T-Butyl Hydroperoxide: The Story, the Science, and the Stakes
Digging Into the History
Chemistry does not evolve in a vacuum. Every major compound has a backstory shaped by technological ambitions and the changing needs of society. T-Butyl hydroperoxide, often abbreviated as TBHP, entered the industrial scene during a period marked by rapid expansion in synthetic organic chemistry. Researchers started searching for strong but predictable oxidizing agents as new pathways in plastics, pharmaceuticals, and coatings took off. The historical context makes sense: once phenol and propylene became routine building blocks after the Second World War, t-butyl hydroperoxide emerged as a valuable tool for pushing certain chemical reactions forward, especially where safer or milder oxidants didn’t cut it. So, behind the product lies a pragmatic response to industrial demand and growing knowledge of how carefully tuned oxidants could transform whole sectors, especially when safer alternatives faltered.
Understanding T-Butyl Hydroperoxide
From a user's perspective, TBHP comes up in labs and factories alike, bridging the academic and industrial. This organic peroxide, with the formula (CH3)3COOH, stands as a colorless liquid. There’s a faint but sharp, ether-like smell to it, so workers need good ventilation. The compound's main chemical clout comes from the peroxide bond — an unstable O–O bond slotted in a branched carbon scaffold. Unlike hydrogen peroxide, TBHP's bigger, bulkier structure allows more control during various oxidation reactions, cutting down side products in processes like epoxidation or radical polymerization. This helps improve yield, which always matters to both researchers and manufacturers. Synonyms you may run into include tert-butyl hydroperoxide, TBHP, and peroxy-tert-butane, reflecting the naming conventions that sometimes trip up even seasoned chemists.
Physical and Chemical Properties in the Real World
TBHP likes to keep things interesting. It holds a low viscosity, which translates to easy mixing, yet brings volatility and fire risk. Flammability and explosive potential top the safety concerns—the material breaks down to release both heat and gaseous byproducts, especially under heat or friction. You’ll often find the commercially available material dissolved in water, roughly at 70 percent concentration, striking a balance between strength and safer handling. The storage temperature can’t swing too high, or TBHP will decompose faster than you might expect. TBHP dissolves in many common solvents (like diethyl ether and alcohols), but it can be persnickety if exposed to reducing agents or transition metals without careful oversight. In an industrial setting, engineers and chemists spend a lot of effort engineering safe process lines, leak detection, and containment, all because of these physical quirks.
Technical Details and Labeling Matters
Anyone dealing with hazardous chemicals knows labeling serves more than regulatory compliance—it actually keeps people safe. TBHP requires hazard diamonds indicating oxidizer risk and the flammable tag. Standard containers get UN numbers and carry prominent warnings for skin and eye damage plus environmental threat. Even in packaging, different concentrations require clear distinction since the risk profile ramps up sharply above 70 percent. Routine monitoring of peroxide concentration comes standard in well-run labs, since even a small drop can indicate slow decomposition or contamination. Documentation doesn’t just protect legal interests; it supports emergency responses and routine audits alike. Good records and strong technical understanding help organizations avoid accidents and minimize downtime.
Methods for Making TBHP
What goes into making t-butyl hydroperoxide? The industry settled on reacting isobutane or isobutylene with molecular oxygen, usually under carefully controlled heat and pressure. Early-day chemists adopted direct oxidation of isobutane as one reliable approach, incorporating metal catalysts (sometimes cobalt or manganese salts) that tip the reaction in favor of hydroperoxide over unwanted byproducts. A flow system, good mixing, and a way to manage heat — this combination paves the road for steady TBHP manufacture. Yet, even now, yields get tangled up in the same issues as before: incomplete reactions, strict moisture exclusion, and product purification. Advances since the 20th century included greener catalysts, lower energy input, and process automation, pushing TBHP towards more sustainable production, but process safety remains an ongoing quest.
Reaction Pathways and Chemical Modifications
T-Butyl hydroperoxide serves as more than just a one-trick pony. In the hands of a practiced chemist, TBHP initiates radical reactions, making it valuable for polymer initiations and oxidizing alcohols, alkenes, and other functional groups. Many prominent epoxidation protocols rely on TBHP to generate epoxides from simple alkenes, unlocking value in everything from adhesives to fine chemicals. Its role in Baeyer–Villiger oxidations helps build valuable esters and lactones. Copper, vanadium, and molybdenum complexes often join the party as catalysts, steering the reactivity in productive directions and minimizing the struggle to isolate the cleanest product possible. Chemical tweaks—such as complexing TBHP with a co-oxidant or quenching agent—help broaden its uses, address shortcomings of selectivity, and tune it for niche applications.
