Tetrachloroethylene: More Than a Simple Solvent
A Walk Through History
Once upon a time in the late 1800s, chemists began pulling apart coal tar and all sorts of curious residues, hunting for powerful cleaning agents and industrial chemicals. Out of this chemical gold rush came tetrachloroethylene, also called perchloroethylene or “perc,” a chlorinated hydrocarbon. Its early years tied closely to the textile revolution and the growing thirst for stain-free clothes. Perc didn’t just show up in a lab — it arrived because real people, with real stains and dirty overalls, needed a more effective cleaning fluid. It soon became the backbone of commercial dry cleaning, pushing older, flammable solvents to the sidelines. Companies invested faith in this transparent, colorless liquid, letting perc ride the wave from the Industrial Revolution into the modern age of consumer convenience.
What Makes Tetrachloroethylene Tick
Seen in its pure form, tetrachloroethylene doesn’t scream danger; it looks much like water. The catch is in the chemistry. This liquid won’t easily catch fire, packs a heavy molecular punch, and evaporates off fabrics without much fuss. There’s a chlorine scent on the nose — sharp, but not quite as aggressive as plain bleach. Its high density keeps it from mixing with water, separating cleanly into its own slick layer. These characteristics give it that magical ability to whisk away greasy grime, oil, and persistent stains, and this set of properties is why it carved out its place in dry cleaning and industrial degreasing decades ago.
The Making of Perc
In manufacturing halls, the route to tetrachloroethylene begins with hydrocarbons drawn from petroleum. Industrial strategies have changed over the years, with several routes staking their claim: some involve the chlorination of hydrocarbons like ethylene, others rely on further chlorinating trichloroethylene. It takes reactors running hot and efficient, carefully metered chlorine, and skilled hands to monitor each stage. Every batch emerges under strict scrutiny, with science chasing purity. Any leftover byproducts can show up as troubles down the line, so industry puts real effort behind refining and purifying each batch.
Names, Labels, and Regulations
Chemists call this stuff tetrachloroethylene or perchloroethylene, and whether chatting with an old hand at a dry cleaner or a hazmat official, those names mean the same thing. Regulations stack up, from U.S. Environmental Protection Agency controls to international standards on labeling, shipping, and storage. Hazmat diamonds, hazard class numbers, and toxic exposure warnings pop up on labels. Workers don’t just swap stories about spills — there are federal standards outlining exposure limits, ventilation requirements, and personal protection. Where rules lagged in the past, new research and a few cautionary tales drove the case for stricter oversight.
How Industry Uses Tetrachloroethylene
For years, walking into a dry cleaner often meant catching a whiff of that unmistakable perc smell. Use didn’t stop at clothes. Factories used it to strip grease from metal machine parts, and it showed up in some adhesives and paint removers. Just as importantly, the properties of perc lend themselves to chemical synthesis of other substances, acting as a starting point for fluorocarbons and certain refrigerants. This versatility fueled demand, especially through the mid-twentieth century, but industries learned the hard way that heavy use without responsible control brings its own headaches.
What Sets Tetrachloroethylene Apart
Perc’s standout feature is its non-flammability — a huge advantage over older solvents that once threatened workers and buildings with fire risk. Add its low solubility in water and quick evaporation, and it becomes clear why cleaning businesses kept tanks of this chemical on hand. It breaks down oil, grease, and waxes in a way soap and water simply can’t. These advantages come bundled with some heavy baggage, though, which researchers have not ignored.
Safety and Health — Knowledge Transforms Practice
Talking honestly about tetrachloroethylene means facing up to its risks. Early dry cleaners and machinists unwittingly breathed vapors for years, long before anyone raised big red flags about chronic health effects. Researchers came forward with evidence linking chronic exposure to dizziness, neurological symptoms, and increased risk of certain cancers. Communities living near contaminated groundwater learned that perc doesn’t just vanish after use — it can seep into drinking water sources, linger in soil, and hang around long after factories close up shop. Shifting health standards forced industry to close the loop, recapture vapors, improve ventilation, and reduce spills. Personal experience in my family’s cleaning business taught me that, even with modern safety helmets and gloves, vigilance is never optional. The old habits — open tanks, minimal protection — changed out of necessity, not convenience.
