Tetramethylguanidine: More Than a Catalyst in Chemistry’s Toolbox
Tracing the Origins: How Tetramethylguanidine Came to Be
Looking back on the birth of Tetramethylguanidine—or TMG for short—you find the classic story of chemical ingenuity fueled by changing scientific needs. In the mid-20th century, industrial and academic laboratories hunted for versatile organic bases that could unlock new reaction pathways. Chemists first synthesized guanidine-based compounds in the 19th century, but those early versions lacked the punch TMG later delivered. Once TMG entered the scene, its strong basicity and manageable handling compared to older, fussier bases grabbed attention. This wasn’t just another compound on a dusty shelf. By the 1960s and 1970s, TMG took root in organic synthesis labs, where need for consistent, reliable results always pushed the limits.
Peering Into the Bottle: Product Overview, Labeling, and Synonyms
The label on a TMG bottle lists it as N,N,N',N'-Tetramethylguanidine. Some chemists simply call it “TMG,” but others refer to it as 1,1,3,3-Tetramethylguanidine, hinting at its structure. A clear, colorless liquid, TMG carries a sharp, fishy odor, thanks to its nitrogen-rich composition. The label typically warns of high alkalinity, advising users about skin, eye, and respiratory hazards. These labels aren’t just for show; every precaution helps, especially in settings where exposure can happen easily. I always double-check the cap and labeling after every pour, because even minor spills demand respect in the lab. Better to learn from the chemical’s reputation than from accidents.
Physical and Chemical Properties: What Makes TMG Tick
TMG packs a strong chemical punch in a small, easy-to-handle package. High boiling but still quite volatile, it mixes thoroughly in water, alcohols, and other common solvents. It stands out in the lab for tremendous basicity—its pKa in water sits around 13.6, rivaling, and often surpassing, other organic bases like triethylamine or DBU. The methyl substitution not only amps up basic strength but steeply cuts down the propensity for side reactions—something you learn to appreciate as you chase high-yield syntheses without foul-tasting complications. Since TMG doesn’t act as a nucleophile under typical reaction conditions, it allows targeted transformations to proceed without fuss or unintended branches.
How Chemists Make It: Preparation and Key Modifications
Most synthesis routes for TMG start with guanidine or its hydrochloride salt. Reacting this with methylating agents such as dimethyl sulfate or methyl iodide builds the hallmark tetramethyl structure. Industrial setups prefer robust, cost-efficient approaches, often recycling solvents and carefully controlling methylation steps to maintain purity. Over the years, tweaks in procedures have emphasized greener choices, like avoiding highly toxic methylating agents wherever possible. Some researchers have even experimented with catalytic methylation using less hazardous feeds, but these remain rare outside specialized studies. As with other high-strength bases, control in synthesis carries weight, because unfinished or overalkylated byproducts can undermine the very properties the user seeks.
Understanding Reactions and the Drive for Modifications
TMG finds its sweet spot as a non-nucleophilic superbase, ideal for deprotonating weak acids or pushing along tough organic transformations like alkylations, condensations, or esterifications. I’ve used TMG to coax sluggish esters into motion where weaker amines would hang back. Its low nucleophilicity prevents the base from directly attacking or modifying substrates unless specifically invited. In rare instances, beyond simple deprotonation, TMG can assemble into coordination complexes with metals—handy when targeting metal-catalyzed reactions that need a basic environment. Scientific literature documents clever modifications, with chemists attaching bulky or functional side groups to tune solubility or base strength. In the end, TMG’s stability often makes it the de facto choice until something better or more specialized shows up.
