T-Butyl Pivalate: Chemistry's Quiet Workhorse
Historical Development: Building on Past Curiosity
In the world of organic chemistry, esters like T-Butyl Pivalate don’t make front-page news. Despite the lack of headlines, their story intersects with major shifts in chemical manufacturing. The journey for T-Butyl Pivalate started as the larger chemical industry searched for reliable building blocks for pharmaceuticals and advanced materials. Chemists in the twentieth century pushed alkylation technology further, and the demand for versatile esters rose. Growth in the paint, fragrance, and polymer sectors fueled exploration of new esters with unique profiles. T-Butyl Pivalate emerged from these developments as a dependable, somewhat overlooked, intermediate. Its story reveals how chemists have relied on incremental advances, steady experimentation, and collaboration between university labs and chemical companies.
Product Overview: Trusty Reaction Partner
T-Butyl Pivalate has become a mainstay in research labs, specialty chemical facilities, and pharma manufacturing. People lean on it as a lightweight, manageable ester with a sturdy backbone. T-Butyl Pivalate bridges simple alcohols and acids, making it useful for moving functional groups around or building up larger molecules in a modular, efficient way. When compared to bulkier or more fiddly esters, it stands out for its predictable behavior and its balance between reactivity and stability. In my own academic days, T-Butyl Pivalate earned a place on the shelf as a compound that did what the label promised—no fuss, no drama, just consistent results when the chemistry called for it.
Physical and Chemical Properties: Working with T-Butyl Pivalate
T-Butyl Pivalate’s physical character speaks to its blend of practical benefits. As a liquid under standard conditions, it’s straightforward to handle, pour, and dispense. The molecule brings together the tert-butyl group—known for bulk and electronic influence—and the pivalate half, which itself resists unwanted side reactions. Its volatility makes it manageable in most standard lab settings. People handling T-Butyl Pivalate notice mild, sweet odors—nothing harsh or overpowering, which is a relief compared to some other esters and acids. Chemical reactivity sits in a useful middle ground; it resists hydrolysis under neutral conditions but can be cleaved with acids or bases when needed, giving synthetic chemists leeway in choosing when to disconnect specific fragments.
Technical Specs and Labeling: No-Nonsense Details
Labels for T-Butyl Pivalate typically indicate purity (usually above 97%), storage temperature, and batch tracking for traceability. Regulatory compliance matters for handling and shipping, so labels include hazard pictograms and safety data. I never encountered a mystery with T-Butyl Pivalate’s labeling—labs make sure documentation spells out hazards, precautions, and storage suggestions. There’s little confusion, and the direct approach helps both new chemists and experienced hands stay safe and focused on the chemistry.
Preparation Method: Steady Chemistry, Reliable Processes
Industrial synthesis of T-Butyl Pivalate centers on the classic esterification reaction. Factories bring together Pivalic acid (2,2-dimethylpropanoic acid) and tert-butanol, typically in the presence of an acid catalyst. Sulfuric acid often acts as the catalyst, driving the equilibrium toward the ester. Removing water as it forms nudges the reaction to completion. This process points to some of the broader strengths of industrial chemistry: repeatable protocols, ease of scale-up, and use of familiar reactants. I remember using Dean-Stark apparatus in an undergraduate lab to remove water and push equilibrium—a real-world demonstration of techniques scaled up in industry. The method reflects over a century’s worth of incremental improvement in esterification as a core method in organic synthesis.
Chemical Reactions and Modifications: A Chemical Chameleon
T-Butyl Pivalate’s utility springs from its role as a protecting group. Organic chemists use it to safeguard carboxylic acids during multi-step synthesis, only to remove it when the time is right by hydrolysis. This trait feels almost like giving a molecule a raincoat for the stormy steps, then peeling it off when the coast is clear. It also enters coupling reactions and may be modified to carry different functional groups down the line. T-Butyl esters in general offer unique stability during grueling reaction conditions, and T-Butyl Pivalate stands out thanks to both the size and electron-donating nature of its tert-butyl component. In academic projects, I’ve seen esterified intermediates pass through oxidative, reductive, and cross-coupling steps in one piece, surviving chemistries that shred more fragile molecules.
Synonyms and Product Names: Beyond the Label
T-Butyl Pivalate pops up in catalogs under various names. People might call it tert-butyl pivalate, tert-butyl neopentanoate, or tert-butyl 2,2-dimethylpropanoate. Sometimes the shorthand “TBP” appears, but context is always important—TBP can mean other chemicals in other settings. Commercial products rarely stray from these core names because the structure and application matter more than branding in this branch of chemistry. Consistent nomenclature avoids confusion, especially given the sometimes tangled web of organic chemistry names.
