Triethyl Borate: Chemistry’s Quiet Workhorse with an Eye on the Future
Historical Development of Triethyl Borate
Not many folks outside of chemistry circles know the story behind triethyl borate. The roots run as far back as the early 20th century, at a time when scientists explored boron compounds for everything from metallurgy to medicine. The compound caught early attention because of its ease of synthesis and its strong, clean flame — early experiments burned it for a distinctive green color, making it a favorite in fireworks. Over time, as organic synthesis picked up steam, chemists started looking for reagents that could add boron into carbon molecules without a mess. Triethyl borate answered that call, offering a balance of reactivity and controllability that’s tough to come by. The development of more advanced refining and separation methods through the 1950s and 1960s made it affordable on a scale large enough for industrial adoption.
Product Overview
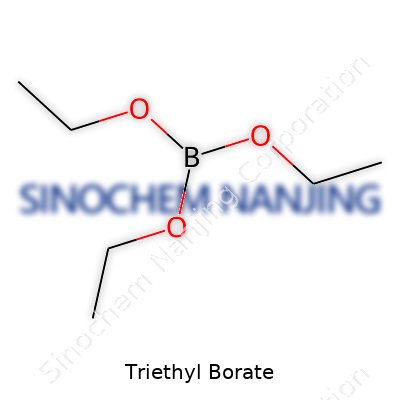
Triethyl borate presents itself as a clear, colorless liquid with a slight, fruity odor. Though it stays out of the spotlight, you’ll spot it wherever laboratories demand a reliable boron source or organic chemists need a mild esterification reagent. Its utility comes from the combination of reactivity, while remaining predictable — it doesn’t surprise users with sudden dangers common to some similar compounds. You’ll find the molecule, called B(OCH2CH3)3, working quietly in adhesives, solvents, and as an additive to impart that signature green flame to pyrotechnics. It may lurk as an intermediate in syntheses that eventually make up additives, resins, or even specialty glasses.
Physical & Chemical Properties
Pull out a small beaker of triethyl borate in a lab, and you’ll notice it flows easily, not sticky or heavy, thanks to its low viscosity and a boiling point hovering near 119 °C. Solubility tells another story — it mixes well with common organic solvents like ethanol, but turn to water and it begins to break down. That hydrolysis is what makes it interesting: add a little water, and over time, you’ll get boric acid and ethanol. This controlled breakdown underlies a lot of its practical use. The molecule is flammable, with a flash point low enough to demand some respect, so thoughtful handling and good ventilation are non-negotiable. Its vapor, if present in significant concentration, carries narcotic properties similar to many other light esters, but exposure rarely reaches levels that would concern the average lab technician with the right precautions in place.
Preparation Method
Chemists have honed a straightforward process to prepare triethyl borate, relying on the reaction of boric acid with ethanol in the presence of a trace amount of acid, usually sulfuric. This classical Fisher esterification method strips water out as the reaction moves forward, often using a Dean-Stark apparatus to catch the moisture and push things along. This route doesn’t just set the standard in the lab — industrial operations use essentially the same chemistry, sizing up with better equipment. Simplicity drives the popularity here; most labs don’t scramble for exotic catalysts or newfangled tricks. The accessibility of raw materials allowed for widespread use, even in places without advanced supply chains.
Chemical Reactions & Modifications
Triethyl borate earns its keep through versatility. Let it hydrolyze, and you land back at boric acid and ethanol. Point it at an alkyl lithium reagent, and it becomes a source for trialkyl borates. The ester group, despite its mildness, doesn’t just hang back. React it with certain strong nucleophiles, and newer, more reactive boron compounds appear. In one context, it’s a gentler alternative for introducing boron into organic backbones, limiting harsh byproducts that complicate purification. With organometallics, it enables formation of boron-carbon bonds, which lay the groundwork for complex pharmaceuticals and specialty chemicals. Tweaking the conditions or pairing it with different alcohols can lead down a new track, creating other borate esters with unique features, each finding a home in industrial niches.
Synonyms & Product Names
If you dig through the literature or chemical catalogues, triethyl borate shows up under several labels. Ethanol, boric acid triester and boron triethoxide come up most often. Each points to the same fundamental molecule — different naming systems reflecting the evolution of chemical nomenclature as understanding deepened. Internationally, it might appear as borate d’éthyle, or simple shorthand like TEB. The product rarely appears under flashy brand names, probably because its function stands clear and its applications stay mostly behind the scenes.
