Tetraethylenepentamine: Chemistry Shaped by Industry and Curiosity
Historical Development
Tetraethylenepentamine, known in workplaces and lecture halls as TEPA, has a backstory rooted in industry’s push for new materials and better processes. During the mid-20th century, chemical manufacturers started noticing patterns—certain amine compounds showed big promise in cleaning up refinery streams and boosting resins for coatings and paints. TEPA arrived as one of those multi-functional molecules, not by sudden invention, but through steady chemical evolution. As refining, adhesives, and polymer industries grew, research teams built on the earlier generations of ethylenediamines and triethylenetetramine, unlocking TEPA’s balance of flexibility and reactivity. The molecule filtered into markets that demanded both complexity and versatility, threading its way beyond labs and into plants across North America, Europe, and eventually Asia.
Product Overview
TEPA offers more than a long, tongue-twisting name. From my own work in industrial settings, it became clear early on that this amine wasn’t just another chemical drifting through supply pipelines. Companies use TEPA in epoxy curing, lubricants, water treatment, and textile processing, counting on its ability to bond or react in just the right places. It often comes as a colorless to yellowish liquid, drawing little attention in the drum but driving big changes on the production line. Chemists and plant operators frequently talk about its “chelate” strength when binding metals or its knack for unlocking performance in adhesives—sometimes a single additive makes or breaks a product. TEPA stands among those few.
Physical & Chemical Properties
Looking at TEPA’s physical and chemical traits brings up a few memorable lab sessions. This liquid’s boiling point sits well above typical room temperature, so process engineers rely on heated systems to pump or distill it. It smells sharply ammoniacal, never letting you forget you’re working with an amine. TEPA dissolves easily in water and many organic solvents, but it also reacts strongly with acids, carbon dioxide, and certain plastics—reminding handlers to approach it with a plan, not just gloves and goggles. With five nitrogen atoms stitched along a flexible chain, TEPA delivers both reactivity and adaptability. Its viscosity can make bulk handling a chore, but that same property gives it an edge in some slow-curing applications.
Technical Specifications & Labeling
Technical specs rarely get the spotlight, but they steer entire projects from start to finish. Standards organizations like ASTM set out purity levels and maximum limits for connected amines, while packaging labels feature clear warnings about its corrosiveness and strong odor. Labels spell out that direct skin or eye contact causes irritation or burns; splash risk isn’t just a footnote. Manufacturers stick to clear drums or containers, often color-coded, since mixing up amines can spoil a batch or even trigger hazardous reactions. In shipping, TEPA counts as a hazardous substance under international regulations, so every crate and tank needs paperwork and clear hazard icons. Reading about accidents from improper labeling or untrained handling underlines the value of those clear standards—every detail aims to prevent injury and keep operations flowing.
Preparation Method
TEPA doesn’t leap straight out of a chemical reactor. Instead, its preparation feeds off bigger, busier plants making ethylenediamine by reacting ethanolamine or ammonia with ethylene dichloride. By pushing those reactions further and adjusting separation columns, operators harvest TEPA alongside smaller ethyleneamines. Column design and temperature control shape the output: too hot and you waste energy, too cool and yields suffer. I’ve seen trial runs where small changes in catalyst type or pressure tipped the product mix, showing that TEPA isn’t just a side product, but sometimes a targeted output when market needs shift. Efficient production methods save energy and reduce hazardous by-products—getting TEPA out with minimal waste remains a point of pride for forward-thinking operators.
Chemical Reactions & Modifications
My own experience with TEPA in the lab showed its double life: eager to bond with acids, metals, or epoxides, but also able to undergo modification. In resin systems, TEPA teams up with epoxy monomers, forming tough, stable polymers after slow, steady curing. This flexibility means there’s constant tinkering—scientists try partial alkylation for new surfactants or react TEPA with fatty acids for corrosion inhibitors. Its ability to form chelates with metals makes it useful in water treatment, grabbing hold of heavy metal ions for removal. Each reaction brings its risks; controlling temperature and stoichiometry avoids unwanted by-products and maintains efficiency, reminding all involved that even “simple” amines carry surprises.
Synonyms & Product Names
Anyone working in purchasing or R&D stumbles over the tangled web of chemical names. TEPA goes by a roster of synonyms like N,N’-bis(2-aminoethyl)ethane-1,2-diamine and others, depending on the naming convention. You’ll spot it in catalogs as “polyamine,” “polyethylene pentamine,” even as a blend in some industrial amine mixes. This name jumble can trip up newcomers—ordering the wrong grade or misunderstanding a formula costs both time and money. Experienced buyers stick to CAS numbers and lean on trusted suppliers to keep things straight. Whenever projects stall over a shipment mix-up or language gap, these naming issues jump to the top of the lesson list.
