Triethylamine: More Than an Industrial Backbone
Tracing the Story: Historical Development
Triethylamine doesn’t have the flash of pharmaceuticals or the household familiarity of acetone, but its story reaches deep into industrial chemistry. Back in the 19th century, chemists like Charles Gerhardt and August Hofmann were laying groundwork for amine chemistry, opening doors for molecules like triethylamine. People forget that these basic nitrogen compounds became crucial as industry scaled up in the 20th century. Triethylamine helped drive innovations in synthetic dyes, pharmaceuticals, and even rubber processing. It’s the sort of compound folks outside the lab rarely notice, yet without it, so many manufacturing threads would unravel.
The Substance Itself: Product Overview
It’s hard to walk through an industrial lab and not come across triethylamine. This compound anchors countless syntheses, prized for its straightforward structure: three ethyl groups reach out from a nitrogen core. I remember the first time I used it in college – a strange, fishy odor, clear as day, but no frills or color. Triethylamine remains a mainstay on chemical benches, a builder’s brick in synthetic organic chemistry. Its value doesn’t come from flash; it comes from reliability and versatility.
What the Numbers Tell Us: Physical & Chemical Properties
Every chemist quickly learns its key traits: colorless, volatile, and carries a pungent smell impossible to forget. Triethylamine boils at about 89°C, a handy figure for separations and distillations. Its density hovers near 0.73 g/mL, lighter than water, floating atop if somehow both ended up in the same vessel. I remember its solubility in water isn’t huge—enough to matter, but not so much that mixtures get tricky. But in organic solvents, it moves freely, making reactions with acyl chlorides or esters quick and efficient. This solvent compatibility is why researchers keep it nearby.
Going by the Book: Technical Specs & Labeling Practices
In any serious lab, bottles of triethylamine carry unmistakable hazmat icons and warnings about flammability. Every workplace sets its own purity standards, aiming for either technical, laboratory, or trace analysis grades. Labels don’t just help with safety; they highlight specifics like water content and percentage purity, which sound dry but shape success or failure in chemical synthesis. Workers depend on clear labeling so nobody grabs the wrong grade for a finely tuned reaction.
Making Triethylamine: Preparation Methods
Manufacturers produce triethylamine using straightforward alkylation routes, reacting ammonia with ethyl chloride or ethanol in the presence of catalysts. These processes crank out large volumes at commercial scale, with companies refining catalyst mixtures for both efficiency and minimal waste. Even now, research teams tweak these methods, hunting for lower-carbon processes and clever ways to turn lower-cost feeds into high-purity product. Anyone who’s worked with older, impure batches knows why refining side reactions has become a priority.
How It Reacts: Chemical Behavior and Modifications
Triethylamine steps up in organic synthesis as a proton sponge or mild base. It soaks up acids liberated in esterifications, peptide couplings, or acylations, quietly pushing reactions where chemists want. The structure keeps it from acting as a nucleophile, so instead of taking part, it clears the path. It’s part of classic reaction work-ups—scavenger, buffer, and sometimes quarrelsome linker when making quaternary ammonium compounds. Specialists have explored its modifications, but in most cases, the triethyl scaffold is enough for its primary job.
Different Names, Same Backbone: Synonyms and Trade Names
Over the years, this compound’s been called by various aliases: TEA, triethylamine, and N,N-diethylethanamine. Some chemical catalogs even list old trade names, but the chemistry community always recognizes the basic structure. Synonyms come and go, but chalk dust and marker ink have commemorated “TEA” on lab whiteboards worldwide for decades.
Staying Safe: Safety Practices and Operational Standards
Triethylamine demands respect in the lab or on the shop floor. Its vapors irritate eyes and the respiratory tract, but its flammability provides the more pressing risk. I’ve seen labs ban open flames anywhere near a TEA workbench. Workers wear goggles and gloves, engineers install fume hoods, and procedures keep big drums tightly sealed. Regulations from US OSHA and similar agencies elsewhere prescribe exposure limits and spill procedures, yet every safety briefing reminds workers: you can’t get casual with TEA.
