Sodium Formate: More Than Just a Chemical Compound
Historical Development: From Lab Curiosity to Industrial Mainstay
Sodium formate didn’t show up on the radar as a must-have ingredient overnight. Early chemists first stumbled across it in the 19th century while exploring the reactions between sodium compounds and formic acid. Many years back, you might find it as just a byproduct in other chemical syntheses, not really the star of the show. Over time, because of the increasing need to treat icy roads, tan leather, and support various chemical syntheses, researchers and industry started to pay closer attention. Its growth closely follows broader trends in science—any time someone needed a stable, water-soluble formate salt, sodium formate kept turning up as the right answer. Its industrial role expanded steadily as manufacturing methods improved and as its benefits over related compounds became better understood.
Product Overview: Everyday Uses Shape Its Relevance
In the real world, sodium formate shows up all over, whether people notice or not. It’s not just sitting in some backroom at a chemical plant. Every cold winter, road maintenance crews load up their trucks with sodium formate-based deicers because it protects the concrete and the environment far better than its old cousin, sodium chloride. In the leather industry, tanners rely on it to improve efficiency and reduce pollution, making hides softer without all the harsh effects of other chemicals. Chemists see it as a key intermediate, especially for producing formic acid or as a reducing agent. It’s a simple-looking material, but its utility stretches far beyond what people see on the label.
Physical and Chemical Properties: The Basics Matter
Sodium formate stands as a white, crystalline powder—sometimes looking almost like table salt—yet it tells a different story in the lab. It dissolves easily in water, which means it’s ready to react or blend wherever needed. The compound’s melting point sits high enough to remain solid in tough conditions but not so high that it resists decomposition in strong heat. Its mild formate odor emerges especially in damp conditions. Chemically, sodium formate ranks as a reliable reducing agent. Once dissolved, its basic pH means that it’s sometimes used to balance acidic solutions. The chemical stability and ease of handling make it less of a headache than some related compounds, which matters if you work in environments that can’t deal with volatile substances.
Technical Specifications and Labeling: Regulations Guide the Details
US and EU regulators set strict guidelines on how sodium formate must appear on labels. Purity requirements remain high, especially if it’s headed into food-processing, pharmaceuticals, or leather tanning. Typical specs demand minimal levels of heavy metals and clear batch traceability. Practical experience says don’t skimp on this—impurities not only mess up results but also affect safety. Accurate labeling reflects everything from lot numbers to shelf life, supporting safety in both lab and field use. This keeps operations smooth and avoids the legal headaches that come from regulatory missteps.
Preparation Method: Simplicity Feeds Large-Scale Production
Most large-scale sodium formate comes from reacting caustic soda (sodium hydroxide) with either formic acid or methyl formate. Earlier, some producers relied on the reaction of sodium carbonate with formic acid, but industry has mostly settled on the caustic soda version for cost-effectiveness and consistent yield. It’s not especially energy-intensive compared to many industrial syntheses. Operators tend to favor batch reactors to keep reaction control tight. Using excess formic acid snags a better purity, which makes downstream cleaning less of a hassle. After that, you’re left with a pretty pure product that only requires filtration, evaporation, and drying.
Chemical Reactions and Modifications: From Bench Chemistry to Industry
Sodium formate doesn’t just sit around once produced. In organic chemistry, it’s a reliable source of formate ions, so it’s been picked up for use as a buffering agent and mild reducing agent in many types of syntheses. In the reduction of certain metal salts, formate salts, including sodium formate, help produce fine metallic powders—think silver or gold—important in electronics and advanced materials research. Its reaction with strong acids quickly produces formic acid, which is useful when only a little formic acid is needed onsite. Sodium formate also plays a role in the production of oxalates, and under catalytic conditions, some researchers use it in transfer hydrogenation, which broadens its reach in fine chemical production.
