Sodium Pivalate: The Story Behind a Quiet Chemical Asset
How Sodium Pivalate Became a Chemist’s Ally
Few people outside chemistry circles ever hear about sodium pivalate, but it plays a bigger backstage role than most think. The sodium salt of pivalic acid, its real breakthrough came not with a headline discovery, but through a gradual buildup of trust by process chemists needing a small, stable base with a knack for getting tough jobs done. Lab notes from the twentieth century show chemists handling everything from pharmaceuticals to synthetic research relying on this compound, often jotting the nickname “trimethylacetic acid sodium salt” in the margin. Over the decades, it passed from one type of process to another, especially in setups that called for a non-nucleophilic base that wouldn’t make a mess by adding itself to reactants. Its role never came with much fanfare, but people in the know reach for it because it just works.
Getting to Know Sodium Pivalate
At room temperature, sodium pivalate shows up as a white, crystalline powder, not much to distinguish from common table salt at a glance. Unlike salt, though, it carries a twist: the three methyl groups packed around its central carbon create a barrier, making it pretty stubborn about reacting in ways that others might. This stubbornness is why chemists like it as a base or in specific organic synthesis tricks. It dissolves without fuss in water, but prefers not to budge much in organic solvents, which makes handling easier when you’re aiming for a clean separation in reaction mixtures. Whether people call it sodium trimethylacetate, sodium neovalerate, or just plain sodium pivalate, the same basic features run through its use.
Digging Into the Physical and Chemical Building Blocks
Every time I’ve handled sodium pivalate, what sticks out is how it melts at a relatively high temperature, well north of 200°C. The powder flows easily, and under the microscope those crystals don’t clump up unless you really let the air get humid. Chemically, those bulky methyl groups mean that, compared to things like sodium acetate, pivalate is less likely to step in and interfere during tricky steps in organic transformations. It doesn’t soak up water from the air much, so storage isn’t a problem—you can stick the bottle on a shelf for a while with no drama. Some of my colleagues have joked that the best thing about sodium pivalate is how it quietly refuses to mess up your day with side reactions.
The Right Way to Prepare and Label It
In most labs, sodium pivalate comes about through a straightforward acid-base reaction. Pivalic acid reacts with sodium hydroxide—two substances with long resumes in industrial and laboratory chemistry. Mix them in water, stir, wait for the neutralization to finish, filter, and you’ve got a crystalline yield of sodium pivalate. The process gives off pivalic acid’s scent, a bit like camphor, but otherwise stays unremarkable. Any chemist worth their salt checks purity with methods like NMR or IR, making sure no leftover acid or sodium hydroxide snuck through. On packages, you’ll find the Synonyms: sodium trimethylacetate, sodium neovalerate, which keep search engines and chemical catalogs consistent. Labels tend to reference its molecular formula, C5H9NaO2, and its CAS number, but for those actually doing the work, it’s the clear handling instructions and storage guidelines that avoid headaches later.
Reactions and Tweaks Chemists Use
What makes sodium pivalate special for many is the way it helps with palladium-catalyzed C–H activation, especially in the pharmaceutical and agrochemical sectors. I’ve seen it used to coax particular reactions along, especially when you want to avoid more reactive or nucleophilic partners like acetate or carbonate. Its structure lets it act as a non-interfering spectator in syntheses—sometimes helping bind metal centers a bit differently, sometimes guiding the formation of new bonds through subtle shifts in equilibrium. Chemists have also fiddled with derivatives, swapping the sodium for lithium or potassium, searching for fine-tuned performance in more ambitious coupling or functionalization reactions. Rarely does sodium pivalate grab the credit, but it’s a behind-the-scenes asset for people scaling up from grams to kilos, aiming to make fine chemicals with less fuss than older, messier reagents used to bring.
Keeping Chemistry Safe and Practical
In the grand scheme of chemical hazards, sodium pivalate rates as a relatively tame substance. I’ve read toxicology reports and never seen red flags flash, especially compared to volatile organics or alkali metals. Good practice still calls for gloves, goggles, and dust control, since any undisciplined powder can irritate skin or lungs in a busy work environment. Disposal guidelines recommend neutralization and dilution, then down the drain for small quantities in most jurisdictions. People in scale-up facilities keep closer tabs, as with every sodium salt, to avoid contaminating water streams. Most risk comes from casual handling, so the best solution is clear labeling, tidy storage, and consistent housekeeping—just the basics of good lab hygiene.
