Thionyl Chloride: A Janus-Faced Powerhouse in Modern Chemistry
Historical Development
Looking back, thionyl chloride didn’t emerge in a vacuum. Early chemists in the 19th century started piecing together the puzzle of sulfur and chlorine compounds, searching for strong chlorinating agents long before the luxury of modern lab equipment. With industry running hard on the back of chemicals that could drive new reactions, labs honed in on thionyl chloride’s promise. By the turn of the twentieth century, methods to make and purify SOCl2 improved, leading to wide adoption not just in academic settings but also throughout pharmaceuticals, dyes, and agrochemicals. The pace of research quickened once it showed the ability to introduce chloride into organic molecules with good selectivity, carving out a reputation as a trusted tool in both large-scale plants and university glassware.
Product Overview
Standing in a chemical storeroom, a bottle of thionyl chloride looks unassuming—clear, mobile, sometimes mistaken for more benign solvents at a glance. Open it, though, and you’re hit with acrid, stinging fumes—this is a signal of the hefty reactivity hiding within each molecule. Thionyl chloride gets called to the bench every day for transforming carboxylic acids into acid chlorides, making those building blocks that fuel everything from plastics to painkillers. What draws attention isn’t just its efficiency, but the way it leaves by-products that mostly walk off as gases, cutting down on messy post-reaction cleanup.
Physical & Chemical Properties
Thionyl chloride presents itself as a colorless to pale yellow liquid, with a sharp, pungent aroma that lingers. The compound boils at about 79 degrees Celsius, below the boiling point of water. Quick to react with water—even atmospheric moisture—it produces sulfur dioxide and hydrogen chloride gases. In the lab, this play between reactivity and volatility keeps both excitement and risk on the table. Chemists know well that thionyl chloride’s structure (SOCl2) brings together sulfur, oxygen, and chlorine in a way that turns it into a potent chlorinating agent. Tackle it without a fume hood, and you’ll find out the hard way how unforgiving its fumes can be.
Technical Specifications & Labeling
On bottles and drums, labels aren’t for show. Every container warns of the dangers: corrosive, toxic, highly reactive, moisture sensitive. Concentration and purity determine how it behaves in a reaction, so industrial suppliers deliver specifications on trace impurities and water content. For anyone handling the material, this information isn’t just legal fine print—it tells you how hard you can push a process, or how much extra drying you’ll need to avoid surprise side reactions. A missed detail often means ruined product or safety incidents, so manufacturers update labeling as regulations and best practices change.
Preparation Method
Making thionyl chloride rarely happens outside specialized industry sites. Its preparation mixes sulfur dioxide and chlorine into a soup over a catalyst—usually a bit of phosphorus pentachloride or a related catalyst. These industrial routes run under controlled temperature and pressure to keep yields high, limit dangerous emissions, and prevent fires or explosions. The old-school lab methods using chlorination of thionyl sulfate no longer measure up for larger demands, with tighter environmental checks and the need to minimize side products pushing for carefully tuned continuous production lines. The manufacturing process itself remains a balancing act between efficiency, environmental responsibility, and cost.
Chemical Reactions & Modifications
In an organic chemistry lab, thionyl chloride’s reputation comes from its willingness to turn acids into acid chlorides—always delivering results cleanly if you respect its quirks. Once the reaction kicks off with a carboxylic acid, the by-products—sulfur dioxide and hydrogen chloride—vent off, avoiding bulky waste. Thionyl chloride plays a unique role outside simple transformations, cropping up in advanced modifications for pharmaceuticals, dyestuffs, and polymers. It can swap out hydroxyl groups, ring up activation steps for functionalization, and act as a dehydrating agent, pulling water from reactions that need to run bone-dry. Many chemists reach for it in battery electrolyte formulation, especially for advanced lithium batteries, tweaking its composition for best performance.
Synonyms & Product Names
Names change but the substance holds steady. In catalogs it goes by SOCl2, thionyl dichloride, sulfurous acid dichloride, and a handful of trade names depending on the supplier. These variations sometimes trip up buyers or students searching for safety data or technical details, so cross-referencing becomes second nature among professionals. The critical point is understanding these synonyms all mean a highly corrosive, water-sensitive liquid that calls for stringent respect during handling.