Safety and Operational Standards: Real-World Handling Counts
Safety around T-Butyl Pivalate feels much like safety around other common organic esters. It calls for gloves, goggles, lab coat, and proper ventilation. Inhalation of vapors is best avoided, and since it is flammable, open flames stay out of the workspace. Compliance with regulations like REACH in Europe or OSHA rules in the US supports both individual chemists and the wider community. Incident reports rarely flag this ester as especially troublesome, but as with any organic solvent, accidental spillage or careless mixing with strong acids or oxidizers can cause headaches—or worse. Industry focus on real, routine safety practices helps avoid hazard escalation, and modern labs tend to bake these safety habits into staff training.
Application Area: Everyday Chemistry’s Support Act
Much of the story for T-Butyl Pivalate unfolds in the background. It acts as a transient intermediate, rarely part of the final product by design. Its biggest impact comes in pharmaceutical synthesis, where chemists must shepherd sensitive intermediates through dozens of transformations. T-Butyl Pivalate’s ability to shield carboxy groups lets researchers stitch together complex molecules, including active pharmaceutical ingredients, step by step. Application spreads into flavors and fragrances, thanks to its mild scent and manageable volatility. In advanced materials work, its structural rigidity from the tert-butyl center brings steric bulk, shaping the outcome of polymerization and cross-linking reactions. Speaking from the classroom, even undergraduate curricula lean on T-Butyl Pivalate and related esters to teach hands-on organic synthesis, reinforcing key lessons about reactivity and protection strategies.
Research and Development: Tweaking, Testing, Advancing
Research groups in industry and academia keep circling back to T-Butyl Pivalate’s underlying strengths—modest reactivity, clean hydrolysis, and resistance to some forms of degradation. Ongoing studies probe its behavior under milder or greener reaction conditions, seeking ways to swap out harsh acids or use alternative solvents like ionic liquids or supercritical CO2. High-throughput screening technologies help researchers profile reaction scopes more quickly, and computational work models how its bulkiness affects kinetic barriers or transition states. Recently, some projects have explored replacing older, more hazardous protecting groups with T-Butyl Pivalate analogs to cut down on unpleasant byproducts or persistent environmental residues. This feedback loop—testing, learning, adjusting—keeps the compound relevant and helps refine process chemistry for both safety and efficiency.
Toxicity Research: Eyes Wide on Health and Environment
Toxicity data on T-Butyl Pivalate shows a profile similar to many light esters: low acute toxicity, potential for mild irritation of eyes, skin, or respiratory tract, and standard caution for flammability. Chronic effects receive less discussion, probably because T-Butyl Pivalate does not pop up in consumer products or direct medical applications. That said, regulatory attention has shifted toward knowing long-term environmental fate—will this chemical persist or break down in wastewater streams? Analytical studies point toward ready hydrolysis in natural conditions, but questions about breakdown products, particularly in large-scale industrial effluent, still spark debate among environmental chemists. Risk assessments push manufacturers to engineer containment solutions, exhaust capture, and waste treatment that minimize release, aiming to keep the compound’s footprint as small as its daily role in synthesis.
Future Prospects: More Value Still to Find
Looking ahead, T-Butyl Pivalate’s track record suggests continued importance, especially in refined pharmaceutical synthesis and sustainable chemistry. Process chemists keep searching for protecting groups and intermediates that deliver selectivity and operational simplicity while fitting with tighter regulatory and environmental demands. Green chemistry principles put pressure on manufacturing to reduce solvent use, energy input, and downstream waste. T-Butyl Pivalate, with its moderate reactivity and compatibility with newer solvent systems, still supports that transition. Policy changes, such as extended producer responsibility and better pollution controls, will likely nudge the chemistry community to revisit and update both synthesis and disposal methods. The quiet strength of T-Butyl Pivalate lies not in dramatic breakthroughs but in the way it anchors small, practical improvements across decades of innovation. The story of this ester reflects the incremental, ongoing effort underneath the chemistry that shapes daily life—and that story is far from finished.
What is T-Butyl Pivalate used for?
