Succinic Anhydride: Understanding Its Roots, Uses, and Where It’s Headed
Looking Back: Where Succinic Anhydride Started
The story of succinic anhydride stretches back through centuries. Succinic acid, its parent molecule, pops up in the pages of early chemistry books from the 16th and 17th centuries, coming out of the distillation of amber—hence the name’s Latin root, “succinum” for amber. The key breakthrough for succinic anhydride arrived in the 1800s, as chemists learned how to dehydrate the acid to make this versatile compound. From there, chemical manufacturers pushed the boundaries in both scale and purity, turning an obscure academic material into one that serves industries from pharmaceuticals to polymers. The growth in synthetic organic chemistry and an expanding plastic industry after World War II planted succinic anhydride squarely in the toolkit of industrial chemists.
What Succinic Anhydride Brings to the Table
Succinic anhydride shows up as a white, crystalline solid. It carries the sharp tang shared by other acid anhydrides, able to sting the nose in a poorly ventilated lab. Its molecular structure—basically two carboxylic acid groups latched together after tossing out a water molecule—gives it a temperamental, highly reactive character. This quality makes it valuable for making all sorts of compounds, from polyesters to agricultural chemicals. In the lab, that dry, crumbly look means it keeps well in a sealed bottle, but give it a chance, it’ll snag water vapor from the air and turn right back into succinic acid. So storage and handling take a steady nerve and reliable lab technique.
Key Physical and Chemical Properties
Succinic anhydride doesn’t need fancy descriptions. If you’ve handled it, you know its melting point hovers just above 120 degrees Celsius, so it’s easy to work with using standard glassware. It dissolves in organic solvents—ether, acetone, and similar common lab fare—but steers clear of water since it reacts instantly. This reactivity with water, alcohols, or amines gives it a strong spot as a platform chemical: once the ring opens, chemists can swing reactions in all sorts of directions from there. Its stability under dry conditions contrasts with its feisty response to moisture, which anyone who’s watched an open bottle clump up on a humid day can attest to.
The Technical Demands and Labeling
Working with succinic anhydride means keeping up with thorough documentation. Labels mark out its hazardous nature: skin and eye irritation are no joke. Tech specs demand a sharp balance between purity and practicality. Labs need it clean to avoid unpredictable byproducts, while industrial users often accept minor impurities to keep costs down. As industrial quality control improved, suppliers listed batch numbers, storage instructions, and clear warnings. I’ve caught more than one newcomer in the lab miss a label and get an afternoon rerun of handling procedures as a result.
The Methods Behind Manufacturing
Chemical plants most often make succinic anhydride by dehydrating succinic acid, which itself comes from both traditional petrochemical routes and more recently, fermentation methods using renewable biomass. Catalysts speed up the water-removal step. Over the years, interest turned toward greener synthesis paths. Today, companies look for bio-based routes, running sugar-rich feedstocks through genetically engineered microbes to churn out succinic acid before anhydride conversion. It’s a bit like watching the industry push to wring every last bit of fossil fuel dependence out of an old textbook reaction.
Chemical Reactions and Laboratory Modifications
This anhydride acts like a key for opening up countless reactions. Organic chemists value how quickly it reacts with nucleophiles—alcohols, amines, or water. You’ll catch it often in an acylation step, slapping succinyl groups onto everything from starch to proteins to give them new chemical features. Where polymer chemistry gets involved, the anhydride forms the backbone (sometimes literally) of easy-to-process resins and polyesters. I remember learning how a well-timed addition of succinic anhydride could tune the flexibility of a polyester resin, making the jump from brittle to tough possible with a tweak in recipe.
Synonyms and Tradename Confusion
Names sneak up differently across countries and industries. Succinic anhydride might get called butanedioic anhydride or succinyl oxide depending on the catalog or application. This can trip up inexperienced buyers and research teams, especially anyone crossing international lines in procurement or regulatory compliance. No one likes discovering two bottles of the same substance under different names halfway through a trial. The nitty-gritty of chemical language—never just about academic precision, but about real dollars and lost time in the wrong order.
