Sodium Succinate: Beyond the Basics—A Candid Look at Its Role in Science and Industry
Historical Development
Curiosity about sodium succinate’s story takes us back to the building days of biochemistry, when scientists sorted through the nuts and bolts of the Krebs cycle. It’s always surprised me how such seemingly mundane compounds unlock big doors in biology. Succinate as a raw molecule first turned heads during investigations into cellular metabolism. Over the years, folks tinkered in labs, combining succinic acid with sodium, which led to a salt that’s easier to handle and more stable in storage. What started in academic circles for dissecting metabolism quickly spiraled into food and pharma factories as the utility of sodium succinate grew clear.
Product Overview
Sodium succinate doesn’t crowd the shelves at grocery stores, but it lives in the background of plenty of packaging lists. It normally comes in granules or powder, white as flour, ready to mix and dissolve. You’d run across it more often if you hang out in food flavor labs or industrial kitchens. In the modern supply chain, there’s a steady call for it—from savory seasonings to injectable solutions in hospitals. Every batch comes stamped with purity grades depending on where it lands, but the core substance doesn’t change: it’s a salt with a reputation for boosting flavor and supporting biochemical experiments.
Physical & Chemical Properties
I’ve handled sodium succinate in both chilly storerooms and bustling lab benches. Its white powder form feels unremarkable to the hand, yet it dissolves in water with a kind of effortless smoothness. The smell doesn’t give much away—there’s nothing pungent or off-putting. Chemically, it carries a simple formula: C4H4Na2O4. It prefers dry, airtight containers, away from moisture, because it can clump if left out. It doesn’t burn easily, but strong heat will break it down. In food applications, folks rely on its neutral taste that turns into an umami punch, which is pretty unique for a compound that chemists once studied mostly under a microscope.
Technical Specifications & Labeling
Clear labeling matters for something that slips into both foods and pharmaceuticals. Sodium succinate typically wears its E-number badge—E363 for the curious. In pharma circles, that changes to formal names and batch numbers to trace every jar back to source. Purity doesn’t just speak to safety; in my opinion, it speaks directly to the respect manufacturers show their customers. With sodium succinate, any sloppiness shows in the results, whether you’re talking food taste or reaction performance in the lab. Accurate weights, expiration dates, and composition details should be front and center—not just for regulators, but for anyone actually using the stuff.
Preparation Method
Industrial prep for sodium succinate usually means neutralizing succinic acid with a strong sodium base like sodium hydroxide. I’ve watched techs adjust the ratios bit by bit to avoid waste or weird byproducts. The process doesn’t demand wild temperatures or exotic materials, which explains its broad reach. The trick doesn’t stop at neutralization—the drying step ensures you don’t end up with a clumpy, damp mess. Different uses—food versus injections—require tweaks in filtration and drying. That’s a step where you see the real difference between raw chemical awareness and attention to human health.
Chemical Reactions & Modifications
Seeing sodium succinate interact with other compounds always brings home the reason it’s found in so many research setups. It plays nicely in buffer solutions, where it can keep pH in check, something every cell line and tissue sample seems to appreciate. In synthetic work, adding sodium succinate can nudge along tricky reactions or serve as a stepping stone for building more complex molecules. There’s also a knack for turning succinate into esters or using it in enzyme studies to monitor how living systems process energy. You can spot it on reaction tables, holding its own as a mild-mannered but vital partner.
Synonyms & Product Names
In catalogs and ingredient lists, sodium succinate keeps a small crowd of aliases. Disodium succinate and E363 are the most common, with scientists often writing just “succinate.” In food circles, it might hide under the umbrella of acidity regulators or flavor enhancers. I’ve tracked plenty of confusion stemming from naming conventions, which highlights the ongoing need for clarity across supply chains and R&D notes. After all, mislabeling leads to mix-ups that could ripple through research or product safety.
