Looking Closer at Styrene: Innovation, Risks, and Reality Beyond the Lab
The Roots and Rediscovery of Styrene
Styrene comes with a backstory that stretches further than most realize. Chemists first noted its existence in the late 19th century, starting with resin extractions from trees like storax. Synthetic methods soon took over, and by the early 20th century, clever minds found safer routes to produce it from ethylbenzene. During World War II, shortages in rubber forced large manufacturers to scale up styrene-based plastics as substitutes, showing how necessity drives creative solutions. Tracking the transition from tree resin to large-scale synthetic production paints a picture of how the search for new materials never stands still, but keeps responding to practical pressures from the world around us.
What Styrene Is—From Monomer to Polystyrene
People know styrene mostly through what it becomes: polystyrene foam, clear plastics, even parts for cars and refrigerators. In its raw form, styrene stands out as a colorless, oily liquid with a sharp, recognizable smell. Polystyrene, which springs from styrene through polymerization, shows just how versatile one molecule can be—providing everything from lightweight packaging to tough cases for home electronics. That journey, from a faint-smelling liquid to everyday solid things, brings chemistry right up against daily life.
Physical and Chemical Features: Telling Details
Anyone who has uncorked a drum of styrene will smell its sweetness and maybe get a slight headache, underscoring the need for careful handling. Its boiling point sits close to 145°C, which spells out why it evaporates under room conditions, filling the air with its distinct odor. Styrene blends well with organic solvents, resists dissolving in water, and tends to stick around in any place where air quality goes unchecked. Its reactivity comes from the double bond in its structure, letting it snap together into long plastic chains or cross-link with other chemicals. Knowing these details shapes how factories design storage and transport, but it also raises health questions for workers.
Straight Talk on Technical Specs and Labels
Walk through any plastics plant and check the drums: labeling reveals more than just a name. Each container carries warnings about its flammability, its vapor’s health effects, and the steps needed to keep the area ventilated. Temperature control prevents runaway polymerization, which once triggered can generate heat and sticky messes. These labels go far beyond compliance—they bear witness to the lessons learned whenever safety slips were met with costly accidents or health claims. Industry standards set by groups like OSHA and the EPA came in response to real incidents, not just theoretical risks.
How Styrene Is Made: Sweat and Ingenuity
Decades back, most styrene came from dehydrogenating ethylbenzene. That step demands high temperatures, metal-based catalysts, and careful balancing to avoid side reactions—details that always felt more like engineering than chemistry to those who’ve worked in the sector. By the 21st century, new routes, such as using bio-based feedstocks, started coming out of university labs. These new approaches aim at cutting greenhouse emissions or reducing fossil fuel use. In research, cleaning up side products and capturing waste heat have become as important as wringing out that last percentage point of yield, showing that innovation in big chemistry takes patience and persistence, not short-term fixes.
How Styrene Reacts and Evolves
Chemists like the way styrene’s carbon-carbon double bond lets it snap into long chains. That quality fuels the production of high-impact polystyrene, acrylonitrile butadiene styrene (ABS), and unsaturated polyester resins. Every modification—whether adding rubber for toughness or blending with vinyl compounds—serves one purpose: to match up with new product demands. Each adjustment means tests for stability, impact resistance, or processability. This cycle goes on because new consumer products and environmental standards push both the boundaries of what’s possible and the responsibilities on those who shape these materials.
Names and Nicknames: More Than Chemistry Jargon
In labs and loading docks, styrene carries different labels: vinylbenzene, phenylethene, styrol, ethylenylbenzene. Commercial packaging sometimes swaps these for trade names, especially when sold as pure monomer versus blended resins. For those managing environmental releases or workplace exposures, accurate identification matters more than jargon, because mistakes echo in regulatory filings and emergency response.
Safety and the Tough Lessons from Industry
Styrene doesn’t forgive carelessness. The chemical’s flammability, its tendency to form thick vapors, and the chronic health issues that can arise from exposure—all demand vigilance. Regulatory guidance, shaped by years of workplace injuries and scientific review, calls for local exhausts, protective gear, and continuous air monitoring. While these requirements sometimes strain budgets, the history of legal settlements and medical claims shows that cutting corners on safety costs far more. Investments in process control, from drum storage to vapor capture, show up as fewer lost workdays and lower insurance payouts, benefits that often go unmentioned in board meetings but matter every day on the factory floor.