Where Scientists Turn to Sodium Pivalate
Most modern references to sodium pivalate crop up in academic journals and patent filings about C–H activation, especially where transition metal catalysts play a starring role. In pharma research, it lets teams try new strategies for building complex molecules by temporarily holding back reactivity in places you’d rather avoid. In polymer chemistry and agrochemical labs, it acts as a helper in building molecules from the inside out—turning what used to need several steps into a single, tidy run. Every time a paper claims better selectivity or yield using a pivalate, the real benefit comes in simpler purification later. Sometimes it gets used in the classroom, too, to show students how bulky counter-ions make a difference that’s both visible and surprisingly dramatic in the course of a reaction.
Digging Deeper With Ongoing Research
The last decade has seen a real jump in chemists experimenting with sodium pivalate-inspired tricks. By tweaking how sodium pivalate interacts with palladium, rhodium, or other transition metals, teams have found ways to open up new C–H bond reactions that could trim steps and cut waste from synthetic routes. Some groups are chasing single-pot processes that skip pesky protection-deprotection cycles, using the gentle influence of pivalate to steer the chemistry toward the desired outcome. The big push in research circles now is finding out just how far the pivalate “template” can stretch, both by modifying its structure and by mixing it with other classes of catalyst partners. What’s striking is that even small shifts—changing the amount, the counter-ion, or how it’s delivered—can lead to patentable breakthroughs in drug or material synthesis.
Sorting Fact From Fear in Toxicology
Compared to solvents and other salts that crowd a chemist’s bench, sodium pivalate lands as a mild presence in toxicity research. Animal studies and occupational exposure have yet to indicate long-term harm at levels seen in normal handling, though chronic exposure always merits investigation. Since it’s not volatile, inhalation risk stays low except if people let dust fly. The pivalic acid part of the molecule shows moderate breakdown in the environment, but it doesn’t seem to persist or bioaccumulate the way heavier organics do. Most incidents boil down to user error—spills, careless mixing, dust inhalation—so training stays as important as the actual material safety data. The general lesson is that routine measures, ventilation, gloves, and masks, give near-total safety in everyday lab routines.
What Comes Next for Sodium Pivalate?
Looking at where the field is heading, sodium pivalate stands to gain from the ongoing wave of “green chemistry” and process intensification. Demand for more targeted, less wasteful syntheses makes the non-nucleophilic, low-toxicity bases more attractive than ever. I see more high-throughput screening happening in both industry and academic labs, with sodium pivalate one of the regular “house bases” for new reaction types. As scientists look for replacements for older, dirtier industrial reagents, pivalate’s profile as an easy-to-handle, safe-by-default chemical matters. Lately, research has been exploring its use in solid-state catalysis and flow chemistry, hoping for applications that cut down on labor and environmental impact. In my own work, the freedom to test process ideas using sodium pivalate without needing heavy safety gear or facing regulatory hassles speeds up development. The field doesn’t need a blockbuster chemical for every challenge—steady, reliable reagents with predictable properties let people do their best work without time lost to troubleshooting. Sodium pivalate fits that bill, and I wouldn’t bet against it showing up in more stories of innovation as the next decade unfolds.
What is Sodium Pivalate used for?
Why Chemists Care About Sodium Pivalate
Ask any synthetic chemist about tools they keep handy, and chances are sodium pivalate will come up. Lab work rarely gets flashy headlines, but this humble salt finds its way into a surprising number of reactions. Sodium pivalate, with its catchy formula (C5H9NaO2), lands on benches worldwide not for its looks, but because it actually gets stuff done — particularly in creating bonds between carbon atoms that open the door to pharmaceuticals, flavors, and advanced materials.
