Sodium Nitrate: Legacy, Science, and the Road Ahead
Historical Development
Sodium nitrate has roots that dig deep into human history, shaping agriculture, industry, and even warfare. In the arid deserts of Chile, massive deposits kicked off the so-called “Saltpeter Wars,” drawing global attention and clashing interests among nations hungry for crop yields and explosives. Chilean saltpeter carried economies for decades, fueling both gunpowder and farming. Most people today will never see a saltpeter caliche by eye, but legacies of those early mining booms echo in fertilizer strategies and the tangled, often painful, stories of local communities displaced for export profit. This chemical story is a mirror for how people chase natural resources, then adapt supply chains when those stores run dry. Synthetic routes took over in the 20th century, but echoes from the nitrate age keep showing up in regional politics and environmental debates.
Product Overview and Everyday Encounters
Sodium nitrate doesn’t usually get a front-row seat in the public consciousness, but its fingerprints show up in more places than most folks realize. Food preservation owes a debt to sodium nitrate’s sturdy ability to stomp out botulism and spoilage. In farming, crops green up under the nitrogen boost found in sodium nitrate–easy to absorb and ready to drive leaf growth. In pyrotechnics, vibrant energy owes a spark to this compound, launching colored flames into night skies for the world’s celebration or caution. Industrialists find peculiar value among reactive mixes and glassmaking. It’s a regular in labs, helping research teams trace chemical patterns or test new soil blends. In every instance, sodium nitrate’s utility isn’t abstract—its reliability lets people manage real challenges, whether that’s preserving bacon or ramping up wheat production.
Physical & Chemical Properties
People who work with sodium nitrate notice its white, crystalline grain, dissolving swiftly in water and melting just above 308°C. The chemical formula, NaNO3, sounds simple, but behind each atom is a long parade of possible reactions. That eager nitrate group acts as an oxidizer, meaning it’s always ready to lend oxygen in a process. Materials like these need careful handling, and the rules here were written the hard way—through factory fires and watery dissolutions that stripped valuable nutrients from soil columns over time. Its hygroscopic nature pulls in moisture from the air, a fact lab techs quickly learn when their carefully weighed samples clump or vanish overnight. Its chemical energy, steady heat behavior, and clear water solubility set it apart from the crowd of white powders filling chem store rooms.
Technical Specifications & Labeling
Guidelines for sodium nitrate shipments leave no room for guessing, especially in a world where shipments crisscross oceans and railways daily. Labels shout warnings about oxidizing potential and spell out purity, contamination levels, moisture content, and bulk density. Only experience in an industrial setting gives you a sense for how much this matters, seeing a fork-lift operator pause before moving a drum marked for sodium nitrate, or a plant manager triple-check atmospheric controls before dosing a chemical blender. Safety data gets relayed to every crew member. Missteps here cut deep, and no one forgets the lesson from a poorly sealed bag or a container found bulging from humidity.
How Sodium Nitrate Takes Shape
Synthetic sodium nitrate mainly starts life in reactors, where sodium salts collide with sources of nitric acid under tightly controlled conditions. Industry shifted hard in the last century, breaking its dependency on natural Chilean deposits after Haber-Bosch revolutionized fertilizer supply. The methods don’t just shape cost trends, they leave an environmental footprint, too. Factories draw energy and release waste, showing how chemical innovation always packs a package of pros and cons. By capturing byproducts, recycling water, and managing nitrogen emissions, manufacturers are learning to close loops. Watching an industry upgrade a legacy facility with scrubbers or a zero-liquid-discharge system, you see chemistry and ecology learning uneasy lessons from each other.
Chemical Reactions & Modifications
In the hands of a skilled chemist, sodium nitrate serves as a backbone for a dozen different reaction pathways. Heated in the company of carbon, it gives up its oxygen to make sodium nitrite and carbon dioxide, a trick used since the dawn of smokeless powders. Mix it with sulfur and charcoal and you have the heart of solid rocket propellants. Beyond that, it assists in the fixation of chromates and in the recycling of precious metals, where its oxidizing bite strips away waste while leaving valuable material. I’ve watched sodium nitrate shift the outcome of glass melts, transforming not just clarity but refractive strength, altering industrial design decisions and product costs.