How Science is Changing the Game
Recent years have seen a surge in efforts for greener chemistry. New solvents, both synthetic and plant-based, challenge the dominance of perchloroethylene. Academic and corporate labs rarely stand still; innovations don’t just chase better stain removal, but methods that keep workspaces healthier and water cleaner. Researchers test alternative methods, lower-toxicity compounds, and improved filtration systems. Some dry cleaners now use liquid carbon dioxide or silicone-based products. Environmental regulations push companies to cut fugitive emissions or switch entirely. The science points clearly: safer and more sustainable options rise as top contenders.
Looking Beyond Today — The Road Ahead
No chemical stays in the spotlight forever. Perc faces tough questions about long-term health, safer work conditions, and what’s left in the environment after decades of use. People in the business know that compliance affects costs, but public trust matters even more. Switching to alternatives won’t happen overnight — machines, skills, and customer expectations won’t change with a snap. Training workers to use new processes, cleaning up old contamination sites, and modernizing regulations take a steady hand and honest conversation among neighbors, lawmakers, and business owners. That’s the only way the dry cleaning world can move forward.
Personal Reflection on Change
My own encounters with tetrachloroethylene always bring back the contrast between convenience and caution. Family stories start with easy fabric care and end with neighborhood health scares, both sides reminding me that progress doesn’t mean erasing the past. Watching regulations catch up, and seeing young chemists push for better answers, I find hope. We can take the lessons learned from the golden age of perchloroethylene, respect the realities of its chemistry, and still demand safer, smarter choices for those who clean our clothes, maintain our machines, and live next door to industry.
What is tetrachloroethylene used for?
Everyday Use in Dry Cleaning Shops
Stepping into a dry cleaner, you can almost catch the distinct, sharp odor of tetrachloroethylene. Dry cleaning depends on this chemical. Most home laundry machines simply won’t remove oil stains from items like business suits or delicate dresses. Tetrachloroethylene, often called “perc” by workers, dissolves grease and dirt without touching water. Dry cleaners across the world use it because it won’t make fabrics shrink or lose color. For someone who’s dropped off wool coats more times than they care to count, the process keeps clothes looking sharp season after season.
Industrial Applications Span Far Beyond Clothing
In factories and workshops, workers break down machinery and rely on tetrachloroethylene to clean metal parts. Oil and sludge find their way into every crack, and most detergents cannot cut through the grime. A quick rinse with the solvent brings parts back to life, ready for assembly or repair. This helps keep crucial equipment working, whether on a busy car assembly line or in the back room of a small appliance repair shop.
Some people may remember the old days when household products and automotive degreasers leaned heavily on such chemicals. Regulations have shifted, but industrial cleaning still depends on it for stubborn contamination.
Role in Manufacturing and Chemical Processing
Tetrachloroethylene doesn’t stop with cleaning. It often gets used to manufacture other chemicals. Factories break it down to produce refrigerants, and sometimes it acts as a carrier for pesticides. Textile mills treat new fabric with it, removing leftover oils from weaving. A friend of mine worked in a fiberglass plant; he told stories of huge tanks filled with the stuff, prepping the glass fibers before spinning them into insulation.
Health Risks and Environmental Issues
Plenty of scientific studies report that exposure to tetrachloroethylene brings risks. Breathing in fumes can cause headaches, dizziness, or sore throats. People working with it every day see higher rates of long-term health problems, including liver and kidney issues. The World Health Organization and U.S. Environmental Protection Agency both consider it a likely human carcinogen.
Concerns run deep in neighborhoods near old dry cleaners or factories. Spills seep into the soil, and groundwater contamination can linger through years. In some towns, cleaning up the chemical pollution becomes a long, expensive process. Homeowners worry about vapor seepage, especially in older buildings that once housed small dry cleaning shops.
