Tetramethylpropylenediamine: More Than Just a Chemical Name
Historical Development
Experienced chemists know that the backbone of any specialty chemical’s story often starts humbly in the laboratory, and Tetramethylpropylenediamine is no different. In the post-war era, as research into amine compounds accelerated, this particular diamine started to turn heads for its unusual symmetry and promise in organic synthesis. Lab journals from the late 20th century mention its role as a selective ligand and as a handy base for tough reactions. The compound’s recognition grew as analytical chemistry advanced, allowing for better understanding and wider adoption. Those early years stand as proof that even niche molecules can build reputations on persistence, rigorous science, and a little serendipity.
Product Overview
Chemists like me appreciate clear function over flair—Tetramethylpropylenediamine delivers both. This compound looks unassuming but steps up as a versatile player. Its symmetrical structure, with four methyl groups crowding the ends of the diamine backbone, creates unique properties. It resists oxidation better than simpler diamines, which extends its life in tough reaction conditions. Colleagues have told me they reach for it when other diamines run out of steam, especially in catalytic cycles needing a little extra punch. From synthesizing specialty polymers to stabilizing reactive intermediates, its practical importance continues to grow as new applications get tested each year.
Physical & Chemical Properties
Get a sample of Tetramethylpropylenediamine in the lab and you’ll find a colorless liquid, mild odor, dense compared to water, and slippery to handle on a glass surface. Its boiling point and solubility in common solvents make it appealing for multiple uses, especially where thermal stability is a must. Methylation at the amino groups bulks up the molecule and lowers its tendency to crosslink, compared to its parent propylenediamine. This matters for certain syntheses, because excessive crosslinking can kill yields. Handling care matters; it reacts strongly with acids, releasing heat, so best to keep it capped and stowed in a cool place.
Technical Specifications & Labeling
Tracking technical specs keeps operations on track, and Tetramethylpropylenediamine usually comes with purity north of 98 percent for lab grade. Density, refractive index, and boiling range help chemists dial in conditions quickly. Labeling in the professional world often draws attention to the chemical’s flammability, inhalation risks, and need for gloves and goggles—no one wants a lab accident from a moment of inattention. Having reviewed MSDS documents over the years, I am always glad when suppliers are explicit about storage and handling, as an extra measure to keep hazardous exposures in check.
Preparation Method
Old textbooks trace the preparation straight to methylation of propylenediamine, usually using a methylating agent like iodomethane under carefully controlled conditions. Industrial routes turn to catalytic hydrogenation or reductive methylation with formaldehyde for bulk quantities. Byproducts can get tricky; chemists need to watch for overmethylation, which reduces yield. It usually demands a delicate balance between reactant concentrations and temperature. Every time I’ve stepped through a methylation, I’ve learned something new—one of the few joys in repetitive lab work.
Chemical Reactions & Modifications
This molecule loves to act as a ligand in metal complexes, especially transition metals, where it stabilizes unusual oxidation states that would otherwise decompose. Its alkylated nitrogens block unwanted side reactions, so in catalytic hydrogenation or condensation, Tetramethylpropylenediamine often serves as a base that won’t gum up the works with side chains. Chemists use it in designing new chelating agents, and sometimes modify it further by introducing functional groups on the carbon backbone. I’ve watched this molecule take on new forms, spawning derivatives for everything from drug discovery to nanomaterials fabrication.
Synonyms & Product Names
Ask around the lab and you’ll hear a few different handles: N,N,N',N'-Tetramethyl-1,3-propanediamine, TMPDA in shorthand, or less commonly, 1,3-Bis(dimethylamino)propane. Chemical suppliers keep those names straight in catalogs, but researchers often just call it “tetramethyl diamine,” rarely pausing to spell it out. Clarity saves money and safety headaches, a lesson most learn early in chemical work.
Safety & Operational Standards
Direct exposure causes skin and eye irritation. Inhalation leaves a burning sensation and, with prolonged contact, headaches or nausea. Long sleeves, nitrile gloves, and chemical splash goggles earn their keep around this material. Ventilation helps too; Tetramethylpropylenediamine is volatile and its fumes can bother even seasoned lab staff. I always stress to students never to underestimate a clear liquid. Labeled containers and well-ventilated storage reduce risk. Company policies call for wastewater neutralization before disposal—responsible handling now keeps the regulators off your back later.