Where Styrene Goes After the Plant
Styrene-based materials fill up everyday life: coffee cup lids, packaging peanuts, insulation, and even parts of sports helmets. Transportation, medicine, toys, and electronics all lean on polystyrene’s clarity, impact resistance, and low cost. Recycling still poses challenges due to contamination, especially when food waste gets mixed up with plastic debris. Still, pressure from consumers and plastics bans are starting to move companies toward closed-loop systems and new forms of recycled resins. Every shift here affects jobs, supply chains, and landfill volumes, tying together choices made in boardrooms and city councils with street-level realities.
Research Focus: Cleaner Routes and Safer Products
Change comes slow in the chemical industry, but research has begun to shift away from just production efficiency toward ecosystem and human health. Universities and public labs run toxicity studies and lifecycle analyses. Emerging areas include developing bio-based styrene precursors, catalysts that work under milder conditions, and new additives to reduce the risk of workplace exposure. Every breakthrough faces a hard test: not just lab results, but real-world trials under commercial conditions. Some efforts stall; others reshape the industry standard over time.
Toxicity and the Debate Around Health
For most workers, the health questions around styrene aren’t theoretical. Stories circulate among plant staff about dizziness, skin irritation, and headaches, which have pushed for improved air monitoring and stricter personal protection rules. Scientific studies produced mixed messages for decades—animal data suggested cancer risks, while broad workforce studies failed to agree. The International Agency for Research on Cancer lists styrene as “possibly carcinogenic,” a label hotly contested by both plastics associations and health advocates. What matters to the public is less about the fine print of classifications and more about the simple need to keep workplaces and neighborhoods safe from spills, emissions, and improper dumping. Clearly, investing in exposure reduction and real-time air testing shouldn’t be up for debate.
The Road Ahead: New Uses and Challenges for Styrene
Demand for lightweight, durable plastics continues to grow, especially in electric vehicles, construction, and medical devices. Manufacturers now face the dual challenge of meeting these needs while cutting environmental harm. Bioplastics, improved recycling, and greener production methods come up often in conferences but still face economic and scale hurdles. People on the ground—workers, residents near plants, local governments—push for more transparency in emissions data and product safety. Styrene’s story, then, becomes less about just filling an order sheet and more about keeping pace with the impacts that its production and use scatter across society. The next generation of research, regulation, and technology will shape both the footprint of the chemical and the trust that underpins its place in modern life.
What is Styrene used for?
The Oddly Familiar Face of Styrene
Step into any living room, office, or car, and chances are good you’re surrounded by objects owing a debt to styrene. This clear, colorless liquid—originally derived from benzene and ethylene—shows up in plastics, synthetic rubbers, and resins. At first glance, the name might sound technical, but the reality sits right on the kitchen table. Coffee cups, refrigerator liners, even the foam packaging cradling electronics during shipping all point back to this single chemical starter.
Why the World Leans on Styrene
Most folks only think about plastic when tossing it in the recycling bin. On the production side, the story starts much earlier, with styrene helping build things like polystyrene and ABS (Acrylonitrile Butadiene Styrene). These plastics show up everywhere, from disposable utensils to safety helmets. The automotive industry leans on ABS for its strength. Kids play with toys made from the same base. Lightweight, strong, and shaped into almost anything, products built with styrene continue to flood the market.
Beyond Disposable Cups
Styrene’s role stretches beyond the obvious. Construction companies use it in insulation sheets, pipes, even certain structural panels. At my last house renovation, I saw how walls can be less drafty and more energy-efficient with the help of styrene-based foam boards, cutting down both noise and heat loss. In medical fields, technicians trust styrene-derived plastics for sterile instrumentation trays and housings, so hospitals can keep patient tools clean and safe.
Don’t forget about the paint can. Chemistry inside the can relies on styrene resins to stick pigment to the wall. These same resins show up in boat hulls and bathtub coatings, adding shine and toughness, so surfaces can handle daily wear.