Where It Lands: Application Areas
The reach of triethylamine is broad—pharmaceuticals, agrochemicals, rubber, textiles, and even fuel processing. Used as a pH buffer and catalyst, it appears in manufacturing dyes and resins, too. In my time in chemical development, I watched TEA turn up from pilot plants making antihistamines to cement mixers for specialty cements. If a process demanded moderate basicity, TEA got the call. The flexibility of its chemistry has put it in formulations both simple and highly specialized.
Pushing Forward: Ongoing Research & Development
Even with decades of history, chemists haven’t run out of ways to tweak, recycle, or upgrade the triethylamine story. Green chemistry teams keep testing routes to lower emissions or substitute fossil-sourced feeds with bio-ethanol. Catalysis research explores if modified TEA derivatives deliver better selectivity or withstand longer runs in continuous reactors. In R&D settings, TEA is being eyed for novel uses in advanced materials or even as part of battery electrolytes. This is where experience meets curiosity—scientists who’ve spent years with standard TEA now search for its hidden potential.
Measuring the Risks: Toxicity Research
Toxicologists have dug deep into triethylamine’s effects. Inhalation causes respiratory discomfort, eye irritation, and high exposures can induce headaches or nausea. Chronic exposure remains a concern in manufacturing, which has prompted extensive studies in rats and mice for carcinogenicity, though the evidence is less concerning than for many other industrial chemicals. Water systems pick up trace amounts, leading environmental scientists to monitor for acute aquatic toxicity. Research hasn’t rested—there’s an ongoing push to find better ventilation systems or personal monitors for facilities using large concentrations.
The Road Ahead: Future Prospects
Triethylamine stands at an interesting crossroads. Society asks for cleaner, safer production—even in workhorse chemicals. Process chemists hunt for lower-impact syntheses, while manufacturers balance economics against regulatory requirements. As the push for sustainability grows, paths open for TEA sourced from renewables or recovered from waste streams. Labs keep searching for efficient ways to recover and recycle it, targeting minimal loss with every batch. In the bigger picture, this old staple keeps pulling its weight, no fanfare needed. It’s proof that chemistry doesn’t need to be glamorous to be essential—it only needs to get the job done, day after day.
What is Triethylamine used for?
The Hidden Chemical Behind Everyday Life
Walk through a hardware store or peek into a pharmaceutical laboratory, and you’ll brush up against triethylamine more often than you think. This clear, sometimes fishy-smelling liquid lands in some of the most surprising places. It’s not something you’ll see on supermarket shelves, but its fingerprints show up everywhere from vitamins to cleaning supplies.
Vital Role in Pharmaceuticals
Many drugs begin life as a jumble of raw ingredients needing the right push to become safe, reliable tablets. Triethylamine steps in as a base during synthesis, helping chemical reactions along, neutralizing acids, and getting medicines closer to what actually helps patients fight disease. Antibiotics, antihistamines, and local anesthetics often trace their history back to processes powered by this chemical. The final product gets purified, but that initial step with triethylamine shapes what ends up in the pill bottle.
Backbone of Chemical Manufacturing
Take a closer look at plastics, especially those used in car dashboards, smartphone cases, or medical equipment, and there’s a good chance triethylamine played a part. Polyurethane foams, which cushion everything from furniture to car seats, rely on this compound as a catalyst, speeding up the reaction that creates the foamy comfort we expect. In the laboratory, I’ve witnessed how a little triethylamine makes reactions go from sluggish to efficient, keeping production running smoothly and people safe from unexpected byproducts.
Supporting Agriculture
We expect our crops to be protected against insects and fungus, and triethylamine helps behind the scenes here, too. Pesticides and herbicides often start as ideas on a whiteboard, but reaching the scale of millions of acres depends on reliable production. Triethylamine reacts with other chemicals, helping form the backbone of these products, so fields stay fertile and harvests feed more people.