Safety & Operational Standards
Handling triethyl borate safely comes from a respect for its volatility and reactivity. The compound’s low flash point places it in the flammable liquid category in most labs and factories. Local regulations line up with that viewpoint, requiring flame-proof storage and careful ventilation. The mild fruity odor can lull folks into a false sense of security, but overexposure — like any lightly volatile compound — can cause irritation or temporary narcosis. Direct skin contact leads to drying and, if inhaled for extended periods, central nervous system effects. Whenever work ramps up above gram scale, splash protection, gloves, goggles, and fume hoods become a necessity. Companies educate workers on spill response and first aid — water cuts through most problems, but fast washing and fresh air always win the day. Waste management focuses on stopping runoff into waterways, since the hydrolysis product, boric acid, poses risks to aquatic life in high doses.
Application Area
Triethyl borate likely finds its widest recognition as a flame colorant, especially among those who ever watched fireworks with a detailed eye. Yet its real impact happens quietly in research labs, fine chemical manufacturing, and specialty glass production. In laboratories, chemists turn to it for smooth introduction of boron into organic frameworks, especially where other approaches introduce too much heat or create harsh reaction byproducts. Its gentle action on carbon-oxygen bonds allows it to serve as a mild dehydrating agent, tucked quietly into protocols for synthesizing more complicated molecules. In industry, it assists in formulations where controlling trace water matters. Some adhesives rely on its estery nature to help improve set times and chemical resistance. Optical glass manufacturers lean on boron content for improved clarity and resilience, and the triethyl form sometimes plays a small role in specific glass-melting steps. Its green flame, while eye-catching, only scratches the surface of its reach.
Research & Development
Chemists and engineers keep pushing the boundaries with triethyl borate, searching for new reaction partners that take advantage of its gentle personality. Research now spins toward more efficient and cleaner boron incorporation into pharmaceutical intermediates, where precise placement of atoms is everything. In academic circles, triethyl borate offers a springboard for exploring sustainable esterification methodologies — the idea: get more mileage from simple chemistry, cut back on harsh conditions and waste. Materials scientists run experiments using triethyl borate as a dopant in glass and ceramics, tuning properties for better performance under thermal or chemical stress. The low toxicity and ready degradation make it a fascinating choice in developing greener synthesis protocols, especially as regulations tighten and customer demands for “clean” chemistry rise. Graduate students focus on its role in organometallic frameworks or look at how subtle changes in its structure impact reactivity and practical application, feeding new insights back into both teaching labs and process innovation.
Toxicity Research
Solid data underscores that triethyl borate poses low acute toxicity by ingestion or skin contact, but as with most chemicals, the chronic picture grows more complex. Animal studies show that at high concentrations, it can suppress central nervous system activity, making drowsiness or temporary loss of coordination the worst-case scenario for accidental lab exposure. Inhalation or substantial spills raise risks — more due to ethanol and related alcohols than the boron core itself. Boron, in larger doses over time, does stress kidneys and reproductive systems in mammals, so regulatory agencies challenge industrial users to minimize long-term worker exposure. Waterways see potential harm, since boric acid — the hydrolysis product — can endanger aquatic organisms above trace levels. Inside industrial and academic safety committees, these numbers feed into guidelines, ensuring controls in place match the genuine risks rather than bureaucratic box-ticking.
Future Prospects
Triethyl borate faces a changing world. Market trends push speciality chemicals to tread lightly on people and planet alike, making its low-toxicity and easy degradability a silver lining for future expansion. As more attention lands on green chemistry and sustainable methodologies, triethyl borate will likely earn a seat at the table for esterification and as a boron source that produces minimal persistent byproducts. Its role might broaden across advanced materials, especially in the development of safer, high-performance coatings and specialty glasses. Emerging tech — energy storage, high-efficiency phosphors, and streamlined synthetic strategies for new pharmaceuticals — may uncover new niches for it. R&D spending keeps shifting toward multipurpose, responsible reagents, and triethyl borate’s legacy as a “utility player” gives it a running start in competing for those dollars. With supply chains and regulations evolving, its accessibility and simplicity just might cement its future as more than an old-school laboratory staple.