Trade-Offs: Convenience and Challenges
Styrene built its reputation on being cheap, easy to work with, and reliable. Those coffee cups and packing peanuts haven’t gone unnoticed by environmentalists, though. Single-use items pile up, and most recycling centers don’t process expanded polystyrene due to low market demand and high processing costs. The EPA lists styrene as a potential human carcinogen, tying high exposure to increased health risks for workers in production plants.
Community groups in cities with styrene factories sometimes raise health concerns. Exposure mostly happens where air quality isn’t closely watched. Decades ago, I volunteered in a factory town where the air often smelled faintly chemical; parents worried, and the state stepped up air monitoring. Workers handling styrene every day use respirators and tight controls for a reason. Safer workplaces make healthier communities.
Solutions in Plain Sight
Companies look for alternative ways to produce familiar products, and researchers are digging into plant-based plastics. No single solution can switch off the global appetite for disposable cups overnight, but better transparency about what goes into these common goods can give consumers more choices. In my own household, we switched to glass storage containers and metal travel mugs, cutting down on single-use waste and oddly enough, saving money.
Policymakers can push for smarter recycling systems that accept more types of plastics. Tougher regulations on plant emissions and occupational safety defend both workers and neighbors. Supporting companies rolling out safer, more sustainable packaging brings new jobs and better products.
Styrene won’t disappear from the modern world soon. Paying attention to its role, both good and bad, means taking an honest look at the price behind everyday convenience.
Is Styrene hazardous to health?
Understanding Styrene and Everyday Exposure
Styrene shows up in more places than most people realize. It’s a key ingredient in producing polystyrene plastics and resins, which means it’s in everything from food containers to insulation and disposable cups. It’s not just something for industrial workers to worry about; just microwaving food in a cheap takeout container could mean exposure, as styrene can seep out of the plastic when heated.
Health Risks Linked to Styrene
Medical research highlights some serious risks. Breathing in styrene fumes can cause irritation of the eyes, nose, and throat. Workers in factories using styrene often report headaches, fatigue, and dizziness. Those who have spent years around high levels sometimes develop trouble remembering things or concentrating.
The longer-term risk always grabs more attention. Cancer agencies have reviewed studies and say there's enough evidence to call styrene “reasonably anticipated to be a human carcinogen.” This classification doesn’t mean one weekend painting a room with styrene-based resins poses an immediate threat, but people who breathe styrene at work for years stand a higher risk of getting leukemia or lymphoma.
Why Styrene Stays in Use
Styrene turns up everywhere because it’s cheap and versatile. Lightweight packaging helps keep food safe and costs down. Companies look for affordable ways to keep products fresh and shipping simple, so styrene-based plastics often win out, even if there are safer options out there.
Regulators walk a tightrope on this issue. Bans don’t come easy, especially when products are woven into so many parts of daily life. It took decades for agencies to fully recognize and regulate dangers like asbestos. Styrene, which appears less dramatic to the naked eye, receives slower action. Regulatory bodies in the United States and Europe have tightened exposure limits for workers, but pollution from plastic waste means most people still encounter low levels regularly.
Protecting Yourself and Your Community
Taking action starts with daily choices. Swapping single-use polystyrene containers for glass or paper can cut exposure. People can talk to local restaurants and shops about switching to safer packaging. At home, skipping the microwave for food that comes in flimsy plastic can help.
For workers, safety courses and protective gear go a long way. Ventilation in factories and regular air-quality checks reduce risks. Employers should keep workers informed about hazards linked to the chemicals on the job. No one should have to trade their health for a paycheck.
Communities can pressure lawmakers to take a closer look at materials allowed in consumer goods and to require clearer warning labels on packaging containing styrene. Stronger recycling programs and new ways to break down plastics would keep more styrene out of landfills and air, cutting back on the background levels everybody deals with.
Looking Forward
People began to understand the dangers of lead paint and asbestos after decades of harm. Styrene hasn’t drawn the same level of outcry, but the science suggests caution makes sense. Government-backed studies, independent reviews, and worker testimonies point in the same direction: lower styrene exposure means better health outcomes for all. As consumer habits shift and green alternatives catch on, demand for risky chemicals should drop, making room for safer, affordable materials that don’t put long-term health on the line.