Application and Research: Where TMG Earns Its Keep
Academic and industrial labs put TMG to work wherever robust, non-nucleophilic basicity is a must. Classic uses include as a catalyst or reagent in the synthesis of pharmaceuticals, agrochemicals, and advanced polymers. When working with complex, sensitive molecules that dislike contamination, TMG brings enough cleaning power to get the job done without causing collateral damage, a rare balance in the crowded family of organic bases. Research groups searching for new reaction conditions or more sustainable chemistry often test TMG as a benchmark—if their greener alternative can match or surpass its performance, it has a real shot at adoption. Some modern applications reach into asymmetric synthesis and organometallic chemistry, where the purity of the reagent shows up directly in product yields and quality.
Safety, Handling, and Toxicity: The Human Side of Chemistry
TMG draws straight lines between possibility and risk. Like any strong base, skin or eye contact means trouble: irritation, burns, and pain. Its volatility means inhalation belongs on the list of workplace risks. Lab veterans wear goggles, gloves, and sometimes full face shields, treating even small bottles with the kind of care earned through hard experience with caustic reagents. Regulatory standards flag TMG as hazardous: storage calls for tightly sealed containers away from acids and moisture. Toxicity research, though somewhat patchy, indicates that high exposure can cause both acute and chronic harm. Long-term studies still need more data, so treating every encounter with caution makes sense—especially as chronic effects might reveal more risks over time. Inexperienced users can benefit from detailed training and room-wide fume extraction, especially given TMG’s ability to sneak into the air.
Future Prospects and the Drive for Safer, Greener Chemistry
TMG’s importance will not fade anytime soon. It provides a level of utility that every crowded synthetic bench or production line respects. Even as regulatory pressure mounts for safer, more sustainable chemicals, TMG still earns its place due to efficiency and reliability. Yet the search for safer reagents never stops. Green chemistry initiatives push researchers to dial down toxicity, cut waste, and rethink energy use. Future work might see TMG reimagined: new derivatives with milder toxicity, improved biodegradability, or recyclable qualities that keep pace with tough regulations. Companies may turn out process optimizations that recover TMG without environmental fallout. Persistent curiosity and careful stewardship—backed by robust toxicity research and incremental regulatory guidance—will shape its evolution. For chemists who balance innovation and responsibility, TMG stands as both a tool and a reminder of the wider responsibilities inherent in chemical science.
What is Tetramethylguanidine used for?
Making Chemical Reactions Happen
Tetramethylguanidine (TMG) stands out in the world of chemistry thanks to one reason: its base strength. Many people may never cross paths with this chemical, but in the world of labs and industrial synthesis, TMG can make or break a reaction. Most chemical processes need a push, and TMG provides exactly that. Its strong basicity allows chemists to spur on various reactions, especially those that standard lab bases just cannot handle. Over the years, its unique profile has found a solid home in the synthesis of pharmaceuticals, agrochemicals, and fine chemicals.
Driving Medicine Creation
Most folks take for granted that a pill exists for every ailment. I’ve watched organic chemists sweat over molecules all night, knowing the work isn’t just about mixing powders but about life and health. TMG often shows up at that workbench. Its base strength lets it pull protons away from molecules, setting off chain reactions. By using TMG, researchers build complex drugs from very simple starting materials. This ability to work with fragile, sensitive molecules opens doors to treatments and cures that wouldn’t exist otherwise.
Cleaner, More Efficient Manufacturing
The industrial sector leans on TMG, not only for its power but also for its efficiency. Many reactions with weaker bases take more energy, more time, and give more byproducts—a real problem if you care about both cost and the environment. Strong bases like TMG cut down on side products and turn out the chemical you want, pure and simple. Cleaner chemistry produces safer workspaces, lowers waste, and makes it possible for manufacturing to keep up with strict modern regulations.
Helping Break Down Barriers in Research
New ideas in chemistry catch fire because of tools that work reliably. TMG is a trusted ally for researchers across synthetic and medicinal chemistry. When experimenting with hard-to-handle compounds, many scientists grab TMG because it does its job without fuss. Its ability to work under milder temperatures makes a real difference. Researchers don’t have to wrestle with harsh conditions and unstable intermediates. This economic use of resources helps academic groups stretch tight budgets further and makes discoveries flow faster.