Other Names on the Label
No two labs seem to settle on the same shorthand. Some old-timers still call it Chile saltpeter or soda niter, names that stir up visions of desert landscapes and rusted railcars. In the trade of food preservation, it appears as E251—a code on ingredient lists that’s easy to miss but packed with history and regulatory care. On the farm, the bag says “Norwegian saltpeter,” even though Norway’s role belongs to hydroelectric-powered synthesis plants that cranked out artificial nitrate decades ago. Each synonym tells a piece of the chemical’s journey, linking ecosystems, trade routes, and kitchen tables across continents.
Staying Safe and Playing by the Rules
Folks sometimes forget that chemicals sold for boosting crops or curing sausage can bite back under the wrong conditions. Sodium nitrate’s oxidizing strength is both its blessing and its risk. Storage guides urge keeping it apart from combustibles and locked away from organic matter, not just for compliance but for peace of mind. The stories of forgotten storerooms that transformed into danger zones, with nitrate dust building into a hazard, get passed down in safety debriefs and regulatory filings. Gloves, eye shields, and good ventilation are more than box-ticking; they’re the result of mishaps learned the tough way. Standards shaped by global agencies like OSHA or the World Health Organization ask teams to test exposure limits, manage spillage, and track worker health for signs of nitrate poisoning.
Where Sodium Nitrate Shows Up
Factory farms, commercial greenhouses, heritage butchers, and fireworks crews all draw on sodium nitrate’s reliable chemistry. In fields, rates of plant growth and visible crop improvements traced their origins to 19th-century fertilization campaigns. Culinary professionals see the good and the downside: nitrate preserves color and flavor, but scientists warn of potential links between nitrite/nitrate cured meats and certain health risks. On a fireworks crew, the right blend delivers show-stopping colors and smoke, and poor mix ratios can turn a celebration into a hazard. Niche uses—catalysis, glass manufacturing, airbag deployers—depend on its predictable, reproducible energy balance. Few chemicals flex across so many sectors, jumping from field to plate to laboratory with equal confidence.
In the World of Research & Development
R&D teams have yet to call time on sodium nitrate. Soil specialists still chase interactions between nitrate presence and soil health, especially as concerns grow around water systems and agricultural runoff. Food safety research tests the fine line between preservation and toxicity, with biochemists mapping how nitrate and nitrite interact with proteins during processing. At the lab bench, new hybrid materials—melts, salts, or composites—employ sodium nitrate as an agent in reactions that need precise oxygen delivery. Scientists continue to investigate improved recovery methods for waste nitrate, along with better purification approaches to reduce contaminants before use in high-purity pharmaceutical or food lines. Tech teams look beyond bulk use, examining nano-scale blends and the potential for sodium nitrate in evolving energy storage and medical diagnostic roles.
Digging Into Toxicity
The toxicity story of sodium nitrate blends risk and context. Most people get exposed through food or water, and typical amounts pass through the body without issue. Excess, though, starts to shift the balance—infants, for example, face specific dangers around methemoglobinemia, turning what should be a safe drink of water into a medical emergency. Population studies wade through murky statistical ground, testing links to cancer risk or blood pressure effects. Regulatory bodies set strict upper limits, not just for public safety but to drive industries toward better monitoring and cleaner production. In places where nitrate runoff contaminates wells, community health hangs on government commitment, infrastructure spending, and honest communication about risk. The dialogue between toxicologists, public health advocates, and chemical producers will decide how sodium nitrate finds its balance in diets and water systems down the road.
Future Prospects and Unanswered Questions
The future for sodium nitrate balances on shifting priorities: feeding growing populations, slashing environmental impact, and redesigning supply chains for sustainability. Synthetic production isn’t going anywhere—but concerns about energy use and emissions drive innovation’s direction. There’s a push for precision agriculture, smarter blending with other nutrients, and alternatives that soften runoff impact. In medicine and diagnostics, researchers test its potential as a controlled source of nitric oxide, hinting at new therapies. Some teams poke at the limits of high-temperature energy storage, using nitrate salts as molten mediums in concentrated solar plants. Eco-friendly processing, regenerative agriculture, and safer food technology continue to shape its next chapter. The story of sodium nitrate keeps unfolding, marking transitions in technology, policy, and daily life, shaped by hard-won lessons from a long and sometimes rocky past.
What is sodium nitrate used for?
The Hidden Ingredient in Everyday Life
Step into any kitchen and you'll probably spot foods that wouldn't last long without a little help. Cured meats like ham or bacon stay pink and flavorful for weeks, thanks in large part to sodium nitrate. Food producers often add it to slow down bacterial growth and keep meat appetizing. I can taste the difference between processed meats cured with nitrate and those that skip it — there's a distinct flavor that comes through, something families and cooks have been drawn to for generations.
