The Real Story on Triethanolamine: More Than a Chemical Footnote
Tracing the Roots of Triethanolamine
Triethanolamine’s story winds through more than a hundred years of industrial growth, touching everything from early textiles to today’s personal care products. Over generations, chemical engineers have kept finding uses for this odd three-armed molecule—often because it solved real-world problems no one else could handle. Early synthetic processes kicked off in the early twentieth century, and you’d find the stuff in everything from textile softening agents to gas treatment systems. Later, the rise of mass-produced consumer goods fueled a fresh demand. Big names in detergents and cosmetics all leaned on triethanolamine because it blended well with both water and oil and stabilized mixtures that would otherwise fall apart. People in the industry still nod to its versatility, admitting that despite lab breakthroughs, nothing else has knocked it completely off the shelf.
What Makes Triethanolamine Different?
If you dig past the complicated names and charts, triethanolamine stands out for a simple reason—it brings both alkalinity and solubility to the table. In my own experience, working with soaps and cleaning products, I always ran into the challenge of keeping fats from clumping. Triethanolamine acted as a bridge, marrying oil and water without drama. Its clear, slightly viscous liquid form means it pours out reliably in the factory and doesn’t throw a wrench into mixing schedules. Boiling just above 330 degrees Celsius, it shrugs off moderate heat; its faint ammonia-like smell gives away its amine heart, but doesn’t overpower like other chemicals in the same family. These physical quirks explain why so many manufacturers keep coming back to triethanolamine instead of riskier or more expensive alternatives.
Demystifying the Specifications and Labeling
Anyone buying triethanolamine on an industrial scale learns quickly that grades and specs can make or break your product. Regulatory labels set the minimum, but real-life application calls for trust in your suppliers. On the shop floor, I’ve seen that purity over 99 percent cuts down on gunk and side reactions, saving hours that would otherwise get lost cleaning tanks and pumps. Labels reflect the amine content, water percentage, and potential presence of traces like diethanolamine—another cousin best kept in check. Workers handling drums of this stuff rely on clear hazard symbols and storage instructions, not because they expect trouble but because accidents with alkaline chemicals leave lasting reminders.
How Triethanolamine Factories Work
The process for making triethanolamine hasn’t changed all that much since its invention. Factories run a continuous reaction between ethylene oxide and ammonia in closed systems, squeezing as much as possible out of each batch. The design demands strict attention to detail. Ethylene oxide isn’t something you want floating free in a workspace, so engineers set up scrubbers and sensors at every stage. Any slip in temperature or ratio throws off the split between mono-, di-, and triethanolamine, so people monitor gauges and pipes with more care than a chef watches a holiday roast. From what I’ve seen on the ground, technicians pride themselves on tweaking the process just right, hitting target yields, and keeping community regulators satisfied.
What Happens Chemically?
Triethanolamine seems unassuming at first glance, but it joins in all sorts of reactions. Its three hydroxy-ethyl arms latch onto acids to make salts or react with fatty materials to form stable emulsions. This behavior fuels the chemical’s reputation as a neutralizer and a buffer. It doesn’t just stop at helping soaps spread evenly; clever modifications insert it into polymers, corrosion inhibitors, and even photographic solutions. In discussions with other chemists, we’ve often marveled at just how many industrial problems bend to triethanolamine’s particular blend of mildness and power. Its ability to swap partners, chemically speaking, opens the door to countless derivatives and custom solutions from adhesives to boiler water treatments.
Names That Point to Its Reach
Mention triethanolamine in a factory and you might get blank stares—unless you also mention TEA, Trolamine, or their foreign equivalents. These synonyms pop up everywhere, from cargo loading docks to safety training sessions. This cluster of names comes from both the chemical’s widespread adoption and its deep roots in traditional manufacturing. Everyone in my circle of chemical engineers and plant managers uses shorthand like TEA because it’s been around for decades, making conversations clearer and keeping paperwork moving.