What is triethyl borate used for?
The Role of Triethyl Borate in Industry
Triethyl borate stands as a sleeper agent among chemicals. Most folks never run into it, but it quietly keeps industries humming. You spot it in everything from specialty glass manufacturing to the making of certain drugs. In my early days dealing with materials for electronics, this compound came up more often than expected—usually tied to cleaning up impurities or prepping things for high-precision work.
This clear liquid walks a tricky line. It feeds chemical reactions. In organic synthesis, you’ll find triethyl borate acting as a reagent, helping push reactions in the right direction, especially where boron’s oddball properties come in handy. For instance, when boron trifluoride gas needs safe handling or precise difference-making power, triethyl borate fills a gap. This matters in pharma and in tech, where minor variation spells big consequences. Purity isn’t just marketing speak—it means safer products and less waste.
Fuel Additives and Fire: The Surprising Side of Triethyl Borate
Triethyl borate doesn’t just play a quiet part in laboratories; it flashes out in the open, literally, when it’s used as a fuel additive. Jet fuels and rocket propellants sometimes receive a dose of it because it helps them light up—fast. Anyone who’s seen a cold-start on a jet engine knows fast ignition isn’t negotiable. Decades ago, when I toured an aerospace plant, an engineer described how tricky it gets to create a reliable spark at altitude—no room for error or frost. Triethyl borate kicks up the reliability factor. It gets the fire started, no questions asked.
Fireworks and Pyrotechnics
This chemical doesn’t only sit in the background of industry. In pyrotechnics, it brings color to the show. Light a compound of triethyl borate, and you get a signature green flame—a calling card for boron. People pushing the limits on firework displays use it to craft colors that don’t show up with basic metals. It’s almost an artist’s trick, blending chemistry and spectacle.
Solvents and Specialty Uses
In lab work, triethyl borate steps in as a solvent, especially where water doesn’t cut it and other organics cause trouble. Processing rare earth materials or textiles for sensitive electronic gear, you sometimes end up needing a solvent that won’t mess with the end product. Triethyl borate fits the bill, though it’s not the cheapest option so it’s reserved for trickier jobs.
Why Safety Needs Attention
Anyone handling triethyl borate has to respect its risks. It carries flammable fumes and, in the wrong hands, can cause trouble fast. History holds plenty of examples where lax chemical storage created disasters, both big and small. It’s not just about putting it in a locked cabinet. Clear labeling, worker training, and up-to-date safety data sheets save lives. Even in my smallest side projects, we followed manufacturers’ guidance closely. That diligence needs to keep pace as new uses for the chemical pop up in more industries.
Perspectives on the Path Forward
This compound’s story isn’t finished. As green chemistry picks up, finding sustainable ways to produce and recycle such chemicals matters more. There’s momentum behind using less hazardous solvents or designing workarounds in synthesis that cut out risky players like triethyl borate. Industry leaders lean on research and information sharing to push change forward. Scrutiny’s a good thing when it steers everyone toward choices that protect both workers and the world downstream.
Is triethyl borate flammable?
Everyday Chemistry Carries Its Risks
Triethyl borate isn’t a household name, but just because you don’t find it under the kitchen sink doesn’t mean it’s harmless. This clear, fruity-smelling liquid shows up in labs, industrial spaces, and sometimes in the hands of people who need its fire-making abilities. That fruity aroma? Don’t let it fool you. This stuff lights up easy and burns hot. If a container spills near an open flame, you’ll see why fire marshals set strict storage rules.
Flammable Means Troubling Flash Points
People often assume clear liquids are safe, maybe confusing them with water. Triethyl borate proves the opposite. It catches fire at around just 22°C (around 71°F)—room temperature for most of the world. That flash point puts it in the “danger zone” for fire risks. Handling this chemical requires the real safety gear: goggles, gloves, flame-proof lab coats, plus an eye on the thermometer. One spark means a classroom or factory could erupt with a flame taller than you.