Best Practices in the Field
Safety doesn’t come from a checklist; it emerges from an alert culture and respect for what TBHP can do in the wrong hands or conditions. Over the years, I’ve seen how seasoned operators become almost superstitious about static discharge, careful dilution, and removing ignition sources. One overlooked spill, or a heat spike in a storage drum, and you’re close to a nasty situation. Proper goggles, gloves, and flame-resistant lab coats matter, but so do routines—the buddy system, regular safety drills, and always keeping neutralizing adsorbents nearby. Everyone who handles TBHP knows to check for leaks, test ventilation, and stow containers in cool, shaded storage. Incidents over decades, from laboratory mishaps to warehouse fires, have led to stricter codes and updated training practice. It’s one thing to tell people “it’s dangerous”—it’s another to drill into real-life what ifs and teach new hires what care really means.
Where TBHP Shows Up
Across industrial chemistry, t-butyl hydroperoxide turns up in familiar and surprising places. The big ticket: its use as a cleaner-burning oxidizer in propylene oxide synthesis, which forms the backbone for polyurethane foams, antifreezes, and specialty solvents. In coatings, adhesives, and elastomers, TBHP sparks the polymerization process longer and more predictably than many old-school initiators. Pharma companies lean on its controlled oxidation for drug intermediates, helping streamline complex syntheses that would otherwise generate more waste. In fine chemicals, TBHP's reactivity gets deployed for making flavors, fragrances, and even agricultural chemicals, as it spares more delicate ingredients from harsher oxidative conditions. In research settings, it stands as a model compound to probe reaction mechanisms and develop safer catalytic systems.
What Research Is Chasing Now
The world never stops asking for safer and greener chemistry. TBHP sits at the center of these debates, both as a tool and a risk. Research efforts focus on reducing byproducts during production, squeezing more efficiency from catalytic systems, and taming decomposition hazards. One trend gaining traction involves embedding TBHP in microencapsulated carriers, aiming to cut down direct exposure and accidental releases. Another line of work goes into customizing transition metal catalysts to get more mileage out of each batch, reducing peroxide waste downstream. Computational chemists increasingly model TBHP’s reactivity to map out new synthetic routes, hoping to cross old limitations in selectivity and speed. Such work doesn’t just appeal to the lab—the potential for more nimble, flexible manufacturing plants hangs in the balance, as companies look to scale new products quickly and safely.
The Facts on Toxicity and Exposure Risks
Spending any time around TBHP makes it clear: this isn’t something to take lightly. Even low-level exposure causes eye and respiratory irritation, and spills on skin lead to burns and blisters. High concentrations introduce real fire and explosion hazards, not to mention chronic effects linked with repeated exposure. Industry guidelines set rigid exposure limits, and for good reason: accidents involving TBHP have led to serious injuries and costly facility shutdowns. Standard safety protocols draw on the lessons of the past, pushing for spill containment, real-time vapor monitoring, and regular hazard reviews. Studies on long-term toxicity keep broadening, especially as regulators scrutinize the byproducts and downstream water effluents. Workers, management, and environmental health staff all share the burden of updating risk assessments and keeping training materials up to date, as TBHP’s toxicity profile nudges companies toward safer alternatives in some applications.
Looking Forward: Future Roles and Promises
Every discussion about chemicals teeters on the edge of what comes next. For TBHP, advances in reactor design and environmental stewardship shape where things head. Efforts crank forward to cut down on energy consumption during manufacturing, recycle unused reagent streams, and devise biodegradable alternatives that keep the best parts of peroxide chemistry without the baggage. With green chemistry taking hold in both academia and industry, researchers lean into life-cycle assessments, cleaner waste streams, and more automation—a future where someone spends less time physically around explosive chemicals, relying instead on in-line sensors or remote management. Still, the fundamentals won’t change: as long as strong, targeted oxidants remain critical, TBHP holds a spot in the toolkit. The real challenge isn't erasing it from the map, but managing its use responsibly and always respecting the lessons learned over decades of hard chemistry and honest mistakes.
What is T-Butyl Hydroperoxide used for?
In the Shadows of Everyday Products
I walked through a manufacturing plant a few years ago. The air smelled clean, but every shelf had labels, and chemists talked shop over the clink of glass. Among those chemicals, I spotted t-butyl hydroperoxide. Folks outside the industry rarely mention it, but without it, many products would look and perform differently. T-butyl hydroperoxide, or TBHP, gets called on for jobs that demand power and precision. It drives reactions behind plastics, coatings, and even pharmaceuticals.