Safety & Operational Standards
Most people never touch sodium succinate in pure powder, but those who do have to respect the usual routines—gloves, masks, and careful scales. In both food and pharma use, standard operating procedures focus on avoiding contamination and accurate dosing, not just out of tradition, but to keep the downstream products safe and predictable. Handling isn’t tricky compared to some reactive salts, but sloppiness can spike sodium levels if folks aren’t careful with calculations. Food safety guidelines and Good Manufacturing Practice help rein in risk, but I’ve always believed the best defense remains a trained and aware staff who know what to expect.
Application Area
You see sodium succinate’s reach in ramen broth packets, injectable drugs, even some personal care items. Its flavor-boosting role in food leans on a gentle, savory quality that can round out artificial taste or bring out natural flavors without hogging the spotlight. In medicine, it can stabilize injectable solutions, acting as a buffer. Chemists and life scientists rely on it for experiment consistency—whether as a buffer or as a model substrate for metabolic research. I’ve even spotted it in industrial fermentations, where it helps feed specialized bacteria that churn out everything from vitamins to specialty polymers.
Research & Development
Current research leans into both refining sodium succinate’s production and broadening its uses. Scientists continue poking at microbial fermentation as a greener alternative to traditional chemical synthesis. That’s driven both by climate concerns and the simple economics of cutting waste. Pharmaceutical development teams test new salt forms and tweak excipient blends to improve drug delivery. In food labs, flavorists pair sodium succinate with other enhancers to reach that elusive “umami” note. I’ve seen some accuracy challenges crop up in analytical methods, which sparks debate over regulatory standards and best practices.
Toxicity Research
On the issue of safety, toxicity research mostly gives sodium succinate a clean bill if used in moderation. It’s a natural metabolite, meaning your body already deals with it as part of everyday metabolism. Animal studies and clinical data show low risk—even so, the lines between doses that help and doses that hurt require strict attention. Acute exposure doesn’t typically create dramatic effects, but chronic high exposure could nudge up sodium intake, and for some patients, that’s no small matter. I’ve learned that keeping an eye on the latest data from toxicity studies, especially long-term ones, is the responsible route for anyone thinking about food or drug formulation.
Future Prospects
Big questions remain about sodium succinate’s future. There’s curiosity about environmental impacts from industrial production, especially given the movement toward “green chemistry.” Biotechnologists are eyeing next-gen bacterial platforms to produce it more sustainably. On the consumer side, demand for clean-label foods might nudge product developers to focus on extraction from bio-based sources instead of sticking with strictly synthetic routes. As for health, researchers dig deeper into metabolic roles—maybe uncovering treatment avenues for certain disorders. In a world hungry for both convenience and safety, sodium succinate’s ability to adapt keeps it relevant. Continued innovation, rigorous oversight, and open data sharing among scientists and manufacturers can push its story into promising directions.
What is Sodium Succinate used for?
What Does Sodium Succinate Do?
Anyone who reads the back of food packaging enough times will run into sodium succinate. Chemists know it as a simple salt made from succinic acid, but out in the real world, sodium succinate finds plenty of jobs from food factories to hospital rooms. The kitchen is a good place to start. Walk down an aisle packed with instant noodles, canned broths, and ready-to-eat meals, and you’ll find sodium succinate playing a key supporting role as a flavor enhancer. It boosts umami, the so-called “fifth taste” so many people love in hearty soups, seafood, and fried rice. Japanese and Chinese recipes use it to give stock a richer, more savory backbone. No surprise that snack makers and professional chefs alike keep it in their arsenal for a flavor punch.
A Trusted Name in Food Science
There's a reason sodium succinate pops up in such a range of foods. It stands out because it works well with a long list of ingredients without making things taste artificial or strange. Returning home from a long day and opening up a can of fish soup, I’ve found sodium succinate helps smooth out harsh fishy notes, making each bite rounder and more satisfying. Food science experts noticed this and rely on it to achieve consistency across batches. Keeping flavors on track can be a challenge in the food business. So, suppliers look to trusted additives that deliver on both taste and safety—sodium succinate clears both bars. The World Health Organization considers it safe at levels much higher than what's used in food, and most digestive systems handle it easily, since it’s a natural part of several biological processes.