Why It’s Still Used
I’ve walked into small pilot plants where engineers swear by triethyl borate for specialty fuel mixtures and some tricky chemical syntheses. It’s useful, fast-evaporating, and gives precise results. Chemists like that it works cleanly—no hidden reactions to mess up product purity. Fire departments don’t like how easily it ignites, so rules stay tight. It has a low boiling point and breaks down at high temperatures, releasing flammable vapors. That’s why leaving its bottle open in a warm space, even for a few minutes, starts to build up a dangerous air-fuel mix that matches what you find inside a lighter.
Flammability in Real Life
Spills aren’t rare. People fumble glassware or miss a cap. Someone bumps a shelf, and the liquid splashes near a spark or hot metal. The flame shoots up instantly—no time to run for help. Water won’t do you much good. Triethyl borate floats; it rides on top of the puddle you just tried to rinse away, spreading flames across the floor and up the benches if you’re not quick with a foam or powder extinguisher.
Rules and Smart Handling Save Lives
I’ve seen college labs pack up outdated bottles and stack them in a corner, assuming nothing happens unless something explodes. This is risky thinking. It takes only a little vapor trapped in a poorly ventilated room to trigger a disaster. A friend of mine once had to clear out an entire classroom after the faintest smell signaled a cap gone loose. Smell doesn’t always come before fire, either.
Limiting the Dangers
Fire codes suggest small bottles, self-closing safety cabinets, and banning heat sources. Sprinklers, chemical fume hoods, and detailed protocols work better than hoping for good luck or quick thinking. For students and researchers out there: pay close attention to labels, always ask for training, and trust safety gear more than your reflexes. Investing in basic vigilance—never storing near oxidizers, locking up flammable supplies, limiting open handling—goes a long way. Even a little knowledge changes the odds: triethyl borate commands respect, not casual use.
What is the chemical formula of triethyl borate?
Digging Into Triethyl Borate
Triethyl borate shows up with the chemical formula B(OCH2CH3)3. Some people shorten it to C6H15BO3. The structure means boron links up with three ethoxy groups—these don’t sound flashy, but they make a big difference in chemistry labs and industrial settings. Years ago in my university’s organic chemistry class, triethyl borate popped up during our synthesis projects. The smell alone—sweet, almost fruity—took over the entire lab bench. Even then, nobody in that room confused which compound we had. Knowing the exact formula kept everyone on the right track, especially as mixing borates gets tricky.
Why Chemical Formulas Count
Out in the world beyond the classroom, getting formulas right means much more than passing a quiz. Picture chemists in pharmaceuticals, electronics, or plastics. Mixing the wrong compound, or grabbing a similar-looking borate, costs time, money, and safety. Each element in that formula brings a specific property—boron’s lightweight, oxygen links everything in the right arrangement, and those ethyl groups make the compound volatile. Without the right structure, everything changes. Working in an adhesives factory, our team relied on suppliers who made labeling formulas a priority; nobody wanted a tank full of the wrong ester blend.
The Role of Triethyl Borate in Industry
Factories use triethyl borate mostly for its ability to play the middleman—helping turn raw boron and ethanol into catalysts and specialized solvents. On the research side, labs count on that formula to run flame tests. I remember the first green flame I saw—triethyl borate was responsible for that pop of color. Teachers look for that in demonstrations, and they lean on the accuracy of the chemical formula to decide if the bottle sits safely on the demonstration cart.
Stakes Around Accuracy
Many accidents in chemical plants start with a silent assumption: the drum contains exactly what it claims. Anyone swapping out a single group in triethyl borate’s formula gets a whole new substance. Different toxicity, different flammability, maybe nothing works as planned. Compliance rules follow this logic—regulations hold factories to unforgiving standards. Suppliers must back up their labeling by testing, and labs cross-check chemical formulas before experiments. I have watched colleagues pause their entire workflow if the certificate of analysis slipped even once.
Solutions for Safer Use
Preventing formula mix-ups takes real steps. Factories install digital inventory systems that track compounds by unique identifiers and full formulas. Warehouses segregate borates based on detailed data, not just brand. Training programs drill staff to memorize the formulas that show up most days. I still keep a laminated copy of key chemical groups by my bench, just in case fatigue kicks in. Schools could encourage students to sketch out chemical structures, not just memorize names, so the logic behind every label sticks. Online verification tools now let people scan a code and pull up the full chemical details, which means fewer costly surprises for everyone along the supply chain.