Where T-Butyl Pivalate Fits Into Industry
T-Butyl pivalate may sound like one of those chemicals buried deep in a warehouse supply list, but it actually has a role that touches more industries than most people notice. In my years working with laboratory supply chains, I saw this compound show up in conversations among industrial chemists, especially those who focus on organic synthesis. T-Butyl pivalate works as a building block in the production of pharmaceuticals, perfumes, and specialty chemicals. When manufacturers design intermediates for medicine, they often look for ways to keep reactions clean and direct. Compounds like T-Butyl pivalate help them get there by protecting reactive parts of molecules during synthesis. Think of it as a chemical cloak: it covers up parts of the molecule that aren't supposed to react yet.
Supporting Efficiency in Chemical Synthesis
Synthetic chemists in pharma prize precision. Shortcuts usually end in wasted material or lost time. T-Butyl pivalate lends a hand as a protecting group for alcohol functionalities in pharmaceutical intermediates. By using this pivalate, they can hit key reaction points in a step-by-step manner. This saves on labor costs and material losses because it shields sensitive parts of molecules until the right moment. Based on numbers I've seen from lab reports, yields can improve by 5-10% per batch when the proper protecting groups come into play. With tight margins the norm in chemical manufacturing, that small bump in efficiency really matters.
Scent Engineers and Custom Fragrances
Most people expect perfumes to rely only on rare flowers or high-end oils. Behind the scenes, designers actually work with hundreds of chemical components to create new scents. T-Butyl pivalate serves as a key intermediate in synthesizing certain esters that have pleasant, sweet, fruity notes. These additives don’t just provide density to fragrances—they help scents last longer. Years ago, a perfumer told me that these molecules act like anchors, allowing the high notes in fragrances to linger longer after a bottle is opened. Keeping those scents stable during production and storage relies on well-made synthetic ingredients such as T-Butyl pivalate.
Concerns and Responsible Handling
Anytime chemicals enter wide-scale use, I think about safety and the potential for misuse. T-Butyl pivalate, like many lab solvents and intermediates, carries risks if handled without care. Prolonged skin contact leads to irritation, and inhalation in poorly ventilated spaces can harm health. Regulatory bodies, including the EPA and OSHA, lay out clear guidance on handling, storage, and disposal. In particular, facilities storing pivalate must equip personnel with gloves, goggles, and proper fume hoods. Unlike some chemicals found in industrial workflows, T-Butyl pivalate does not appear on federal lists for persistent bioaccumulation or high acute toxicity, but that doesn't mean safety measures get ignored.
Pushing Toward Greener Alternatives
Chemical manufacturers face growing pressure to adopt greener, safer reagents. Some companies now explore alternatives or modified processes aiming to reduce reliance on volatile organics like T-Butyl pivalate. Screening safer protecting groups or leveraging biosynthetic pathways could ease environmental impact over the coming decades. Consulting with sustainability experts and updating lab protocols make sense for facilities keeping an eye on long-term compliance and worker safety.
What is the molecular formula of T-Butyl Pivalate?
Understanding T-Butyl Pivalate
T-Butyl pivalate rarely pops up outside a lab, yet its structure draws attention from anyone who’s spent time studying organic chemistry. It’s an ester formed from pivalic acid—otherwise known as trimethylacetic acid—and tert-butyl alcohol. The ester bond forms when the carboxyl group from pivalic acid links up with the alcohol.
It’s easy to get lost in chemical names, so let’s look closer at what’s really in this compound. Pivalic acid alone has the formula C5H10O2. If you’ve worked with it before, the smell is recognizable and its bulkiness jumps out, thanks to three methyl groups crowding one carbon atom. Mix in tert-butyl alcohol, which adds C4H10O to the mix, and you think the story is straightforward. Yet in ester formation, water leaves the scene, so the final formula cuts out two hydrogens and one oxygen. That lands us at C9H18O2.
Why Structural Clarity Matters
Anyone in the pharmaceutical or fragrance business knows bulky esters like t-butyl pivalate affect both chemical reactivity and physical properties. I remember the first time I ran a GC-MS analysis on a mixture containing such esters—sharp, reliable peaks, but stubborn persistence on the column. That’s the pivalate backbone doing its thing.
Toughness pays off in harsh conditions. Many synthesis routes count on robust esters to weather basic or acidic environments without falling apart. Shelf stability rises, degradation slows, and products don’t spoil so fast. Chemical suppliers around the world list t-butyl pivalate in catalogs because plenty of researchers and manufacturers count on it as a model bulky ester or a protecting group scaffold.