Meeting Safety and Operational Standards
Handle succinic anhydride with respect and well-ventilated lab space. Those white crystals irritate skin, eyes, and lungs. The Safety Data Sheet reads like a warning sign, and the injuries need serious attention. Proper gloves and goggles are standard; a spill on bare skin feels like running into an angry patch of poison ivy. As environmental regulations toughened up, disposal and emissions guidelines clamped down too. Companies set up closed systems and strict monitoring, keeping emissions in check and staying out of regulators’ crosshairs. In my own lab experience, minor mishaps around open containers drove home the reason for bending over backward to follow protocols—otherwise the risk outweighs the reward in a hurry.
Areas of Application: Where Succinic Anhydride Leaves Its Mark
Look at the reach of succinic anhydride and you get a glimpse into the interconnected world of modern chemistry. Its biggest uses come in making polymers and resins, especially alkyd and polyester types, tailored for coatings, adhesives, and textiles. It’s also a critical link for making surfactants and some biodegradable plastics that land in medical devices or packaging. On top of that, pharmaceutical firms use it to craft key intermediates when building up complex molecules for drugs—everything from antibiotics to anti-inflammatories. Baselining applications further, food processing also dips into succinic anhydride derivatives, but regulations clamp down hard, and only with careful process controls. Anyone who’s ever worked in a chemical plant knows these connections aren’t accidental—it’s about squeezing as much value from a versatile molecule as possible.
Research, Development, and Innovation
In the research world, succinic anhydride stands as a launchpad for academic exploration and licensing deals. Much focus lands on greener synthesis processes—cutting down carbon output, ditching toxic solvents, or swapping out fossil-derived feedstocks. Many academic groups push for microbial platforms that take waste plant material and turn it into high-purity succinic acid, then swing cleanly over to the anhydride. In my own time in the university lab, collaborative groups chased patents around these bio-based tweaks, trying to offer industry something scalable, cost-effective, and friendly to both workers and the environment. These efforts pay off not only for sustainable chemistry but for building stronger global supply chains less prone to oil shocks and geopolitical swings.
Toxicity Studies: Knowing the Risks
Toxicologists have looked closely at succinic anhydride, juggling industrial benefit against potential harm. Acute exposure causes irritation—a fact no seasoned chemist overlooks. Studies show repeated low-level exposure might affect respiratory health or skin, especially without proper PPE or exhaust systems. Animal studies show that high doses can trigger inflammatory responses, but regulatory agencies draw clear lines to prevent excessive worker contact. No widespread chronic toxicity has emerged, but the safe path runs through careful workplace design and ongoing exposure monitoring. Workers trust that companies stay ahead of any risk, knowing full well that small lapses can turn into lawsuits or regulatory headaches. For community exposure, well-run plants and regulatory oversight have kept emissions under control, and no public health warnings have surfaced at the concentrations used in manufacturing.
Where Things Are Heading Next
Peering into the future for succinic anhydride, interest builds in making everything more sustainable. Companies line up investments in bio-based production, aiming for lower carbon footprints and safer working conditions. Flexible, biodegradable plastics created from succinic anhydride–based polymers look set to carve out a bigger chunk of the green packaging market. Specialty chemicals for electronics, personal care, and new medicine candidates stand to benefit as well. Regulatory agencies will likely keep a close watch, especially as more applications touch foods, consumer products, and health care. It turns out even something as old as succinic anhydride keeps finding new relevance—caught between chemical tradition and the push for sustainable industry. As new markets and environmental standards raise expectations, the future for this compound looks as challenging as ever, but also full of promise for those willing to innovate.
What is Succinic Anhydride used for?
A Hidden Ingredient in Everyday Life
Succinic anhydride rarely grabs headlines, yet its presence touches plenty of industries. This white, crystalline compound often gets its start in the lab from maleic anhydride, then heads off to factories making things that people use or encounter every day.
Producing Plastics and Polymers
A huge chunk of the world’s succinic anhydride transforms into specialty resins and polymers. These materials show up in our homes through plastic handles, paints, and even adhesives. Manufacturers use it to make plasticizers, which keep plastics flexible instead of brittle. Epoxy hardeners also rely on it, making possible all the sturdy coatings and glues that stay put rather than crumbling within months.