Driving Force in Chemical Reactions
Industries rely on TBHP for its strong oxidation capabilities. In plain language, it helps add oxygen to other molecules efficiently. Companies use TBHP to kickstart polymerization—the process that turns simple chemicals into the plastics we use every day. Think drinking bottles, insulation, toys, or even protective films. None of this would reach shelves if someone didn’t start the process, and TBHP handles that behind the scenes.
Helping Medicines Take Shape
In labs, TBHP doesn’t just sit in a corner collecting dust. It’s involved in making active pharmaceutical ingredients. I remember reading about labs using it to build certain antibiotics and anti-inflammatory drugs. Chemists count on it to introduce oxygen into complex molecules, making medicines safer and more effective for patients. The process isn’t always straightforward, so experience and a steady hand matter as much as good raw material.
Tough and Unforgiving—Handling Matters
This compound packs a kick. TBHP is reactive and can catch fire or explode if handled without care. Workers need solid training, protective gear, and constant monitoring. When regulations get ignored, accidents happen, risking lives and property. I recall a story from a friend who worked around peroxide-based chemicals. Strict protocols and weekly drills kept people safe, but even a quick slip could cause disaster. Plants that handle TBHP invest in safety for a reason.
Environmental Impact and Safety
People get concerned about what rolls out of chemical plants. TBHP’s high reactivity means any spill or leak demands attention. Many facilities have scrubbers, containment zones, and tightly controlled handling processes. That’s not just about rules—it’s about respect for communities and the environment. A nearby waterway turned murky after a spill years ago, and the cleanup effort showed how risky improper chemical management gets. After stricter oversight, incidents like that dropped noticeably.
Alternatives and Looking Ahead
Industries look for safer or greener alternatives. TBHP’s utility keeps it in rotation, but researchers develop catalysts and milder oxidizers all the time. Some smaller outfits, especially in Europe and Japan, experiment with replacements. The big hurdle remains balancing performance, cost, and safety. For now, TBHP stays in play, but it’s smart to keep asking how we can do better, keep people safe, and reduce environmental impact.
Is T-Butyl Hydroperoxide hazardous to handle?
Chemicals Demand Respect, Not Panic
Every day in the lab or on the factory floor, familiar routines can mask the hidden power of what sits in our bottles and tanks. T-Butyl hydroperoxide, or TBHP, stands as a clear example. A small container of this stuff changes the vibe. You can spot the warning labels for a reason. TBHP isn’t the most notorious chemical in industrial use, but turn your back on its risks and you’ll remember it the hard way.
Fire and Health: Double Edged Danger
Touching TBHP drops you into two worlds of concern: things that burn, and things that burn you. Its fire risk jumps out first. TBHP breaks down easily—and when it does, the reaction gives off a lot of heat. Temperatures as mild as a sunny day can push it into explosive territory. People who’ve seen a peroxide-driven fire remember the speed and violence. It doesn’t just burn; it feeds flames with pure energy, making firefighting nearly hopeless until the fuel runs out.
Breathing in fumes or letting it touch your skin brings health risks on top. TBHP isn’t gentle. My first real experience with it came when a colleague, distracted and rushed, skipped changing his gloves. Not long after, he noticed his skin turning white and burning. Turns out, TBHP eats away at tissue and stings the eyes, mouth, and lungs if you’re not careful. Research links exposure to headaches, dizziness, and in higher doses, damage to the lungs or liver.
Why People Still Use It
TBHP makes itself useful across the chemical industry. Makers of plastics, pharmaceuticals, and specialty chemicals put it to work for oxidation reactions that few alternatives can match. Whether you’re pushing a tough molecule to react or trying to fine-tune a drug process, TBHP offers results that keep people coming back. It’s part of a club: strong oxidizers like hydrogen peroxide and organic peroxides call for the same careful attitude and planning.
Smart Handling Protects People
I’ve spent enough time around strong peroxides to see what happens when the basics slip. TBHP asks for respect—a locked cabinet, a cool storage room, gloves made for chemical splash resistance, and goggles that actually fit your head. Spill control supplies should sit close by and the training can’t just check a box. Real practice, drills, and honest talk about near-misses build a safety culture people trust.