Application Area
Polymer chemists love Tetramethylpropylenediamine for its action as a chain modifier in polyurethanes and other specialty plastics. Catalysis research leans on its ability to buffer reaction environments, especially where selectivity trumps speed. In analytical labs, it helps stabilize reactive intermediates, making complicated syntheses possible in a single flask. Some electroplaters use it as an additive to improve metal deposition. I’ve seen it patch up reaction bottlenecks that stymied entire research programs. Most impressive has been its ability to reinvent itself as new fields emerge, often just waiting for a curious chemist to try it in a novel setting.
Research & Development
University labs dig deep on Tetramethylpropylenediamine’s role in transition metal complexation, yielding new catalysts for challenging organic transformations. Startups in green chemistry have picked up the compound for low-energy syntheses, particularly where non-toxic alternatives rank high on the priority list. Over the last decade, papers report its use in making organometallic frameworks and sensors for environmental monitoring. Dedicated research teams look for ways to reduce cost, waste, and energy in the synthesis of TMPDA, which brings economic and environmental wins. Chemists share findings at conferences, comparing results and pushing the field forward one problem at a time.
Toxicity Research
While not the most toxic amine in the chemical arsenal, Tetramethylpropylenediamine deserves respect. Animal tests—sometimes referenced, though debated on ethical grounds—show low acute toxicity, but repeated exposure raises questions about reversible nervous system effects. Chronic data remains sparse; so used with caution, it shouldn’t build up in organisms under typical lab conditions. Disposal strategies stick to neutralization and dilution, minimizing contamination to water or soil. Reviews in recent years encourage more rigorous study, especially as new uses and higher-tonnage production could increase occupational exposures. I teach new lab staff to treat every unfamiliar compound as a potential hazard, a lesson rooted as much in experience as in the findings of safety journals.
Future Prospects
Shifting regulations and rising demand for smart specialty chemicals shape the next chapter for Tetramethylpropylenediamine. Emerging markets in battery technology and specialty coatings offer new testing grounds. With society pushing for sustainable materials, research turns toward greener syntheses and safer formulations, which TMPDA could fit with the right tweaks. As more labs rely on machine learning to screen reagents, its flexibility in synthesis and catalysis keeps it firmly on the must-try list. For every old compound, markets and technologies reframe its value. Tetramethylpropylenediamine remains a punchy example that, with a few smart changes, yesterday’s specialty could become tomorrow’s staple.
What is Tetramethylpropylenediamine used for?
Understanding the Role of Tetramethylpropylenediamine
Tetramethylpropylenediamine, more commonly abbreviated as TMPDA, holds a specific yet crucial place in scientific and industrial labs. Many folks outside chemistry circles would probably walk past it without a glance, yet I remember the first time I came across TMPDA during a late-night graduate lab work session. My supervisor handed me a small, tightly sealed vial, warning me, “This stuff opens doors.” She wasn’t talking about literal doors but about opportunities in chemical synthesis.
In the lab, TMPDA acts as a strong organic base. It steps up to grab protons during synthesis, which sounds technical, but in practice, it’s often used to get things moving in reactions that otherwise stall. Chemists like it because it does its job cleanly without adding too much bulk or complexity. That matters for making complicated molecules — the sort used for new medicines or advanced materials — where every bit of simplicity counts.
TMPDA in Action: Making Something Out of the Ordinary
TMPDA isn't a household chemical like bleach or acetone, yet its reach extends far into daily life. In research and development for pharmaceuticals, TMPDA lends a helping hand in building blocks for drugs. I watched my colleagues piece together tiny fragments of a potential cancer therapy, and TMPDA was a part of the critical step, gently coaxing one molecule to join another without chaos. It supports these fine-tuned reactions by scooping up stray bits that might throw things off track.