Shaping Medicine and Agriculture
Some pharmaceutical companies use succinic anhydride to craft intermediates, which later become key ingredients in active drugs. These might treat infections or regulate enzymes in the body. Modern agriculture pulls from the same pool, using it to build herbicides and plant growth regulators. Without these, crops may not thrive and yields may suffer, especially in places battling pests or poor soil.
Improving Food Quality
The food sector has found its own ways to squeeze value out of succinic anhydride. It pops up in food additives that stabilize flavors and improve texture. Some modified starches and sweeteners, which keep sauces smooth or help baked goods hold together, owe a little of their performance to this compound. Its safety in food applications depends on careful regulation and strict production controls. The U.S. Food and Drug Administration allows certain uses but keeps a sharp eye on exposure.
Chemistry Driving Industry
Paints, dyes, detergents, and lubricants all tap into the chemical’s reactivity. The anhydride group bonds well with other molecules, creating compounds that perform much better than their starting ingredients. This means tougher paint finishes, longer-lasting lubricants, and dyes less likely to fade in sunlight. Chemical engineers and formulators depend on it for consistent results.
Keeping Workers Safe
Anyone handling succinic anhydride in a factory or lab should stay careful. Skin and eyes react badly to its dust. Long-term studies point to irritation more than chronic diseases, but with so many chemicals around, companies stay prepared. Good ventilation, ventilation, and protective gear play a daily role in factories that see big volumes of the stuff.
Green Chemistry and What Comes Next
There’s a push for greener ways to make chemicals, and bio-based routes to succinic anhydride are taking off. Instead of starting with petrochemicals, producers ferment renewable sugars, cutting down on carbon emissions. Some startups already supply bio-succinic acid, which gets turned into the anhydride using less energy and fewer toxic reagents. That holds promise for those aiming to shrink the industry’s footprint.
Focus on Safety and Sustainability
Regulators continue to track the uses and health impacts of succinic anhydride. Industry leaders publish new findings about safety, making progress more transparent. In my years visiting factories that handle the stuff, I’ve seen how simple steps, like updating training and sharing hazards, keep workers healthy and boards at ease. Bringing chemistry out of the shadows, discussing its benefits and risks plainly, helps everyone—from shop floor to scientist—make smarter choices.
What are the safety precautions when handling Succinic Anhydride?
Working With Something That Bites Back
Succinic anhydride gets used in a bunch of everyday industries, from pharmaceuticals to making plastics. Its uses highlight its importance, but handling it without thinking can land someone in trouble. I remember an old lab supervisor drilling one point into everyone's head: always respect splashes, dust, and vapors, because they don’t play favorites. Eyes and lungs complain the loudest, but the skin notices just as quickly.
Eye and Skin: Not Just Minor Irritations
Even the keenest chemist can get caught off guard by a dash of powder in the air. Succinic anhydride stings like crazy on exposed skin, giving burning or reddened patches, and it really gets dangerous in the eyes. The corrosive bite of this compound makes thick gloves, goggles, and a fitted lab coat more than just box-checking items—they serve as the frontline defense. But not all gloves are equal: natural rubber, nitrile, or neoprene gloves block this stuff best.
Clean Air and Smart Storage
The sharp smell hints at the chemical’s volatility. Trust those fumes as a warning sign. Good labs take ventilation seriously, and a proper fume hood does more than protect from odors—it shields lungs from harm, especially if the powder's airborne during weighing or mixing. Before starting, double-check the fume hood: sash sitting low, airflow working.
Every container needs a cap. Moisture in the air starts breaking succinic anhydride down, often creating heat and sticky mess. I once watched someone rush and tip a jar, only to discover that cleaning up a spill in the open air creates a world of regrets—strong odor, itchy skin, and that uneasy knowledge that bad habits just caught up.
Training: The Ingredient That Doesn’t Expire
A lot of accidents come down to a simple lack of know-how. It doesn’t take long to read the safety data sheet, but actually understanding what the warnings mean requires hands-on training. Practice with the eyewash station and safety shower. Know where to toss contaminated gloves. Regular rehearsals help keep instinct sharp; nobody wants to fumble when something stings or burns.