Industry experience lines up with science. Data from government reports show most TBHP accidents happen during transfer, mixing, or storage—often when safety habits get sloppy or shortcuts creep in. In my work, I’ve seen big wins just from regular audits: walk the storage room, check labels and expiration dates, quiz the team about emergency plans. Companies that build a habit of these steps cut down their emergencies and keep everyone healthier.
Room for Better Solutions
People keep working on safer alternatives and better containment tech for TBHP. Some specialty tasks already use less hazardous chemicals or processes, but not every reaction has a swap. Hospitals and fire departments push for more public info and faster response training, which matches what I’ve learned from tough experiences—everyone benefits from stronger communication across teams.
The gear, habits, and policies around TBHP make or break safe handling. Every bottle of peroxide carries the same silent warning: know your stuff or pay the price. In my book, that kind of risk opens the door for new technology, stronger teamwork, and more open conversation about what works and what doesn’t on the front lines of chemical handling.
What are the recommended storage conditions for T-Butyl Hydroperoxide?
Handling a Volatile Chemical in Real Life
T-Butyl hydroperoxide, often found in industry labs, raises an eyebrow for anyone who’s seen what happens if it gets out of hand. Storage isn’t about formality or following a checklist; it’s about keeping people and places safe from real danger. This chemical burns hot, and it doesn’t take much for things to escalate. Every measure counts because stories of near-misses echo around workshops and laboratories. No one forgets the sharp scent, nor the stories of heat and flame.
Temperature Matters Every Day
Most folks working with this chemical know the drill: keep it cool, keep it steady. Above 30°C, t-butyl hydroperoxide can turn from a stable liquid into a risk. Higher temperatures increase pressure inside storage containers and raise the odds of leaks or explosions. I’ve learned that a flammable liquid like this belongs in a dry, well-ventilated spot, away from heaters, direct sunlight, or any source of ignition. Even fluorescent lights in a cramped closet don’t help the situation.
A cool storeroom, fitted with temperature monitors, earns its keep over time. Refrigeration sometimes works, but only if there’s no risk of condensation that would drip water into the chemical. Any water increases the danger — this isn’t a risk worth taking for the sake of convenience.
Containers Keep Trouble at Bay
T-Butyl hydroperoxide prefers steel with a protective lining or high-quality polyethylene. Glass can crack, and cheap plastics degrade over time. Reliable seals, not just “good enough,” make the difference between a routine month and an emergency call. Regular checks for bulging drums or odd smells have stopped incidents before they started. Over the years, I learned not to trust containers just because they look fine from the outside.
Separate from the Wrong Neighbors
Oils, acids, metals—mixing these with t-butyl hydroperoxide led to disasters in the past. Even a lazy worker who tossed containers onto the same shelf could unknowingly set up a chemical reaction. Guidelines call for designated cabinets, and I’ve seen places label and color-code every drum and bottle just to cut confusion. It’s not overkill. A cleaner, organized storeroom means everyone heads home safe.
Labels and Training Save Lives
In some shops, fresh hires got a safety talk before touching a single bottle. Labels shout out danger and handling instructions in big, clear print. I’ve seen how one person’s mistake—pouring chemicals down the wrong drain or leaving lids loose—can shut down a whole floor. Quick, informed responses come from good habits and proper drills, not luck.
Planning for Emergencies
Spill kits, fire extinguishers, eyewash stations—anyone storing this chemical needs more than a first-aid kit. Local fire codes set rules, but real experience comes from monthly reviews and emergency drills. Alarm systems, staff training, and written checklists earn respect after the first close call.
The Role of Regulations
OSHA, NFPA, and other authorities spell out requirements, but lived experience shapes day-to-day choices. Inspections keep teams honest, but trust builds from watching coworkers follow best practices because they know what’s at stake. People with a long history working around t-butyl hydroperoxide don’t cut corners, because the risks aren’t abstract—they’re hard facts.
How should spills or leaks of T-Butyl Hydroperoxide be managed?
T-Butyl Hydroperoxide and the Risks on the Floor
T-Butyl hydroperoxide stands out as a strong oxidizer. Some call this chemical tricky because contact with flammable materials or certain metals can spark fires or explosions. Once a spill hits the floor, the clock starts ticking. Fumes start rising, and even brief exposure can irritate the eyes, skin, or lungs. Nobody wants a chemical burn or a trip to the emergency room from a routine clean-up.
Immediate Steps: Set the Scene for Containment
Grab control before chaos takes over. The first thing anyone in the area must do is move away from the spill if possible and alert other workers. I remember seeing a spill at a small research lab; the quick-thinking tech kept us safe by announcing the situation and hitting the ventilation controls in one smooth move. A good emergency plan gets everyone out of harm's way before things escalate.