Ink and Cleaners: Everyday Encounters
Metal parts in factories and well-printed magazines both owe something to triethylamine. Industrial cleaners count on its ability to dissolve stuck-on grime, especially where water alone doesn’t cut it—think machine parts or stubborn residues. In printing, it adjusts the acidity of inks, so each magazine page comes out sharp, not smeared. I’ve seen how even small mistakes in preparation lead to wasted batches, so careful measurement matters.
Worker Safety and Environmental Concerns
Triethylamine packs power, but that same potency raises red flags in workplaces. Inhalation can irritate eyes and lungs, and high concentrations may cause bigger health problems. From my days shadowing a chemical plant safety officer, strict ventilation, proper gloves, masks, and well-maintained storage standards stood out as the only way to keep handlers out of trouble. Emergency plans, regular drills, and strict waste controls keep risks down for everyone, not just the people in chemical suits.
Building A Safer Future
The world produces more specialty chemicals every year, putting a premium on safety and sustainability. Cleaner production techniques, better containment, and new synthetic methods are always on the drawing board. As research piles up, scientists aim to find safer alternatives for more hazardous processes, but until then, real progress means consistent monitoring, responsible supply chains, and honest reporting. That way, the chemical’s benefits reach workers and consumers without risk spinning out of control.
What are the safety precautions when handling Triethylamine?
Real Hazards, Real Life
Anyone working in a lab or plant with triethylamine knows it isn’t some mild-mannered solvent. This chemical packs a punch. It smells like rotten fish, and even pulling open the bottle can sting the eyes. More than that, inhaling its vapor burns the nose, the lungs, and reminds you just how quickly a normal day turns nasty. Reports show regular exposure leads to headaches, dizziness, and even breathing problems. In fact, the American Conference of Governmental Industrial Hygienists set a low limit for airborne concentrations—only 1 part per million for an 8-hour average. That number isn’t chosen at random; real people got sick before those rules existed.
PPE Matters—But It’s Not The Whole Story
Triethylamine soaks through regular latex gloves fast. Nitrile or butyl rubber holds up longer, so I always double-check my PPE before starting work. Goggles aren't optional; they’re the line between a safe shift and an emergency eyewash trip. Splashing triethylamine in the eye leads to severe irritation, blurred vision, and if things go wrong, permanent damage. Lab coats, long sleeves, and even face shields play their part. Not every workplace puts these out, so it’s on each of us to track down the right equipment, whether management pushes it or not.
The Risk Lurks in the Air
Opening a bottle can release enough vapor to fog the whole bench. Fume hoods aren’t a luxury. The vapor rises fast, floats across workspaces, and brings a fire risk with it—triethylamine has a flashpoint at just 20°C. Just thinking back to a time when someone used it outside the hood gives me chills: one stray spark from a static discharge, and the bottle’s a firebomb. All electrical equipment should be spark-safe. Labs and storage spaces need ventilation that moves serious air volume and has alarmed detectors for leaks.
Storing Without the Guesswork
Leaving a bottle out isn't just sloppy work. Triethylamine corrodes some metals and soft plastics. I’ve seen shelving degrade after a year, leaching brownish residue. Stainless steel holds up, but regular iron does not. Storing in well-marked, tightly sealed containers in fire-proof cabinets reduces headaches and complaints later. Although it feels like overkill, grounding your storage containers reduces the risk of static discharge setting off a fire.
Accidents Happen—Respond Fast
Even with every precaution, spills happen. Triethylamine doesn’t mix with water; it runs across floors and gets into drains. Spilling it means evacuating staff, notifying emergency squads, and locking down the area. I keep the local hazmat response number saved in my phone after a close call. Large spills demand foam or powder extinguishers, not water—spraying water spreads the vapor and plays right into the fire hazard.