Safety & Operational Standards
Any amine deserves respect, and TEPA’s no different. In several facilities I’ve walked through, specific handling rules cover everything from splash barriers to closed transfer lines. The MSDS drills home that skin contact causes burns and that vapor inhalation brings coughing or worse. Workers know to suit up with chemical-resistant gloves, goggles, and face shields, setting up emergency showers and eye wash stations nearby. Teams look for leaks or spills around pumps and valves, since a little TEPA goes a long way in spoiling a shift. Ventilation and air monitoring back up PPE, since poor airflow lets vapors build up quickly. Training remains constant—new hires and old hands alike get reminders and refreshers as standards and equipment improve. Observing how quickly accidents happen in too-hasty setups drove home the point: standards save lives, not just liability headaches.
Application Area
TEPA’s reach spans everything from water treatment to paints and resins. Epoxy resin curing has become one of its strongest suits, with TEPA enabling coatings to harden at room temperature into films that stand up to heavy wear and chemical attack. In lubricants, its nitrogen structure helps fend off engine corrosion or stabilize oil compositions under stress. In water treatment, TEPA-based chelating agents help snatch up heavy metals like copper or lead from industrial streams. Textile firms use it in dye fixing, improving colorfastness and lowering defects—something I saw firsthand on the production lines. Agrochemical industries spin TEPA into new adjuvants and plant-protection mixes. Each use rests on TEPA’s blend of reactivity and chain length, with manufacturers adjusting formulations to hit performance targets or regulatory thresholds.
Research & Development
Research into TEPA never sits still. In industrial chemistry forums and at technical conferences, teams share work on novel derivatizations or new fields for TEPA blends. Green chemistry keeps growing in focus, with researchers hunting for ways to synthesize TEPA more efficiently or use safer feedstocks. I’ve followed publications exploring its use in biomedical fields, such as carrier materials or gene transfection agents, though toxicity limits broad adoption. Some groups mix TEPA with silica or nano-materials, hoping for improved catalysts or adsorbents for environmental cleanups. Grant programs and private start-ups look for breakthroughs that stretch TEPA’s abilities or shrink its environmental cost—curiosity keeps feeding new cycles of experimentation.
Toxicity Research
TEPA’s double-edged performance comes with toxicity risks that aren’t swept under the rug. Studies show that direct contact with TEPA burns skin and eyes, while repeated vapor exposure harms lungs or causes headaches. Animal studies alert regulators that chronic exposure brings organ stress, making workplace controls as vital as gloves. The demand for greener, safer chemicals has kept industry and regulators working on smarter labels and better exposure limits. Recent years brought more research into TEPA’s fate in wastewater or soil, underlining the push for treatment or containment before discharge. Each report and lab test feeds into risk assessments, shaping policy and driving equipment upgrades or process tweaks.
Future Prospects
Looking ahead, TEPA’s future hinges on how demand tracks with regulation and innovation. Epoxy and water-treatment sectors continue to pull for reliable supplies, yet calls for environmentally benign amines are getting louder every year. Synthetic biology promises alternative feedstocks; process intensification stands to shrink waste and energy use. Researchers aim to unlock new uses, from smart materials to more selective chelating agents tuned for remediation in mining or clean water. Growth in bio-based and recyclable chemicals holds opportunity and challenge—TEPA might evolve or get replaced in formulas, but its impact on industry isn’t fading soon. From factory floor to R&D bench, TEPA keeps chemists on their toes, ready for the next round of change.
What is Tetraethylenepentamine used for?
What Is Tetraethylenepentamine
Few folks walk around talking about tetraethylenepentamine in day-to-day conversation, but that doesn’t mean it isn’t a big deal. This chemical, better known as TEPA, shows up across a bunch of industries. If you’ve ever used a cleaning product, worked in a factory that produces plastics, or even marveled at the smooth surface on metal tools, there’s a good chance TEPA played a role somewhere along the line.
Why Do Companies Use It?
TEPA acts as a backbone for making chemicals known as amines. This may sound a little technical, but the truth is, almost every field that deals with manufacturing and processing leans on these amines for something important. TEPA helps oil companies process crude oil better by neutralizing acids during refining. Without it, the process slows down and machines get damaged more quickly.