Navigating Safety Standards in Real Workplaces
Staying safe around triethanolamine matters not just because of rules, but because little slips add up fast in busy plants. Skin contact stings, especially on chapped hands, and overexposure from splashing can lead to headaches or respiratory irritation. I’ve worked in settings where extra gloves and careful face masks were non-negotiable. Spills get neutralized right away—not only to limit exposure, but to stop residue from interfering with finished product quality. Industry standards like OSHA and REACH require regular training sessions, safety data sheets on every wall, and emergency showers within easy reach. No matter how comfortable folks get, old-timers pass along cautionary stories about what happened the one time someone didn’t double-check a valve.
Everyday and Specialized Uses
Triethanolamine has a hand in products most people use daily, though few know it. From shaving creams to liquid laundry detergents, its stabilizing roles mean foams don’t collapse before the job’s done. Walking the aisles of any large store, I see its effects in cleaner glass, fresher-smelling carpets, and longer-lasting latex paints. It steps beyond household goods too—refineries lean on it to scrub acidic gases from exhaust streams, textile mills soften fibers with it, and metal manufacturers include it in cutting fluids to keep machines running longer. Each industry leans on triethanolamine’s chemical flexibility, but they share the common truth that without it, product performance drops and maintenance costs go up.
What’s Happening in Research and Development?
On the research front, labs continue searching for green pathways and improved formulations. People want alternatives with lower toxicity, and innovators keep tweaking the triethanolamine molecule to suit cleaner industrial cycles—less waste, more recycling. Some teams experiment with biobased production, skipping fossil feedstocks completely. Others develop smarter blends tailored for niche markets like medical lubricants or advanced coatings. The academic journals buzz with new findings about improved biodegradability and reduced skin irritation, hinting at a future where traditional chemicals get replaced, or at least meaningfully upgraded. Several colleagues say the ingredient may eventually fade from cosmetic uses as stricter regulations roll through, but in manufacturing, its deep integration means change won’t come overnight.
Diving into Toxicity Research
Toxicity research on triethanolamine paints a picture with plenty of shades of gray. Generally, scientists agree the compound poses low acute toxicity when handled correctly, but there are signals worth heeding. Chronic exposure, especially in high concentrations, raised alarm bells about skin sensitization and, in rare animal studies, tumor formation when contaminated with secondary amines. The European Chemicals Agency keeps it listed for ongoing review, and studies keep probing for potential links to long-term health effects in repeated cosmetic applications. While no global health agency has sounded a total ban, industry insiders watch regulatory trends closely. From my own experience, anyone working hands-on with this chemical keeps up to date on safety protocols and doesn’t hesitate to swap in alternatives if too many red flags pop up.
Future Directions and the Road Ahead
Triethanolamine’s future depends on the push and pull of regulation, consumer demand, and technical innovation. Companies that build safer alternatives face the tough challenge of matching triethanolamine’s cost-effectiveness and performance. Some markets, like electronics and renewable energy, could see more advanced surfactants taking its place, nudged along by public pressure and environmental legislation. In the personal care aisle, shifts toward cleaner labels and organic branding will probably chip away at its dominance over time. No matter how the landscape shifts, the industrial backbone it provides keeps it relevant longer than many newcomers. I’ve seen how deeply embedded it is in factories and supply chains—a change will take years of planning, testing, and retooling. For now, that familiar clear liquid keeps finding new ways to stick around.
What is Triethanolamine used for?
Everyday Chemistry Hidden in Plain Sight
Triethanolamine isn't a name most folks run across unless they're squinting at the back of a shampoo bottle or picking up paint for the living room. This chemical has a knack for weaving itself into everyday life without kicking up much fuss. Triethanolamine belongs to the family of the amines and alcohols, giving it the power to both stabilize things and play nice with lots of other ingredients.
Sudsy Showers and Clean Faces
Take a quick look at personal care shelves—face cleansers, shaving cream, and bubble bath—all rely on something to help blend oil and water. Triethanolamine steps in here. It helps soap lather up by keeping fat molecules suspended in water. This makes showers feel a little silkier, skin cleaners wash off smoothly, and shaving creams spread right.