Sourcing and Safe Use
Accessing high-purity t-butyl pivalate gets easier year after year. Most chemical vendors focus on purity since labs need consistently clean stock to avoid reaction pathway hiccups. Safety data sheets urge proper ventilation for storage, and I never go without goggles and gloves. Many esters volatilize pretty fast—they don’t care about your sinuses—so sealed containers matter just as much as dry benches.
Applications and Future Directions
As processes become greener, chemists look into esters both as solvents and as testbeds for new catalysts. T-butyl pivalate’s resistance to hydrolysis means it stands out when aggressive reagents enter the fray. Some experimental setups use it specifically to probe reaction selectivity. If organic students groan about memorizing molecular formulas, this molecule teaches why structure ties so closely to chemical behavior and why shortcuts don’t exist in real-world synthesis.
Identifying the formula doesn’t just tick a box for exams. It helps everyone from product engineers to researchers avoid missteps, keep costs in check, and stay safe. As industries update old product lines with modern chemistry, accurate understanding of compounds like t-butyl pivalate is essential for progress.
Is T-Butyl Pivalate hazardous or toxic?
Unpacking T-Butyl Pivalate’s Reputation
T-Butyl Pivalate rarely shows up in the news, but it’s not an obscure laboratory relic. In chemistry circles, this organic ester gets respect as a reagent—one of those chemicals that can either keep things running smoothly or, if ignored, make a mess. So, let’s talk about the real story behind its toxicity and hazards, using a grounded approach based on both the facts and a practical sense shaped by working with industrial solvents and esters.
Breaking Down the Science
T-Butyl Pivalate has a chemical formula of C9H18O2. On the surface, many organic esters like this don’t ring alarm bells, especially compared to volatile solvents such as acetone or benzene. Still, the absence of urgent warnings doesn’t mean it’s as safe as dish soap. Labs and industrial users won’t gamble on wishful thinking. Material Safety Data Sheets (MSDS) lay out the hard truths: eye and skin irritation counts among the common risks. If you ever spilled a small drop of organic solvent on your skin, you’d recognize the quickly rising irritation—a burning or tingling, maybe even red splotches if ignored. I’ve seen small accidents escalate when personal protective equipment gets skipped, even “for just a moment.”
Breathing in the vapors is another issue. Volatile esters sometimes trick people with mild smells or the lack of obvious smoke. There’s no poison warning like methyl isocyanate or hydrogen sulfide, but repeat exposure to vapors can wear down your lungs. According to data compiled by the European Chemicals Agency and the U.S. National Library of Medicine, t-butyl esters may lead to coughing, sore throats, or dizziness after high exposures. That’s not rarefied theory—anyone who has worked repeatedly around open solvent containers knows headaches or wooziness can creep in.
Toxicity: More Gray Than Black or White
Few chemical safety debates are black and white. T-Butyl Pivalate does not register on lists for acute toxicity or chronic carcinogenicity. Still, that doesn’t mean it’s harmless. If swallowed in quantity, it may upset digestion, even leading to nausea or vomiting. Some animal studies used for regulatory reviews reveal only mild toxicity when compared to high-hazard chemicals. Workers in chemical plants or large labs listen to the phrase “use in well-ventilated areas” for a reason.
Disposal and leakage pose additional concerns. Because it is an organic solvent, T-Butyl Pivalate will not mix with water but may float or linger on top—posing a risk to aquatic habitats. Environmental regulatory agencies track these chemicals not just for their immediate toxicity, but for their resistance to breaking down in nature. Stories of solvent leaks harming groundwater drive home why proper containment matters. In my years working with chemical storage, I’ve seen barrels marked “non-toxic” spark frantic cleanup efforts after tipping over.
Practical Steps Toward Greater Safety
Complacency invites trouble. Gloves, goggles, and ventilated workspaces aren’t optional extras—they make the difference between a minor scare and a real emergency. Proper labeling and chemical storage slow down accidents. Training employees to recognize the symptoms of overexposure, and teaching by example, lifts workplace safety beyond hollow memos.
On the regulatory side, governments require up-to-date hazard labels and reporting of quantities in storage. These rules do more than keep inspectors happy—they nudge everyone to track their chemicals, double-check seals, and review spill protocols. For disposal, working with certified chemical waste handlers ensures these solvents don’t find their way into rivers or soil where they could linger.
Final Thoughts on Responsible Use
T-Butyl Pivalate sits in that broad middle zone. It isn’t a panic-inducing toxin, but it’s not the sort of liquid you’d want splashing on your hands or running down a drain. Workers and home chemists alike benefit from treating it with the care reserved for anything that stings the nose and irritates the skin. The underlying lesson: respect for the chemical keeps surprises to a minimum. Emphasizing clear procedures and open conversations builds real trust, both in the lab and beyond.