Synonyms and Product Names: Reading Between the Lines
Chemistry isn’t always user-friendly when it comes to names. Sodium formate might also appear as “sodium methanoate” in textbooks or regulatory filings. Older texts refer to "formylic acid, sodium salt." The name might vary, but whether listed as E237 in food processing or plainly as “sodium formate” on an MSDS, it’s the same chemical. International trade sometimes introduces local spelling preferences or translations, which can complicate procurement or regulatory reporting if you’re not paying attention.
Safety and Operational Standards: Keeping Risks in Check
People sometimes take sodium formate’s presumed safety for granted, but every chemical carries its baggage. Direct skin or eye contact can lead to mild irritation. The bigger exposure risks come with dust, which is why wearing gloves and proper eye protection is just smart business. Industrial and lab protocols usually call for solid ventilation and dust management. Larger spills don’t pose catastrophic hazards to the environment but should be managed promptly to avoid run-off or unnecessary waste disposal costs. Wastewater treatment plants can handle it with standard techniques, which underlines why local regulations still oversee its use closely.
Application Area: Real-World Use Cases Spell Out Value
Real experience shows sodium formate’s influence everywhere from roadways to manufacturing lines. Ice control on city streets stands out as a success story—it proved itself less corrosive, friendlier to groundwater, and safer around animals compared to older salts. Leather tanners count it as a mainstay because it boosts the efficiency of tanning baths, reducing both cost and environmental impact. In the chemical industry, it’s an important intermediate, especially for making formic acid. Oil and gas drilling outfits rely on sodium formate brines to keep wells flowing efficiently, even in deep, high-pressure environments. Research teams have turned to it in specialty applications, including solar cell production, thanks to its chemical reducing ability. Its presence in food preservation pops up more rarely but remains legal under certain food safety codes.
Research and Development: Scientists Still Dig Deeper
Interest in sodium formate doesn’t just fade after high school chemistry. Researchers keep looking for new applications and greener production pathways. Some teams focus on turning biogenic waste into formic acid derivatives, including sodium formate, which brings circular economy ideas closer to reality. Others look at how blending sodium formate with other deicers or tanning agents could cut costs even further for big operations. There’s been work on catalytic uses too, including in hydrogen storage, since sodium formate can decompose to release hydrogen under certain conditions. This feature makes it a focus in the search for better hydrogen carriers and safer chemical storage.
Toxicity Research: Safety Realities and Environmental Caution
People sometimes assume sodium formate’s low toxicity means it’s entirely harmless. Animal studies show the compound breaks down quickly in the body, converting to natural metabolites, with only minor impacts unless exposures are very high. Chronic exposure, especially by inhalation on the job, remains a concern in certain settings, which underscores the importance of up-to-date occupational hygiene measures. Environmental researchers track its fate in soil and water to ensure it doesn’t build up or cause harm to local ecosystems. So far, evidence says it degrades readily, which puts it ahead of many related chemicals, but researchers don’t want to be caught off-guard by long-term surprises.
Future Prospects: Where It’s Heading
Sodium formate's future looks tied to sustainability trends and smarter manufacturing. As demand grows for safer, more effective de-icing products, its market footprint won’t be shrinking. Synthetic chemists are finding new ways to unlock its value in energy storage and catalytic cycles, which suggests sodium formate could play a bigger role in clean technology. Advances in recovery and recycling, especially from industrial waste streams, show promise for closing the loop in chemical manufacturing. Whenever industry or researchers look for a stable, affordable, and versatile compound, sodium formate’s track record almost always puts it on the shortlist—and the story is nowhere near finished.
What is Sodium Formate used for?
A Closer Look at an Underappreciated Workhorse
Most people walk past shelves of cleaning products or bags of de-icer at the hardware store and never hear the name sodium formate. But the white, salt-like powder quietly pulls more weight than the label suggests. I learned this first during a stint at a small construction site, scraping stubborn ice off a delivery ramp in the predawn dark. I tossed sodium formate on the ground and watched as the slush cleared, making it safer for everyone showing up before sunrise.