My own experience comes from standing in the shower, watching foam bubble up from a dime-sized dollop. Not magic, just chemistry. Without effective emulsifiers like triethanolamine, those bubbles wouldn’t hang around.
Staying Fresh: Cosmetics Rely on Chemistry
The make-up aisle is another place where triethanolamine gets busy. Lipsticks, foundations, mascaras—these need to keep their shape, color, and texture for months. Triethanolamine tightens up the formula, fights off clumps, and helps keep preservatives even throughout the bottle or tube. Stability in these formulas helps cut waste too, since less product needs tossing thanks to spoilage.
Cleaners, Paints, and Plants
Home improvement jobs often call for paints or polishes. Here, triethanolamine kicks in as a buffer. It holds the pH of paint steady, so colors stay true and cans keep for longer stretches on the shelf. In polishes, this chemical smooths the wax and water mixture, letting it glide on countertops and furniture.
Even lawns and gardens get a dose. Herbicides and fertilizers can clump up or clog sprayers, so a little triethanolamine helps those blend properly and cover every blade of grass.
Health Concerns and Risk Checks
No free ride comes with chemicals in the spotlight, so safety should stay part of the conversation. The US Food and Drug Administration and similar agencies worldwide allow limited use of triethanolamine in cosmetics, but levels are capped. Constant exposure in high amounts has raised questions, especially since under some conditions, it can react with certain ingredients to form nitrosamines—compounds linked to cancer in animal studies.
So what should people do? Reading product labels gives consumers power. Brands that follow transparent sourcing and safety testing earn more trust. The science community keeps an eye on formulations, pushing for clear answers and safer blends.
Greener Ways Forward
Plenty of companies now invest in alternative formulas that cut down on potential risks without scrubbing away performance. Ingredient innovation continues, with startups and research labs testing plant-based options that mimic the effects of triethanolamine. As a result, we get goods that work just as well—sometimes even better—with fewer question marks.
Transparency, science-backed safety, and pushing for cleaner chemistry steer the conversation in a positive direction. Triethanolamine serves a purpose, but staying up to speed on research keeps people and the planet in mind. That’s how trust in everyday products stays strong.
Is Triethanolamine safe in cosmetics?
What Triethanolamine Does in Your Lotion
Open up a bottle of moisturizer, sunscreen, or even face wash, and you might spot triethanolamine on the ingredient list. This chemical plays a big part behind the scenes. It helps balance pH levels and keeps formulas feeling smooth. Without it, creams would separate and wouldn’t feel quite as nice on the skin. In a world where folks want products that look, feel, and smell appealing, companies rely on ingredients like this one.
Concerns Around Everyday Chemicals
Seeing a strange name in the list of ingredients can cause some worry. Triethanolamine—often shortened to TEA—has been looked at by safety groups for years. Some research raised flags related to skin irritation and allergic reactions. Smearing something on your face that leads to redness or breakout isn’t what anyone wants.
In the U.S., the Food and Drug Administration (FDA) doesn’t ban triethanolamine from cosmetics, but they do keep an eye on how companies use it. The ingredient shows up most often in very low concentrations. European regulators set stricter limits than the U.S., especially because TEA can sometimes react with certain preservatives to create trace amounts of nitrosamines—impurities that can pose real health risks over years of daily use.
The Science Behind the Warnings
Long-term studies led scientists to check out what happens after repeated exposure. In high doses or with ongoing contact, TEA can irritate skin or cause allergic reactions—these findings pushed some experts to recommend lower amounts in anything applied to the body. The International Agency for Research on Cancer listed certain nitrosamines as probable carcinogens, fueling more calls for caution.
The actual data on people who use regular, over-the-counter cosmetics at home paints a less alarming picture. Rarely does irritation pop up from the tiny amounts found in face wash or moisturizer. Most reactions trace back to heavy use or to pre-existing skin sensitivity. It’s not about single exposures; concerns crop up with heavy, long-term use from multiple products each day.