The Dark Side: Safety and Environmental Concerns
No tool is perfect. TMG brings downsides, too. Inhaling or touching this compound isn’t without risk. Like many strong bases, it burns on contact and can cause lasting harm. Strict handling procedures and good training protect workers and students alike. After reactions finish, careful waste management avoids polluting water and soil. Current practices include neutralizing spills and collecting residues for proper disposal, helping to meet both legal rules and the higher bar of social responsibility.
Looking Ahead: Safer and Smarter Chemistry
The chemistry world keeps searching for even greener solutions. Alternatives with less risk and waste catch attention, but TMG sets a tough benchmark. Newer bases might rise to the challenge or perhaps improved safety equipment will let users keep reaping benefits with fewer risks. Funding for research always helps, and tighter rules from regulators push everyone to stay sharp. Chemists sharing ideas, labs working together, and manufacturers supporting better training all form part of a blueprint for safer chemical use.
Is Tetramethylguanidine hazardous or toxic?
The Straight Facts
Tetramethylguanidine, often called TMG, turns up in chemical labs, manufacturing lines, and specialty syntheses across the globe. Chemists appreciate how it boosts reactions in making medicines and advanced materials. Most people rarely hear its name, but those who work with it can’t afford to ignore its warning labels. TMG isn’t exactly something you want splashing on bare skin or leaking around the workplace.
Health Hazards Hit Home
Anyone who’s spent time in a lab knows the unsettling smell that hints at more than just a mild irritant. TMG gives off fumes you remember: peppery, almost stinging. It doesn’t just irritate the nose and throat. Studies from the National Library of Medicine show that inhaling its vapors causes coughing, dizziness, and headaches. Direct contact means chemical burns, especially on eyes, skin, or the lining of your mouth. One bit of research from Germany’s Federal Institute for Occupational Safety and Health points out that concentrated TMG can damage mucous membranes and trigger allergic reactions after repeat exposure.
Lesser-known risks include possible liver or kidney stress for workers in frequent contact. I remember colleagues who reported odd, persistent rashes after a week handling it with torn gloves. Disposal blunders can also spell trouble outside the lab; TMG doesn’t break down easily in water, so spills can hit downstream systems, affecting aquatic life.
Data Over Decades
Despite these serious concerns, TMG doesn’t rank alongside notorious toxins like mercury, lead, or benzene. Acute poisoning proves pretty rare with proper handling, and no evidence shows it causes cancer in humans, according to the International Agency for Research on Cancer. Still, that’s no green light to get careless. The American Conference of Governmental Industrial Hygienists assigns low exposure limits—less than 1 part per million in air—because even small amounts can cause trouble.
The Safety Gaps
I once saw a rookie intern ignore the safety briefing, assuming common gloves would block anything. At the end of the shift, she complained about her fingers tingling and red blotches. TMG goes straight through many types of latex—nitrile or neoprene offer better barriers. Lab fume hoods cut the immediate risk, but leaky bottles and careless wipes track TMG where it shouldn’t go. Chemical suppliers often ship TMG in tightly sealed glass bottles, and for good reason. An open bottle can turn a safe workspace into a problem in minutes, with vapors slowly filling closed rooms.
Emergency rooms and poison control centers carry specific procedures for exposures. Rapid rinsing, eye washes, and medical evaluation stand as the front lines. I’ve learned it’s not enough to trust just one method of control. Combining personal protective equipment, ventilation, clear training, and chemical-resistant storage creates a much stronger defense against accidents.
Building Solutions
Better labeling, more visible data sheets, and hands-on safety training push back against complacency. Supervisors must set an example, stopping work when safety routines break down. Shifting away from TMG toward safer, less volatile chemicals can work for some reactions, though many chemists still prefer its results. Finding green alternatives or designing processes that use smaller amounts limit overall exposure. Regular monitoring and spill drills catch small mistakes before they spiral into real emergencies.