Safer Practices and New Technology
More companies now use sealed machines to limit workers’ contact with the chemical. Training and protective gear are standard in responsible businesses. Manufacturers look for alternative solvents made from silicone or liquid carbon dioxide, though those can cost more and work less efficiently. Some dry cleaners switched to “wet cleaning,” a high-tech version of washing that relies on special detergents and computer controls instead of hazardous chemicals.
People living near industries or old dry cleaners want to see stricter monitoring of soil and groundwater. City leaders and cleanup crews turn to advanced filtration and soil vapor extraction. Programs that help small businesses upgrade their cleaning equipment also play a part, lowering community exposure.
The Future of Tetrachloroethylene
Tetrachloroethylene’s place in industry highlights the balance between convenience and health. The chemical keeps more than a few shirts crisp, but costs tied to pollution should not fall on workers or neighbors. As a community, investing in safer methods and smart regulation pays off in cleaner air, safer neighborhoods, and peace of mind for everyone who just wants to pick up their laundry without a second thought.
Is tetrachloroethylene hazardous to health?
The Chemical Behind Dry Cleaning
Walk into just about any dry cleaner, chances are you’re breathing in traces of tetrachloroethylene. This solvent, also called perc, removes grease from clothes without water. Factories and auto shops also use it to scrub metal parts. At first glance, it seems to get the job done, no fuss. Truth is, there’s a reason workplaces keep safety sheets taped to the wall: tetrachloroethylene sticks around in the air and clings to clothing. Some folks bring their suits home and notice a sharp, sweet smell. That’s not just the scent of clean clothes—it means molecules of perc, which can make their way into your household air and sometimes even your bloodstream.
Health Risks in Plain Sight
Back in the day, nobody gave chemical fumes much thought. Workers rarely wore masks. I remember my uncle, who did repairs at a small dry cleaning business, telling stories of headaches and throat irritation after long shifts. Now, science catches up more quickly. Study after study has found that even low doses can irritate eyes, skin, and lungs. Higher exposures bring on dizziness, sleepiness, and trouble with memory. Folks working directly with this solvent have built up enough evidence to suspect links with increased risk of certain cancers, including esophageal and bladder cancer.
The International Agency for Research on Cancer has flagged tetrachloroethylene as a possible carcinogen. The U.S. Environmental Protection Agency calls it likely to cause cancer in people. Lab tests point to DNA changes in animal cells. That’s not speculation—real data links long-term exposure to real disease. My neighbor’s family owned a dry cleaner; he shared about friends who battled chronic kidney or liver issues after years on the job. Concerns go beyond workers. Apartment buildings near older dry cleaning shops have faced indoor air tests showing higher levels than outdoors, especially in basements.
Environmental Impact and Community Concerns
Tetrachloroethylene doesn’t just vanish after use. It can seep into soil and groundwater if handled carelessly, leading to polluted wells and rivers. Ingesting contaminated water threatens community health and can take decades to clean up. In parts of the United States, city councils have forced closures and expensive remediation projects for businesses that let perc spill into the ground. These aren’t minor costs, either—they add up for both taxpayers and business owners.
There’s a human side to cleanup, too. I’ve met tenants who received eviction notices after health officials found high vapor levels in their building, the results of forgotten tanks or corroded pipes leaking chemicals. Once the solvent gets into the structure, airing out the place doesn’t fix the problem. Removal becomes a costly, drawn-out affair that disrupts lives.
Moving Toward Safer Solutions
Workplaces can make a difference by swapping out old machines for closed-loop systems that recover most of the solvent. Switching to water-based alternatives or professional wet cleaning can cut down exposures for staff and customers. Today’s regulations in Europe and California crack down on emissions or ban perc in new dry cleaning shops. Masks, gloves, and proper ventilation should form the routine, not the exception, in places where legacy equipment remains.