Safety & Operational Standards
Safety with thionyl chloride starts well before anyone cracks open a container. Even in small-scale academic settings, personal protective gear—acid-resistant gloves, full face shields, fume hoods—becomes non-negotiable. Accidents have happened when people underestimated the volatility or forgot how quickly it can react with water. Industry goes further, designing entire rooms for safe transfer and storage, using scrubbers to clean escaping gases, and training staff on emergency neutralization protocols. International regulations keep tightening, pushing for detailed exposure monitoring, spill response plans, and transport engineering that keeps containers from compromising under accidental shocks. Safety science learns from real-world mishaps, not just textbook scenarios, reinforcing the need for continuous vigilance.
Application Area
Walk into the product development office of a pharma company or a specialty materials plant, and thionyl chloride regularly makes the list for starting new syntheses. Drug makers use it for intermediates, dyestuff manufacturers for new colorants, agrochemical engineers for herbicide raw materials—the reach never stops expanding. Lithium battery development often lands on thionyl chloride because of its performance in non-aqueous electrolytes, powering sectors from defense to remote sensing. Its range stretches beyond chlorination: surface treatment, polymer processing, and even semiconductor fabrication rely on unique reactions only thionyl chloride can accomplish. The compound stands both behind the scenes and front-and-center in industries chasing innovation.
Research & Development
Innovation in using thionyl chloride doesn’t mean running old reactions in new places. Today’s chemists tinker with lower-waste protocols, recycling systems for unreacted reagent, and greener alternatives for solvents during reactions. Research pushes for ways to recover spent materials, especially as attention mounts on the environmental impact of halogenated chemicals. Projects at the intersection of chemistry and sustainability explore replacing thionyl chloride or blending it with additives that cut release of toxic by-products. Analytical chemists keep refining methods to monitor trace levels in finished products, cracking down on contamination risks for sensitive applications like electronics or pharmaceuticals. The R&D process doesn’t stand still—competition and tighter regulation mean constant searching for safer, cleaner, and more efficient uses.
Toxicity Research
Any conversation about thionyl chloride eventually turns serious when health risks come up. Its toxicological profile calls for respect. Researchers found clear evidence of corrosive injury to tissue and lungs long ago, and accident reports fill pages in chemical safety bulletins. Chronic exposure risks led to legal limits for workplace exposure and strict guidelines for air monitoring. Modern studies focus not just on acute exposure, but on the long-term breakdown products that factor into environmental safety. As environmental science advances, so does pressure for less hazardous alternatives, better personal protective equipment, and more effective fume handling systems. Toxicologists push for animal-free test methods and more real-world exposure modeling, using computational approaches to predict and prevent harm.
Future Prospects
Thionyl chloride’s role in high-value chemistry looks secure, but the ground keeps shifting underfoot. As environmental rules grow more complex and industries face pressure to lower their emissions, the focus sharpens on reducing its footprint—recovering and recycling the chemical, developing safer process designs, and investigating alternatives where possible. The battery industry, in particular, keeps driving demand upward, even as research pushes for next-generation compounds that offer better performance with fewer hazards. On the academic side, teaching safe handling joins creative attempts to demystify its dangers for students stepping into synthetic chemistry for the first time. Looking ahead, expect to see thionyl chloride stick around—not as a relic, but as a compound forced to adapt in a world demanding safer, cleaner, and more responsible chemistry.
What is Thionyl Chloride used for?
A Chemical With a Big Role
Thionyl chloride shows up in many places where you’d least expect it. At first glance, this simple compound, made up of sulfur, chlorine, and oxygen, doesn’t look like much. In real-world factories and labs, though, thionyl chloride pulls a lot of weight. You find it at the center of important steps in pharmaceutical production, electronics, and even some consumer products. My own years working in a chemistry lab taught me that this compound always gets attention from both researchers and health and safety managers. Thionyl chloride has a tough personality—reactive, a bit intimidating, but essential if used with respect.
Pharmaceuticals and Synthesis
Medicine relies on precise chemistry. Thionyl chloride helps make certain medications by transforming alcohols into chlorides, a move that helps build complex molecules. Drugs such as antibiotics or treatments for high blood pressure often go through a stage where thionyl chloride gets involved. Chemists count on this reaction to run efficiently and consistently. The pharmaceutical world moves fast, and thionyl chloride keeps things rolling by offering a reliable way to move from one stage of production to the next.