Tetramethylguanidine demands respect and sound judgment in every step, from storage to cleanup. It seldom causes catastrophe among trained workers, but its hazards remain real and constant. Ignoring the risks means inviting problems that affect not just chemistry but personal health and water safety beyond the lab walls.
What are the storage conditions for Tetramethylguanidine?
Why Storage Choices Matter
Dealing with chemicals such as Tetramethylguanidine means showing real respect for their properties—anyone with time in a laboratory or an industrial setting knows this. Storing something like Tetramethylguanidine doesn’t just protect the material; it prevents nasty surprises. This base reacts fast and strong with acids and moisture in the air. If you’ve ever cracked the lid on a bottle only to get hit with a sharp, amine-like vapour, you know how volatile it can be if left unchecked.
Specific Precautions for Tetramethylguanidine
Keep bottles in a cool, dry area—heat ramps up volatility, and humidity pulls water into the mix. Lab setups that cut corners and store this compound under a sink or near water lines are a recipe for incidents. A locked chemical closet, separated from acids, oxidizers, and sources of water, makes for a much safer bet. I’ve seen facilities lose thousands of dollars’ worth of inventory to simple dampness getting in through a cracked cap or loose seal.
Tetramethylguanidine will break down, especially if light gets in. Shielding it from sunlight slows that process. Amber glass bottles do the trick. It also pays to check that caps have working seals—sometimes cheap screw tops let air creep in, which accelerates hydrolysis and contamination of the chemical.
Safety Gear Isn’t Optional
Hands-on work with this chemical calls for eye protection and gloves. Once, a new colleague thought skipping gloves for “just a minute” was no big deal. That small lapse led to skin irritation, and they had no one to blame but themselves. If you’ve ever spilled Tetramethylguanidine, you know mopping up without gloves isn’t worth the risk. Spills also remind us of another important point: keep chemical absorbent nearby in every storage area.
Monitoring for Problems
Inspections catch problems early. I’ve helped spot bottles with crusty threads or weird odours before anything serious happened. Label every container with the date it’s received and last checked—no guessing how long a bottle might’ve sat hidden at the back of the shelf. Rotating stock and using older material first is more than best practice; it keeps things tidy and safe.
Improving Chemical Management
Practical steps go a long way. Staff training that doesn’t just check regulatory boxes helps everyone feel confident in what they’re doing. It’s far from rare to find newcomers surprised by how much time goes into cleaning up storage spaces and keeping logs. That detail-driven approach keeps everyone healthy.
Researchers can also use chemical inventory software to flag old stock, remind about expiration dates, and link safety data sheets. Tight organization keeps the pressure down during inspections and makes emergency cleanups smoother.
Final Thoughts on Responsibility
Tetramethylguanidine isn’t forgiving. Storage conditions matter. Following these habits by choice, not just mandate, keeps everyone safer and protected from both health risks and costly mistakes. A little vigilance and respect for detail pays off every time.
What is the molecular formula of Tetramethylguanidine?
Looking Beyond the Letters: What Tetramethylguanidine Means
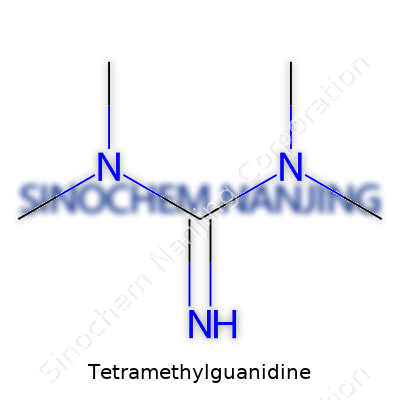
Tetramethylguanidine, short-handed in labs as TMG, comes with a straightforward molecular formula: C5H13N3. The numbers and letters point to a molecule built from five carbons, thirteen hydrogens, and three nitrogens. On paper, it looks simple. In practice, the impact stretches much further.