Industries developing specialty polymers and coatings also find value in TMPDA. High-performance paints or coatings for electronics won’t win awards for glamour, but they rely on the tough, reliable chemistry that bases like TMPDA can provide. TMPDA helps chemists manage the conditions just right, which is especially important during large-scale production. This hands-on experience with TMPDA taught me that even small shifts in how reactions proceed can spell success or failure for materials used in products like wearable sensors, advanced cables, or high-durability paints.
Safety and Environmental Considerations
Working safely with TMPDA is non-negotiable. It’s a chemical you want to respect — gloves, goggles, and good ventilation have become second nature to me. Several industry reports highlight that, without proper training or handling, the vapor and liquid can irritate skin and eyes. Dumping it down the drain isn’t an option; responsible disposal with hazardous waste collection is the only path forward. Companies must keep their teams trained and aware, especially as regulations become tighter around chemical waste.
As society keeps an eye on the environmental footprint of laboratory chemicals, TMPDA hasn’t escaped scrutiny. Developing methods to recycle or neutralize leftover TMPDA remains at the front of conversations between chemists and safety officers. Academic groups have begun sharing greener pathways for reactions that cut down on chemical waste. These practices don’t just protect the environment; they save money and build trust with communities living near chemical plants.
Where Things Go from Here
Tetramethylpropylenediamine has earned its spot in the toolkit of chemists and industrial labs. Its power to encourage tough reactions and support innovation in medicine and materials continues to matter. The focus now shifts towards safer use and smarter disposal. It’s up to the community—researchers, industry leaders, and environmental advocates—to keep TMPDA helpful and safe for everyone who depends on what comes out of modern chemistry.
What is the chemical structure of Tetramethylpropylenediamine?
Getting Into the Details
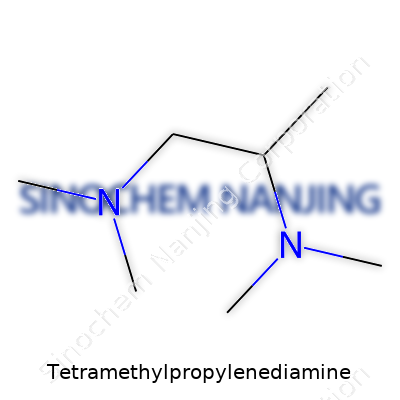
Tetramethylpropylenediamine, known in some chemistry labs by the shorthand TMPDA, has a structure that gets attention for its utility and versatility. The basic framework revolves around a propane backbone with two amino groups attached — one on each end. To make things interesting, chemists have attached four methyl groups to the nitrogens. This bulks up the molecule and changes how it behaves in real reactions.
The IUPAC name is N,N,N',N'-Tetramethyl-1,3-propanediamine. That mouthful basically means: take a three-carbon chain (propane), stick a nitrogen on each end, and on each nitrogen, put two methyl groups. The connectivity looks like this: (CH3)2N–CH2–CH2–CH2–N(CH3)2. This kind of design isn't just for show; it has major effects on both how stable the molecule is and how it interacts with other chemicals.
Experience From the Lab Bench
During my years in organic synthesis, I saw TMPDA showing up more than a few times. It often steps in for other amines when reliable, non-nucleophilic base strength is needed. Its methyl groups create a shield of sorts, making the nitrogens less reactive toward electrophiles, but those lone pairs still grab protons when called upon. In air- and moisture-sensitive reactions, TMPDA's structure means it doesn't muck up delicate processes by introducing unexpected side products.
Chemists prize its stability. The methyl arms do more than pad out the molecule; they keep the nitrogens from attacking in unwanted side reactions, which helps the main reaction run more cleanly. Think about it: fewer random products mean a happier chemist and less frustration during purification.
Why Structure Shapes Uses
Looking at the structure, every methyl group on those nitrogens serves a purpose. Each one blocks overcrowding and prevents hydrogen bonding, which can change solubility. TMPDA dissolves in organic solvents with ease, which lets it get involved in a wider range of reactions compared to other, more water-loving diamines.
Industry uses don't always get flashy write-ups, but TMPDA plays a role in the making of dyes, polymers, and catalysts. In some cases, fine-tuned structure like TMPDA's helps make specific ligands for metal-catalyzed reactions. The shape and electronic profile set by the tetra-methylation lets chemists build precision tools for tough applications — things like pharmaceutical intermediates, agrochemical precursors, or advanced materials.