Building ventilation matters. Never try to manage volatile chemical leaks without firing up all exhaust fans in the room. Strong airflow moves fumes out fast and limits the chance for anyone nearby to breathe in hazardous vapors.
Personal Safety: Suit Up
Anyone facing a T-butyl hydroperoxide leak must toss on the right set of gear: chemical goggles, gloves made from nitrile or neoprene, a lab coat, and if needed, a face shield. The chemical burns through regular work gloves in no time. Respirators rated for organic vapors keep noses and lungs safe if the amount spilled is large or the air feels heavy with chemicals.
Containment: Controlling the Spread
Quick damming stops the puddle from crawling farther along the floor. Materials like towels, pads, or sand work well. Skip sawdust, paper, or rags made from natural fibers. T-butyl hydroperoxide reacts with organic material. A fire in the middle of a spill only makes things worse. People sometimes overlook this, and factories have gone up in flames as a result.
For larger spills, special absorbent pads designed for chemicals are worth every penny. In schools, labs, or warehouses storing this oxidizer, stock these pads and review spill-cart supplies every quarter. It’s not enough to have gear gathering dust; up-to-date material can save hours of cleanup and maybe a life.
Clean Up and Disposal: Finish Strong
Once the liquid gets soaked up, scoop and transfer everything—pads, sand, or loose debris—into a proper waste drum lined with heavy-duty plastic. Never throw chemical waste in regular trash bins. Contact the environmental health and safety office, as they arrange for hazardous waste pickup. Every drum or bag should get labeled clearly so no one accidentally handles it without protective equipment.
Splashing water directly on a fresh spill causes heat, possible splattering, and increases danger. Instead, rely on absorbent materials first, only rinsing the area thoroughly with water once all residue clears. For ground-level spills outdoors, form a berm with earth to keep the chemical from reaching nearby drains.
Local agencies demand incident reports for most chemical leaks, no matter the size. People often see paperwork as a nuisance, but these records point out weak spots in training, storage, or facility design. Continuous improvement comes from learning, not luck.
Building a Broad Culture of Awareness
Regular safety drills keep team members sharp. Written instructions stuck to spill kits or on laboratory walls help everyone stay calm under pressure. New hires appreciate a mentor walking through emergency steps long before they face a real spill. No shortcuts—practice counts for more than instructions buried in a manual.
Fire extinguishers suited for chemical fires should never get stashed in a corner. Walk through exits and equipment locations every month so that response time drops when someone really needs help. Storage zones need strong ventilation, flame-proof cabinets, and solid containment trays.
T-butyl hydroperoxide spills don’t just test protocols—they reveal if a workplace pays attention to detail, values preparation, and looks out for every person who comes through the door. Building a mindful culture beats luck every time.
What are the main safety precautions when working with T-Butyl Hydroperoxide?
Real Risks In Real Life
Anyone who works in a chemical lab long enough knows you don’t mess around with organic peroxides. T-Butyl hydroperoxide, a staple for certain oxidations and polymerizations, belongs to the type of stuff that scares you into caution. Most accidents I've heard about traced back to folks moving too fast or skipping simple safety steps. This chemical sits on the shelf looking innocent, but it harbors explosive potential—literally. It releases oxygen fast. A small mistake can spark a violent reaction or serious fire.
Personal Protective Gear Is Non-Negotiable
When I first used T-butyl hydroperoxide, my supervisor double-checked my gloves. I thought he was overdoing it, but one spill shut that thought down. This compound eats right through thin gloves. Heavy nitrile or neoprene gloves stay intact under exposure. Safety glasses only do so much; full face shields keep splashes out of your eyes. Chemical-resistant lab coats stay a step ahead of light cotton, and closed-toed shoes finish the line of defense.
Keep It Cool—Literally
Many new chemists learn temperature’s importance by cleaning up broken glass. T-butyl hydroperoxide turns nasty at higher temperatures, so refrigerators rated for flammable storage become your best friend. Heat, even sunlight through a window, speeds up decomposition, which means the build-up of gas pressure and a possible blast. I always store this stuff in small, clearly labeled containers. Large quantities in a single bottle increase the risk if something goes wrong.
No Sparks, No Static, No Smoking
I once saw a stray static shock ruin a good day in a chemistry lab. Peroxides ignite from sparks, friction, or impact—common in busy labs. Grounded equipment and anti-static mats make life easier. Never open bottles near open flames, hot plates, or electrical devices that spark. Throw “No Smoking” signs in the mix and enforce them. T-butyl hydroperoxide fumes creep up fast and don’t check ID at the door.