Making Smarter Choices in the Aisle
If you’re like me, standing in the store squinting at labels, it’s easy to feel overwhelmed. At home, I always make note when I try something new. Redness or itch means it goes straight in the trash. Listening to your skin makes all the difference. For folks with sensitive skin or allergies, skipping products with TEA or keeping use to a minimum can help.
Companies and watchdogs have found ways to make products safer. Some swap TEA out for gentler ingredients or use tighter rules on how much they add. Pay attention to regulations—many places require manufacturers to keep amounts well below those linked to health risks. A little homework pays off; trusted brands tend to follow guidance from groups like the FDA or European Commission.
Good science and transparency matter. Demand for clear information has pushed brands to label better and cut back on questionable additives. In the long run, reading and making informed decisions beats worry. If a product causes trouble, plenty of reliable alternatives line the shelves. People deserve both safe products and clear answers.
What are the potential side effects of Triethanolamine?
What Is Triethanolamine?
Triethanolamine often shows up in personal care and cosmetic products. You will spot it on ingredient lists for skin creams, lotions, shaving foams, or shampoos. This compound plays a role in balancing pH and creating lather. Its presence in many bathroom shelves and shower racks sparks questions about health and safety.
Skin Irritation and Allergic Reactions
Experience has shown that some people develop red or itchy skin after using creams or soaps with triethanolamine. Rashes or tiny blisters pop up for those who have sensitive skin or existing allergies. Research from the American Contact Dermatitis Society points out that this ingredient can spark allergic contact dermatitis. Products for sensitive or broken skin often give more trouble than those on healthy, intact skin.
Eye Discomfort
Shampoos that run into the eyes usually sting. Surveys on users of drugstore shampoos put eye discomfort down to triethanolamine. The substance may lead to mild burning or watering if it gets in the eyes. These reactions disappear quickly for most people, but users with eye conditions or contact lenses notice the effects more. The FDA sets strict concentration levels to lower this risk, but even small amounts hit hard in unlucky individuals.
Respiratory Effects
People working in factories or salons deal with triethanolamine powders or aerosols in the air. Inhaling dust or fumes irritates the nose and throat, causing some to cough or get a scratchy throat. Heavy exposure has triggered asthma symptoms in a handful of reported cases, mostly in industrial or occupational settings. The American Conference of Governmental Industrial Hygienists tracks these events and rates triethanolamine as a substance to handle with good ventilation.
Concerns About Carcinogenic Byproducts
Long-term cancer risk often enters conversations about cosmetic chemicals. Lab tests suggest that nitrosamines, a class of chemicals linked to cancer, can form when triethanolamine mixes with certain preservatives. The European Commission banned some of these preservatives in products with triethanolamine. Safety experts urge consumers to read labels and avoid using combinations of products that might raise this risk.
Environmental and Reproductive Effects
Sewage treatment plants find triethanolamine in greywater running off from homes and salons. Studies from the US Geological Survey show that neither high concentrations in streams nor drinking water usually turns up. Most reports list minimal risks for reproductive health, yet scientists keep checking for long-term effects since so many people use these products daily.
What Can Consumers Do?
Choosing products with clear labels helps. Anyone with skin issues or allergies can patch-test a new cream or lotion before daily use. Salons and factories keep good airflow or use masks for protection. Doctors recommend switching to products without triethanolamine if symptoms flare up. Advocates for safer cosmetics push for companies to offer cleaner ingredient lists and for regulators to update rules as new science comes out.
Final Thoughts
Triethanolamine has a place in modern routines, but it’s good practice to stay alert, read ingredients, and trust how your skin and breathing feel. Personal experience, mixed with honest reporting on side effects, supports making informed decisions about daily products.
Is Triethanolamine harmful to the environment?
The Ubiquity of Triethanolamine
Triethanolamine shows up a lot more in daily life than many realize. You find it in shampoos, detergents, industrial coolants, and even some household cleaners. Its main task? Balancing pH and helping mix oil and water. Easy to see how something so versatile gets widely used.
What Happens After the Drain?