What are the main properties of Styrene?
What Makes Styrene Stand Out
Styrene grabs attention across the plastics and chemical industries because it adapts without too many restrictions. Born as a colorless, oily liquid, the first thing many notice is its distinct, noticeable smell—a bit like sweet gasoline, easy to spot if you’ve been around a plant or a workshop. Factories shape it into many forms, turning it into countless products. Without styrene, most of us would not enjoy insulated coffee cups or lightweight vehicle parts.
Physical Strengths and Weaknesses
Take a look at how styrene handles heat and pressure. Its boiling point sits at just under 150 degrees Celsius, which puts it above many common organic solvents. Anyone melting down raw materials sees it hold for a decent stretch before vaporizing. Meanwhile, the glass transition temperature—where plastics like polystyrene turn from brittle to flexible—lands at roughly 100 degrees Celsius. This matters when thinking about packaging or medical trays that need to stay tough at room temperature.
Pour styrene and you find it flows smoothly, less viscous than syrup but not as runny as water. Because of this, mixing or pumping doesn't take special equipment. It evaporates faster than oil, so keeping containers sealed cuts down on losses and health risks—styrene vapors don’t do the lungs or nervous system any favors in closed spaces.
Chemical Nature
Styrene features a handy double bond in its structure. Chemists recognize this as a site where action starts. That double bond drives its use in making plastics—particularly when linking up into long chains called polymers. In my own lab experience, I saw students amazed as pure, liquid styrene turned solid, showing its reactive nature. Combine that with its aromatic ring—a set of six carbon atoms arranged in a stable circle—and you have a molecule eager to transform, yet stable enough for storage if you add stabilizers.
Benefits and Challenges in Industry
Styrene forms the backbone for polystyrene, ABS plastic, and synthetic rubber. Manufacturers love it because the final products stay clear, resist snapping under stress, and don’t weigh much. Lower density translates to shipping advantages: more product, less freight cost. A few decades ago, the rise of take-out containers and bike helmets happened because of reliable, moldable styrene-derived plastics.
Safety and environment worries follow styrene everywhere. It burns easily, releasing noxious smoke, which puts pressure on factories to store and handle it with care. Disposing of waste or recycling polystyrene isn’t straightforward—landfills fill up fast with foam products. Long-term exposure to vapor can hurt workers’ health, sparking stricter ventilation and monitoring rules. In my town, local regulators now require chemical plants to post regular air-quality reports because of styrene’s presence.
Looking Forward: Improvements and Solutions
Companies searching for less polluting manufacturing routes have started exploring bio-based styrene production. Breakthroughs in polymer recycling also gain traction. Mechanical recycling used to ruin clarity, but new approaches keep recovered plastic almost as clear and solid as original. Swapping out disposable foam for reusable or compostable alternatives has picked up momentum in packaging, slowly reducing the styrene footprint in consumer waste.
Styrene’s story holds lessons about progress and responsibility. It earned its important spot in everyday life because of flexibility and strength, yet calls for care at every stage—from production to trash pickup. Balancing convenience with health and environmental impact defines how people move forward with old chemicals like styrene.
How should Styrene be stored and handled?
Taming a Risky Chemical
Styrene shows up in plenty of products people use every day, from packaging materials to auto parts. Anyone who's worked in a plant knows styrene's sharp, sweet odor long before they read a safety sheet. Even if the stuff plays a big role in modern industry, the health hazards stick out. Breathing high levels of its fumes can cause dizziness, tiredness, and irritation. Over time, handling it the wrong way leads to bigger trouble for both people and the environment. That’s why folks on the factory floor feel nervous if upper management cuts corners.