What Sodium Pivalate Actually Does
Sodium pivalate works as a base. In simpler terms, it pulls protons away during certain steps in organic reactions. In cross-coupling chemistry, this makes a big difference. You see it in C–H activation reactions, where it's used to help break and reform chemical bonds that would otherwise dig in their heels. Recently, organic chemists have relied on sodium pivalate with palladium or copper catalysts to make those reactions work better, smashing open new ways to build complex molecules. A 2017 paper in the Journal of the American Chemical Society highlighted how swapping out bulkier, less convenient bases for sodium pivalate made tough reactions tick faster, with less gunk left over.
Sodium Pivalate in the Real World
Lab scientists often remember the first time they swapped sodium acetate for sodium pivalate and watched their reaction go from stubborn sludge to clear completion. Industry teams use it to cut down on waste and hit targets for purity, since some other bases create byproducts that slow everything down. It’s safer to handle than stronger bases like sodium tert-butoxide, which fizz and pop when they touch water. For the research labs that dream up tomorrow’s cancer drugs or crop protectants, just having a base that cooperates in air and damp conditions cuts headaches and costs. From personal experience in a pharma research group, I saw how switching over to sodium pivalate helped push experiments over the finish line and made tricky steps far more predictable.
The Environmental Angle
Too many chemical reagents come with a list of hazards that run longer than their benefits. Sodium pivalate holds up just fine — it melts easily, doesn’t raise red flags for skin contact, and stays stable on the shelf. In greener chemistry, where making less waste is half the challenge, sodium pivalate makes a difference by reacting cleanly and offering a milder footprint. The U.S. Environmental Protection Agency’s Safer Choice criteria stress picking reagents that keep waterways and air cleaner. Labs trying to shrink their environmental impact often add sodium pivalate as a go-to option, especially compared to organic bases that evaporate and linger in the air.
Looking for Better Chemistry
Every field needs unsung heroes, and sodium pivalate fills that role for many in synthetic chemistry. Researchers still push for even milder, safer bases that tap into renewable sources instead of petrochemicals. Investment in process safety training, greener chemical sourcing, and smart waste disposal all play a part. For now, the impact shows up every time a research team adds sodium pivalate and walks away with a cleaner, faster, more predictable reaction — freeing up lab time for bigger challenges instead of cleaning up messes from yesterday’s chemistry.
What is the chemical formula of Sodium Pivalate?
The Chemical Formula Up Close
Chemistry has a way of giving us simple answers to big questions. Sodium pivalate’s formula is C5H9NaO2. This stands for a sodium salt made by replacing the hydrogen atom in pivalic acid with a sodium atom. Pivalic acid brings in a bulky structure, and the sodium connects straight to the carboxyl group. That combo makes sodium pivalate more than just another salt—it has its own personality in the lab.
Why the Formula Matters in Everyday Chemistry
Knowing the formula C5H9NaO2 isn’t just a trivia win. Think about the work it does in chemical reactions. In organic synthesis, sodium pivalate often helps in coupling reactions, especially for building carbon chains without unwanted side reactions from smaller, “stickier” ions. The sodium part brings good solubility in water, while the bulky pivalate group adds selectivity that many synthetic chemists rely on.
My background in chemistry labs tells me that formulas like this one guide a lot of what we do on the bench. If a reaction goes off track, checking whether you have the right salt can quickly narrow down the cause. Building reliability in research means trusting simple facts like formulas, as mistakes in weights or concentrations add up fast.
Where Sodium Pivalate Shows Up
It’s not rare to see sodium pivalate on reagent shelves—especially in pharmaceutical labs. Take the development of some antibiotics or specialty polymers. The compound’s formula makes it a favorite as a base in palladium-catalyzed coupling reactions. Nothing fancy; chemists just need confidence that Na (sodium) brings the charge, and that the pivalate part keeps the molecule from breaking down too easily under heat.
Quality always matters in these settings. Medical and material scientists depend on consistency, so any batch of sodium pivalate should match the correct molecular weight based on its formula. Lab techs need to trust that C5H9NaO2 won’t surprise them with impurities.