TEPA does more heavy lifting in the coatings business, too. It reacts well with epoxy resins, which are used to make coatings for floors, pipes, and even wind turbines. Folks who work in construction depend on tough, long-lasting coatings to protect steel from rust, and TEPA helps make those coatings bond and cure just right.
Everyday Products Have Ties to TEPA
Most people might not see the link, but TEPA helps bring us better detergents and cleaners. Its strong cleaning and binding abilities give household formulas extra boost. Factories use it during wastewater treatment. TEPA acts as a binder, helping trap impurities that could otherwise end up in our rivers and streams. Clean water matters to everyone–from kids playing in parks to farmers irrigating crops.
Safety Matters With TEPA
This chemical isn’t for the faint of heart. TEPA has a strong smell and can cause skin irritation or breathing problems if someone handles it carelessly. Folks in plants learn quickly to use gloves and masks when dealing with it. The Environmental Protection Agency and OSHA both set guidelines to keep workers out of harm's way, but mistakes do happen. Better ventilation and regular safety drills go a long way. There’s plenty of research into substitutes that can reduce risks, though right now, many businesses still find TEPA tough to beat for certain jobs.
Finding Greener Alternatives
Years ago, not many people cared about where industrial chemicals ended up, just as long as nothing exploded. Now, consumer demand for safer and more sustainable products grows each year. Companies pour money into research to find greener additives and safer substitutes for chemicals like TEPA. Some labs experiment with blending bio-based amines or engineering alternative epoxy hardeners, but each option brings its own challenges with cost and performance.
Moving Toward Safer Solutions
The push to keep workplaces healthy and protect the environment has changed the game for chemicals like TEPA. Industry leaders partner with universities and government groups to reduce exposure and waste. Open communication about risks helps everyone down the supply chain–from those mixing chemicals to workers applying epoxy–stay informed and safe. TEPA may not be a household name, but the discussions around its responsible use touch all of us in ways big and small.
What are the safety precautions when handling Tetraethylenepentamine?
The Substance and Why It Demands Attention
Tetraethylenepentamine, often called TEP, pops up a lot in industry. You’ll see it in epoxy hardeners, lubricants, and cleaning chemicals. People who work with TEP know it doesn’t mix well with relaxed habits or a lack of preparation. My own time around chemical environments drove home an important lesson: you can’t afford shortcuts when dealing with any chemical with a reputation for causing skin burns and breathing issues.
Why PPE Can't Be Skipped
TEP loves to jump onto your skin or into your eyes if given half a chance. Its sticky, persistent nature makes prevention better than any cure. Gloves made of nitrile or neoprene give solid protection—you feel more confident reaching into a drum or wiping up a spill knowing your hands won’t soak up harmful stuff. I remember working a shift where someone underestimated a tiny leak. He skipped his goggles. One splash later, he spent the afternoon flushing his eye at an eyewash station, lucky to avoid lasting damage.
Eye protection really isn’t optional. Safety glasses with side shields or, better, full goggles, handle the risk. Long sleeves, lab coats, and chemical-resistant aprons shield arms and legs. Fumes don’t stay put, either. Good ventilation stops invisible danger from building up. Running local exhaust or pushing work into fume hoods makes any task safer and keeps your lungs happy.
Handling Spills and Waste with Respect
TEP doesn’t just fade away after a spill. Even a few drops eat into surfaces and hang in the air. Every crew benefits from clear steps: confine the spill, soak it up with approved absorbents, and double-bag it for disposal. Never sweep or dry-wipe, since this raises dust and risk. In my past, a spill across a shared workspace led to confusion. The right absorbent made cleanup simpler, and nobody tried to cut corners twice after that day.
Waste management isn’t about ticking boxes. Improper disposal brings fines, but more importantly, it damages trust with the community and hurts the environment. TEP waste gets isolated and sent off with other hazardous chemicals—marked, locked up, and ready for professionals.
Training, Practice, and Team Awareness
Training can’t just live in a binder on the shelf. Regular updates and hands-on practice give everyone muscle memory for bad situations. I’ve seen fresh staff run through spill drills beside people with years in the field, both groups admitting later they learned something new. Signage, safety sheets taped to the wall, and routine reminders keep risks top-of-mind.
My own respect for routine safety walks changed after watching a seasoned supervisor check storage racks for leaks and out-of-date labels. One missed label nearly led to mixing incompatible chemicals—a mistake no one wanted to repeat. Discussion and double-checks really keep up a safety culture.