Staying informed and asking questions about dry cleaning can protect local air and health. Businesses can help keep customers and employees safe by training staff, maintaining equipment, and keeping up with changing laws. Making the safe choice pays off for everyone: cleaner air, fewer long-term health scares, and clothes that come home without a chemical tagalong. Folks deserve to know what they’re breathing in, so simple awareness becomes the foundation of preventing harm.
How should tetrachloroethylene be stored?
Why This Chemical Matters in the Real World
Tetrachloroethylene, often seen in dry cleaning shops and degreasing operations, worries me more than most people realize. This stuff's got a long track record for sticking around in the air, working into soils, and even finding its way into drinking water. I remember reading about a small laundromat where workers had health complaints—headaches, dizziness, skin issues—just because someone left the storage area open after a delivery. Protecting health means taking storage seriously, every single step along the way.
Barrels Aren’t Enough: Containers Built for Chemicals
Storing a chemical like tetrachloroethylene relies heavily on simple, durable containers. Only steel drums with tight seals or thick-walled plastic tanks really provide enough strength. Weak points in cheap materials let fumes escape or moisture creep in, which spells trouble. I’ve seen cases of mild steel corroding around the lids, which lets vapor escape even if the barrel looks okay on the outside.
Each barrel should sit completely sealed any time it isn’t in use. The workers handling these containers often notice a sharp smell if someone fails to put the cap on snugly. That’s not just unpleasant—it signals a real risk for airborne exposure.
Cool, Dry, Well-Ventilated Spaces Save Lives
Heat and humidity become enemies in chemical storage. Temperatures over 24°C (75°F) speed up evaporation, sending more vapor into the air. Damp basements can rust the best tanks. Windless rooms let fumes collect, so storage areas must allow plenty of air movement. From jobs I’ve worked in old factories, I know that a fan or a window often makes as much difference as a warning label. If the smell lingers, the storage isn’t set up right.
Labels and Leaks: Looking Beyond the Obvious
Every drum or tank requires a clear, permanent label. No scratched writing, no faded printers—labels must list the chemical name and hazard warnings big enough for a tired delivery driver to read on a rainy day. Emergency responders count on this during accidents. I once saw fire marshals spend precious minutes looking for a label while a small leak pooled under an unmarked drum. That delay could have turned tragic in the wrong circumstances.
Leaks and spills happen even if people try their best. Good practice means keeping absorbent pads, chemical-resistant gloves, and a container of baking soda or vermiculite nearby. Relying on a mop or a towel never cuts it. I’ve seen clean-up crews struggle without the right gear, and that just spreads the mess around.
Separate from the Rest, Outside Direct Sun and Sparks
Tetrachloroethylene belongs nowhere near open flames, heaters, or electrical panels. Sparks and solvent fumes don’t mix, and static electricity sometimes builds up even on cool days. Setting dedicated shelves or fenced sections in a warehouse keeps the drums away from accidental bumps or heat sources. I’ve visited shops where chemicals sit behind a simple barrier, keeping workers from stacking tools or boxes on top of them. Small steps make big differences when accidents start with a lazy shortcut.
Training: More Than Just a Legal Requirement
No rules or manuals beats watching someone show the right way, so every worker—new or old—deserves hands-on training with these chemicals. People ignore paper signs after a week, but they remember spills, stings, and rough coughs for months. Managers who treat safety training as a yearly chore soon find out how costly a single mistake can get. Regular refreshers and open conversations about near-misses help everyone stay prepared.
Storing tetrachloroethylene in the right spot protects not just the workers, but their families, neighbors, and the water they all depend on. I’ve seen too many avoidable close calls to believe otherwise.
What are the environmental impacts of tetrachloroethylene?
Tetrachloroethylene and Its Real-World Uses
Tetrachloroethylene, often called perc, sees everyday use in dry cleaning shops and industrial degreasing. Walk by the back door of a local dry cleaner, and you might catch a distinct, sharp smell — that's perc. For decades, it helped keep shirts crisp and metal parts oil-free. Industry saw perc as a strong solvent, but outside the laundry and the workshop, problems grew.