Electronics and Batteries
Most folks never think about what makes their electronic devices tick, but thionyl chloride leaves fingerprints on many of them—especially in lithium batteries. Companies turn to lithium-thionyl chloride batteries because they pack a lot of power into a lightweight design and last for years. Some wireless alarm systems, water meters, and medical devices depend on batteries with thionyl chloride inside them. This chemical mixture can handle tough environments and keeps electronics running longer without frequent battery swaps. In remote areas, or in devices where reliability matters more than anything, these batteries often get the call.
Pesticides, Plastics, and Beyond
For every product in your kitchen or garage, there’s a journey from raw materials to the finished product. Thionyl chloride shows up in the early steps for insecticides, dyes, and some plastics. Its ability to swap out certain atoms in molecules means manufacturers can tweak the properties of a product long before it ever hits a store shelf. Without it, many everyday items would look or perform very differently.
Risks and Solutions on the Table
Using thionyl chloride comes with clear risks. It gives off toxic fumes and reacts quickly with water—creating hydrochloric acid and sulfur dioxide. Working with it calls for careful training, good ventilation, and real attention to detail. I remember a day in the lab when a small spill set off alarms, sent everyone out for fresh air, and reset our attitude about taking shortcuts. That memory still sticks as a warning: even common chemicals bring hazards if you skip the basics.
Industry leaders have started to look at safer alternatives, and new engineering controls have decreased the odds of accidents. Switching to closed systems, using better protective gear, or looking for less reactive chemicals show real promise. Regular training and tougher safety standards keep the worst problems in check.
What the Future Could Hold
Demand for batteries keeps growing, and rules around chemical safety keep getting tighter. Experts and manufacturers want safer, greener options, but thionyl chloride will probably keep its place for a while—at least until a better substitute comes along. The path forward means handling this tool with the caution and skill it deserves, not cutting corners, and always looking for smarter ways to get the job done.
What are the safety precautions when handling Thionyl Chloride?
Thionyl Chloride: More Than Just a Chemical
Anyone who has worked in a lab or a chemical plant knows some substances demand extra respect. Thionyl chloride stands out for its reactive nature. It pushes people to pay attention, plan ahead, and make smart decisions every time it comes out of storage. My personal experiences, along with stories from colleagues, highlight just how critical these precautions can be.
The Hazards Behind the Bottle
Inhalation of Thionyl chloride vapors burns the nose and throat. A single spill can rapidly produce choking, acidic fumes. Old stories from expired fume hoods come to mind—alarms shrieking, staff scrambling to evacuate, and the unforgettable sting in the eyes. This isn't just overkill by safety managers. This reagent hydrolyzes instantly, producing hydrochloric acid and sulfur dioxide, both highly corrosive and dangerous to airways and skin.
Personal Protective Equipment: The Real Barrier
Some days, it might be tempting to grab only safety goggles and regular nitrile gloves. With Thionyl chloride, that won't cut it. Splash-resistant goggles, a full face shield, a chemical-resistant apron, and double-layer nitrile or even butyl gloves block the worst risks. When my colleague suffered a minor splash, even through doubled gloves, only quick access to a safety shower and proper PPE stopped a trip to the ER. Long sleeves, fitted lab coats, and closed shoes complete the armor. I always prefer slight discomfort from extra gear over burnt skin any day.
The Fume Hood: Not Optional Equipment
Ventilation saves lungs. Thionyl chloride belongs inside a properly functioning fume hood every time. Working in open lab space guarantees vapor exposure, especially if a bottle tips or a reaction overreacts. Regular maintenance routines—checking airflow, confirming alarms—become habits worth sticking with. I’ve learned to test the hood first with a bit of smoke or tissue before even opening the bottle. Shortcuts here invite problems.
Storage and Handling: No Shortcuts Allowed
Thionyl chloride lives in tightly sealed glass or chemical-resistant bottles, far from water, heat sources, or any substance that can react with acids. Give it its own storage cabinet if possible. Never return unused portions to stock; cross-contamination transforms a bad day into a disaster. I’ve seen labels dissolve under chemical drips, so I always double-check labeling and use secondary containment trays. It keeps everything visible and catches leaks before they wreak havoc.