Solutions That Make a Difference
Better containers, clearer labeling, and routine replacement of safety gear shift a workplace from guesswork to confidence. Real-time air monitoring and more accessible emergency showers or eyewash stations bring peace of mind. Choosing less hazardous alternatives when possible also lowers risk, but when TEP use can’t be avoided, planning and honest communication help everyone go home without a scratch.
Lasting safety comes from people noticing details and respecting the challenge that TEP brings. Building habits, investing in training, and never ignoring odd smells or unexpected spills shape a safer environment for everyone on the team.
What is the chemical formula and structure of Tetraethylenepentamine?
A Closer Look at Tetraethylenepentamine
Tetraethylenepentamine—often shortened to TEPA—draws little fanfare outside chemistry labs, but its impact spans industries that need tailored molecules for reliable results. Its chemical formula, C8H23N5, hints at a backbone loaded with both carbon and nitrogen, and this formula brings it into play from manufacturing to agriculture. View it physically, and its viscous, clear-to-yellow liquid stirs up a room with a heavy ammonia odor. The actual structure? Imagine a chain of ethylene units, with amine groups checking in every so often. A working chemist visualizes this as NH2–CH2CH2–NH–CH2CH2–NH–CH2CH2–NH–CH2CH2–NH2. These alternating amine and ethylene units set it apart. More branches mean more opportunities for chemical bonding, which translates to value in the real world.
Why Structure Matters
Structure makes all the difference. Tetraethylenepentamine stands as a polyamine, crammed with five amine groups attached through ethylene links. These amine groups make it more flexible and reactive, especially next to its chemical cousins like diethylenetriamine or triethylenetetramine. My own early years in industrial chemistry showed how important it is to pick the right compound for the task. Choose TEPA, and its sprawling structure allows it to latch onto more components than shorter amines. This single decision can stabilize a catalyst or even ease the cleanup of acidic spills in a plant. The hands-on side of industrial chemistry teaches that if you want consistent performance, you don’t skip understanding the fine structure behind the formula.
Everyday Impact and Specific Uses
TEPA may hide behind the scenes, but it finds its way into common products. Resin and epoxy manufacturers rely on it for curing agents—a role that depends on those nitrogen atoms ready to grab on during polymerization. The same properties come into play for water treatment: TEPA neutralizes acids and binds with heavy metals, helping clean contaminated streams or industrial runoffs. The agriculture world looks for TEPA’s help as a chelating agent, grabbing onto micronutrients so crops absorb them more efficiently. I’ve watched colleagues in coatings production underline the importance of using TEPA as an intermediate: its molecular layout helps create stable emulsifiers that keep paints smooth and workable for longer.
Challenges and Responsible Management
Every powerful tool poses a challenge. With TEPA, the concentration of amine groups means high reactivity, sometimes leading to hazardous byproducts if mishandled. Chemists and engineers walk a fine line, measuring conditions closely to avoid runaway reactions. In my experience with chemical plant audits, strict adherence to safety data sheets makes all the difference. Strong odor and skin irritation also underscore the need for protective gear—goggles, gloves, proper ventilation—no shortcuts. Environmental concerns prompt tighter rules on discharge and emissions. Real solutions focus on improved containment, continuous monitoring, and investing in recovery systems. Purposeful recycling and regeneration processes can turn spent amines from waste into reusable resources.
Room for Progress
As demands for safe and sustainable chemicals grow, the way forward means smarter engineering and leaner processes. TEPA’s unique structure delivers benefits, but success depends on responsibility at every stage. Ongoing research explores alternatives with reduced toxicity and innovations in process design that limit byproducts. Industry leaders see gains where careful monitoring and training keep workers safe and reduce environmental burdens. The story behind TEPA reminds us that in chemistry, understanding the nuts and bolts is what keeps progress moving in a way that works for everyone.
How should Tetraethylenepentamine be stored?
Why Safe Handling Matters
Tetraethylenepentamine, or TEPA for short, might sound like it belongs in a chemistry textbook, but it shows up in plenty of real-world settings—from epoxy curing rooms to water treatment plants. In my early years working at a coatings manufacturer, nobody handed me a manual on how to keep chemicals like TEPA out of trouble. I learned from mentors and, more often than not, from close calls. This stuff is no friend to bare skin or careless storage. It gives off strong fumes and causes nasty irritation if you get sloppy.