How should T-Butyl Pivalate be stored?
Understanding the Risks
T-Butyl pivalate sits on many chemical shelves for a reason. It offers valuable uses in labs, but ignoring safe storage could lead to real hazards. This liquid brings a flammable nature and can irritate skin, eyes, and lungs. In my past work with specialty chemicals, I saw far too many times where folks ended up scrambling because the basics were overlooked. Even a harmless-looking bottle disrupts entire labs if care drops for a moment.
Location and Containment
Keeping t-butyl pivalate in a well-ventilated, cool area reduces the risk of dangerous vapor buildup. Warm, stuffy storage speeds up evaporation and builds internal pressure. I once saw minor swelling in a poorly placed bottle; the simple mistake could have turned ugly. Select a chemical cabinet specifically designed for flammable materials—one that stays away from direct sunlight, any heat sources, and the casual traffic of the main workspace.
Don’t put it near acids, alkalis, or oxidizing agents. A splash, tiny leak, or even vapor mixing could ignite or corrode, which spells trouble. Proper segregation keeps “mix-ups” to a minimum, and clear labeling helps everyone stay on the same page.
Containers and Seals
Bottles and containers matter more than some people might think. I know someone who skimped on this, using an old food jar, then panicked when a sticky ring built up and the cap warped. Always choose an airtight, chemical-resistant bottle with screw-tight lids. Never trust casual plastic or recycled containers that can soften or degrade over time.
Labels must list the chemical name, concentration, hazards, and date received. Bold ink and chemical-resistant tape go a long way. If anything leaks, the cleanup becomes straightforward and quick, reducing exposure and confusion.
Temperature and Air Quality
Stable room temperature stops pressure swings inside bottles and prevents unwanted chemical reactions. Nothing beats an air-conditioned space or a ventilated fume hood for indoor chemical work. Fumes released at higher temperatures often escape and irritate lungs faster than expected. Staying aware of local fire codes keeps insurance headaches far away too.
Essential Personal Protection
Opening a bottle of t-butyl pivalate? I always put on chemical-resistant gloves, goggles, and a lab coat, even if it feels repetitive. Skin and eye irritation comes fast, with just a droplet or a quick waft of vapor. I once heard of a spill that left someone’s hands tingling for hours—simple gloves would have stopped that in its tracks.
Planning for Emergencies
Fire extinguishers designed for chemical fires belong near storage spaces. Learning where the emergency shower is, as well as keeping spill kits topped up, makes sure everyone reacts fast if mistakes happen. Emergency drills, practiced with the real risks in mind, keep the team ready and confident. Taking shortcuts only puts people and property at risk.
Looking Ahead
Constant review of safety data sheets goes hand in hand with good storage practice. Supervisors and lab teams should talk about chemical hazards often, update documentation, and encourage questions. Safer habits form when everyone feels responsible for each other. It’s not about paranoia—it’s about protecting the people who put chemicals like t-butyl pivalate to productive use every single day.
What are the physical and chemical properties of T-Butyl Pivalate?
Getting to Know T-Butyl Pivalate
It’s tough to ignore how much small, specialized chemicals shape the world around us. T-Butyl Pivalate, known in labs by the name tert-butyl neopentanoate, falls in that group. The name might not come up in everyday conversation, but anyone who has spent time staring at beakers knows its power as an ester. In practical terms, this compound comes across as a clear, colorless liquid, putting off a smell somewhere between fruity and solvent-like. For chemists scanning a bottle, the molecular formula C9H18O2 says it all: a blend of a t-butyl group with a pivalate backbone.
Physical Properties That Matter
Density, boiling point, and volatility aren’t just facts for a safety sheet; they tell you how the stuff acts in the real world. T-Butyl Pivalate has a boiling point that sits around 142-144°C, which means it doesn’t just vanish in a warm room. Its density weighs in at about 0.865 g/cm³, close to many other organic liquids. You pick up a flask—lightness jumps out. The low viscosity flows naturally, which makes transferring or mixing easy.
Anyone who tries dissolving it in water is in for a struggle. This ester prefers hanging out in organic solvents like diethyl ether or ethanol. Working in the lab, it’s clear that water and T-Butyl Pivalate barely mix—shaking won’t get the job done. This immiscibility puts limitations on some applications but opens doors for others.