Keeping Roads and Airports Running
Sodium formate steps in as a de-icer with a friendlier face than rock salt. Traditional chloride salts corrode bridges, highway rebar, and car undercarriages. Sodium formate breaks up ice efficiently with less damage to concrete and steel. It doesn’t leave a slippery, polluted mess behind, so airports turn to it during snowstorms. This isn’t just about convenience — thousands of flights depend on safer runways, and cities avoid million-dollar repair bills tied to salt corrosion. Agencies in North America and northern Europe have used it to reduce maintenance costs, and some airports have noted fewer issues with electrical systems after making the switch.
Needed in Leather Production
Beneath the surface of a new leather belt or car seat, sodium formate helps out again. Tanneries use it to adjust pH during the chrome tanning process. This keeps the leather supple and prevents it from stiffening. Many European tanneries favor sodium formate because it delivers consistent results and reduces the release of hazardous byproducts. Better-managed tanning benefits leather workers, the rivers near processing plants, and, eventually, consumers who expect their shoes to last.
Working for Our Health and the Environment
Nearly every chemical formula relies on steady, predictable reactions. Chemical manufacturing companies use sodium formate as a reducing agent — a substance that helps spark the right change without adding new risks. Pharmaceuticals often need a safe reagent for certain steps, and sodium formate fills the role, contributing to the quality and cost-effectiveness of antibiotics and specialty drugs.
Cleaner energy production ties into this story. When oil fields need a brine to help push crude from deep underground, operators look for salts that won’t damage drilling equipment, harm the local water table, or spark regulatory nightmares. Sodium formate, collected after the process, delivers a lower environmental burden. If you’ve ever relied on heating oil or worked with engineers trying to limit groundwater pollution, you probably see why practical options matter.
What Could Be Better?
Demand keeps climbing as cities grow, more cars hit the pavement, and technical standards get tighter. That adds pressure on suppliers and makes it tougher to ensure every batch meets safety expectations. Some manufacturers invest in stricter quality checks and pursue cleaner production routes that limit waste. Researchers keep searching for additives to lower costs or boost performance in harsh climates, focusing on real-world conditions over theoretical improvements.
Trade groups and regulators could do more to share test results and lessons from field applications, giving buyers better data. By grounding choices in solid evidence and firsthand experience, companies and cities can use sodium formate wisely, cutting down risks and saving money without sacrificing progress.
Is Sodium Formate safe for humans and the environment?
How Sodium Formate Shows Up in Everyday Life
Sodium formate pops up in different corners of daily life. It keeps airport runways free of ice during the winter months, helps in leather tanning, and works as a buffering agent in the labs. The name might not sound familiar to most shoppers, but many have indirectly crossed its path. Over the years, questions about its impact on health and the world outside our doors keep cropping up. It’s worth digging into these concerns with clear eyes instead of losing the signal in chemical jargon.
Human Health and Direct Contact
Next to the kitchen sink or pantry, sodium formate has no role. It rarely finds its way into the food chain, which lowers the risk of unintentional consumption. Still, jobs in airport operations, tanneries, and factories sometimes push people into closer contact. The compound generally causes little to no irritation with brief, small-scale touch or skin exposure. If large amounts make contact, skin or eye irritation can develop. Reliable sources like the U.S. National Library of Medicine and European Chemicals Agency confirm that with personal protective equipment (gloves, goggles), any risk sharply drops. As someone who has worked in environments with chemical use, I learned early on that gloves and eye protection become as necessary as any tool in the shed.
Swallowing or inhaling sodium formate in bulk can lead to problems – nausea, headache, or even metabolic disturbances at high doses. Medical literature shows these situations almost never occur outside of mishaps at industrial facilities. I’ve stood on job sites where vigilance and regular safety drills do more to protect workers than any warning posted on a wall.
Impact on Nature and Wildlife
The bigger worry for many people revolves around what sodium formate does after it washes off icy runways or factory floors. Compared to chloride deicers, which collect in soil and waterways and harm plant roots and aquatic habitats, sodium formate breaks down quickly. University studies and government agency reports show that microbes in the soil convert it to carbon dioxide and water. This breakdown process limits buildup and reduces threats to plants, fish, and amphibians.