Training Makes the Difference
No amount of signage can make up for lazy training. Routine drills for spills, burns, and fires using triethylamine are more than box-ticking paperwork. When a reaction runs hot or a bottle breaks, panic is the real enemy. Every new hire needs to walk through the actual process, not just skim an email once. Reviewing real-world incidents sharpens everyone’s awareness. In the end, the safest labs are the ones where every voice—from student to supervisor—gets heard when it comes to dangerous chemicals like triethylamine.
What is the chemical formula and structure of Triethylamine?
The Chemistry Behind Triethylamine
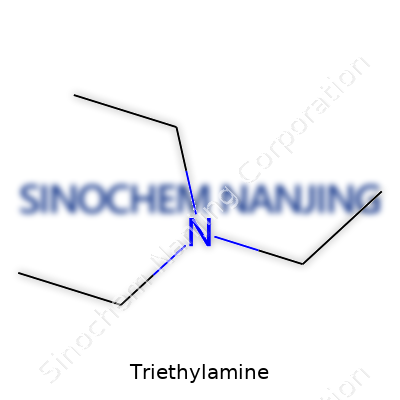
Triethylamine goes by the formula C6H15N. If you get down to the basics, it packs three ethyl groups – CH2CH3 – latched onto a nitrogen atom. Picture a central nitrogen with three arms, and each arm stretches out into a simple two-carbon chain. Not a fancy look, but it gets the job done. The structure looks like this: N(CH2CH3)3 – that’s the full spread. It’s what you’d call a tertiary amine. Forget about extra hydrogen on there; all available spots around the nitrogen are claimed by those carbon chains.
Triethylamine’s Place in My Lab Experience
Smell a whiff of fish and ammonia while handling transparent, volatile liquids, and odds are you’ve met triethylamine. In my own time mixing solvent batches or prepping organic reactions, this chemical tends to show up, often as a helper—like removing a stubborn acid out of the way. Sometimes it’s the little things that count in a reaction: triethylamine steps in to keep pH sweeter and nudges chemicals on the path to something new. It’s one of those go-to bases sitting on the shelf, easy to pour or pipette, and it saves time because it mixes clean even at room temperature.
Safety and Handling: Not Optional
This liquid doesn’t play nice. Long hours in a university lab taught me that skipping gloves and goggles becomes memorable for all the wrong reasons. Skin itches, air gets sharp, and accidents land you borrowing the eye-wash. The vapor makes its presence known fast, and it travels. According to the United States Environmental Protection Agency (EPA), triethylamine vapor can impact mucous membranes and the respiratory tract at low concentrations. Labs need solid airflow and careful storage, far from acids and ignition sources, to keep things predictable.
Industrial Use and Environmental Impact
Factories line up triethylamine for much more than academic chemistry. Paints, coatings, pharmaceuticals, and rubber plants use it for everything from neutralizing acids to prepping compounds for further reactions. Bulk use brings responsibility—leaks and spills head into water or air if nobody pays attention. On a larger scale, regulatory bodies keep a close eye on how much gets used and how it’s released to keep air and water around manufacturing plants within safe limits. Data from the Occupational Safety and Health Administration (OSHA) shows workplace exposure limits and reflects the need for strict safety protocols, not just for workers but for communities nearby.
Getting Ahead of the Risks
Labs and plants can keep risks in check with real training and better monitoring, not just a binder on the wall. Workers remember stories longer than rule sheets—sharper training, routine equipment upgrades, and transparent reporting help keep everyone safer. It pays to teach new hands what triethylamine means under the hood: not just the formula, but how to spot leaks, act fast on spills, and protect colleagues. On the industry side, updated venting systems, spill containment, and robust emergency planning should come ahead of production demands. Building a culture of respect for chemicals like triethylamine matters far beyond one company or one lab—it's one piece of keeping science sustainable.
How should Triethylamine be stored?
Why Triethylamine Demands Respect in Storage
Triethylamine isn’t just another bottle on the chemical shelf. Anyone who’s had experience in a lab or a manufacturing plant knows the unmistakable rotten fish smell—hard to ignore even from a sealed drum. It’s flammable, it catches fire easily, and simply leaving it on a bench is an invitation for trouble. I remember a time during my early lab days when a single uncapped vial cleared a building floor in minutes. Most people only make that mistake once.