Potential Issues and Smarter Solutions
A lot of researchers complain about source reliability. Not every supplier makes sodium pivalate with the same level of purity. Contaminants sneak in, sometimes making reactions unpredictable. I’ve seen projects delayed just because the salt didn’t work as expected, which is frustrating on a team level.
A practical fix is sticking with trusted chemical suppliers and always verifying what’s in the jar using spectroscopy or melting point checks. Even better, chemists push for more transparency in sourcing, including detailed certificates of analysis with every shipment. This goes a long way in getting reproducible results and cutting down on wasted effort.
The Bigger Picture
The story of sodium pivalate’s formula isn’t just about numbers or chemistry jargon. It reveals how even small molecular differences change what happens in the lab. Whether working with pharmaceuticals or plastics, having reliable, clearly defined materials lets creative science happen safely and speedily.
Is Sodium Pivalate hazardous or toxic?
What Actually Is Sodium Pivalate?
Sodium pivalate doesn't draw much attention compared to everyday chemicals, but it does play a role in labs and some industrial settings. Chemists tend to use it to speed up reactions, not because it’s flashy, but because it gets the job done. Anyone who’s mixed up a batch in an R&D lab knows that its white powder looks pretty unremarkable. If you don’t work in chemistry, you probably haven’t crossed paths with it.
What Science Tells Us About the Risks
Concerns often come from lack of information, and sodium pivalate’s safety profile isn’t as widely talked about as household substances like bleach. Digging into available toxicology and safety assessments, sodium pivalate generally doesn’t set off huge alarm bells compared to more notorious chemicals.
Swallowing or inhaling large amounts probably won’t do wonders for your health, but most people won’t run into this scenario unless they’re working with it directly. A report from Sigma-Aldrich points out it can irritate the eyes, skin, and airways if mishandled. Basic lab safety—goggles, gloves, a bit of common sense—usually covers things.
Long-term toxicity presents a far more interesting conversation. Research on animals found that derivatives of pivalic acid can cause certain health effects, including effects on the liver and kidneys, if large amounts circulate through the body. The body doesn’t break down pivalate the same way it handles something like vinegar, but typical lab exposure tends to fall far below those thresholds.
Sodium Pivalate and Environmental Impact
Spill a bunch in the environment and you get a different story. The chemical doesn’t break down quickly, so it could persist in water or soil. Regulations such as the EU’s REACH program monitor chemicals that stick around or build up in living things. At this point, sodium pivalate doesn’t top those regulatory hit-lists, but good stewardship means keeping it out of the drain or landfill in large amounts. Anyone treating sodium pivalate as ordinary trash isn’t respecting either the chemical or the land beneath their feet.
Safety in Practice
Experience working with industrial chemicals reminds me that many risks come down to how someone handles the material. Sodium pivalate looks harmless, but thinking you can ignore protocols is where trouble can start. Any powder that irritates the airways deserves respect in the form of ventilation, dust masks, and keeping containers tightly closed. Stories pop up in safety reports about folks skipping gloves and then dealing with rashes, not because sodium pivalate is some hidden poison but because good habits keep everyone safe.
What Should Companies and Labs Do?
Reliable procedures make all the difference. Material Safety Data Sheets spell out hazards and aren’t meant to gather dust on a shelf. Supervisors lead by example, making sure that labels stay visible, clean-up materials are nearby, and staff know what not to mix together. Training doesn’t have to be a boring checklist—just straight talk about what can go wrong if steps get skipped.
What Could Improve Safety Further?
Continuous training and regular review of chemical inventories help a lot. Lab managers investing time to teach new team members about sodium pivalate’s actual risks, not just theoretical ones, see fewer accidents. Companies can push for greener chemistry options, or at least safer packaging and storage solutions.
Anyone who’s handled chemicals for years knows that honest, practical information prevents both danger and panic. Sodium pivalate isn’t a monster. Treat it with respect, keep updated on research, and problems shrink before they start showing up.
How should Sodium Pivalate be stored?
Why Sodium Pivalate Gets Extra Attention
Sodium pivalate often shows up in chemical labs and some industrial settings. It’s not something you want to take lightly, since handling mistakes can lead to big problems for both people and the environment. I’ve worked in a lab where one small error with chemical storage turned into hours of clean-up and stacks of incident reports, so I know how quickly things can spiral. Choosing the right spot and the right container shouldn’t feel like chores—it’s about making sure you don’t land you or your colleagues in trouble.