Air, Water, and Soil: The Cost Beyond the Shop
Air quality suffers in neighborhoods close to dry cleaners or chemical plants. Tetrachloroethylene escapes as vapor and lingers, especially indoors. Inhaling even low-level fumes makes people dizzy and irritates the lungs. Research from the United States Environmental Protection Agency ties long-term exposure to nervous system effects, and high levels can damage the liver. Children move through the day near homes and parks, so even small leaks add up.
Water doesn’t get off easy, either. When perc drips onto concrete or spills out, it sinks through the soil. Its chemical makeup lets it slip past many water filters. As it travels, it spreads underground, sometimes reaching public wells. A report from the Agency for Toxic Substances and Disease Registry tracked perc in city tap water, where people unwittingly used it for cooking and bathing. I grew up in a town that struggled for years to clear perc from our groundwater. The fix ran into the millions, and the ticking clock put a burden on families and local health boards.
Lingering in Nature
Unlike oil or alcohol, perc doesn’t break down quickly in nature. In soil, the compound can hang around for years, trickling into streams and moving through the food web. It’s heavy enough to settle deep underground, where cleanup is tricky. Birds and fish exposed to perc suffer liver and kidney damage, behaviors shift, and populations drop. This happens quietly, over years, without much notice in the rush of bigger environmental news.
Looking for Solutions
No big city fixes this issue overnight. Stricter rules help, of course, but they do not close all the gaps. Dry cleaners in California, for example, began switching to safer solvents after state law banned new perc-based machines. That change alone kept thousands of gallons out of the air and water, though old equipment still operates in many parts of the world. More chemical storage training and regular leak checks catch slow drips before they build up. Public health departments have begun tracking neighborhoods with high perc readings, letting families know when water is risky.
Alternative methods offer hope. Liquid carbon dioxide and silicon-based cleaners step in for laundry jobs, sparing the air and water. These options cost more, and small business owners worry about the price, but creative state incentive programs help with the swap. In the end, informed shoppers and local leaders drive these changes — direct questions and everyday choices set the pace for what happens next at the corner cleaner and in the neighborhood well.
Everyday Choices Shape the Environment
Living with chemicals like tetrachloroethylene pushes us to weigh convenience against health and safety. Relying less on hazardous solvents means cleaner water, air that welcomes a deep breath, and parks where kids can run without worry. Each safer choice builds on the last, offering hope for both people and nature down the block and far beyond.
How can tetrachloroethylene exposure be minimized?
Understanding the Real Risks
Tetrachloroethylene, often called PERC, doesn’t get much attention outside the dry-cleaning industry. I first realized its weight when I visited a family friend’s small dry-cleaning shop and noticed strong chemical smells hanging in the air. PERC cleans clothes well, but at a price: studies from the International Agency for Research on Cancer link this solvent with increased cancer risk and nervous system issues. Even short-term exposure—the kind laundry workers experience daily—can lead to dizziness, headaches, and respiratory irritation. Over time, risks pile up.
Better Ventilation Means Safer Air
Walking into any shop or facility using this chemical, the first thing I look for now is how the air feels. Cross-ventilation works wonders. American Conference of Governmental Industrial Hygienists recommends using local exhaust ventilation in places where PERC gets used or stored. Fresh air cuts down on those eye-watering fumes and helps workers avoid breathing in dangerous amounts day after day.
Switching Up the Solvents
No one likes a lecture on going green, yet some alternatives speak for themselves. Wet cleaning and liquid carbon dioxide tech handle delicate fabrics without PERC’s toxic punch. EPA lists wet cleaning among safer choices for businesses making the switch. I’ve talked to cleaners who moved away from PERC and found customers appreciated the cleaner air and lack of harsh chemical smell. State and city governments around the US now push financial incentives for shops embracing these safer methods.
Appropriate Storage and Handling
Old drums of this solvent leaking out back? That’s an accident waiting to happen. Sealed containers, kept in cool, well-ventilated areas, keep leaks at bay. OSHA recommends storing PERC away from heat and sunlight to prevent pressure build-up and spills. Any time a drum gets moved, using gloves and goggles cuts down on skin and eye contact. Good habits here make a real difference in stopping accidents before they happen.