Keeping Styrene Stable
Styrene doesn’t just evaporate quickly—it’s pretty reactive. The wrong kind of spark or even a hot day can kick off dangerous reactions. Anyone who worked a summer shift knows the stress when storage tanks heat up. Tanks and drums holding styrene stick to certain basics: cool, shaded spaces out of direct sun, away from open flames or heat sources. Metal tanks often use inert gas blanketing, usually nitrogen, to keep out oxygen and stop polymerization. The last thing anyone wants is a runaway reaction.A facility I visited in Texas kept their styrene stores under constant temperature checks. Workers took pride in daily logbooks. A thermometer’s cheap compared to the fallout from bad storage. Proper venting and fire protection gear surrounded those tanks, too. This isn’t just about box-ticking for inspectors – most workers have read stories about explosions caused by pressure build-up or shoddy maintenance in another town. That keeps people vigilant, not rules written by folks in distant offices.
Handling: Every Move Matters
Transferring styrene raises the stakes. Spills, splashes, toxic clouds – one mistake creates big headaches. Personal protective equipment comes standard: gloves resistant to chemicals, splash goggles, flame-retardant clothing. Old hands always warn the rookies that skin contact won’t just sting, it could lead to long-term health issues. Ventilation can’t be an afterthought – working in a closed room with volatilizing styrene is a recipe for trouble.During transfer or mixing, grounding and bonding equipment stops static sparks. More than one plant has had an incident tied to static electricity. It only takes one person slipping up or missing a frayed wire.
Emergency Planning Makes the Difference
It’s easy to forget about emergency plans until alarms blare. Facilities storing styrene keep spill kits, eye wash stations, and clean-up procedures handy. Firefighting crews practice with foam instead of water, since water sometimes spreads the chemical or increases the vapors. Training matters. A friend worked a fire at a plastics warehouse – he said the quick response, not fancy gear, kept it from turning into a regional disaster.Routine drills and open communication help stop panic during real emergencies. New hires should practice evacuation and know exactly where to find breathing masks. Local responders benefit from knowing exactly what the plant stores before disaster strikes.
Room for Improvement
Some facilities invest in early leak detection and constant air quality checks, using the latest monitors. Plant owners who care about their teams spend money on prevention, not just cleanup.The rise of stricter local regulations pushes companies to keep higher standards. Nobody wants to be the next headline about health risks or environmental damage. At the end of the day, safe storage and handling come down to treating people’s health and safety as more important than short-term savings. No shortcut matches the peace of mind earned from straightforward care and respect for this risky material.
Is Styrene environmentally friendly?
Understanding Styrene’s Impact
Styrene shows up in plenty of places—take-away coffee cups, insulation panels, packaging peanuts. The world uses millions of tons every year for plastics like polystyrene and resins for boats or bathtubs. The stuff got popular because it’s lightweight and affordable, and I’ve seen it make life simpler in some ways. Looking at whether it’s environmentally friendly isn’t about nitpicking science terms—it’s about real world effects and what happens after we toss it in the trash.
What Styrene Means for the Planet
Science shows polystyrene sticks around in the landscape. It doesn’t break down easily, and I’ve spotted white cups and broken foam coolers at too many campsites after busy weekends. This plastic fragments into tiny pieces—microplastics—which float in waterways and wash up on beaches. Researchers found polystyrene microplastics inside fish and seabirds. That alone says this material does real harm to wildlife. Even in the air, producing styrene can release emissions that affect both health and climate.
Health Worries Connect to Everyday Use
Beyond hurting wildlife, styrene’s vapors become a problem for workers in factories. The American Cancer Society lists styrene as “possibly carcinogenic.” Over several years, people exposed on the job have higher risks of certain cancers and nervous system issues. Some countries have started setting tougher safety rules for workers, but plenty of places fall short. At least for users, the exposure is generally low, but hot coffee in polystyrene cups can leach chemicals. Food safety agencies keep an eye on the limits, though safer alternatives take that concern away up front.
Styrene and Recycling: Not So Simple
I remember being told “just recycle it” whenever I asked about those foam trays at the grocery store. Truth is, most recycling centers don’t handle polystyrene. These foams cost too much to collect and ship, and lots end up in the trash anyway. The EPA estimates recycling rates for post-consumer polystyrene hover under 5%. Compare that to paper or basic plastics and the gap is obvious. Without infrastructure that handles it better, that packaging winds up buried or burned, releasing even more toxic pollution.