Food uses only scratch the surface. For years, farmers relied on more traditional manure, but large-scale agriculture turned to chemical options for predictable crops. Sodium nitrate gives a steady hit of nitrogen, the same element that plants need for lush growth. Cornfields and wheat stretches soak up this compound, especially in places where organic fertilizer can’t keep up. The American Midwest, for example, leaned heavily into chemical fertilizers to produce record harvests. It's a classic story: one tool, several jobs.
The Role in Industry and Science
If you’ve watched fireworks or road flares light up the sky, you’ve seen sodium nitrate in action. This chemical acts as an oxidizer, supplying oxygen so the bright colors and loud pops can happen. In the classroom, I remember mixing it in small science demonstrations, where its ability to feed quick, lively reactions left a big impression. Explosives manufacturers depend on reliable oxidizers for stable, predictable results. Working in industry, I learned that consistency and safety mean everything, and sodium nitrate brings both.
Glass and ceramics manufacturers also turn to this compound, using it to influence melting points and produce smooth, clear finishes. This isn’t something people often picture when they think of chemicals — but careful tweaking of ingredients shapes everything from smooth windowpanes to heat-resistant dishes. I’ve spoken with glassmakers who insist on high-grade chemicals; without them, quality just slips.
Concerns and Solutions
All these applications mean sodium nitrate ends up in a wider environment than many realize. Runoff from farms hits rivers and streams, contributing to issues like algal blooms. These bursts of plant life use up oxygen and threaten fish. The Environmental Protection Agency keeps tabs on nitrate levels for this very reason. I’ve seen community meetings where local fishers and farmers debate how to protect water without losing crop yields. Newer precision agriculture uses sensors and software to cut down on extra fertilizer, helping reduce contamination and waste.
Some health advocates worry about sodium nitrate in processed meats, linking it to possible health risks if eaten in big amounts. The World Health Organization flagged it as a concern in relation to certain cancers. Grocery shoppers now look for nitrate-free labels, and companies have started offering more alternatives. I know friends who stick to fresh, unprocessed meats for this reason, especially when shopping for their kids.
The Need for Balance
Sodium nitrate can boost crop yields, preserve Sunday brunch bacon, and help science work safely — but the trick comes down to using it wisely. Whether you’re running a farm, making glass, or planning a family meal, the choices around this single chemical reach further than they seem. Sustainable practices, transparent labeling, and smart innovation show there’s room to keep the benefits without stacking up the risks. Solutions aren’t simple, but the choices and information are right in our hands.
Is sodium nitrate safe for consumption?
What You’re Really Eating
Every time people reach for cured meats like bacon or hot dogs, sodium nitrate tags along for the ride. It keeps the rosy color in meat, and blocks bacteria that can cause sickness. For decades, food companies leaned into sodium nitrate as a preservative. On grocery shelves, you’ll find it lurking in deli turkey, bacon, ham and even some cheeses.
Looking Past the Hype
Food safety scares come and go all the time, and sodium nitrate always sparks big debates. Consumer groups warn about it, and scientific studies go back and forth. So it helps to cut through the noise and focus on real risks. Large reviews from trustworthy sources like the World Health Organization and U.S. Centers for Disease Control and Prevention connect steady nitrate intake mostly to processed meats, not leafy greens or home cooking. Too much processed meat over years links to higher rates of colorectal cancer, and nitrates play a part in those studies, but no single ingredient takes all the blame.
The Real Health Concerns
What makes sodium nitrate tricky lies in what happens after eating. In the human body and during high-heat cooking, it can turn into nitrite. Nitrite in turn helps form nitrosamines, which can damage cells and bump up cancer risk under certain conditions. Cooking methods matter—a pan full of sizzling bacon usually packs higher levels of nitrosamines than sliced deli turkey.
It’s worth noting that vegetables have even higher nitrate amounts than processed meats, yet nobody rings alarm bells over salads. That’s because fresh vegetables come with vitamin C and compounds that fight nitrosamine formation. Meat products lack those natural shields.
FDA Oversight and Daily Intake
The U.S. Food and Drug Administration checks sodium nitrate and sets clear limits on how much can legally go in food. Food labels must display nitrates, and most brands keep amounts lower than health guidelines allow. Recent surveys from the CDC and Harvard School of Public Health say that for most people in the U.S., sodium nitrate exposure from food falls below risk levels.