Spill Plans and Training Save Lives
Unpredictable things happen, no matter the preparations. Every team member benefits from regular spill response training, knowing exactly where neutralizer kits and fresh air exits are located. I make it a point to quiz new staff on emergency eyewash locations—the few seconds saved matter in real emergencies. In a real amped-up situation, pre-printed spill charts, clear communication, and drills let people react with muscle memory.
Waste Disposal: Out of Sight, Not Out of Mind
After the experiment wraps up, leftover Thionyl chloride can't go down the drain. Dedicated chemical waste containers, clear labeling, and prompt pickup by trained disposal staff prevent unwanted reactions and fines. I came to appreciate scheduled hazardous waste pickups after seeing what happens when forgotten bottles leak over time. Responsible waste management builds trust in the system and keeps everyone working confidently.
Real Safety Means Real Vigilance
Chemical safety isn't just paperwork. Thionyl chloride reminds people to respect what they handle. Clear rules, proper gear, regular training, and precise storage bring peace of mind. In my experience, cutting corners never leads anywhere good—sticking to precautions protects people, research, and the lab itself.
What is the chemical formula and molecular weight of Thionyl Chloride?
Knowing the Numbers: Formula and Molecular Weight
Thionyl chloride brings a simple formula to the table: SOCl2. Each molecule contains a sulfur atom, an oxygen atom, and two chlorine atoms. Counting up the atomic masses – sulfur at 32.07 g/mol, oxygen at 16.00 g/mol, and chlorine at 35.45 g/mol for each atom – the math lands the molecular weight at about 118.97 g/mol. That’s more than just a number for labs or factories. Knowing the molecular weight matters every time someone wants to make a measured chemical reaction or ship out a drum of the stuff.
Why This Data Matters Beyond the Lab
Anyone who’s run a chemical process knows the formula and weight guide how much to buy, how to store it, and how much to react for maximum effect. Thionyl chloride can turn benign environments dangerous. It decomposes into toxic gases like sulfur dioxide and hydrogen chloride in the presence of water. The right molecular weight keeps chemists on track with safe handling measures.
Take storage. Many stories run through chemical plants where a wrong guess on the weight of a shipment led to containers buckling from built-up gas or a runaway reaction. Thionyl chloride’s formula reminds users to avoid water and stay ready for leaks.
Packed With History and Hazards
This chemical holds a steady spot in history books because it changes careers and technologies. Some decades ago, storing thionyl chloride in glass brought costly spills from unnoticed cracks. Now, manufacturers keep it in specific steel cylinders that stand up to corrosion.
Safety professionals pay attention to routine tasks as simple as weighing thionyl chloride. I remember a safety seminar where the trainer demonstrated how even a teaspoon dropped into a wet beaker could fog up an entire room. The same demonstration made it clear: never lose sight of the numbers.
Facts Guide Smart Handling
Thionyl chloride’s dangers aren’t exaggerated. Exposure can burn skin, eyes, and lungs, so personal protective equipment is necessary. Fume hoods and proper ventilation go along with chemical goggles and gloves. Each employee training makes clear: know the formula, remember the weight, and respect the chemistry.
Reactions that rely on SOCl2 – like turning carboxylic acids into acid chlorides – scale up in pharmaceutical plants and research labs all over the world. Each time, chemists run the math, double-check the weights, and never cut corners.
Room for Improvement and Solutions
Better education around the basics goes a long way. New hires who learn hands-on are less likely to skip steps or overlook dangers. Labeling that prints the formula and weight front and center builds a habit of mindfulness in the warehouse and at the bench.
Research looks at substitutes for thionyl chloride, but for certain essential transformations, it doesn’t have a rival. Until alternatives take over, strict storage, thoughtful transport, and respect for its properties keep teams and communities safe. Sharing stories and data, not just warning labels, helps everyone work with confidence and caution.
How should Thionyl Chloride be stored?
The Nature of the Chemical
Thionyl chloride isn’t something anyone can shrug off in a lab or warehouse. This colorless to yellowish liquid, with its sharp, suffocating odor, has a reputation for reacting vigorously with water—not even humid air goes unnoticed. What happens next? It releases hydrogen chloride and sulfur dioxide gases—both nasty for people and damaging to anything unlucky enough to get in the way. That makes negligent storage a shortcut to disaster.
