each time a researcher pours triflic acid into an aluminum tray, they watch the fizzing and might even catch a whiff of sour gas. That’s the sound and smell of money burning away within seconds.
The Staying Power of Borosilicate Glass
Most glassware in labs comes in borosilicate, known for its toughness against regular acids. But triflic acid doesn’t care much about tradition. With concentrated triflic acid, etching starts quickly, and long-term use produces surface fogging. Whatever goes in comes back with particles that can ruin sensitive experiments. I keep a mental list of the beakers lost along the way—the lesson sticks.
What Actually Works?
Some lucky labs have access to high-purity quartz glassware—it’s more resistant, mainly if heating stays gentle. PTFE-lined containers, when checked and swapped at the first sign of wear, also hold up. Cost and limited shelf life make these solutions tricky for folks who don’t run huge budgets.
Over the years, chemists have pushed manufacturers to design more compatible specialty gear. Today, you find tools with PTFE coatings or even dedicated quartz vessels for handling the worst of the worst. Even so, very few labs can standardize all their hardware around these tough materials, so researchers learn to treat triflic acid like a loaded weapon.
Best Practices: Safety and Common Sense
The real answer for safe handling lies in training and strict protocols. Teams train under supervision, always double-checking before transferring acids. Safety shields stand by for any open transfer, spills get neutralized fast with sodium bicarbonate, and everyone keeps emergency gear close. Eye protection and heavy gloves become standard wardrobe.
Mistakes rarely end with minor expense. My own early years involved mopping up a small spill that etched a hole through a supposedly “acid-resistant” container. The cost ran far beyond a replacement flask—cleanup, air filter replacement, wasted experiment hours, and added anxiety in the team.
Sharing these stories keeps lessons alive. Triflic acid impresses with power, but successful chemists respect it for the danger. Whenever labs edge toward risk, they pause and swap in the right materials, no shortcuts allowed. As long as that mindset stays, people get to push chemical boundaries—and get home safe.
How is Trifluoromethanesulfonic Acid typically supplied and packaged?
Supplying a Powerful Reagent
Trifluoromethanesulfonic acid, known as triflic acid, turns heads in any chemistry lab. It brings enormous strength to the bench thanks to its superacid properties—some call it rocket fuel for chemical reactions. Strong acids like this don’t just show up on a shelf without some thought. The process that gets triflic acid from a chemical plant to the hands of a skilled chemist is both careful and strict for a reason.
The Appearance and Risks
Triflic acid stands out even before doing anything. This colorless liquid loves water, but add moisture and you set off violent reactions. Exposure to the eyes, skin, or lungs can cause severe burns or possibly permanent harm. I’ve watched a bottle of triflic acid chill in its own secondary container, everyone around it acutely aware of the risk. Not many substances demand this level of respect, and that’s before you even uncap the vial.
Packaging That Means Business
Manufacturers supply triflic acid in vessels designed for both safety and chemical stability. Commonly, suppliers use amber glass bottles to keep this acid guarded from stray light—trouble starts when photons and strong acids mix, especially in long-term storage. The glass needs to be thick and the seal airtight; triflic acid fumes crawl into the air at the slightest excuse, destroying seals that aren’t up to the job. Some companies turn to Teflon-lined containers for added peace of mind since Teflon resists attack by strong acids far better than basics like polyethylene.
Volume depends on the likely use. In research labs, 25-gram-to-500-gram bottles do the trick. For manufacturers needing industrial quantities, heavy-duty drums or metal cylinders—internally lined with fluoropolymers—step in. Each container gets clear, standardized hazard labels. Many companies layer multiple containers to lower the odds of leaks or accidents during shipping, especially when traveling across country or around the world.
Shipping With Care
Shipping regulations play a big part. The United Nations assigns triflic acid a number on their hazardous substances list: UN 3265. Regulations get picky about what kind of truck, packaging, and paperwork travel with those bottles or drums. You need crews who understand how to handle and respond if something tips over or cracks. In my years working with dangerous liquids, the paperwork and inspection process takes as long as measuring out the acid itself.