How to Build a Safe Plate
From personal experience, cutting back on ham sandwiches and swapping in grilled chicken nudges people toward safer eating patterns. Most registered dietitians echo that advice: keep processed meats as an occasional treat, and fill plates with a bigger mix of leafy greens, beans, baked fish, and roasted veggies. Families can choose nitrate-free or “uncured” labels at the store, though these products swap sodium nitrate for celery powder—a natural source of nitrates that delivers a similar hit.
Schools, hospitals, and cafeterias across the country also shift their menus in healthier directions—more whole grains, fewer hot dogs. Simple swaps add up. Longstanding habits like pairing bacon with orange juice or a tomato slice actually help, since vitamin C slows down harmful nitrosamine formation in the stomach.
What to Watch in the Future
Science always marches forward. Big studies now track long-term eating patterns, not just single ingredients, to see how diet shapes health. Groups like the American Cancer Society and the National Institutes of Health spread balanced messages: less processed meat, more plants, and a focus on whole foods. Nobody needs to panic over an occasional hot dog at a baseball game, but building a weekly menu around fresh, minimally processed food sets a sturdier foundation.
What are the potential health risks of sodium nitrate?
Life at the Grocery Store: More Than Just Bacon and Deli Meat
Sodium nitrate pops up in lunch meat, bacon, hot dogs, all sorts of foods lining the racks at the supermarket. If you’ve glanced at the back of a pack of cured meat, you’ve probably seen it listed. Companies use sodium nitrate to help processed meats keep their color and stretch their shelf life. That pink ham and bright red salami would look a lot grayer without it.
The problem is, eating a lot of foods with added nitrates may carry real risk. Most people aren’t reading ingredient lists closely or thinking about the chemistry behind their sandwich filling. Growing up in a house where cold cuts showed up at every summer BBQ, nobody sat me down to explain nitrates. Later in life, hearing more about potential hazards snapped me awake.
What the Doctor Sees in the Research
Decades of medical studies point toward a link between sodium nitrate in processed meats and a higher chance of developing certain cancers, especially colorectal cancer. The World Health Organization places processed meats in the same category as tobacco and asbestos for cancer risk. Not every scientist agrees on the exact level of risk, but the trend pops up enough to cause concern.
Part of the problem lies in what happens inside the human body. When we eat nitrate-preserved foods, our stomach acids and bacteria can turn the nitrate into nitrite. That then can react with other components in the gut, creating nitrosamines, substances shown in lab tests to damage DNA and promote tumor growth. Lab animal studies and big population studies both suggest this is not a harmless byproduct. The American Institute for Cancer Research and the American Cancer Society caution against eating much processed meat for this reason.
Hidden Strains: Heart and Metabolic Health
Cancer isn’t the only story. Some research tracks regular nitrate intake to a higher chance of developing high blood pressure and heart disease. One study from Harvard found people who ate processed meats on a regular basis (such as a few slices every day) faced higher risks of these health problems than those who stuck with fresh meats.
It’s not just about nitrates either. Foods carrying these preservatives often contain a lot of added salt, saturated fat, and other preservatives. These combo meals tend to push up cholesterol and blood pressure over time, both classic foes for heart health. As a kid who loved pepperoni pizza, I had no clue repeated exposure might set up trouble down the road.
What Ordinary People Can Do
No one needs to swear off deli sandwiches for life, but paying attention to processed meat intake goes a long way. Preparing meals at home using fresh chicken, fish, beans, and vegetables slashes exposure to added nitrates. For sandwich lovers, some brands offer nitrate-free or “uncured” versions. These choices use natural sources like celery powder, but even those can wind up producing nitrites during processing. The safest bet looks like limiting any processed meat to an occasional treat instead of an everyday staple.
Kids tend to pick up eating habits from adults, so showing better choices sets a pattern for younger generations. As someone who inherited a love of hot dogs and subs, I can’t say I’ve quit completely, but these days a turkey or veggie wrap feels like a safer habit. Knowledge here means better decisions, so knowing about sodium nitrate risks lets everyone protect their long-term health—without having to ditch every guilty pleasure.
How is sodium nitrate stored and handled safely?
The Reality of Working With Sodium Nitrate
Sodium nitrate pops up everywhere, from fertilizers to food preservation. I have worked with chemicals in manufacturing and laboratory environments for years, and sodium nitrate always came with a clear message: treat it with respect. This compound, an oxidizer by nature, can pose real dangers if handled carelessly. Stories in the news about fires in chemical warehouses often trace back to improper storage of substances just like this.