Final Thoughts on Getting It Right
Triethyl borate’s formula means clarity, reliability, and safety for everyone who opens the bottle. It’s a simple but critical bit of knowledge—proof that, in chemistry, every atom matters. Accuracy lifts up the entire system, from the research bench to industrial-scale reactors. For anyone in the business of chemistry, keeping a sharp eye on formulas stands as the best investment you can make.
What safety precautions should be taken when handling triethyl borate?
Understanding the Risks
Most people never hear about triethyl borate unless they're working in a lab or a chemical plant. This clear liquid packs more punch than its appearance suggests. It can catch fire without much warning, sends fumes into the air that can irritate your throat, and once spilled, leaves behind a mess that’s both flammable and toxic. I’ve seen new lab techs underestimate chemicals because they seem harmless at first glance—clear liquids don’t seem as threatening as bright acids or foul-smelling solvents. That mindset gets folks into trouble.
Personal Protective Equipment: Not Just for Show
Walking through a chemical storeroom, you’ll notice gloves, goggles, and face shields hanging by the doors. These aren’t decorations. Triethyl borate can sting the eyes or raise welts on skin. Standard practice calls for splash-proof goggles and nitrile gloves, not latex ones (latex sometimes breaks down with certain chemicals). I once watched a careless colleague use no gloves with a similar ester—he wound up in the emergency room after spilling some on his arm. Even short exposure can end up burning.
Besides hands and eyes, the lungs need protection. Triethyl borate fumes can trigger headaches or nausea. Good labs always use a fume hood when pouring or mixing this compound. In crowded workshops or older chemistry labs, open windows and fans offer some help, but they’re no substitute for an actual hood.
Fire Dangers No One Wants to Test
Fire remains the top threat. Triethyl borate flashes at temperatures lower than boiling water. One misplaced spark or open flame could turn a routine task into a disaster. I’ve seen long-time coworkers forget this, lighting a Bunsen burner across the room, only to get hit by a vapor flashback. Store this liquid far from heat sources—think steel cabinets marked with flame hazard labels.
Water will not put out a fire here. Having carbon dioxide or dry chemical extinguishers ready saves lives. Sometimes people figure a sink or safety shower can douse any trouble. If this stuff starts burning, dumping water only spreads the fire. Fact-checking reports from old accidents, most injuries came because folks used the wrong tools during a crisis.
Routine Handling and Smart Storage
Triethyl borate belongs in secure, tightly sealed bottles. Don’t ever keep these near acids or bases; unwanted reactions can burst bottles or send gas into the room. I’ve worked in cramped storerooms where chemicals crowd each other on shelves. One bump, and a leaking bottle can leave people coughing through the rest of their shift. Organization isn’t just housekeeping—it cuts down on accidents.
Every container should carry a clear label: hazard diamond, date received, and an emergency contact number. Without up-to-date labeling, confusion reigns when you actually need fast answers.
Training Makes the Difference
No clever engineering control replaces plain old practice and education. Anyone handling triethyl borate needs hands-on training. I’ve met new hires who skim safety sheets, nod in meetings, then freeze up at the first spill. Prepared teams know where to run, who to call, and what gear to reach for before things go sideways. In my shop, we run regular drills: spill containment, emergency showers, and escape plans.
Triethyl borate plays a role in synthetic chemistry, fuel additives, and specialty glass, but it only stays safe if folks stop rushing and take precautions. Remember, the real danger isn’t the liquid in the bottle—it’s forgetting what that bottle can do when you drop your guard.
How should triethyl borate be stored?
Stability Comes Down to More Than a Label
Triethyl borate isn’t the sort of chemical you leave on a shelf and forget about. It’s a clear, flammable liquid, often used for specialized reactions or as an additive in manufacturing. But anyone who’s spent time with lab inventory knows that so many accidents stem from misplaced bottles or ignored warning signs. Questions about safe storage don’t just safeguard property; they protect the lives of coworkers and prevent environmental headaches far down the line.
Fire Hazards Call for Serious Precautions
Take a whiff of triethyl borate – its sweet smell might seem harmless, yet it masks one of its biggest dangers: high flammability. Fires start in an instant in labs crowded with heat sources, so anything this volatile belongs in a tightly controlled area. Ordinary storage cabinets rarely cut it. Flame-proof lockers vented properly cut down the risks. I’ve worked in places that tried using leftover metal cabinets, but all it takes is one missed spark, and a minor mistake burns down a year's worth of work.