I grew up near a river impacted by road salt runoff each winter. Water quality dropped, and fish vanished for months. Over years of watching this cycle, I could see why cities now search for safer deicers. Tests run by the EPA and Canadian environmental labs highlight that sodium formate’s lower toxicity makes it a better option. Still, dumping vast amounts of any chemical onto the ground deserves careful management.
Making Sensible Choices
Every chemical comes with trade-offs. Deciding to use sodium formate means looking at application rates, alternatives, and how to handle spills responsibly. Practical training for those working with it stands as the first layer of protection. Cities and companies that stick to recommended amounts and build containment systems stop leaks before they leave the parking lot or factory gate.
People and the planet share the same water, soil, and air. Sodium formate offers a softer touch compared to more stubborn pollutants, but wise use matters. Solutions always depend on attention to detail, routine checks, and people who care enough to do the job right. As with most safety questions, it isn’t just about the chemistry, but how we handle our responsibility to each other and to the environment we all count on.
What are the physical and chemical properties of Sodium Formate?
Physical Nature That Stands Out
Sodium formate often takes the form of white powder or colorless crystals. Its texture looks a lot like fine table salt—only far less tasty, of course. Pick up a handful, and you’ll notice how easy it dissolves in water. You can even stir it in cold water without much trouble, which says a lot about its solubility. This makes it handy for practical applications: from deicers for airport runways to leather tanning.
Boiling it down to details, sodium formate’s melting point lands around 253°C. The substance tends to absorb moisture from humid air, making it slightly deliquescent. This can turn it a bit clumpy if you leave it out of a sealed bag. You’ll rarely catch any odor from pure sodium formate, and most folks would have a tough time telling it apart from similar white salts just by looking at it.
Chemical Properties That Drive Its Usefulness
Pop sodium formate in water, and it breaks apart straight away—forming sodium ions and formate ions. This property shows why it makes a reliable buffering agent in chemistry labs. You won’t find it putting up much of a fight when involved in chemical reactions, either.
Sodium formate, being a salt of formic acid, brings a slightly basic solution when you dissolve it in water. That’s why wastewater treatment plants consider it when they want to buffer solutions and control pH levels. It acts as a reducing agent too, which comes in handy for various reductions in industry. Mix it with strong acids, and the formic acid gets released again.
Heat up sodium formate, and it breaks down into formic acid and sodium oxalate. At even higher temperatures, you’ll find it decomposing further into sodium carbonate and gases such as hydrogen and carbon monoxide. Chemists take advantage of its predictable reactions for industrial synthesis, especially in the dye and textile sectors.
Practical Impact and Safety Concerns
Factories use sodium formate for more than just chemical reactions. In the winter, spreading it on icy roads keeps people safe without damaging concrete or corroding vehicles as much as old-fashioned rock salt. The substance also serves as a feed additive and sometimes as a preservative, although anyone using it in food should remember that regulation varies widely among countries.
On the safety front, sodium formate brings relatively low risk. It doesn’t carry major toxic effects at the concentrations people use commercially. Long-term exposure or large amounts could irritate the skin, eyes, or throat, so gloves and goggles make sense if you handle bulk quantities.
Challenges in Handling and Solutions
Sometimes sodium formate picks up enough water from the air to turn sticky, which complicates storage. Factories solve this by storing it in airtight bins and controlling humidity—simple steps that make a difference in product shelf life. Since it dissolves so easily, spills need fast attention to keep coverage confined.
As industries look for more eco-friendly solutions for deicing and chemical manufacturing, sodium formate sits near the top of the list. Researchers continue testing its full potential, but evidence points to real gains when it replaces more hazardous chemicals.
How should Sodium Formate be stored and handled?