Keeping It Cool—Literally and Figuratively
Heat is a dangerous friend to this chemical. Temperatures above room temperature push the risk up, and exposure to direct sunlight or hot pipes only raises the odds of vapors leaking out. I’ve seen storage rooms designed with thick walls and cooling units running nonstop, specifically for organics like triethylamine. Metal storage cabinets with explosion-proof fans prevent vapors from building up. Regular fans don’t cut it, because one spark from a motor, and you can have more than a ventilation problem.
Ventilation and Vapor Control: Non-Negotiable
Any area set aside for this chemical needs serious ventilation—movable air keeps vapor levels down. Good ventilation systems aim the airflow away from workers to exterior vents. At a facility I toured last year, the engineers insisted on local exhaust right at the drums, making sure anything that escaped got caught at the source. Relying on general air movement only puts everyone’s safety at risk. Proper vapor detection alarms add another layer, catching leaks before noses have to.
Container Material and Labeling—Get It Precise
Storing triethylamine in the wrong container is asking for leaks. Only containers rated for flammable, basic liquids make the cut—steel drums with tight, spark-proof seals or heavy-duty polyethylene bottles for smaller amounts. Some older plastics can break down, so checking with the material safety data is critical. Each bottle or drum should show clear hazard labels and a date. I’ve seen labels scrawled in faded marker—it only takes one mix-up to cause a costly mistake.
No Ignition Sources Near Storage
Triethylamine’s low flash point means that a spark from a nearby outlet or even static electricity on a dry day packs plenty of risk. Safe storage areas keep electrical panels, forklifts, cell phones, and any other spark-makers far away. I’ve watched teams install grounding straps from drums to the floor, making sure static doesn’t build up. Something as simple as a grounded metal strip can make all the difference.
Regular Checks and Training Matter
Everyone on site should know the emergency procedure for leaks and fires. Regular training avoids panic on bad days. Inspection routines catch drips, bulging containers, or torn labels long before they turn into emergencies. During an audit inspection I witnessed at a midsize chemical plant, checklists caught a corroded cap that could have brought a world of trouble. Good habits around this chemical are built through experience and attention to detail.
Smart Policies Backed by Reality
Following the best practices for storing triethylamine saves more than money. It protects lives, property, and the environment. Storage isn’t about ticking boxes on a compliance sheet—every control, from ventilation to labeling to regular checks, keeps accidents from becoming stories on tomorrow’s news. No one remembers the day nothing happened, and that’s really the mark of a storage plan working as it should.
What are the potential health hazards of Triethylamine exposure?
Understanding How Triethylamine Affects People
Triethylamine shows up in places where folks make plastics, some medicines, and fuel additives. It’s got a strong, fishy smell, easy to notice even at low levels. Each time someone breathes it in, touches it, or gets exposed at work, there’s a real risk to health that doesn’t always get enough attention.
Immediate Risks on the Job
People who work with triethylamine tend to notice nose and throat irritation first. Eyes start watering, skin itches, and sometimes gets red. Even a short time around this stuff can trigger headaches or dizziness. Inhaling higher amounts sometimes brings on breathing trouble, chest tightness, or asthma-like symptoms. Emergency rooms see patients whose lungs took a hit after being exposed, especially in job sites with poor ventilation. It’s not a mild nuisance—this chemical pushes the body’s limits with just a little too much contact.
Longer-Term Health Impacts
Prolonged exposure builds up more problems. The liver and kidneys work overtime to get rid of triethylamine, but they can only do so much. Stories from workers describe tiredness and poor appetite after many months. Research studies connect long-term inhalation with chronic cough and chronic bronchitis.