After the rinse or cleaning session, triethanolamine makes a journey from the sink or washing machine right into the wastewater system. Here’s the catch: not all treatment plants completely break it down. According to the U.S. Environmental Protection Agency, municipal wastewater treatment systems remove only some portion of triethanolamine, letting the rest leak into rivers and lakes.
Impacts on Aquatic Life
Once in the waterways, triethanolamine can hurt small creatures at the bottom of the food chain. Fish and other aquatic organisms may experience trouble with normal body processes if they have long exposure to even low doses. The European Chemicals Agency flagged this, listing aquatic toxicity as a concern, especially when the chemical breaks down into ammonia, which further stresses fish and insect larvae.
Soil Health and Long-Term Troubles
Gardeners and farmers can run into indirect problems. If wastewater sludge rich in triethanolamine residues gets spread on fields, the soil can end up with unwanted chemical loads. Healthy soil teems with microbes, and these microbes sometimes don’t handle chemical intruders all that well. In my own backyard, I once tried using compost that came from a municipal waste site—plants grew slower, and the usual signs of earthworm activity dropped. Only after switching back to untreated local compost did things improve. Official studies point to similar concerns: excess chemical residues can reduce soil fertility by hammering down beneficial bacteria.
Beyond Wildlife: Human Exposure
While the immediate worry focuses on animals and soil, humans sit in the chain of events, too. Drinking water sources downstream of wastewater plants sometimes show trace amounts of triethanolamine. The numbers usually stay well below safety limits, yet no long-term studies spell out what regular low-level exposure means for health over decades.
Making Better Choices
We’ve come to rely on convenience and low-cost products. That means looking the other way when certain ingredients never quite vanish from the system. Getting serious about the issue calls for modernizing wastewater treatment. Advanced setups like membrane bioreactors capture more pollutants, but most towns still stick with older designs.
On a personal level, choosing household and grooming goods that skip unnecessary chemicals helps cut demand. I started checking labels for “plant-based” surfactants, and the difference in my home’s chemical load was noticeable within a year. Advocacy also matters—asking local councils and water boards to publish data about effluent quality can spark change.
Industry doesn’t have to lose, either. Companies can swap out problem ingredients with safer alternatives, especially since consumer pressure in the last decade has nudged some big brands to clean up formulas. In the end, protecting waterways and soils lets wild and domestic systems stay robust for future generations. Real change starts with what’s going down our drains right now.
Can Triethanolamine be used in food products?
A Closer Look at Triethanolamine
Triethanolamine pops up in many places: cosmetics, household cleaners, industrial applications. Most folks probably never spot it on a label. It serves as an emulsifier, a stabilizer, or to manage pH levels. The technical buzz can obscure a simple question: does it belong in food?
Safety and Regulatory Details
The U.S. Food and Drug Administration sets tough rules about what goes into food. Triethanolamine appears on lists for indirect food additive uses: things like adhesives and coatings, but not for direct consumption. Ingredient approvals in this area often focus on chemicals that could migrate from packaging during storage. Still, some global regulatory groups keep it off their food ingredient lists entirely.
Science often sets the stage for these decisions. Animal studies link high doses of triethanolamine to liver and kidney changes, though most exposure scenarios involve much lower amounts. Human studies remain limited. The World Health Organization does not recognize triethanolamine as a food additive. Its presence in food often results from accidental contamination, not purposeful formulation.
Lived Experience in Food Production
Anyone involved in food manufacturing learns to prioritize consumer safety above all. Even the hint of risk can turn manufacturing standards upside down. I once worked alongside a quality assurance team that spent weeks tracing the source of a chemical contaminant. That experience showed how effortlessly even trace chemicals can become a big deal—quickly.
Consumers and manufacturers alike should trust that an ingredient holds up to close inspection. The absence of triethanolamine from ingredient lists in food is not about paperwork. It’s about real safety questions that don't have clear, reassuring answers. Foods undergo some of the strictest ingredient reviews across any industry. The stakes matter more than convenience or cost.