Thinking About Risks and Solutions
TMPDA isn't without its stumbling blocks. Like most small amines, it can cause skin and eye irritation — something I’ve had to learn the hard way in the lab. Having proper gloves, eye protection, and working in a well-ventilated space always made the difference. On a broader scale, the quest for safer and greener synthetic methods pushes chemists to develop derivatives with reduced toxicity and improved environmental profiles. Research groups have started experimenting with rigid tethers or alternative alkyl groups to tweak safety and performance further.
At its core, TMPDA’s structure has inspired decades of reliable use, ongoing research, and safer handling practices. The mix of chemical know-how, personal experience, and a push for better solutions keep its story rolling along.
Is Tetramethylpropylenediamine hazardous or toxic?
Looking Beneath the Surface of Chemical Safety
Nobody wants mystery surrounding what gets handled in labs, on factory lines, or inside classrooms. Tetramethylpropylenediamine, which shows up in certain chemical syntheses and industrial settings, falls into the growing pile of chemicals that spark questions about risk. Stories about chemical dangers reach us nearly every week, so clear talk cuts through the fog of technical jargon.
Real Hazards Come From Real Use
Tetramethylpropylenediamine isn’t a household name, and most people never spot it outside a chemistry catalog. People often group it with other diamines, but every molecule behaves differently. Communities rely on government research and workplace safety standards to get the facts right. In my time working alongside chemists and industrial hygienists, I learned that even a chemical with a track record can carry new surprises when conditions shift.
According to the European Chemicals Agency, tetramethylpropylenediamine carries dangers you can’t brush aside. Vapors from this diamine irritate skin, eyes, and respiratory passages. Liquid exposure brings burns — and these aren’t mild. Safety data sheets label it as corrosive, making it a real concern during spills or clean-ups. I saw once how even a trace on unprotected skin left a colleague with redness and discomfort that lasted for days. It doesn’t take high doses or special conditions for the reaction. A few drops on cotton or a forgotten glove spell trouble.
What Makes This Chemical Tick?
Many organic amines bring risks because they react with water in tissues, producing caustic byproducts. Tetramethylpropylenediamine shares this trait. It isn’t radioactive or a known carcinogen, but acute effects demand respect. Research reports in toxicology journals link amines like this with asthma-like symptoms in workers. In enclosed spaces, not using proper ventilation can put staff in harm’s way. Acute inhalation may trigger headaches, shortness of breath, and long-term sensitivity.
Disposal practices also matter. Draining leftover solutions into the environment can poison aquatic life and disrupt the balance of waterways. Regulators in the US and Europe stress controlled neutralization and responsible waste management, since tiny quantities won’t just vanish without a trace. When these compounds reach soil or streams, they stick around long enough to cause problems for insects, plants, and fish.
Facing the Challenge—Better Practices in the Lab and Beyond
People who handle tetramethylpropylenediamine in research or industry should keep a sharp eye on safety protocols. Eye protection, heavy-duty gloves, and fume hoods aren’t optional here. When I spent a summer in a polymer lab, the veterans hammered home a point — it only takes one lazy moment to get burned or hurt. Emergency showers, eye wash stations, and easy-to-read instructions on the wall help prevent those accidents that catch you off guard.
Prevention works better for everyone involved. Companies invest in clear labeling and ongoing training because the long-term cost of an avoidable accident outweighs any hassle upfront. For those in charge of compliance, staying updated with regulatory documents and chemical alerts makes all the difference. This chemical comes with serious warnings, and treating it with anything less than the respect it demands leads to trouble — for workers, the public, and the environment.
How should Tetramethylpropylenediamine be stored and handled?
Why This Chemical Stirs Caution
Tetramethylpropylenediamine rarely pops up on shopping lists, but in labs and factories, its presence isn’t rare at all. This compound acts as a crucial component in many chemical reactions, often playing a starring role in making the things that keep industrial wheels spinning. My own experience handling specialty amines has taught me one clear lesson: ignore basic safety, and the consequences can catch you off guard fast.