Understanding Sodium Pivalate’s Risks
Let’s be clear: sodium pivalate isn’t as notorious as strong acids or high explosives, yet it carries its own hazards. Its white, crystalline powder can irritate the respiratory system and eyes. It reacts if exposed to moisture. Once, in a shared research space, someone left a loosely capped bottle out on a humid day. The clumping and faint chemical smell sent us scrambling. Every person in that room learned quickly—just because a compound looks harmless, doesn’t mean you should treat it casually.
What A Good Storage Setup Looks Like
Sodium pivalate belongs in a cool, dry place. Humidity will wreck your stock and might even start a reaction inside the bottle. A friend once tried to store their supply near a window that caught the afternoon sun—big mistake. Direct light and fluctuating temperatures turned their bottle into a clumpy mess. Stick with sealed, airtight containers, kept clear of any area where there’s a chance of leaks or spills from water or acids.
My best tip? Use tough, chemical-resistant packaging. Glass or good quality plastic works. Label everything with the date it arrived and a clear hazard sticker. If someone you work with misses a label, it doesn’t take long for confusion to set in. I once found a jar in the back of a shared cabinet with nothing but a sketchy, faded label. That kind of mix-up invites serious accidents.
Forget the habit of stacking different chemicals together just to save shelf space. Put sodium pivalate far from acids, oxidizers, and materials that could trigger a reaction. Fumes can linger, and chemicals have a way of finding their way into unexpected places through shared air or sneaky leaks. Never store open bottles on top shelves; even careful hands can slip, and then, everything comes crashing down—literally.
Simple Steps Make a Difference
Routine checks do more than fill up inspection logs—they prevent bad surprises. Every couple of weeks, I pull out all my chemical stocks and double-check lids, labels, and shelf stability. It may sound tedious, but a quick five-minute inspection beats a ruined experiment or a health scare.
Separate cleanup materials should stay close by. Spills need immediate attention. Keep gloves and goggles right where you can grab them. Don’t only rely on written rules; instead, make them your daily habit, just like washing your hands before leaving the lab.
People sometimes forget about disposal, treating it like someone else’s job. Hazardous waste bins belong close to where you handle sodium pivalate. Don’t just pour leftovers down the drain. Local regulations spell out clear protocols for chemical waste; follow them to the letter.
What Good Storage Achieves
By storing sodium pivalate carefully, you lower health risks, cut down on waste, and keep research running smoothly. Safe storage isn’t just about following rules on paper—it’s about respect for your own wellbeing and everyone around you. The best labs I’ve seen run on trust and shared responsibility, not on luck or crossed fingers.
Where can I purchase Sodium Pivalate?
Understanding Sodium Pivalate and Its Uses
Sodium pivalate plays a part in organic synthesis, medicine, and even some advanced lab work in universities. In my days working alongside research chemists, requests for compounds like sodium pivalate would show up whenever someone started a project involving esterification reactions or needed certain molecular tweaks. So, finding the right source moves the whole lab forward—or stalls it if search efforts go wrong.
Where Can You Find It?
Buying chemicals like sodium pivalate isn’t something most folks do at the local pharmacy or hardware store. Companies serving labs and universities, such as Sigma-Aldrich, Thermo Fisher Scientific, and TCI, keep compounds like this in stock. Orders usually go through institutional accounts, and buyers need to provide proof the chemical is for legitimate research or industrial purposes. There’s no way around the paperwork. Retail channels like Amazon and eBay rarely carry specialized chemicals and, when they do pop up, quality standards and origin can’t be traced. Sticking with the established suppliers keeps lab teams safe from surprise contaminants or mislabeled bottles.