Why Ingredient Choices Matter
Food isn’t just fuel — it’s tied to health, trust, and community. People expect food companies to use ingredients that offer nutritional value and pose no risk. Each ingredient on a food label tells a story. Many additives have roots in chemistry, but those used in food go through harsh scrutiny.
Triethanolamine might work well in making creamy lotions, but that doesn’t translate to the dinner table. Most food-grade emulsifiers and pH buffers, such as lecithin or citric acid, maintain a strong reputation built on both evidence and consumer trust. Keeping a safe distance from industrial chemicals in recipes protects both brands and the people who eat their food.
Moving Toward Safer Alternatives
Anyone crafting food recipes ought to seek out ingredients with a proven history of safe use. Sourcing teams address similar puzzles every day: what works well, keeps production cost in check, and doesn’t raise health concerns? Unlike in industrial chemistry, food companies can't cut corners by turning to obscure compounds with uncertain safety profiles.
Stronger regulations and more informed buyers continue to nudge the industry toward natural and thoroughly-tested ingredients. Sticking with trusted, food-safe emulsifiers blocks confusion and protects everyone involved. The drive for transparency is not a trend; it’s the foundation for real trust and healthier products.
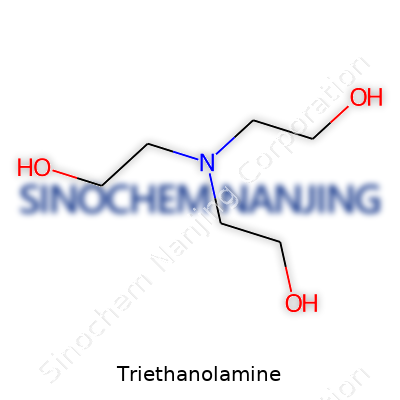
| Names | |
| Preferred IUPAC name | 2,2',2''-Nitrilotriethanol |
| Other names |
TEA Trolamine Tris(2-hydroxyethyl)amine Triethylolamine |
| Pronunciation | /traɪ.ɪˌθæn.əˈloʊ.ə.miːn/ |
| Identifiers | |
| CAS Number | 102-71-6 |
| 3D model (JSmol) | `JSmol.loadInline("data/mol/triethanolamine.mol")` |
| Beilstein Reference | 1819805 |
| ChEBI | CHEBI:18373 |
| ChEMBL | CHEMBL1539 |
| ChemSpider | 5647 |
| DrugBank | DB03300 |
| ECHA InfoCard | 03b577da-63d8-4673-8239-cf8c916bb9fa |
| EC Number | 203-049-8 |
| Gmelin Reference | 8224 |
| KEGG | C01876 |
| MeSH | D019369 |
| PubChem CID | 7618 |
| RTECS number | KL9275000 |
| UNII | 803I5X27HF |
| UN number | UN2491 |
| Properties | |
| Chemical formula | C6H15NO3 |
| Molar mass | 149.19 g/mol |
| Appearance | Colorless to pale yellow viscous liquid |
| Odor | Ammonia-like |
| Density | 1.13 g/cm³ |
| Solubility in water | miscible |
| log P | -1.0 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 7.8 |
| Basicity (pKb) | 4.1 |
| Magnetic susceptibility (χ) | -7.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.482 |
| Viscosity | Viscosity: 450 mPa·s (at 20°C) |
| Dipole moment | 5.08 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 362.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1196.78 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4190.5 kJ/mol |
| Pharmacology | |
| ATC code | D01AE24 |
| Hazards | |
| Main hazards | Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. Harmful if swallowed. |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Flash point | 179°C |
| Autoignition temperature | 335°C |
| Lethal dose or concentration | LD50 Oral Rat 6400 mg/kg |
| LD50 (median dose) | “5000 mg/kg (rat, oral)” |
| NIOSH | TRO |
| PEL (Permissible) | 5 mg/m³ |
| REL (Recommended) | 1.0% |
| IDLH (Immediate danger) | 1000 mg/m3 |
| Related compounds | |
| Related compounds |
Diethanolamine Monoethanolamine Tetrakis(hydroxymethyl)phosphonium sulfate |