The Backbone of Modern Chemistry Labs
Not all molecules get to shake up a lab's protocol, but TMG earns its keep. TMG serves as a strong, non-nucleophilic base, and sometimes it stands as one of the most cost-effective ways to drive certain reactions. Organic chemists, especially those synthesizing complex rings and chains, grab this molecule to make weak acids give up their protons. I still remember the first time I uncapped a bottle in my grad school days. Just a whiff tells you this chemical demands respect—sharp, pungent, sticks in your memory like ammonia. Good ventilation and gloves weren’t optional.
Not Just for Academics—Industry Cares Too
TMG helps pharma companies cut down on by-products. Cleaner reactions mean fewer headaches during purification, faster development, and lower production costs. This matters most when companies try to bring a drug to market quickly. The clock never stops for medicinal chemistry—using TMG, with its crisp molecular punch, can save a week in a busy synthesis campaign. Speed here can turn into a real advantage for patients waiting on the next breakthrough therapy.
Tall Claims Need Real Safety
TMG has a double-edged personality. That molecular formula makes it efficient, but user mistakes can bring trouble. It burns the skin, stings if it hits your eyes, and it vaporizes more easily than one expects. Chemical accidents often boil down to overconfidence or cutting corners. Science needs more than good formulas; it needs stubborn respect for risks.
Cleaner Reactions, Cleaner Earth?
Many chemists call for “green” chemical methods. Traditional strong bases often come with waste streams that strain sewage systems and final disposal. TMG, because of its efficiency, often lets chemists work with lower volumes, helping to cut down on some of the harsher side-products. Still, manufacturers can work to improve recycling systems, reducing the environmental load even more. A closed-loop recovery process for solvents and bases offers promise. The best labs invest not just in cleaner chemicals but in better ways to reclaim them.
The Lesson of the Formula
C5H13N3 only tells part of the story. The advantages, risks, and responsibilities go far beyond the mere sum of atoms. TMG’s practicality in the chemical world has shaped new methods, delivered new compounds, and shaped expectations for efficiency and safety. As a scientist, watching this molecule in action reminds me that behind every formula, people stand—curious, cautious, and, ideally, committed to making every reaction count for something bigger than a simple yield.
How should Tetramethylguanidine be handled safely in the laboratory?
Why Tetramethylguanidine Demands Respect
Tetramethylguanidine (TMG) shows up pretty often in organic labs. It’s a powerful base and that comes with risks. Anyone who has uncapped a bottle knows its vapor hits you fast, burning the nose and eyes. TMG absorbs through the skin, so touching a droplet turns into a real hazard. Respiratory exposure lands you with serious irritation or worse. I’ve worked with it during late-night synthesis, and the urge to rush without proper gear almost always leads to regret. Every experienced chemist has stories about shortcutting safety, sometimes ending with a chemical burn or frantic trip to the eyewash station.
The Gown, Shield, and Air: Setting Up for Safety
Every TMG project starts before popping open the vial. A fitted lab coat and safety goggles never feel like enough, so add a splash-proof face shield if there’s a chance of vigorous mixing or transfer. Nitrile gloves offer some protection, but double-layering them keeps hands safer, since TMG chews through glove material. Changing gloves often, especially after a splash, avoids painful surprises. Controlling exposure starts with the work zone. Even a slight whiff signals poor ventilation—so move to a certified fume hood, pull the sash low, and avoid leaning inside. Fume hoods aren’t only for the worst cases; plenty of routine TMG steps release enough vapor to demand extra airflow.
Smart Moves with Storage and Handling
I’ve seen bottles of TMG stuffed on overcrowded shelves or left open on benches. Spill risk climbs with this kind of sloppiness. TMG belongs in a tightly sealed container, kept away from acids, oxidizers, and anything reactive, since it can trigger nasty reactions. A cool, dry spot in a corrosives cabinet works best. Always label the container with both name and hazard classification. This avoids mix-ups—because nothing derails experiments like cleaning up the wrong chemical.