PPE and Regular Medical Checks
In my experience, personal protective equipment matters. Gloves, goggles, and NIOSH-approved respirators shield workers from splashes and vapors. It’s not glamorous, but it works. Regular health checks for those spending all day around PERC help catch problems early, before symptoms get worse. Employers stepping up to pay for these checks show staff that their health comes first.
Community Awareness and Training
Education plays a big role. Sharing knowledge among workers and managers about safe use, proper disposal, and signs of overexposure builds a circle of protection. The CDC recommends employers train staff on emergency steps, storage, spill handling, and correct PPE use. In shops I’ve seen, those who talk openly about these risks catch small problems before they grow.
Looking Ahead
Tetrachloroethylene gave the laundry world a cleaning boost, but health experts all point toward safer ways to do business. Every little change—better ventilation, careful storage, PPE, switching cleaning systems—chips away at risk. Everyone deserves clean clothes and clean air, side by side.
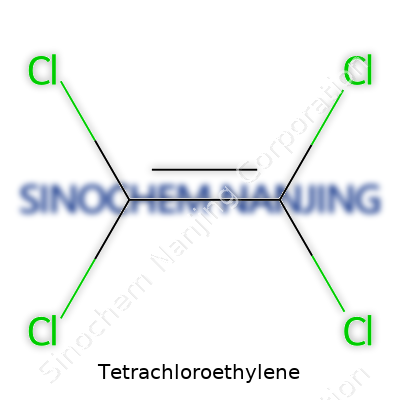
| Names | |
| Preferred IUPAC name | Tetrachloroethene |
| Other names |
Perchloroethylene PCE Ethylene tetrachloride Tetrachloroethene Carbon dichloride Perc |
| Pronunciation | /ˌtɛtrəˌklɔːrəˈɛθɪliːn/ |
| Identifiers | |
| CAS Number | 127-18-4 |
| 3D model (JSmol) | `13@C(Cl)(Cl)C(Cl)Cl` |
| Beilstein Reference | 1200263 |
| ChEBI | CHEBI:27382 |
| ChEMBL | CHEMBL1409 |
| ChemSpider | 11700 |
| DrugBank | DB06733 |
| ECHA InfoCard | 100.001.032 |
| EC Number | 602-028-00-4 |
| Gmelin Reference | 60718 |
| KEGG | C06530 |
| MeSH | D014026 |
| PubChem CID | 31373 |
| RTECS number | KX3850000 |
| UNII | 8ZG23ZG5XU |
| UN number | UN1897 |
| Properties | |
| Chemical formula | C2Cl4 |
| Molar mass | 165.83 g/mol |
| Appearance | Colorless liquid |
| Odor | Ether-like |
| Density | 1.622 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.40 |
| Vapor pressure | 18.5 mmHg (20°C) |
| Basicity (pKb) | Not a base |
| Magnetic susceptibility (χ) | −9.6×10⁻⁶ |
| Refractive index (nD) | 1.505 |
| Viscosity | 0.89 cP (20 °C) |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 290.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -165.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -266.0 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AX06 |
| Hazards | |
| Main hazards | Harmful if inhaled, causes skin and serious eye irritation, may cause cancer, suspected of damaging fertility or the unborn child, causes damage to organs through prolonged or repeated exposure, toxic to aquatic life. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H351, H319, H336, H411 |
| Precautionary statements | P201, P202, P261, P280, P308+P313, P305+P351+P338, P337+P313, P405, P501 |
| Autoignition temperature | 480 °C |
| Lethal dose or concentration | Inhalation-rat LC50: 27,200 mg/m3/4H; Oral-rat LD50: 2,620 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2620 mg/kg (oral, rat) |
| NIOSH | NIOSH: TWA 100 ppm (670 mg/m3) |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
Trichloroethylene Perchloroethylene Chloroform Carbon tetrachloride |