Keeping the Substance Dry and Secure
Sodium formate seems pretty harmless at first glance. It’s a white powder, not some fuming or bubbling chemical you’d see in a science fiction movie. Still, experience working around industrial and lab chemicals has shown me one rule wins every time: never judge a compound by its looks. Moisture gets to sodium formate fast. If it sits exposed or the storage room feels muggy, the powder can clump, turn mushy, or react in ways nobody wants. Sealed containers, tight lids, and a cool, dry spot make all the difference. Walk into any chemical supply warehouse and you’ll spot sturdy plastic drums or heavy-duty bags, stacked off the floor and far from steam lines or leaky pipes.
Avoiding Exposure—For People and the Product
Gloves and goggles aren’t just for show. Even though sodium formate doesn’t eat through skin, it dries it out and can irritate eyes. The real trouble starts when someone forgets to clean up a spill or brushes dust off a workbench with bare hands. Proper handling takes discipline. Wash hands before and after working with it. Don’t eat or drink near storage areas. Signage helps, but real safety comes from habits built over time.
Understanding Chemical Interactions
Mixing sodium formate with the wrong materials leads to fire risks or toxic fumes. Years ago, a colleague got a scare by storing sodium formate near strong acids “just for a minute while reorganizing.” Acids and formates can react to let out carbon monoxide — a silent, deadly gas. Storing away from acids, oxidizers, and even some common cleaning agents isn’t just a recommendation. It’s a lifeline. Written protocols on shelf assignments might sound boring, but I’ve seen them prevent headaches and accidents time after time.
Training and Preparation
No one wants to read another bland safety pamphlet, but hands-on walkthroughs make protocols stick. Taking a new team member through storage rooms, opening containers safely, and practicing spill clean-up creates real muscle memory. Quick-access spill kits, proper labeling, and clear instructions posted nearby keep everyone on track. Any confusion about material safety or handling should get answers, not guesses. Supervisors earn their keep most on days when things nearly go wrong—but don’t, because teams stay sharp and prepared.
Simple Steps That Matter in the Long Run
Labeling and routine inspections can catch storage problems before small issues grow. A weekly check for leaks, damage, or moisture in containers uncovers potential trouble before it escalates. Inventories aren’t just for accounting—knowing exactly what’s on hand clears up confusion if an emergency hits. Ingredients matter, too; only keep sodium formate of known quality with up-to-date records and safety data sheets on file.
Staying Ahead of Problems
Over the years I’ve watched teams that take shortcuts end up with product loss or avoidable injuries. The ones that treat chemical safety as a top priority get better results every time. Sodium formate doesn’t cause big problems on its own, but improper storage and careless handling stack small risks into major ones. The best solution always begins with respect for the tools and materials in front of you, and sodium formate is no exception.
What are the main industries that use Sodium Formate?
Deicing Roads and Runways
Anyone who’s watched city trucks scatter salt on icy highways might not realize the story doesn’t stop with rock salt. Many airports depend on sodium formate to keep runways safe during long winters. I’ve seen how standard road salt can corrode concrete and metal—prompting big airports to turn to sodium formate. It gets the job done without eating away at critical surfaces or wrecking vehicles. Planes need to take off on time, and sodium formate lets ground crews clear ice efficiently without worrying about long-term damage. The material also breaks down fast, so it doesn’t sit in the environment like old-school salts.
Leather Tanning and Processing
Leather work has been around for centuries, but modern tanneries rely on chemicals to speed up the process and produce softer hides. Sodium formate helps in the chrome tanning step, where animal skins move from raw material to durable leather. By keeping pH levels steady, it makes chrome work without damaging the hides. I’ve talked to folks in the leather industry who deal with massive volumes each day. Small disruptions in chemical balance can mean ruined products. Sodium formate helps them avoid costly mishaps and turn out leather that lasts.
Oil and Gas Drilling
Oil and gas companies drill deep underground, facing tricky conditions with high pressure and temperature. In these muddy, shifting holes, they use sodium formate to help create “drilling mud.” This keeps boreholes from collapsing and controls the pressure as they work. I’ve read reports showing drilling fluids with formates reduce corrosion and lower the risks of blowouts. Teams on rigs rely on tough, predictable materials. Sodium formate gives them confidence in unpredictable underground spaces, making tough jobs a little more manageable.