Safety and Responsibility
Chemical suppliers don’t just ship to anyone with a credit card. Oversight agencies—including OSHA in the US and ECHA in Europe—put rules in place to safeguard buyers and the public. That’s why I’ve always recommended that curious hobbyists and students avoid trying to order laboratory-grade compounds on their own. Transactions often need an institutional email, a business address, and sometimes even a description of the intended use. This way, suppliers knock out most inappropriate requests and lower the risk that chemicals wind up in illegal or dangerous hands. Working in research, I saw firsthand that reliable vendors want a paper trail on every purchase.
Beyond Buying: Storage and Handling
Sodium pivalate, like many laboratory salts, calls for storage under dry, sealed conditions—moisture causes clumping or chemical changes over time. I remember mistaking a bottle of sodium pivalate left open over the weekend—it had changed consistency, so the results in the next synthesis didn’t match our calculations. So, always store chemicals according to the Safety Data Sheet and make sure colleagues do the same.
Finding the Right Supplier
Those working in academia or industry should start with a procurement team or check existing vendor agreements. The organization probably already works with a couple of approved chemical suppliers. Research shows that trying to save a few dollars using non-vetted sources can end up more expensive due to ruined experiments or extra waste disposal fees. Shopping directly from established chemical companies offers more certainty—each bottle comes with a certificate of analysis, letting you verify identity and purity right away.
Building Knowledge and Trust
With the internet full of “click here to buy” links, sticking to well-known brands isn’t just a cliché. In research and manufacturing, one bad experience with an unreliable supplier can throw off a whole semester’s work or, worse, endanger safety. Training and experience taught me that time spent vetting suppliers beats extra time troubleshooting failed reactions. Connecting with colleagues who have made similar purchases adds another layer of trust and can shortcut the buying process. In this field, reliable supply means reliable science.
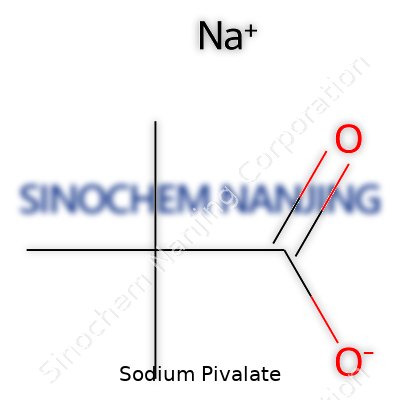
| Names | |
| Preferred IUPAC name | Sodium 2,2-dimethylpropanoate |
| Other names |
Sodium trimethylacetate Sodium 2,2-dimethylpropanoate |
| Pronunciation | /ˌsəʊdiəm pɪˈveɪleɪt/ |
| Identifiers | |
| CAS Number | 630-08-0 |
| 3D model (JSmol) | `CC(C)(C)C(=O)[O-].[Na+]` |
| Beilstein Reference | 1721397 |
| ChEBI | CHEBI:91214 |
| ChEMBL | CHEMBL3305766 |
| ChemSpider | 86614 |
| DrugBank | DB14118 |
| ECHA InfoCard | 13e744b9-b1c6-4aa6-8f75-c89c3e0d8cdf |
| EC Number | 205-488-0 |
| Gmelin Reference | 59864 |
| KEGG | C18636 |
| MeSH | D013007 |
| PubChem CID | 8666 |
| RTECS number | AJ3675000 |
| UNII | 142W4J12K2 |
| UN number | UN3276 |
| CompTox Dashboard (EPA) | DJWYHZOVHDLVBT |
| Properties | |
| Chemical formula | C5H9NaO2 |
| Molar mass | 110.11 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 0.980 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.9 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 11.21 |
| Magnetic susceptibility (χ) | -41.0×10^-6 cm³/mol |
| Refractive index (nD) | 1.397 |
| Viscosity | 125.5 mPa·s (25 °C) |
| Dipole moment | 1.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -576.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2056.4 kJ/mol |
| Pharmacology | |
| ATC code | V03AB21 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Lethal dose or concentration | LD50 (oral, rat): > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose) Oral rat: > 2000 mg/kg |
| NIOSH | SNAP8310000 |
| PEL (Permissible) | No OSHA PEL established |
| REL (Recommended) | 5g |
| Related compounds | |
| Related compounds |
Potassium pivalate Pivalic acid Pivalamide Pivaloyl chloride |