Risks Nobody Should Ignore
Sodium nitrate doesn’t explode on contact, but its ability to fuel fires stands out. Combining it with anything flammable — like sawdust, paper, or even clothing fibers — can set the stage for disaster. When I started in the lab, the first thing hammered into me was how nitrate salts can intensify flames and cause burns that don’t quit. Anyone working around large quantities knows that avoiding cross-contamination is essential. One slip, and a leak can become a risky situation much faster than most expect.
The Details of Sensible Storage
Storing sodium nitrate well begins with location. Keep it far from organic and combustible materials. At the plant where I worked, our storerooms carried strict separation rules. Nitrates always claimed a cool, dry corner — nowhere near oily rags, cleaning agents, or anything that could catch fire. Shelves stayed sturdy and non-wooden, metal bins stayed clean and tightly closed, and humidity got monitored with a hawk’s eye. Moisture, even a small amount, can cause the salt to clump or liquefy, raising the risk of spill or unwanted reaction. Temperature swings also cause problems, increasing the chance of decomposition and release of dangerous gases.
Personal Experience Shaped by Safety Training
Personal protective equipment made a major difference in how I approached these materials. At the start of every shift, we put on our gloves—nitrile over latex for extra protection—along with goggles and lab coats. Even after years, I never skipped the simple steps. Small splashes irritated skin and eyes; inhaling the dust caused coughing fits or nosebleeds. Risk may look small, but it’s always there.
We kept sodium nitrate containers at or below shoulder height, with clear labeling and proper hazard signs. Any spill called for immediate cleanup using noncombustible absorbents. A friend of mine once saw a careless worker sweep nitrate dust under a bench with a paper towel. The fire marshal didn’t appreciate that one bit. All empty bags and containers got washed out before disposal, just in case.
Regulations Back Up Daily Habits
OSHA and the EPA both recognize sodium nitrate as a substance calling for special handling. Facilities must keep full documentation and train workers regularly. One audit caught our team off guard with surprise spot checks — and I’m glad they did. Routine drills helped us prepare for the rare but real risk of fire or spill. Many accidents come from complacency, not ignorance.
Looking for Solutions and Smarter Practices
The best way forward involves dedication to regular training, updates to material safety datasheets, and clear communication across departments. Simple investments in ventilation and humidity control gear can prevent major headaches down the line. If community emergency crews know what’s on-site and where, their response sharpens in a crisis.
In my experience, the safest environments come from teams where everyone takes responsibility, not just the manager on duty. Respect for sodium nitrate means more than rules; it’s a daily mindset. As more industries rely on chemicals, people’s safety both inside and outside factories depends on consistent application of these smart habits.
What industries commonly use sodium nitrate?
Fertilizer: Boosting Crop Yields and Food Security
Agriculture relies on a handful of key chemicals, and sodium nitrate plays a big role in that ecosystem. Farmers use it as a quick source of nitrogen, applying it to soils that struggle to feed crops what they need. Nitrogen fuels leaf and stem growth, so it’s tough to talk about food supplies without recognizing the impact of these fertilizers. In drought-prone regions, the ability to get a reliable harvest sometimes comes down to using the right inputs, and sodium nitrate keeps food shelves fuller around the world.
Chemicals and Explosives: More Than Just Mining
Sodium nitrate goes beyond farms. Factories turn to it for making explosives—think mining, quarrying, and even road construction. It works as an oxidizing agent, providing fuel so that explosives can do their job breaking through hard rock. The need for minerals, metals, and construction materials continues to grow, and sodium nitrate sits in the middle of that process. Some might picture explosives as a rough business, but safety and performance stem from a solid chemical foundation. That means sodium nitrate indirectly helps build roads, tunnels, and even the concrete beneath our feet.
Food Preservation: Tradition Meets Modern Manufacturing
People have relied on sodium nitrate in the kitchen for centuries, especially to keep meats safe and tasty. Before refrigeration, salting and curing with compounds like sodium nitrate offered a reliable way to prevent spoilage. Even today, you’ll find it listed on the back of bacon or salami packages. It keeps meats pink, just like folks expect, and slows down bacteria that can cause sickness. Modern food processing uses much less of it than in the past, but some traditions haven’t changed, especially in regions where dry-cured hams, sausages, and similar foods remain a local staple.
Glass and Ceramics: Finishing Touches and Functional Beauty
Artisans and engineers both value sodium nitrate for producing glass and glazes. In glassmaking, it removes bubbles and keeps the product clear. Ceramics manufacturers get smoother, brighter finishes for tiles and pottery thanks to chemical reactions that happen during firing. This side of sodium nitrate doesn’t get as much attention as food or farming, but every time someone admires stained glass or walks on a shiny tile, chemistry deserves a nod.