Role Beyond the Kitchen
Nutrients and metabolic intermediates based on succinate matter in medicine and research, too. Doctors and pharmacists have used sodium succinate in intravenous solutions to help balance blood pH or treat acute metabolic acidosis—a situation where the body can’t process acids properly. Emergency room staff reach for it when a patient’s situation gets critical. Some forms of steroids, such as hydrocortisone sodium succinate, depend on this salt to relieve inflammation quickly and blend well with the patients’ blood. Researchers even look at succinate salts in studies about energy production and cell metabolism, hoping for ways to tackle illnesses linked to mitochondria.
Potential Issues and Safer Choices
Big companies sometimes go overboard with sodium succinate, going after as much flavor as possible in snacks or processed foods. Many nutritionists warn that excess sodium in diets, no matter the source, can increase blood pressure and put the heart at risk. Watching out for all forms of sodium, including those hiding in salts like sodium succinate, matters for people with kidney or heart issues. I look for low-sodium versions of my favorite packaged soups because manufacturers listen if enough of us speak up.
Balanced use makes sense. Flavor manufacturers and food processors can keep exploring alternatives that use potassium instead of sodium, or use natural ingredients with a richer umami profile such as mushrooms, tomatoes, or seaweed. In medicine, hospitals already weigh risks and benefits before reaching for any sodium-based salt. Home cooks and professionals both benefit from using sodium succinate sparingly and thoughtfully.
The Takeaway: Everyday Chemistry, Wisely Used
Sodium succinate sits quietly in ingredient lists, but behind the scenes, it shapes the taste of comfort foods and supports modern medicine. Like many tools in science and cooking, a thoughtful approach works best—one that keeps health in mind and doesn’t lose sight of what really matters: real flavors, safe meals, and transparent choices.
Is Sodium Succinate safe for consumption?
Understanding Sodium Succinate
Sodium succinate shows up in ingredient lists for foods like instant noodles, frozen dinners, and even snacks that claim an extra “umami” flavor punch. This food additive, a salt derived from succinic acid, earns its spot in the kitchen for enhancing savory taste and acting as a stabilizer. Many cooks, bakers, and food manufacturers trust it to bring depth and richness to recipes. Regulatory authorities, including the U.S. Food and Drug Administration and the European Food Safety Authority, both recognize sodium succinate as generally safe for eating, supporting the idea that toxic effects don’t show up at the typical levels people eat.
Why Some Consumers Raise Concerns
Questions around sodium succinate often focus on safety in the long-term, especially if someone consumes many processed foods daily. Some worry about allergic reactions or negative interactions with other food additives. Succinic acid, the main building block, actually turns up in many plants and animals. Our own bodies make it as part of the process of turning food into energy. Eating a moderate amount from food rarely leads to trouble.
Scientific Studies and Real-World Outcomes
A review of animal studies and clinical trials shows that sodium succinate passes easily through the digestive system and breaks down to smaller, harmless molecules. Reports outside the lab echo these findings; most healthy adults notice no health effects after eating normal amounts. Adverse reactions remain quite rare. Occasionally, some people report sensitive stomach or hives, but strong allergic reactions do not turn up often in the medical literature.
Current evidence from several countries supports that sodium succinate falls within the safe zone, unlike additives linked to hyperactivity or metabolic issues. The Joint FAO/WHO Expert Committee on Food Additives backs this safety rating, and global regulators set limits much higher than typical diet patterns reach.
Balancing Flavor with Health
Like many flavor enhancers, sodium succinate improves palatability but can encourage folks to seek out processed foods with higher added salt and fat. Over time, eating too many prepackaged meals contributes to more serious health problems, such as raised blood pressure or excess weight gain. In my own kitchen, checking labels and sticking to “whole” foods where possible keeps my family on a healthier path. Flavors matter, but so does the larger food pattern.
Anyone with a history of allergic reactions or digestive sensitivity should keep an eye on food labels and choose trusted brands. For the broader public, sodium succinate doesn’t jump out as a major risk. If you’re concerned about food additives, making more meals at home, shopping for simple ingredients, and reading packaged food labels can make a big difference.