Ventilation Makes A Real Difference
A fume hood becomes more than just a suggestion with this chemical. I’ve watched seasoned researchers go out of their way to avoid pouring T-butyl hydroperoxide on open benches, instead working under strong vents. The vapors can irritate lungs and eyes within seconds. It doesn’t take much for vapors to accumulate if things get stuffy. Keeping work areas well-ventilated reduces the odds of dangerous concentrations and nasty surprises.
Cleaning Up And Disposing Safely
I can’t count the number of times someone took a shortcut with chemical waste. With T-butyl hydroperoxide, lazy disposal invites disaster. Dedicated peroxide disposal containers save lives. Neutralizers help, but you have to know which ones actually work. Paper towels soaked in this chemical catch fire on their own. Label everything—every beaker, every flask, every bit of waste.
Training And Awareness Matter More Than You Think
No policy beats a culture of vigilance. Training sessions about storage, handling emergencies, and correct spill responses keep everyone ready. Newcomers learn to respect the chemical before ever popping a cap. Share accident stories—not to scare, but to make the risks real. Risk assessments help catch hidden dangers before a project gets underway.
The Responsibility Sits With All Of Us
Lab safety isn’t just procedure; it’s personal. When working with T-butyl hydroperoxide, everyone has a story where precaution saved the day. Take these lessons seriously, and you keep dangerous incidents off your list.
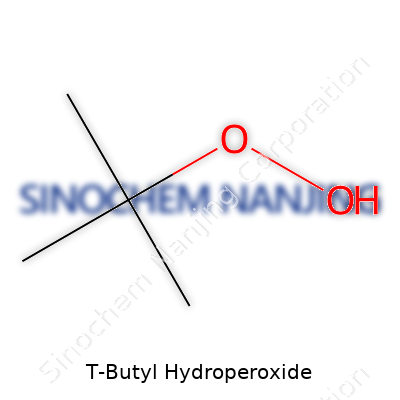
| Names | |
| Preferred IUPAC name | 2-methylpropan-2-yl hydroperoxide |
| Other names |
TBHP tert-Butyl hydroperoxide tert-Butyl peroxide t-Butyl hydroperoxide t-BuOOH |
| Pronunciation | /ˌtiː ˈbɜːr.tɪl haɪˌdrɒk.sɪˈpɜːr.ɒksaɪd/ |
| Identifiers | |
| CAS Number | 1314-29-2 |
| Beilstein Reference | 3581592 |
| ChEBI | CHEBI:53088 |
| ChEMBL | CHEMBL185086 |
| ChemSpider | 16215 |
| DrugBank | DB03850 |
| ECHA InfoCard | 100.102.341 |
| EC Number | 'EC 201-254-7' |
| Gmelin Reference | 2045689 |
| KEGG | C06507 |
| MeSH | D014445 |
| PubChem CID | 6410 |
| RTECS number | RD3325000 |
| UNII | 7E4PHP5N1D |
| UN number | UN3109 |
| CompTox Dashboard (EPA) | DTXSID2020606 |
| Properties | |
| Chemical formula | C4H10O2 |
| Molar mass | 90.12 g/mol |
| Appearance | Colorless liquid |
| Odor | pungent |
| Density | 0.94 g/cm³ |
| Solubility in water | Miscible |
| log P | 0.3 |
| Vapor pressure | 23 mmHg (20°C) |
| Acidity (pKa) | 13.0 |
| Basicity (pKb) | Basicity (pKb): 12.8 |
| Magnetic susceptibility (χ) | -5.1·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 1.32 mPa·s (25 °C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 209.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -161.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –3397 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H272, H302, H314, H332, H335 |
| Precautionary statements | H242, H302, H314, H332, H335, P210, P220, P221, P260, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P330, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2-OX |
| Flash point | 15 °C (59 °F; 288 K) (closed cup) |
| Autoignition temperature | 410 °C (770 °F; 683 K) |
| Explosive limits | Lower 2.5%, Upper 18% |
| Lethal dose or concentration | LD50 oral rat 382 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 382 mg/kg |
| NIOSH | NIOSH: EZ3150000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of T-Butyl Hydroperoxide: 0.07 ppm (OSHA Ceiling) |
| REL (Recommended) | 2-8°C |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
tert-Butyl alcohol tert-Butyl peroxide Hydrogen peroxide |