Mixing, Transferring, and the Devil in the Details
Transferring TMG with syringes adds a new layer of risk, especially since static electric sparks or simple mistakes lead to dangerous spills. Use dry glassware, since TMG reacts badly with moisture. A slow, steady hand prevents splashes. After working, wipe surfaces with the right solvent and dispose of any soaked towels in dedicated waste jars. Some labs set up spill kits nearby, stocked with absorbents and neutralizers designed for caustic materials. Even the quick jobs deserve spill preparedness.
Disposal and Cleaning That Don’t Cut Corners
Lab veterans know disposal rules shift from campus to campus, but dumping TMG down the drain causes real headaches for everyone downstream. Used TMG belongs in a labeled hazardous waste container, destined for professional collection and treatment. Cleaning up calls for patience—a common sense flush isn’t enough. All surfaces and tools need a solvent wash. Even the air needs purging before leaving the lab for lunch.
Training, Reminders, and Respect for the Unknown
No single rulebook covers every accident. Training sessions and refreshers build muscle memory for emergencies. A printed chart on the wall helps avoid panic. New students need walk-throughs before their first TMG task; peer mentoring picks up details handbooks miss—like how a tiny drop rolling down gloved fingers stings for hours if ignored. Regular check-ins with lab partners keep everyone thinking ahead, not just reacting.
Culture Shapes Lab Safety
TMG is unforgiving, pushing everyone to stay sharp. Building a lab culture that prizes caution over speed leads to finished projects and uninjured hands. Real safety means watching each other, sharing “what went wrong” stories, updating protocols, and reporting close calls. Having spent years sharing benches, I’ve learned that you never get used to the hazards, but you can grow a habit of always double-checking before reaching for that bottle.
| Names | |
| Preferred IUPAC name | 2-tert-Butyl-1,1,3,3-tetramethylguanidine |
| Other names |
1,1,3,3-Tetramethylguanidine TMG |
| Pronunciation | /ˌtɛ.trəˌmɛθ.əl.ɡwəˈnɪːdɪn/ |
| Identifiers | |
| CAS Number | 107-47-1 |
| Beilstein Reference | 1634307 |
| ChEBI | CHEBI:39075 |
| ChEMBL | CHEMBL1542 |
| ChemSpider | 10988 |
| DrugBank | DB04209 |
| ECHA InfoCard | 03a9e962-9a2e-43d7-b672-a70b8a508d73 |
| EC Number | 204-698-2 |
| Gmelin Reference | 84123 |
| KEGG | C01874 |
| MeSH | D017964 |
| PubChem CID | 11261 |
| RTECS number | KV5250000 |
| UNII | N8APG8I8BQ |
| UN number | UN3272 |
| Properties | |
| Chemical formula | C5H13N3 |
| Molar mass | 115.19 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Ammonia-like |
| Density | 0.926 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | -0.22 |
| Vapor pressure | 0.5 mmHg (20°C) |
| Acidity (pKa) | 13.6 |
| Basicity (pKb) | 4.2 |
| Magnetic susceptibility (χ) | -33.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.458 |
| Viscosity | 1.08 cP (20°C) |
| Dipole moment | 2.19 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -30.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3406 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H302, H314 |
| Precautionary statements | P280-P261-P303+P361+P353-P304+P340-P305+P351+P338-P310 |
| NFPA 704 (fire diamond) | 3-1-2-A |
| Flash point | -6 °C |
| Autoignition temperature | 430 °C |
| Explosive limits | Explosive limits: 1.5–9.7% |
| Lethal dose or concentration | LD50 oral rat 190 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 56 mg/kg |
| NIOSH | JEM |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Tetramethylguanidine: "Not established |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
1,1,3,3-Tetramethylthiourea 1,1,3,3-Tetramethylurea Formamidine |