Practical Solutions for Informed Choices
Pushing for more transparency in labeling would help consumers who want to limit sodium succinate or any additive. The food industry could also offer more low-additive and natural-flavor options to meet growing demand. Dietitians and health educators play a key role in explaining why certain additives like sodium succinate get approval and providing tips for building a balanced diet.
Safety always comes down to context and real use, not individual ingredients in isolation. Sodium succinate shows few risks at realistic intake levels, but smart eating means knowing both what’s in your food and how often it’s landing on your plate.
What are the side effects of Sodium Succinate?
Looking at Health Risks In Plain Terms
Sodium succinate shows up in some medications and food products. On a nutritional label, most people skip over it. Doctors sometimes use it to treat certain medical conditions, such as metabolic acidosis, but it’s usually administered in hospitals rather than at home. The truth is, chemicals with “sodium” in the name can set off alarm bells for a lot of folks—mostly because of stories about bloating or blood pressure problems. But sodium succinate works a bit differently from table salt, and the risks it brings deserve a practical look.
Common Reactions: From Mild to Uncomfortable
Stomach complaints probably top the list. People might notice some nausea, or a bit of stomach pain after consuming large amounts. Once, after sampling a new electrolyte supplement, I felt queasy for a few hours. A look through the supplement’s label pointed to sodium succinate, and further reading confirmed that some folks get stomach issues from this additive, especially if the dose is too high. It’s not the kind of thing most people notice after a single, small serving, but put enough into your system, and your gut might push back.
Too much sodium from any source can increase thirst or leave you feeling bloated, especially for people sensitive to sodium or those already on a low-salt diet for medical reasons. I’ve met seniors who get concerned about swelling in their ankles if they sneak an extra bag of chips. For them, every gram of sodium matters, and sodium succinate adds to that daily total.
More Serious Effects: The Heart’s Warning Signs
Blood pressure can go up. The risk grows larger for patients with heart problems or kidney conditions. The American Heart Association reminds people that excess sodium puts extra pressure on blood vessels. This isn't just theoretical. My neighbor struggled with high blood pressure for years. After switching to some “low-sodium” foods, her readings stabilized, but hidden sources like food additives almost tripped her up. Even if sodium succinate plays a small role compared to regular salt, in someone with chronic conditions, that little bit adds up. It might bring headaches, dizziness, or confusion for some.
Who Faces Higher Risk?
Kids, pregnant women, and older adults face more risks than healthy adults if they take in too much sodium succinate. Kids’ kidneys can’t handle overload as easily, and pregnant women have delicate electrolyte balances. People with kidney disease or heart conditions can’t excrete sodium easily, so these groups deserve extra caution. Medical guidelines, including from the FDA and European Food Safety Authority, have not flagged sodium succinate as dangerous for most people, but they still recommend keeping an eye on all sodium intake.
Smart Solutions: Staying Safe with Sodium Succinate
Reading the ingredient list matters. Most people never see sodium succinate in their foods in high amounts, but processed foods and some supplements include it. For anyone with health concerns, talking to a pharmacist or doctor about safe levels makes sense. They can help track total sodium, catch hidden sources, and suggest alternatives where available. Cooking at home with fresh ingredients sidesteps most sodium additives completely. And for products with sodium succinate, sticking to serving suggestions prevents most trouble before it starts.
With information, people make decisions that fit their health needs. For those who check labels and know their own limits, sodium succinate rarely becomes more than a line on a label. For people with sensitivities or chronic conditions, a bit of caution with hidden sodium can keep health problems under control.
Is Sodium Succinate natural or synthetic?
Understanding the Source
People often ask whether sodium succinate comes from nature or if it’s cooked up in a lab. This is a fair question. Food additives tend to make us uneasy, especially when their names sound more like chemistry than cooking. Sodium succinate shows up in broths, seasonings, and packaged snacks. It keeps foods tasting savory and can even cut the sharpness of acid. Many food labels call it a “flavor enhancer,” which feels vague—so where does it really come from?