Chemical Nature and its Clear Behavior
Some esters like to react at the drop of a hat; T-Butyl Pivalate shows a steady hand. Hydrolysis can break it down under acidic or basic conditions, giving back alcohol and carboxylic acid—basic textbook chemistry. This reaction can speed up with heat or strong solutions, which I learned during a warm summer in the lab. Drop a dash of acid, let the mix cook, and you’ll see the ester bonds snap.
Stability against oxidation gives safe handling—no surprise decompositions. Still, it isn’t bulletproof. Strong acids or bases will chew through it, especially if left to sit. Its vapor can catch fire with the right spark, and the flash point tends to land around 35°C. From a safety perspective, that means good ventilation and no open flames—things drilled into you in chemical safety training.
Why People Pay Attention
People want T-Butyl Pivalate for more than one reason. Its use in making flavors and fragrances stands out. The subtle fruity notes come through in many lab-made aromas. In industrial work, it pops up as a starting material for more complex compounds. The predictable solubility and controlled reactivity let chemists handle it for lab syntheses, pilot plants, and scale-up batches. Its footprint in the pharmaceutical and fine chemical markets comes from being just reactive enough to transform, but not so touchy that it causes headaches during storage.
Looking at Challenges and What's Next
Concerns about safe use keep coming up. With any volatile organic compound, there’s a need for tight control—good fume hoods, safe storage, and careful monitoring. Waste treatment is crucial; esters spilling into groundwater don’t break down fast and can hang around. Teaching new chemists to respect its properties, rather than fearing them, pays off. Shifting toward greener synthetic paths or using it in closed-loop systems may ease some environmental worries.
All said, T-Butyl Pivalate keeps showing up on chemical shelves for good reason. It handles well, does its job, and, if respected, stays safe around people who know what they’re working with.
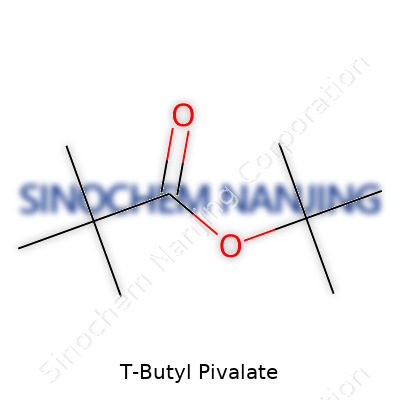
| Names | |
| Preferred IUPAC name | 2,2-Dimethylpropyl 2,2-dimethylpropanoate |
| Other names |
Pivalic acid tert-butyl ester tert-Butyl pivalate tert-Butyl trimethylacetate Trimethylacetic acid tert-butyl ester |
| Pronunciation | /tiː ˈbɜːtɪl pɪˈveɪleɪt/ |
| Identifiers | |
| CAS Number | 540-31-0 |
| Beilstein Reference | 1201578 |
| ChEBI | CHEBI:89374 |
| ChEMBL | CHEMBL513998 |
| ChemSpider | 11636082 |
| DrugBank | DB22056 |
| ECHA InfoCard | 19d1bce4-f0e2-457d-b2b9-d206b92da6c2 |
| EC Number | 211-047-0 |
| Gmelin Reference | 585118 |
| KEGG | C21112 |
| MeSH | C089697 |
| PubChem CID | 12328730 |
| RTECS number | EJ5950000 |
| UNII | 4Y97Q9XE0E |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID7020708 |
| Properties | |
| Chemical formula | C9H18O2 |
| Molar mass | 160.26 g/mol |
| Appearance | Colorless transparent liquid |
| Density | 0.861 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 1.97 |
| Vapor pressure | 0.7 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | Basicity (pKb) = -2 |
| Magnetic susceptibility (χ) | -8.62 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.398 |
| Viscosity | 1.7 mPa·s (20 °C) |
| Dipole moment | 2.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 239.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -599.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6885.7 kJ/mol |
| Pharmacology | |
| ATC code | ATC code not assigned |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: "H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 71 °C |
| Autoignition temperature | 370 °C (698 °F; 643 K) |
| Lethal dose or concentration | Lethal dose or concentration: LD₅₀ (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose) of T-Butyl Pivalate: Oral, rat: >5000 mg/kg |
| NIOSH | NA7125000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 150-300 ppm |
| Related compounds | |
| Related compounds |
Methyl pivalate Ethyl pivalate Isopropyl pivalate Tert-butyl acetate Pivalic acid Tert-butyl alcohol |