Wastewater Treatment and Environmental Cleanup
Not every use of sodium nitrate shows up on store shelves. Wastewater treatment facilities depend on it to manage unwanted chemicals in runoff and sewage. By feeding bacteria that break down ammonia or help remove toxic metals, sodium nitrate helps prevent pollution from making its way into rivers and lakes. Clean water, often taken for granted, gets a boost from these chemical helpers.
Challenges and Safer Alternatives
Relying on any single chemical creates risks. Fertilizer run-off can lead to algae blooms and dead zones in lakes and oceans. Preserving food with sodium nitrate raises concerns when used in excess, as byproducts like nitrosamines can pose health risks. Keeping these industries safe means regular checks, precise dosing, and movement toward greener options. In farming, for example, better soil testing and improved irrigation lower reliance on chemical fertilizers. In food, curing with natural alternatives or reducing overall sodium content responds to modern health concerns.
Looking Ahead
Sodium nitrate continues to shape everything from dinner tables to city skylines. The balance between practical need and responsible use calls for transparency, regular monitoring, and a willingness to adopt new approaches. Supporting strong science education, robust safety standards, and honest discussion helps industries and communities stay prepared for changes ahead.
| Names | |
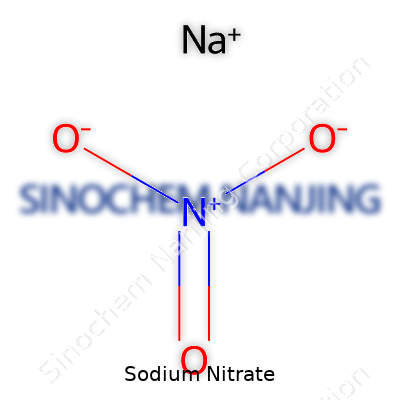
| Preferred IUPAC name | Sodium nitrate |
| Other names |
Chile saltpeter Nitratine Soda niter Norwegian saltpeter |
| Pronunciation | /ˈsəʊdiəm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 7631-99-4 |
| Beilstein Reference | 358990 |
| ChEBI | CHEBI:76233 |
| ChEMBL | CHEMBL1355 |
| ChemSpider | 11021 |
| DrugBank | DB06754 |
| ECHA InfoCard | ECHA InfoCard: 036c7d56-1b7d-418d-8b2f-f139586b64ff |
| EC Number | 231-554-3 |
| Gmelin Reference | Gmelin Reference: **1454** |
| KEGG | C00208 |
| MeSH | D009687 |
| PubChem CID | 24268 |
| RTECS number | WC5600000 |
| UNII | O5QNCJYY9P |
| UN number | 1498 |
| Properties | |
| Chemical formula | NaNO3 |
| Molar mass | 84.994 g/mol |
| Appearance | White, crystalline solid |
| Odor | Odorless |
| Density | 2.26 g/cm³ |
| Solubility in water | 82.3 g/100 mL (25 °C) |
| log P | -3.7 |
| Vapor pressure | 1 mm Hg (865°C) |
| Acidity (pKa) | pKa ≈ -1.3 |
| Basicity (pKb) | pKb > 7 |
| Magnetic susceptibility (χ) | +250.0e-6 cm³/mol |
| Refractive index (nD) | 1.586 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 116.5 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | −467.85 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –467.21 kJ/mol |
| Pharmacology | |
| ATC code | V03AB13 |
| Hazards | |
| Main hazards | Oxidizing; may intensify fire; harmful if swallowed; may cause respiratory, eye, and skin irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS03,GHS07 |
| Signal word | Warning |
| Hazard statements | H272, H319 |
| Precautionary statements | P210, P220, P221, P264, P270, P280, P301+P312, P305+P351+P338, P306+P360, P370+P378, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 0, Instability: 0, Special: OX |
| Autoignition temperature | 380°C (716°F) |
| Lethal dose or concentration | LD50 Oral Rat 1267 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1267 mg/kg (oral, rat) |
| NIOSH | SDW23000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Sodium Nitrate: Not established |
| REL (Recommended) | 500 mg/m³ |
| IDLH (Immediate danger) | 6300 mg/m3 |
| Related compounds | |
| Related compounds |
Nitric acid Sodium nitrite Potassium nitrate Ammonium nitrate Sodium chlorate |