Diving Into Succinate’s Origins
Succinate itself forms during cellular respiration in all living things, even humans. Our bodies cycle it for energy. Some foods—tomatoes, mushrooms, certain meats—hold small amounts of succinate, and that’s where people get the idea sodium succinate might be natural. But the version found in industrial food production isn’t pressed from mushrooms or tomatoes. Factories make it by fermenting sugars through bacteria or yeast, or by chemical synthesis. Both routes involve technology and processing you just can’t duplicate in a home kitchen.
The Safety Angle
Every time a new food ingredient appears, the safety question comes up. Sodium succinate has been studied and cleared by food safety authorities worldwide. It’s considered non-toxic at typical amounts found in foods. Eating plenty of tomatoes or drinking bean broths gives you succinate too. Problem is, industrial food uses sodium succinate at much higher concentrations than you’ll find in homemade soup. Regulators keep an eye on thresholds to avoid overconsumption, but the lack of transparency on food labels can still worry shoppers.
Natural Isn’t Always Better
Marketing has leaned heavily on “natural” to sell everything from yogurt to supplements. People feel safer with ingredients straight from the earth. Still, nature alone doesn’t guarantee safety. Some mushrooms with natural toxins can be dangerous, while many “synthetic” ingredients pass strict health checks. Sodium succinate’s journey from raw biomass or chemicals, through fermentation or synthesis, to white crystalline powder shows the blurry line between natural and synthetic. For most food additives, including sodium succinate, it’s about the process rather than just the starting point.
Looking at Food Labels and Industry Practice
Trying to shop intelligently means learning how food gets to the grocery store shelf. I sometimes stand in the aisle, flipping over containers to read the ingredient lists. Sodium succinate rarely jumps out as a red flag, but it doesn’t offer much clarity. “Flavor enhancer” is as opaque as it gets, especially when you have dietary restrictions or allergies. Some companies flag fermentation on their packaging to ease concerns, though the effect is often more about marketing than science.
Building Trust and Transparency
The main friction point isn’t sodium succinate itself. People want to know not just that something is safe but that they understand what they’re eating. As a shopper, I’d rather see straightforward language on labels. Listing the process—fermentation of plant sugars—tells me more than just a chemical name. Companies and regulators could step up with better consumer education, explaining where these ingredients come from and how they’re used. No one wants to feel tricked into eating something unfamiliar.
Where We Go From Here
Tug-of-war between “natural” and “synthetic” distracts us from the bigger question: does this ingredient make our food safer and tastier, without introducing real health risks? As a food lover and a label-reader, I trust facts, not hype. More honest information and stricter oversight can help everyone make better choices at the table.
What foods contain Sodium Succinate?
Sodium Succinate: The Hidden Additive
Most people don’t pay much attention to the ingredients list on a bag of chips, a can of soup, or a packet of instant noodles. I tend to scan those lists, not just to see what’s familiar, but to watch out for unassuming substances like sodium succinate. It’s one of those food additives that matter more than folks think, because it’s not just a filler — it tweaks both flavor and shelf life.
Where You Find Sodium Succinate
Processed savory foods are the biggest carriers. You’ll spot it in instant noodles, ramen seasoning packets, and powdered soup bases. Restaurants use ingredient mixes packed with it for broths and sauce concentrates. That sharp, savory kick in some snack chips or meat-flavored crackers often comes thanks to this additive, lending a rich, rounded profile known as “umami.”
If you like seafood-flavored snacks — dried squid strips or prawn crackers — check the label. Manufacturers add sodium succinate here to give a deeper, more mouthwatering taste. Canned and packaged seafoods like tuna or crab, especially those labeled as “flavored,” get some succinate magic to hold flavor and boost appeal, especially if the product needs to travel far or last longer on the shelf.
Moving to the Hot Foods
Frozen dumplings, sausages, and some grilled meats carry sodium succinate to pump up the savory notes. Modern food processing turns to this ingredient for soups, gravies, and even certain gravied potpies, because it’s cheaper and more stable than extracting flavors from fresh stock.
Why Would Anyone Care?