Simple Habits, Big Impact
Working clean isn’t about checking off rules—it’s about coming home with the same number of eyes and fingers you started with. Keeping hands away from the face, sealing up bottles, and not walking away from an open jar can mean the difference between just another workday or an urgent care visit. Never eat or drink near the workbench, and stash phones far from chemicals.
Waste: Treat It Like It Bites
This isn’t trash for the usual landfill. Used wipes, gloves, or leftover powder should go into labeled hazardous waste bins. Improper disposal can ruin equipment, send strange odors through a building, or endanger waste handlers who never signed up for exposure. I once saw a bottle left in a regular bin—within hours, it had eaten right through the lining. It's better to sort and label waste right after finishing the work.
Fresh Thinking Saves More Than Just Paperwork
Respect for succinic anhydride doesn’t require fear, just clarity and care. Smart habits, practical gear, and healthy skepticism for shortcuts keep the lab safe for everyone. Mistakes stay fewer and memories stay good.
What is the chemical formula of Succinic Anhydride?
Understanding the Basics: What Is Succinic Anhydride?
Succinic anhydride stands out in the world of organic chemistry. Known for its white crystalline form and strong, penetrating odor, this compound has a chemical formula of C4H4O3. The structure gives it a reactive edge, with two adjacent carbonyl groups joined in a five-membered ring, making it a prime ingredient in more than just academic experiments.
Everyday Uses and Real-Life Impact
During undergraduate lab days, working with succinic anhydride taught me how versatile some chemicals can be. This compound goes beyond textbooks, earning its stripes in industries like pharmaceuticals, polymers, and even agriculture. Each sector finds a reason to reach for it.
Manufacturers use succinic anhydride as a core reagent for synthesizing compounds such as succinimide. Succinimide shows up in medicines, especially anticonvulsant drugs for epilepsy. Antibiotic production sometimes starts with this same building block. When companies break down big molecules—like starches—using succinic anhydride, they end up with food additives that help keep your favorite snacks fresh, textured, and shelf-stable. From headache medications to everyday snacks, the reach goes further than most people think.
Industry Demands and Worker Safety
Production plants regularly handle significant quantities of succinic anhydride. Its broad application means that improving safety protocols directly affects thousands of workers. Inhalation or skin contact can irritate or even damage tissue. Wearing the right gloves or masks, ensuring robust ventilation, and clear hazard labeling—each measure prevents long-term harm. My time volunteering at a local science outreach program taught me that simple demonstrations about chemical safety can change habits for the better, especially for those just entering technical fields.
The Future: Sustainability and Waste Reduction
Making succinic anhydride once relied mostly on fossil fuels, adding another layer to the talk about sustainability in manufacturing. Advances now let companies turn to biosynthetic pathways, using plant-based sources or even waste materials to extract succinic acid, then convert it to the anhydride form. This shift cuts down on environmental impact and supports the move toward greener chemistry. Responsible sourcing doesn't just help the planet; it often brings cost benefits for factories over time, especially as pressure from environmental agencies rises.
Knowledge Builds Trust
Chemicals often get a bad rap. Getting to know the facts—like the chemical formula C4H4O3 for succinic anhydride—can empower people to ask smarter questions about the products in their daily lives. Sharing accurate information from trusted references, such as peer-reviewed journals and regulatory bodies, reinforces public awareness. It reminds me how important credible sources remain, especially in a world full of quick takes and rumor-driven headlines.
Moving Forward: Open Communication and Lifelong Learning
Improving chemical literacy opens doors for safer workplaces, better environmental choices, and stronger consumer confidence. Whether a person manages a lab, teaches high school chemistry, or just wants to understand ingredient labels, clarity about compounds like succinic anhydride keeps progress moving in the right direction. Staying curious and informed beats fear-based thinking every time.
How should Succinic Anhydride be stored?