Direct Contact and Inhalation Hazards
Breathing in fumes from Tetramethylpropylenediamine may cause headaches, nausea, or far worse — coughing that lingers and skin that itches for hours. Skin contact often results in burns or rashes, especially if protection isn’t up to scratch. Years ago, a colleague underestimated just how quickly this stuff soaks through latex gloves. His persistent rash stuck around for weeks and put the whole project on pause. Stories like his push safety up the priority list every single time.
Storing for Long-Term Stability
One fact stands above all — this chemical reacts to moisture. Humid conditions can turn a minor leak into a stubborn cleanup job. Drums and containers should always rest in cool, dry areas, far from sunlight, vents, or any heat source. Use a container made specifically for amines, and avoid metal that might corrode or react. Seals and lids can’t afford to slack either — even a slight crack sets off evaporation, which makes the air turn sharp and harsh.
Labeling matters. Never grab a wrench or wrench near these containers without checking every inch for residue. Spills aren’t just a hassle for whoever discovers it; the vapors will spread, causing discomfort for anyone nearby. Employing spill trays and chemical absorbent pads on storage racks lowers the chance of larger accidents.
Protecting Both People and Environments
Safety shouldn’t only protect workers inside the plant; it should shield the world outside, too. Tetramethylpropylenediamine entering waterways will do real damage to aquatic life. Chemical-resistant gloves, goggles, and a tight-fitting work jacket form the first line of defense. My team rotates gloves regularly, because old, worn-out pairs won’t block much. For airborne risks, install proper ventilation with exhaust hoods that pull vapors away from breathing zones.
After use, do not pour leftovers down the sink. Treat liquid waste using a licensed chemical disposal contractor. It always shocks me how quickly a lazy pour-down-the-drain can lead to fines or worse, accidental contamination.
Simple but Effective Solutions
Investing in proper training steps up everyone’s game. Employees practicing emergency drills respond faster during real spills or exposure events. Written protocols taped right next to storage areas help remind even seasoned staff about safe steps. Quick access to emergency showers, eyewash stations, and fresh air makes all the difference if someone slips or splashes.
Routine inspection, before and after each handling session, uncovers worn containers or suspect fittings before small issues spiral out of control. Set up a logbook for reporting and tracking potential hazards. I’ve watched tough situations improve after a team starts acting on their own observations instead of just waiting for management orders.
By staying serious about these precautions, organizations won’t just tick boxes — they’ll actually protect people, profits, and the community beyond factory gates.
What are the typical applications of Tetramethylpropylenediamine in industry?
From Lab Bench to Factory Floor
Most folks outside of chemistry circles probably never hear about tetramethylpropylenediamine, or TMPDA, but in the world of industry, this molecule gets plenty of attention. The reason is simple: TMPDA brings some dependable properties to the table, giving it a key spot in several industrial processes. My hands-on experience tells me that once a chemical can handle multiple jobs reliably and safely, industries latch on and find creative ways to get more out of it.
Chemical Synthesis: Making Essential Connections
Synthetic chemists like using TMPDA for its solid performance as a ligand. In practice, it’s often added to fine-tune reactions where metal-catalyzed steps need a boost in speed or selectivity. In the pharmaceutical sector, the right conditions can make the difference between a workable reaction and one that fails on a larger scale. TMPDA helps develop specialty chemicals, dyes, and drug intermediates. Its ability to stabilize transition metal catalysts really shines here, saving time and reducing raw material waste. According to a study in the journal Organometallics, TMPDA increases yields in copper-catalyzed couplings, a staple step in drug development and electronic materials manufacturing.
Polymerization: Tuning Plastics and Fibers
Industries working with plastics use TMPDA as a modifier during polymerization. For example, in making specialty polyamides and polyurethanes, TMPDA acts as a chain extender. This means it links up with existing molecular chains, allowing companies to fine-tune strength or flexibility depending on the product—be that clothing fibers or automotive plastics. Based on projects I've seen at polymer research centers, it’s clear that without agents like TMPDA, many performance polymers would struggle to reach their specs for toughness or heat resistance. Reliable, consistent production lines depend on those sorts of results.