The Importance of a Cool, Dry Zone
Heat works against safe storage, especially with amines like TEPA. Too warm, and you risk pressure building up in containers or, worse, changes to the chemical itself. Moisture sneaks in and can trigger reactions you do not want to clean up. In our warehouse, we always set aside a ventilated area away from direct sunlight and made sure humidity stayed low. Industrial-grade, climate-controlled cabinets make a lot of sense for this reason. Storing TEPA next to other chemicals creates surprises nobody wants. Incompatible substances, like acids or oxidizers, need plenty of distance—mixing them can lead to fire risks or toxic clouds. Mixing hazard classes is a shortcut to a long day.
Container Choices: Simple, But Critical
TEPA comes in drums or IBCs (large plastic tanks with metal cages), and these need regular checks. A leaky valve or cracked lid can mean exposure or environmental mess. Metal containers with chemical-resistant liners do well, but we always checked that labels hadn’t faded or fallen off. One time, a mismarked barrel led a colleague to attempt an improper transfer. That’s how accidents start. The lesson stayed with me: labeling is simple, but skipping it guarantees confusion.
Ventilation Is Not Optional
Some folks assume screwing a cap on tight solves the issue, but TEPA vapors can seep out slowly and stay around. Fume hoods, exhaust fans, and open shelving—instead of crowded closets—save lungs and keep inspectors happy. I remember our plant made a switch from old closed cabinets to open wire racks in a ventilated corner. The stubborn, lingering odor finally faded, and people stopped getting headaches on their way through the section.
Emergency Prep Counts Every Day
You never see a spill coming. Absorbent pads, chemical-resistant gloves, face shields, and eyewash stations give a fighting chance if things go sideways. After a splash incident, no one argued about the cost of extra safety gear again. Clear walkways and visible safety data sheets cut down panic time when every second counts.
Why Regular Training Sticks
Newcomers need hands-on safety drills, not just pamphlets. Watching a supervisor calmly handle a mock spill drives home the real risks and right moves. Even the most seasoned workers appreciate refreshers—complacency sneaks in faster than you’d think. Our crew gathered for quick check-ins monthly. Hurt pride from safety quiz blunders stings less than a chemical burn.
Looking Ahead With Smart Investments
Companies who take shortcuts with storage usually pay later—through fines, shutdowns, or medical bills. Investing in high-quality, well-ventilated storage, clear labeling, and real training pays itself off quietly over years. It protects the people doing the work, the community nearby, and keeps expensive chemicals where they belong. I’ve learned that good storage isn’t about following rules for the sake of rules. It’s about coming home safe every night, with nothing to explain to your family except how dull your workday was.
What are the potential health hazards of Tetraethylenepentamine exposure?
What Is Tetraethylenepentamine?
Tetraethylenepentamine, sometimes just called TEPA, plays a role in making epoxy hardeners and the plastics found in things like pipes, paint, and flooring. Most people don’t handle it every day, but for those in industrial work, even a little exposure can matter.
Immediate Health Problems
People who have worked with amines like TEPA know the risks don’t always wait. Getting some of this chemical on your skin can leave a person with burns that take weeks to heal. Inhaling even low amounts in poorly ventilated places triggers headaches, dizziness, or a burning sensation in the nose and throat. Anyone who’s cut corners on gloves or safety glasses learns fast: the stinging in eyes or the patches of raw, red skin aren’t worth saving a minute.
Long-Term Hazards
Long stretches around TEPA bring their own challenges. Workers who breathe in too much vapour over months can end up with asthma symptoms, a cough that just won’t quit, or permanent shortness of breath. Breathing chemicals triggers the immune system in odd ways. In my time touring older epoxy factories, veteran workers described tightening chests and itchy skin that only started after years on the job.
Skin allergies become a big struggle, too. TEPA acts as a sensitizer. That means even small amounts over time set off rashes, hives, or welts—sometimes stopping people from ever working around amines again. There’s research linking repeated TEPA contact to more severe conditions, like occupational asthma, which can last a lifetime. In places lacking proper air handling or without break rooms for changing out of dirty coats, workers suffer most.
What Science Tells Us
Tetraethylenepentamine falls under the family of chemicals called polyamines, well-known for causing irritation. Health agencies like the U.S. National Institute for Occupational Safety and Health (NIOSH) recommend limiting how much TEPA anyone can breathe in during a typical work shift. Medical studies consistently show that high exposures spike rates of chemical burns, airway swelling, and chronic dermatitis.
The World Health Organization warns about even more subtle risks. Overexposure links to liver and kidney issues over long years on the job. Though not a proven carcinogen, TEPA still raises red flags in safety circles. Many industrial accidents come down to poor training or equipment failures. I’ve spoken to chemical plant managers who stressed that, without regular training, even veteran workers sometimes forget the basics—like changing clogged filters or double-gloving for spill cleanup.