Understanding Succinic Anhydride’s Nature
I’ve spent a good deal of time around labs and storage rooms, and I’ve seen firsthand that safe chemical storage isn’t just about following a checklist. Succinic anhydride sounds intimidating, but store it the right way and it’s manageable. This chemical starts as a white crystalline solid. Give it access to moisture, and it quickly turns to succinic acid. That by itself might clog up your process, but worse, the substance also causes irritation to skin, eyes, and respiratory passages. If you let it react with water or strong bases, it can release heat, making storage conditions especially important.
Picking the Right Container
Choose containers that truly seal out moisture. Keep succinic anhydride in tightly closed glass or specifically designed plastic containers. Loose lids spell trouble. I’ve seen cracked containers after someone tried using an old repurposed jar—don’t cut corners here. Manufacturers ship this material in airtight packaging for a reason. Any leaks allow water vapor inside, ruining quality and risking a corrosive mess. Don’t use metal unless you know the alloy resists acid; corrosion isn’t just a pain to clean up—it can mean contamination of your whole batch.
Conditions Make All the Difference
Don’t stash succinic anhydride in a hot corner of the warehouse or a sunlit cabinet. Store it in a cool, dry, well-ventilated space. Heat accelerates its tendency to react, especially in humid climates. Even a sudden summer heat wave can make sealed containers sweat inside. Try to target a steady, moderate temperature if you want your supply to last. Forget about shelving it above tanks of acids, bases, or oxidizers. Mixing up your storage space sometimes leads to spills and reactions, and it’s rarely by design. Give this substance its own shelf, ideally with labels that stand out—no one should grab the wrong jar by mistake, especially under pressure during busy shifts.
Know the Risks—Protect People and the Supply Chain
People can underestimate how dangerous accidental exposure gets with this chemical. No one wants to pull open a container only to come face-to-face with a waft of harsh fumes. Always use gloves and splash-proof goggles. In a small tech startup lab, I watched a team skip goggles during a late-night cleanup, thinking it would make things quicker. That single shortcut left someone with a serious eye injury. No shortcut makes up for risking your health.
Even the best-labeled containers need regular checking. Over time, seals degrade or get knocked out of place. Assign someone the responsibility to check storage every week—I’ve seen problems go unnoticed just because everyone thought someone else was on it. Promptly disposing of compromised containers prevents the spread of contamination and protects staff. For spills, keep spill kits nearby and train staff on their use. Fires are rare but possible in case of contact with strong oxidizers, so never keep it near oxidizing agents and ensure the right fire extinguisher is on hand.
Prevention Pays Off
Reviewing your chemical inventory frequently and providing real-world training on handling and storage means fewer costly accidents or ruined materials. Label every container, teach the hazards, and store succinic anhydride away from water, heat, and incompatible chemicals. This approach delivers safer workspaces and better outcomes for everyone involved.
Is Succinic Anhydride hazardous to health or the environment?
Where Succinic Anhydride Shows Up
Succinic anhydride has carved out a niche in the worlds of polymers, pharmaceuticals, and resins. Factories use it to create plastics and certain prescription drugs. Working in a chemical plant taught me that even routine chemicals can pose trouble, depending on how people handle or dispose of them. I’ve seen the shipping drums stamped with hazard labels that demand respect, not fear.
The Health Concerns
Most folks never run into succinic anhydride outside of a professional setting, but for people in manufacturing or labs, exposure becomes a real concern. This compound brings the potential to irritate airways, skin, and eyes. I remember a technician who let his gloves slip during an equipment check and walked away with red, stinging hands. Succinic anhydride reacts with water—including the moisture on skin—producing succinic acid, which has a bite. The fumes aren’t much better. Inhaling its dust in a poorly ventilated room leads to coughing and discomfort.
Prolonged exposure with little protection pushes risks higher. Respiratory issues, eye inflammation, and even the possibility of longer-term effects pop up in some scientific literature. Even though plenty of substances are more hazardous, complacency here leads to injury or days missed from work. The solution isn’t complicated. Fresh air and good safety gear do most of the heavy lifting, but not every workplace keeps standards as high as they claim. Training and enforcement still make a difference.
Environmental Impact
Chemicals always find their way somewhere. Spills, improper storage, and poor disposal all create routes to air, water, or soil. Succinic anhydride reacts fast with water, so big releases into rivers or lakes don’t hang around as the original chemical for long. That doesn’t mean no risk—its acidic reaction products can mess with local water chemistry and aquatic life. I’ve lived near industrial riverbanks long enough to recall fish die-offs after even small pH shifts.