One concern for the eyes: Triethylamine can slowly eat away at the cornea. Workers in factories sometimes develop “blue haze vision,” where things look foggy for days or weeks—an early warning sign that often gets ignored. The risk is greatest for those who work several years in older plants, especially if eye protection isn’t standard.
Community and Environmental Concerns
Neighborhoods near manufacturing plants worry about chemical leaks. Spills during transport or storage release fumes into the air and seep into water. People who don’t even work with triethylamine sometimes face mild headaches or nausea when a wind shift brings vapors into town. Because this chemical floats around easily, keeping it away from drinking water matters—a lesson local governments learn when emergency teams get called for chemical spills.
Evidence and Solutions That Stick
Respected science journals and agencies such as the CDC have flagged triethylamine as hazardous. Their studies show that high exposure leads to irritated airways, organ stress, and even possible nerve problems. Small companies sometimes downplay fears, but it’s clear workers need more than a simple face mask. Real improvements happen when companies install local air extractors, airtight process lines, and invest in proper gloves and goggles. Regular training keeps safety culture alive on the floor. Medical checkups spot trouble before it turns serious, giving people the chance to step away when symptoms pop up. In the community, it helps when plants set up strong odor controls, prompt spill cleanups, and transparent air monitoring. Neighbors deserve the right to know what’s in the air and water near their homes.
Why Protecting Workers and Neighbors Pays Off
I’ve spent time talking with safety teams in chemical plants. One thing rings true: accidents and illnesses cost a lot more than prevention. When workers get sick or a spill hits a nearby park, everyone’s trust erodes fast. People want the right information and clear action steps, not complicated reports after the fact. Simple, clear safety procedures, regular health screenings, and honest communication keep risks in check and help everyone sleep a little easier, knowing their work and home stay safe from hazardous exposures.
| Names | |
| Preferred IUPAC name | N,N-Diethylethanamine |
| Other names |
N,N-Diethylethanamine TEA Triethylamine anhydrous Diethylethylamine |
| Pronunciation | /traɪˌɛθɪl.əˈmiːn/ |
| Identifiers | |
| CAS Number | 121-44-8 |
| 3D model (JSmol) | `/wiki/Special:EntityData/Q422547.sdf?flavour=gmmlx` |
| Beilstein Reference | 635873 |
| ChEBI | CHEBI:35797 |
| ChEMBL | CHEMBL1425 |
| ChemSpider | 8036 |
| DrugBank | DB00137 |
| ECHA InfoCard | 100.000.621 |
| EC Number | 203- 004-2 |
| Gmelin Reference | 790 |
| KEGG | C00490 |
| MeSH | D014248 |
| PubChem CID | 8003 |
| RTECS number | YE0175000 |
| UNII | BKOGFKQOJQZVIE |
| UN number | UN1296 |
| Properties | |
| Chemical formula | C6H15N |
| Molar mass | 101.19 g/mol |
| Appearance | Colorless, volatile liquid with a strong ammonia-like odor |
| Odor | Ammonia-like |
| Density | 0.726 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | 2.30 |
| Vapor pressure | 56 mmHg (20 °C) |
| Acidity (pKa) | 10.75 |
| Basicity (pKb) | 3.25 |
| Magnetic susceptibility (χ) | -13.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.398 |
| Viscosity | 0.37 mPa·s (at 20 °C) |
| Dipole moment | 0.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 254.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -137.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2020.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H302, H311, H314, H336, H412 |
| Precautionary statements | P210, P261, P271, P280, P304+P340, P305+P351+P338, P311, P337+P313, P303+P361+P353, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 2-3-0 |
| Flash point | -7 °C (19 °F; 266 K) |
| Autoignition temperature | 215 °C |
| Explosive limits | 1.2% - 8% |
| Lethal dose or concentration | LD50 oral rat 730 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 730 mg/kg |
| NIOSH | KI5775000 |
| PEL (Permissible) | 25 ppm |
| REL (Recommended) | 4 ppm |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Diethylamine Triethanolamine Trimethylamine Tripropylamine |