Keeping the Labs Safe
Storage and handling dominate discussion for anyone unpacking a shipment of this acid. Fume hoods, chemical-resistant gloves, and eye protection become non-negotiable. A chemical like this doesn’t forgive mistakes. Suppliers add safety data sheets with every order—sometimes updated within the year to reflect new research or regulations. Workers review every step, from carrying a bottle across the lab to neutralizing spills.
Building Better Safety (and Saving Money)
Some labs have asked suppliers to switch to smaller bottle sizes. Smaller bottles don’t just limit exposure; they cut waste if you only need a little for one project. Returnable containers offer another option, letting users send back empty—but never washed!—bottles for safe industrial cleaning or responsible recycling. These changes boost safety and trim costs, letting labs do more with less controversy or downtime.
Looking Ahead
The world demands stricter safety standards every year, especially around chemicals like triflic acid. Packaging technology keeps moving too. I expect smarter seals, tamper-proof lids, and improved warning systems. People who work with such acids benefit every time a supplier thinks harder about both safety and convenience. It’s about trusting not just the acid, but the people handling each step from factory to fume hood.
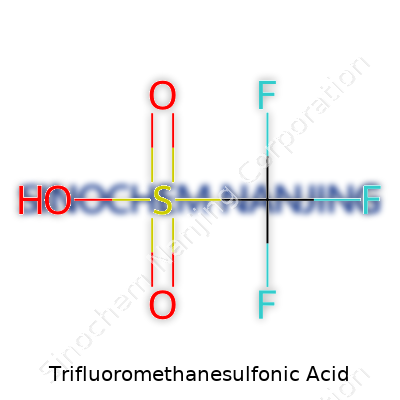
| Names | |
| Preferred IUPAC name | trifluoromethanesulfonic acid |
| Other names |
Triflic acid Methanesulfonic acid, trifluoro- Trifluoromethanesulphonic acid CF3SO3H Trifluoromethanesulfonate acid |
| Pronunciation | /traɪˌflʊərəʊˌmiːˈθeɪnˈsʌlfɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 1493-13-6 |
| 3D model (JSmol) | `3DModel: CCCS(=O)(=O)C(F)(F)F` |
| Beilstein Reference | 1718734 |
| ChEBI | CHEBI:5580 |
| ChEMBL | CHEMBL50475 |
| ChemSpider | 5116 |
| DrugBank | DB14108 |
| ECHA InfoCard | 100.004.875 |
| EC Number | 214-986-9 |
| Gmelin Reference | 82104 |
| KEGG | C02327 |
| MeSH | D014273 |
| PubChem CID | 61357 |
| RTECS number | YW5450000 |
| UNII | 88X2R5435S |
| UN number | UN3265 |
| Properties | |
| Chemical formula | CF3SO3H |
| Molar mass | 150.07 g/mol |
| Appearance | Colorless to slightly yellow liquid |
| Odor | Pungent |
| Density | 1.696 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | -2.1 |
| Vapor pressure | 14 mmHg (20 °C) |
| Acidity (pKa) | -14 |
| Basicity (pKb) | -14.7 |
| Magnetic susceptibility (χ) | -40.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.333 |
| Viscosity | 1.5 cP (20°C) |
| Dipole moment | 1.492 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -963.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1566.9 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage. Reacts violently with water. Corrosive to metals. Toxic if inhaled. |
| GHS labelling | GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 0, Instability: 2, Special: - |
| Flash point | 42 °C |
| Autoignition temperature | 400 °C (752 °F; 673 K) |
| Lethal dose or concentration | LD50 (oral, rat): 200 mg/kg |
| LD50 (median dose) | LD50 (median dose): 200 mg/kg (rat, oral) |
| NIOSH | NA9100000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | No REL (Recommended Exposure Limit) has been established. |
| IDLH (Immediate danger) | 30 ppm |
| Related compounds | |
| Related compounds |
Methanesulfonic acid Nonafluorobutanesulfonic acid Perfluorooctanesulfonic acid Triflic anhydride Trifluoromethanesulfonate |