Ventilation and Containment
Anyone who has worked with thionyl chloride keeps the bottle far from open air and water sources. The right storage area offers good ventilation, simple as that. An enclosed space with airflow from acid-resistant ductwork limits fume buildup and keeps workers from accidental exposure. A vapor-tight cabinet, with proper latching, stops any leaks. Anyone walking by should never get a whiff of the stuff unless there’s been a mistake.
Material Compatibility
Steel containers start corroding fast, so the choice usually lands on glass bottles or stainless steel drums lined with protective material. Even the smallest gasket, if made from rubber or plastic not rated for thionyl chloride, will surrender to deterioration. This isn’t about ticking off a checklist; it’s the simple fact that one oversight can mean container failure and an emergency no worker wants to face.
Temperature and Location
Stores keep thionyl chloride somewhere cool and dry. The range stays between 15 and 25 degrees Celsius, well away from direct sunlight or sources of heat. Too much warmth or fluctuating conditions speed up decomposition and make vapors a bigger threat. Facilities handling large volumes set up the storage zone far from entry points, breakrooms, and operations that handle water or alcohols.
Labeling and Access Control
A bottle or drum should declare its contents—unmistakable with warning labels and hazard symbols. This isn’t only for the regulators; tired eyes, hurried hands, or new recruits deserve a fighting chance to avoid a costly mix-up. Only trained personnel get access to the storage rooms. In a small lab, that means a locked cabinet. In industry, it's a dedicated, monitored storage building with a short list of who’s allowed inside.
Spill Preparedness and Emergency Planning
Any site that handles real volumes of thionyl chloride stands ready for trouble. Spill kits with neutralizing agents—soda ash, lime, or specialized materials—sit within easy reach. Protective equipment, like chemical-resistant gloves and goggles, hang on the wall. Teams set up emergency showers and eyewash stations just a few steps from the storage area. In my experience, even the best planning pays off when a careless move or an unseen leak tests the system.
Why Accountability Matters
The importance of these steps—ventilation, compatible materials, temperature, labeling, access—isn’t just compliance or common sense. The risks are real: skin burns, chronic respiratory damage, corrosive destruction of property, and regulatory penalties all hang in the balance. Serious storage standards build trust, keep the workplace safer, and protect the people doing the work. Even a smaller spill can eat through flooring and equipment in no time, and ignoring the basics means putting livelihoods and reputations on the line.
Improvement and Oversight
Periodic checks, honest drills, and real engagement—these habits help keep everyone alert. Automation and electronic monitoring work, but so does paying attention to normal smells, container conditions, and minor leaks. No system beats staff who know the risks and pay respect to the job. From small university labs to full-scale manufacturing plants, everyone benefits when thionyl chloride is given the respect it deserves from receipt through every moment on the shelf.
What are the hazards and first aid measures for Thionyl Chloride exposure?
Why Thionyl Chloride Packs a Punch in Labs and Industry
Thionyl chloride deserves respect on the shop floor or in a research lab. This clear-to-yellow, smelly liquid reacts fiercely with water, throwing off choking clouds of hydrochloric acid and sulfur dioxide. You might spot it in chemical manufacturing, dye production, or even old-school lithium batteries. Use always grows where companies chase lighter, energy-dense batteries and specialty plastics.
Getting even a drop in your eyes, on skin, or into your lungs can send you scrambling for help. One whiff tells you just how nasty it is—immediate coughing, stinging, tearing, and a sense that something’s gone badly wrong. Having worked near fuming acids and toxic gases, I’ve seen that underestimating the volatility of chemicals like this ends quickly in the ER.
Main Hazards—What Makes It Dangerous
Let’s talk about what actually happens during exposure. Aerosols hang in the air, irritating eyes, throat, and lungs in seconds. Direct skin contact burns fast, and even a small spill brings on blisters and deep tissue damage. Thionyl chloride even works through clothing, melting through fibers and clinging to the skin.
It’s corrosive inside and out. Swallowing a mouthful can cause severe burns down the esophagus and stomach, often leading to complications that only emerge hours after the fact. Breathing in lots of vapor leads fast to breathing problems, pulmonary edema, and—if first aid falls short—serious, permanent harm.
Large spills in closed rooms expose everyone around. Inhaling those acidic fumes, especially for folks with asthma or lung disease, runs up the risk of severe complications. For those running production lines or working in research facilities, even brief lapses on PPE set the stage for accidental splashes or unintended releases.