Real Solutions That Protect People
Companies serious about safety fit every workspace with strong air extraction fans and hand-washing stations. Nobody should have to buy their own gloves or goggles. Training matters most. People learn best from real-world demonstrations, not dry slides. Bringing in folks who've worked with TEPA helps drive home just how fast a mistake can happen.
Workers who move through places where TEPA gets stored should see labels and safety sheets everywhere, not locked in a supervisor's office. Medical checkups do more than catch early signs of damage—they keep health at the center of every shift. Companies willing to rotate jobs, give time away from the most hazardous spots, and offer paid breaks see fewer injuries. Engineers constantly tinker with machinery to reduce leaks and spills, keeping exposure low from the start.
Listening to those closest to the risk—floor managers and seasoned workers—often leads to the best ideas. Simple changes like better locker rooms, easy-clean surfaces, or gloves that actually fit make everyone’s shift safer. Health and safety depend on steps taken every day, not just on paper.
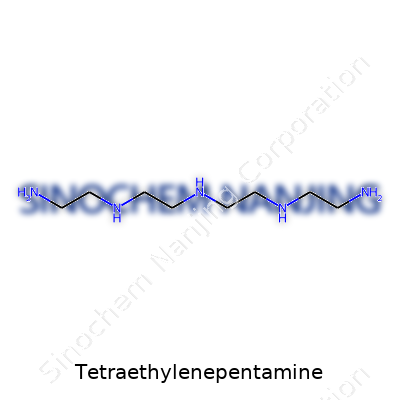
| Names | |
| Preferred IUPAC name | 2,2′,2′′,2′′′-(Ethylenediaminetetra-yl)tetraethan-1-amine |
| Other names |
TEPA Tetraethylene pentamine N-(2-Aminoethyl)ethane-1,2-diamine Pentamine 3,6,9,12-Tetraazatetradecane |
| Pronunciation | /ˌtɛtrəˌɛθaɪliːnˈpɛntəˌæmiːn/ |
| Identifiers | |
| CAS Number | 112-57-2 |
| 3D model (JSmol) | `JSmol` 3D model data (as a string) for Tetraethylenepentamine is: ``` CCNCCNCCNCCN ``` This is the SMILES string representing the molecular structure for use in JSmol and similar molecular viewers. |
| Beilstein Reference | 1710827 |
| ChEBI | CHEBI:39176 |
| ChEMBL | CHEMBL1239815 |
| ChemSpider | 10998 |
| DrugBank | DB01841 |
| ECHA InfoCard | EEEH0193NN |
| EC Number | 203-986-2 |
| Gmelin Reference | 7783 |
| KEGG | C14345 |
| MeSH | D014155 |
| PubChem CID | 15318 |
| RTECS number | TY2000000 |
| UNII | 6F539Q39WV |
| UN number | UN2320 |
| CompTox Dashboard (EPA) | DTXSID9020156 |
| Properties | |
| Chemical formula | C8H23N5 |
| Molar mass | 189.30 g/mol |
| Appearance | yellow liquid |
| Odor | Ammonia-like |
| Density | D=0.988 g/cm3 |
| Solubility in water | miscible |
| log P | -1.02 |
| Vapor pressure | 0.3 mmHg (25°C) |
| Acidity (pKa) | 9.70 |
| Basicity (pKb) | 5.35 |
| Magnetic susceptibility (χ) | -48.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.502 |
| Viscosity | 23 mPa·s (20 °C) |
| Dipole moment | 3.38 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 388.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -267.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4013.7 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause an allergic skin reaction, toxic to aquatic life. |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H314, H317, H319, H332, H412 |
| Precautionary statements | P280, P261, P264, P271, P272, P273, P302+P352, P333+P313, P305+P351+P338, P310, P321, P362+P364, P363, P391, P501 |
| NFPA 704 (fire diamond) | 3-1-0 |
| Flash point | 170 °C (338 °F; 443 K) |
| Autoignition temperature | 285 °C (545 °F; 558 K) |
| Lethal dose or concentration | LD50 oral rat 2100 mg/kg |
| LD50 (median dose) | 2,100 mg/kg (rat, oral) |
| NIOSH | SN4290000 |
| PEL (Permissible) | 5 mg/m3 |
| REL (Recommended) | 1 mg/m3 |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Diethylenetriamine Triethylenetetramine Pentaethylenehexamine |