On land, smaller leaks seep into soil, where they can throw off microbial life or plant growth. Luckily, this compound tends to break down rather than persist, though repeated or careless dumping ramps up the threat. There isn’t yet a big case of succinic anhydride damaging local wildlife for years, but that doesn't give industry a pass to skip proper controls. My experience says sloppy storage or shortcuts in waste handling have a way of snowballing—and cleanup costs multiple times more than safe disposal up front.
Tackling the Issues
Industry stands to learn from places that get safety and disposal right. Regular worker training, visible warning labels, and well-maintained personal protective equipment all help. I’ve found that giving employees a voice to report unsafe conditions without fear of reprisal goes farther than fancy compliance certificates.
On the environmental front, it boils down to secondary containment, emergency spill plans, and pressure to treat or neutralize waste before it escapes offsite. Modern plants invest in sensors and monitoring to catch leaks early, but old facilities often lag behind. Regulators and watchdogs, in my experience, play a crucial role where company motivation thins out. Real teeth behind environmental rules, transparent incident reporting, and community right-to-know policies keep everyone honest.
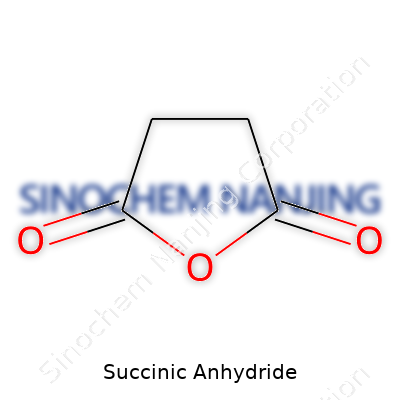
| Names | |
| Preferred IUPAC name | 2,5-Dioxotetrahydrofuran |
| Other names |
Butanedioic anhydride Dihydro-2,5-dioxofuran Succinic acid anhydride Succinyl oxide 2,5-Furandione |
| Pronunciation | /ˈsʌk.sɪ.nɪk ænˈhaɪdraɪd/ |
| Identifiers | |
| CAS Number | 108-30-5 |
| Beilstein Reference | 1207002 |
| ChEBI | CHEBI:17921 |
| ChEMBL | CHEMBL1402 |
| ChemSpider | 54619 |
| DrugBank | DB03624 |
| ECHA InfoCard | 100.006.232 |
| EC Number | 2.6.1.19 |
| Gmelin Reference | 1434 |
| KEGG | C01082 |
| MeSH | D013429 |
| PubChem CID | 7923 |
| RTECS number | WS8225000 |
| UNII | 0P6K7HM2UF |
| UN number | UN2215 |
| Properties | |
| Chemical formula | C4H4O3 |
| Molar mass | 100.07 g/mol |
| Appearance | White crystalline solid |
| Odor | Pungent |
| Density | 1.43 g/cm³ |
| Solubility in water | Decomposes |
| log P | -0.4 |
| Vapor pressure | 0.04 mmHg (25°C) |
| Acidity (pKa) | 4.2 |
| Basicity (pKb) | -6.55 |
| Magnetic susceptibility (χ) | -54.0e-6 cm³/mol |
| Refractive index (nD) | 1.487 |
| Viscosity | 1.056 mPa·s (at 40°C) |
| Dipole moment | 2.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -587.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1795 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB26 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H334, H335 |
| Precautionary statements | P261, P280, P304+P340, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2-W |
| Flash point | 205°C |
| Autoignition temperature | 445 °C |
| Explosive limits | LEL: 1.1%; UEL: 6.6% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1780 mg/kg |
| LD50 (median dose) | LD50 (median dose): 500 mg/kg (rat, oral) |
| NIOSH | SN1225000 |
| PEL (Permissible) | PEL: 0.5 mg/m3 |
| REL (Recommended) | 25 kg |
| Related compounds | |
| Related compounds |
Succinic acid Maleic anhydride Glutaric anhydride Phthalic anhydride Adipic acid |