Finding Smarter Alternatives
Communities and some countries are phasing out single-use foams. Local bans in California and New York started turning up before COVID, and plenty of restaurants moved toward paper, bamboo, or compostable plastics. There is a cost—that’s true—but businesses and shoppers start getting used to bringing their own mugs and containers. My own coffee thermos cost less in the long run than a year’s worth of takeout cups. Bioplastics made from plants aren’t perfect, with their own quirks and limits, but ditching persistent pollution feels better.
Solutions Beyond Personal Choices
Fixing this problem isn’t a job for consumers alone. Governments need to set clear rules so that companies pay attention beyond profit. Extended Producer Responsibility puts more burden on manufacturers to collect and recycle what they sell. Some cities use cleanup fees that go straight into river and park maintenance. Scientists focus on developing new biodegradable materials that actually work without leaving microplastic behind.
Deciding if styrene is environmentally friendly means looking at the full picture. The evidence points to real problems for nature and people. It's hard to call a product green if it leaves a mark for generations. Rethinking what we use and demanding real solutions keeps our parks, water, and neighborhoods a bit cleaner for the next crowd coming along.
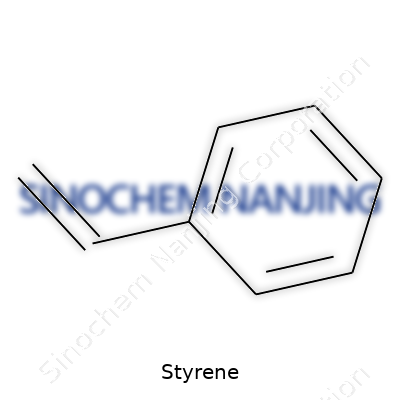
| Names | |
| Preferred IUPAC name | Ethenylbenzene |
| Other names |
Ethenylbenzene Vinylbenzene Phenylethene Cinnamene Styrol Phenylethylene |
| Pronunciation | /ˈstaɪəriːn/ |
| Identifiers | |
| CAS Number | 100-42-5 |
| 3D model (JSmol) | MPDGIYIXWLWPIH-UHFFFAOYSA-N |
| Beilstein Reference | 1460404 |
| ChEBI | CHEBI:27487 |
| ChEMBL | CHEMBL941 |
| ChemSpider | 5356 |
| DrugBank | DB03096 |
| ECHA InfoCard | 03bfa3e1-2981-4b14-96d7-1cd8254c2869 |
| EC Number | 202-851-5 |
| Gmelin Reference | Gmelin Reference: 51237 |
| KEGG | C00403 |
| MeSH | D013333 |
| PubChem CID | 7501 |
| RTECS number | WL3675000 |
| UNII | UN1107QTW8 |
| UN number | UN2055 |
| Properties | |
| Chemical formula | C8H8 |
| Molar mass | 104.15 g/mol |
| Appearance | Clear, colorless to yellowish, oily liquid with an aromatic odor |
| Odor | Aromatic, sweet |
| Density | 0.909 g/cm³ |
| Solubility in water | 0.3 g/L |
| log P | 2.95 |
| Vapor pressure | 0.61 kPa (20 °C) |
| Acidity (pKa) | 41.2 |
| Basicity (pKb) | 14.10 |
| Magnetic susceptibility (χ) | -6.7 × 10⁻⁹ |
| Refractive index (nD) | 1.546 |
| Viscosity | 0.763 cP at 25°C |
| Dipole moment | 0.92 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 146.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | ΔcH⦵298 = -4540 kJ/mol |
| Pharmacology | |
| ATC code | V04CX06 |
| Hazards | |
| GHS labelling | Warning, H226, H315, H319, H332, H361, H372, H304 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H226, H304, H315, H319, H332, H335, H361, H372, H412 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P272, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P312, P314, P331, P337+P313, P362+P364, P370+P378, P403+P235, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 52.0 °C |
| Autoignition temperature | 490 °C |
| Explosive limits | 1.1–6.1% |
| Lethal dose or concentration | LD50 oral rat 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Styrene: "5000 mg/kg (oral, rat) |
| NIOSH | 100-42-5 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | 700 ppm |
| Related compounds | |
| Related compounds |
Ethylbenzene Polystyrene Cinnamaldehyde Divinylbenzene |