Most food labels list sodium succinate under food additives, which doesn’t mean much unless you know its number — E363 in Europe. It’s considered safe by food regulators, but anyone eating a high-sodium diet for years ends up at greater risk for health issues. I’ve seen relatives struggle with hypertension, made trickier by lots of restaurant and packaged foods loaded with sodium from several sources, including sodium succinate.
Why the Food Industry Relies on It
Quick flavor without burning time or resources makes for cheap, tasty food. Customers want convenience and reliable taste — and sodium succinate delivers. For decades, it flew under the radar while more familiar sodium compounds like MSG drew attention. Now, with folks reading labels more carefully, awareness is rising.
Balancing Flavor, Health, and Transparency
Sodium succinate won’t disappear from foods any time soon. People crave quick meals, and the food industry depends on low-cost flavor. Yet, balancing taste and health does take some effort. Food brands could offer more transparency about additives and encourage customers to cook whole foods when possible. As a home cook, I find laying off the pre-packaged flavorings and leaning on vegetables, herbs, and real meat makes a clear difference in both taste and how I feel after a meal.
Ways Forward
Anyone looking to cut back should stick with fresh food and limit highly processed products. If you reach for packaged snacks or soups, aim to check the label and get to know what’s inside. Better decisions start with knowledge — not just about sodium succinate, but about all the extras that tuck into our food for flavor, cost-saving, and convenience. The more you understand about your own pantry, the more control you have over your health and what goes on your plate.
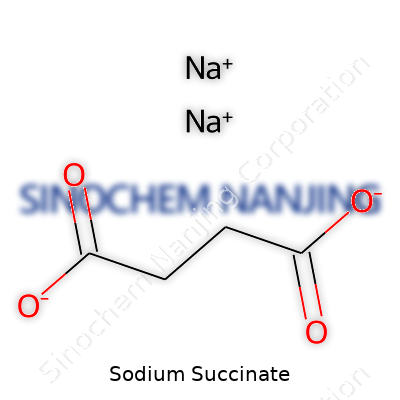
| Names | |
| Preferred IUPAC name | disodium butanedioate |
| Other names |
Disodium succinate Butanedioic acid disodium salt Succinic acid disodium salt |
| Pronunciation | /ˈsəʊdiəm səksɪneɪt/ |
| Identifiers | |
| CAS Number | 150-90-3 |
| 3D model (JSmol) | `CC(=O)C(=O)[O-].[Na+]` |
| Beilstein Reference | 1720474 |
| ChEBI | CHEBI:9175 |
| ChEMBL | CHEMBL1372 |
| ChemSpider | 21427 |
| DrugBank | DB09163 |
| ECHA InfoCard | ECHA InfoCard: 03b3917c-c2b3-47ae-aa32-0a7cba1cc403 |
| EC Number | EC 204-701-5 |
| Gmelin Reference | 6831 |
| KEGG | C00432 |
| MeSH | D013438 |
| PubChem CID | 23665760 |
| RTECS number | WS0250000 |
| UNII | C151H1V75CA |
| UN number | UN3261 |
| Properties | |
| Chemical formula | Na2C4H4O4 |
| Molar mass | 118.09 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.610 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.12 |
| Vapor pressure | Negligible |
| Acidity (pKa) | pKa1 = 4.21, pKa2 = 5.64 |
| Basicity (pKb) | 2.6 |
| Magnetic susceptibility (χ) | -36.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.420 |
| Viscosity | 2.0 - 3.0 mPa.s (20°C, 2% solution) |
| Dipole moment | 2.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 155.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -971.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1576.8 kJ/mol |
| Pharmacology | |
| ATC code | B05XA15 |
| Hazards | |
| Main hazards | May cause eye, skin, and respiratory tract irritation. |
| GHS labelling | GHS07, Warning, H319 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Autoignition temperature | 630 °C |
| Lethal dose or concentration | LD50 (oral, rat): 8,400 mg/kg |
| LD50 (median dose) | Mouse oral LD50: 10,000 mg/kg |
| NIOSH | WSH4570000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 250-500 mg/kg |
| Related compounds | |
| Related compounds |
Succinic acid Disodium malonate Disodium maleate Sodium fumarate |