Chemical Manufacturing
Sodium formate isn’t just a final product—it feeds into bigger things. Chemical plants use it to make formic acid and oxalic acid, which pop up in everything from cleaning products to pharmaceuticals. Industry veterans look for starting materials that react cleanly and leave little waste behind, and sodium formate fits the bill. I’ve noticed companies favoring it in processes where quality and yield really matter. The push toward more eco-friendly operations keeps giving sodium formate more room in formulas where reliability counts.
Textile Dyeing
Dye houses turn plain fabric into colorful clothes, sheets, and more. Sodium formate gets involved to keep dyes even and prevent fading. From what I’ve seen in textile plants, a quality finish matters a lot—no one wants shirts that bleed color in the wash. Sodium formate evens out the dyeing process and gives finished products their staying power. These companies look for chemicals that handle high volumes and deliver the color customers see on store shelves.
Cutting Costs and Improving Safety
Across these diverse sectors, sodium formate offers a safer, often cheaper alternative to harsher chemicals. Deicing with sodium formate means fewer potholes and less metal rust, leading to lower repair bills for cities and airports. In oilfields and tanneries, safer chemical handling cuts accident rates and keeps more workers healthy. The industries using sodium formate aren’t just thinking about today’s problems—they’re watching how it fits into longer-term goals for cleaner, more sustainable practices.
Looking Ahead
Innovators in each of these fields keep searching for ways to improve safety, performance, and environmental results. As regulations tighten and companies compete on greener strategies, I see sodium formate carving out a bigger place on chemical supply lists. Leaders in infrastructure, energy, and manufacturing won’t just rely on tradition—they’ll keep adapting ingredients like sodium formate to solve new challenges.
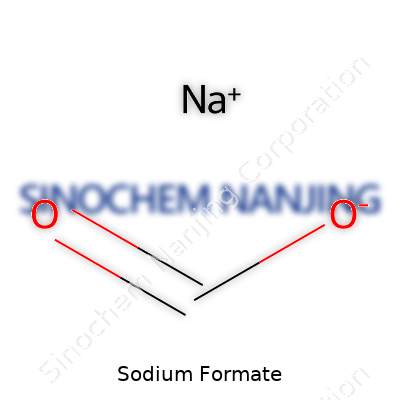
| Names | |
| Preferred IUPAC name | Sodium methanoate |
| Other names |
Sodium methanoate Formic acid sodium salt |
| Pronunciation | /ˈsəʊdiəm ˈfɔːr.meɪt/ |
| Identifiers | |
| CAS Number | 141-53-7 |
| 3D model (JSmol) | `data/mol/Sodium-formate-molecule.jmol` |
| Beilstein Reference | 1739904 |
| ChEBI | CHEBI:29377 |
| ChEMBL | CHEMBL1351 |
| ChemSpider | 4876 |
| DrugBank | DB09294 |
| ECHA InfoCard | 100.006.376 |
| EC Number | 205-488-0 |
| Gmelin Reference | 787 |
| KEGG | C02432 |
| MeSH | D013007 |
| PubChem CID | 27206 |
| RTECS number | NM7520000 |
| UNII | 9NEZ333N27 |
| UN number | UN2818 |
| Properties | |
| Chemical formula | HCOONa |
| Molar mass | 68.01 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.92 g/cm³ |
| Solubility in water | Very soluble |
| log P | -4.01 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.75 |
| Basicity (pKb) | 8.37 |
| Magnetic susceptibility (χ) | −20.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.42 |
| Viscosity | Low to medium |
| Dipole moment | 0.925 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 91.6 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -567.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -635.0 kJ/mol |
| Pharmacology | |
| ATC code | V03AB06 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | Sodium Formate: 1-0-0 |
| Autoignition temperature | > 500 °C |
| Lethal dose or concentration | LD50 oral rat 11,200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 11,200 mg/kg |
| NIOSH | UN1604 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL-TWA 5 mg/m3 |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Formic acid Potassium formate Calcium formate Sodium acetate Sodium oxalate |