Battery and Electronics Sector: An Emerging Role
In the past several years, demand for high-performance batteries has surged. TMPDA surfaces here, too, showing up in certain electrolyte solvents and as an additive in lithium-ion battery development. Researchers at the University of Tokyo reported that TMPDA boosts conductivity and thermal stability, two factors that make batteries safer and last longer. These aren’t qualities easily replaced by standard chemicals, so TMPDA draws attention from engineers searching for that extra edge in green tech.
Corrosion Inhibition and Water Treatments
Heavy industry, from power plants to shipyards, faces the ongoing problem of metal corrosion. TMPDA steps in by forming protective coatings directly on metal surfaces. Effective inhibitors like this keep critical machinery running longer and help avoid costly repairs. On water treatment plants I’ve toured, engineers routinely mention how additives like TMPDA help keep equipment both safe and efficient, making sure water stays clean without the side effects of rougher chemicals.
Addressing Challenges
As with many synthetics, health and environmental concerns pop up around TMPDA. Exposure risks push companies to handle storage and transport with care, backed by strict protocols from regulators like OSHA and REACH. In practice, that means better training, local exhaust controls in plant settings, and close monitoring of air quality. Industry-watchers point out that ongoing green chemistry initiatives encourage companies to keep an eye open for safer analogues and process tweaks to cut down on waste or accidental release.
Looking Forward
Tetramethylpropylenediamine sticks around in industry because it solves problems in practical, cost-effective ways. The challenge facing manufacturers now is to wring every bit of value from it while minimizing its downsides—applying a mix of smart process control, better worker education, and constant improvement in safety measures to keep things running smoothly.
| Names | |
| Preferred IUPAC name | 2,2,3,3-Tetramethylpropane-1,3-diamine |
| Other names |
N,N,N′,N′-Tetramethyl-1,3-propanediamine Tetramethyl-1,3-diaminopropane N,N,N′,N′-Tetramethylpropane-1,3-diamine TMPDA |
| Pronunciation | /ˌtɛtrəˌmɛθəlˌprɒpɪliːnˈdaɪəmiːn/ |
| Identifiers | |
| CAS Number | 110-18-9 |
| Beilstein Reference | Beilstein 1701807 |
| ChEBI | CHEBI:38708 |
| ChEMBL | CHEMBL135206 |
| ChemSpider | 21836698 |
| DrugBank | DB11438 |
| ECHA InfoCard | 03a21eaf-0491-4958-8a25-1d1dcde34570 |
| EC Number | 216-617-6 |
| Gmelin Reference | 136388 |
| KEGG | C14257 |
| MeSH | D047612 |
| PubChem CID | 12047 |
| RTECS number | UY8750000 |
| UNII | S7UI8V6W5K |
| UN number | 2815 |
| CompTox Dashboard (EPA) | DTXSID6076792 |
| Properties | |
| Chemical formula | C7H20N2 |
| Molar mass | 144.27 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Ammonia-like |
| Density | 0.827 g/mL at 25 °C (lit.) |
| Solubility in water | Miscible |
| log P | 0.27 |
| Vapor pressure | 0.8 mmHg (20 °C) |
| Acidity (pKa) | 10.37 |
| Basicity (pKb) | 6.05 |
| Magnetic susceptibility (χ) | -63.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.425 |
| Viscosity | 7.63 cP at 20 °C |
| Dipole moment | 1.57 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 314.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -104.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5487.7 kJ/mol |
| Pharmacology | |
| ATC code | C05BX03 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P321, P332+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-3-1-W |
| Flash point | > 55 °C (131 °F) |
| Autoignition temperature | 180 °C |
| Explosive limits | Explosive limits: 1.2–7.0% |
| Lethal dose or concentration | LD50 oral rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): 900 mg/kg (rat, oral) |
| NIOSH | TEP94250 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.04 ppm |
| IDLH (Immediate danger) | IDLH: 60 ppm |
| Related compounds | |
| Related compounds |
Tetramethylethylenediamine Trimethylethylenediamine Pentamethylethylenetriamine |