What To Do—Quick First Aid Makes All the Difference
Stay clear-headed. Remove contaminated clothing immediately, because thionyl chloride won’t stop eating through fabric and skin. Wash affected areas with plenty of water—fifteen minutes isn’t overkill. If it lands in the eyes, use an eyewash station and hold lids open, letting the water flush away every trace.
If vapors hit your lungs, get to fresh air. For bigger spills, evacuate, and make sure responders suit up with chemical-resistant gloves, eye protection, and supplied-air respirators. Avoid letting anyone collapse in the contaminated space, since thionyl chloride vapors settle close to the ground.
In case of swallowing, don’t try to vomit—get medical attention right away. Drinking milk or water may help dilute the acid, but only if it doesn’t delay the ambulance. Medical staff usually need oxygen, pain relief, and sometimes advanced airway support.
How to Make Handling Safer
Training makes all the difference. Nobody picks up thionyl chloride without knowing where the eyewash, safety shower, and closest exit are located. Chemical-resistant gloves, face shields, and full-body suits block splashes. Good ventilation or closed systems suck away corrosive fumes before they reach anyone’s breathing zone.
Strong labeling and storage rules cut down on accidents. Never store thionyl chloride near water or moisture, since even a tiny leak can start a deadly reaction. Chemical spill kits stay close at hand—fast response ends up saving limbs and lives.
It pays to keep the Material Safety Data Sheet visible and make sure every technician can recite first aid steps from memory. In workplaces that drill for emergencies, people respond faster and get back on their feet, even after something goes sideways.
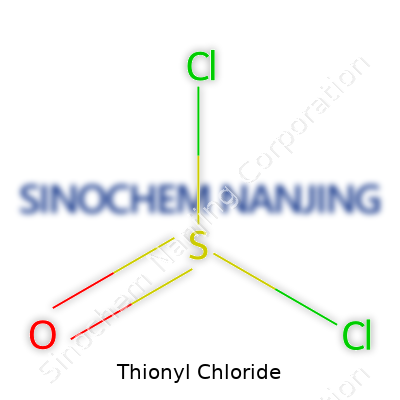
| Names | |
| Preferred IUPAC name | Thionyl dichloride |
| Other names |
Sulfur oxychloride Thionyl dichloride Sulfurous oxychloride SOCl₂ |
| Pronunciation | /ˈθaɪ.ə.nɪl ˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 7719-09-7 |
| Beilstein Reference | 3510663 |
| ChEBI | CHEBI:29767 |
| ChEMBL | CHEMBL1376 |
| ChemSpider | 109804 |
| DrugBank | DB11343 |
| ECHA InfoCard | 100.028.931 |
| EC Number | 233-045-4 |
| Gmelin Reference | 878 |
| KEGG | C19198 |
| MeSH | D013887 |
| PubChem CID | 24636 |
| RTECS number | WS4900000 |
| UNII | Q9L3BD3Y1K |
| UN number | UN1836 |
| Properties | |
| Chemical formula | SOCl2 |
| Molar mass | 118.97 g/mol |
| Appearance | Colorless to pale yellow fuming liquid |
| Odor | Pungent, suffocating |
| Density | 1.638 g/cm³ |
| Solubility in water | Reacts |
| log P | -0.08 |
| Vapor pressure | 19 mmHg (20°C) |
| Acidity (pKa) | -7 |
| Basicity (pKb) | -7.05 |
| Magnetic susceptibility (χ) | -33.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.515 |
| Viscosity | 1.23 mPa·s (at 25 °C) |
| Dipole moment | 1.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –297 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -296 kJ/mol |
| Pharmacology | |
| ATC code | V09AX04 |
| Hazards | |
| Main hazards | Causes burns, toxic if inhaled, reacts violently with water releasing toxic gases. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H331, H314, H317, H335 |
| Precautionary statements | Precautionary statements: "P223, P260, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-0-2-W |
| Autoignition temperature | > 140 °C (284 °F; 413 K) |
| Lethal dose or concentration | LD50 oral rat 141 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 2600 mg/kg |
| NIOSH | NIOSH: WS4900000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Thionyl Chloride: "1 ppm (parts per million) as a ceiling limit |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 20 ppm |
| Related compounds | |
| Related compounds |
Sulfur dichloride Sulfur monochloride Sulfuryl chloride Phosphoryl chloride Phosphorus trichloride |