No Moisture, No Mistakes
Triethyl borate reacts with water, slowly breaking down into ethanol and boric acid. Pouring it next to a sink or in humid storage rooms just invites trouble. Moisture sneaks past cracked seals or faulty vents. Desiccants stand as a good line of defense, especially for folks working in older facilities with less climate control. It's easy to undervalue a well-taped cap or a desiccant bag until you deal with gummed-up lids and spoiled batches.
Avoid Breathing Fumes
Anyone who’s ever cracked open a bottle knows the sting that comes from inhaling vapors. People often cut corners: bottle it up quickly, and ignore the fume hood when pulling a sample. After walking out with a headache once, I always favor extra ventilation. Keeping bottles tightly closed with a solid seal, labeling them with expiration dates, and stashing them away from direct sunlight steers clear of unintentional exposure. Good habits build safety culture just as much as any written protocol.
Don’t Store With Strong Oxidizers
Mixing up chemical storage creates potent hazards. Triethyl borate sitting next to oxidizers like potassium permanganate puts the entire storage area at risk. I’ve seen storage plans gone wrong simply from putting incompatible chemicals an arm’s reach from each other. Separate shelves — even across the room — go a long way. It may slow down workflow a bit, but it spares everyone long hours of incident reports and cleanup.
Disposal and Emergency Gear Should Never Be Afterthoughts
Chemical fires and spills happen, even to careful folks. Storing absorbent materials and a fire extinguisher nearby, not across the building, keeps emergencies manageable. Having a written procedure, running safety drills, and making sure every shift knows where the gear lives — this is what stops panic. It’s not just a rule; it’s what you do to keep everyone heading home healthy.
Smarter Storage Isn’t Optional
Keeping triethyl borate safe calls for more than a locked cabinet. The risks move quietly but can turn any day upside down if ignored. Over the years, careful planning and simple habits have proven their worth time and again. People matter more than convenience, so putting in the extra care pays off — every bottle, every time.
| Names | |
| Preferred IUPAC name | Triethoxyborane |
| Other names |
Borate ester Boron triethoxide Triethoxyborane Triethyl orthoborate |
| Pronunciation | /traɪˈɛθ.ɪl ˈbɔː.reɪt/ |
| Identifiers | |
| CAS Number | 121-46-0 |
| 3D model (JSmol) | `3DModel:JSmol:C(COB(OCC)(OCC)OCC)` |
| Beilstein Reference | 3900175 |
| ChEBI | CHEBI:51848 |
| ChEMBL | CHEMBL138870 |
| ChemSpider | 5086 |
| DrugBank | DB14162 |
| ECHA InfoCard | ECHA InfoCard: 100.003.850 |
| EC Number | 203-742-5 |
| Gmelin Reference | Gmelin Reference: 1361 |
| KEGG | C18673 |
| MeSH | D002270 |
| PubChem CID | 7922 |
| RTECS number | EY5450000 |
| UNII | NJ4J2A8A4I |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C6H15BO3 |
| Molar mass | 130.94 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet |
| Density | 0.885 g/cm³ |
| Solubility in water | Soluble |
| log P | 0.94 |
| Vapor pressure | 2.6 mmHg (20°C) |
| Acidity (pKa) | 18.0 |
| Basicity (pKb) | 17.32 |
| Magnetic susceptibility (χ) | −52.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.398 |
| Viscosity | 1.326 mPa·s (at 25 °C) |
| Dipole moment | Triethyl Borate dipole moment: 1.63 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 269.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -498.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1977 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | No ATC code |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319 |
| Precautionary statements | P210, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-4-1-W |
| Flash point | \-10 °C |
| Autoignition temperature | 402 °C |
| Explosive limits | 1.8% - 8.6% |
| Lethal dose or concentration | LD50 (oral, rat): 3,200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 7,000 mg/kg |
| NIOSH | WT1750000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Triethyl Borate: 15 ppm (TWA) |
| REL (Recommended) | REL (Recommended Exposure Limit) of Triethyl Borate: **2 mg/m3** |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Trimethyl borate Tripropyl borate |

