Sodium Dithionite: A Closer Look at a Versatile Reducing Agent
A Journey Through History
Sodium dithionite came about through a search for a more effective way to reduce dyes in textile factories before the twentieth century ended. Chemists working with varying sulfite and SO2-based compounds noticed that sodium dithionite—previously a scientific curiosity—could actually pull oxygen out of certain solutions with impressive speed. The textile industry, hungry for anything that could make indigo and vat dyes easier to handle and more vibrant, jumped on this newfound compound. By the mid-1900s, sodium dithionite plants turned what started out as small-batch chemistry into a crowded landscape of industrial tanks and pipes. Factories across Asia, North America, and Europe all found a reason to keep producing this reliable powder.
What Sets Sodium Dithionite Apart?
Many know sodium dithionite as a reducing agent that comes as a white, crystalline powder—though if you keep it around, moisture makes it break down and clump up fast. The scent of burnt matches hovers over an open bag, which doesn’t surprise anyone who knows sulfur chemistry. Sodium dithionite dissolves in water quickly, forming a clear solution that feels almost neutral but tends toward the alkaline. Because of its strong reducing ability, the chemical likes to react with oxygen in air, making any storage or transfer a race against time.
The Science Under the Hood
Physically, sodium dithionite looks simple, but chemically it packs a punch. With a molecular formula of Na2S2O4 and a molar mass just shy of 175 grams per mole, this compound contains both sodium and sulfur in a unique arrangement. It stays stable at lower temperatures and in dry, airtight containers, but hit it with water or a spark and you get rapid decomposition—releasing sulfur dioxide and other byproducts best avoided. The solid breaks down quickly under acidic or heated conditions and loses its strength when exposed to air, underscoring the need for careful handling. The chemistry giving sodium dithionite these properties traces right back to its two sulfur atoms, linked tightly together and tuned to transfer electrons with ease. That’s the source of its reducing power, making it useful for stripping oxygen or other unwanted groups from a wide range of molecules.
Methods of Making Sodium Dithionite
My first encounter with industrial sodium dithionite came in a paper mill, where flammable gases and odd-smelling tanks marked every step of its manufacture. Factories often start with sodium bisulfite and reduce it using powdered zinc or sodium formate as the electron donor. Large tanks churn this mixture while keeping the temperature in check—overshoot, and runaway reactions spoil the batch. Filtering removes the leftover sludge, and evaporators finish the product into a solid white powder. Some methods use SO2 as a starting point, mixing it with sodium carbonate and a reducing agent to get to the same result. Every process feels like a balancing act: too little water and the powder clumps, too much oxygen and everything goes to waste.
Technical and Regulatory Touchpoints
Chemical supply chains make a big deal about labeling and purity for a reason—too many industries live or die by reagent quality. Sodium dithionite, sold under names like sodium hydrosulfite or dithionous acid disodium salt, usually comes in bags with warning labels about heat, air, and water. Regulations vary by region, but nobody in their right mind lets this stuff near food or direct human contact. Labs go through strict protocols for handling, storage, and disposal. Drum storage gets priority, and workers monitor humidity with diligence, since anything above a low threshold can trigger unwanted reactions. Labeling touches on batch purity, but also details like residual zinc or iron—trace amounts that can matter for high-precision dye work or specialty paper coatings.
Common Synonyms and Names
Across the globe, sodium dithionite gets called by a mix of trade and chemical names. Older chemists stick with “sodium hydrosulfite,” a term reflecting an earlier, less-precise grasp of its structure. Product brochures in textile regions often use “dithionite,” while less formal vendors just call it “sodium hypo.” Each name comes with its own community customs, safety lingo, and cautionary tales. In research papers, the IUPAC name appears: disodium oxido-oxo-sulfanylidene-λ4-sulfanylium. Most chemists don’t bother, preferring the terms that keep communication quick.
Reactions and Chemistry Lab Modifications
Every chemistry teacher I knew in school warned us about sodium dithionite’s thirst for oxygen. That same trait makes the compound valuable for reducing dye intermediates, stripping colors out of recycled paper pulp, or cleaning old paintings. The basic reaction releases sodium sulfate, a product of little value in itself but harmless to most wastewater systems. Sometimes chemists tweak the molecule by pairing it with stabilizers or blending with other reducing agents to stretch its shelf life. These combinations allow for slower release in some processes, such as ore flotation or advanced material synthesis, where timing and strength in electron transfer matter more than raw power.
Risks and Best Practices in Handling
Anyone who has spent hours around open tyvek bags or handled spills in a warehouse knows sodium dithionite is tricky—mix it with water and you can generate heat and gases like sulfur dioxide that burn the nose and lungs. OSHA and similar agencies require gloves, eye protection, and fresh air, especially in mixing rooms. Safety showers get tested more than once a week. It pays to have every employee know what to do with a cloud of SO2 or a spilled pile—neutralizing with lime and never washing straight into a drain. Fire risk stays low except if strong oxidizers are around. Insurance inspectors insist on separation from anything that increases the chance of ignition or accidental decomposition, and experienced supervisors never let dusty bags pile too high.
Applications That Rely on Sodium Dithionite
For a decade, sodium dithionite turned up in every textile plant I visited, always as the go-to solution for reducing complex dyes down to the colorless, soluble forms that stick to fiber and then turn brilliant and permanent after exposure to air. Paper manufacturers use it to de-ink recycled material, restoring brightness to lower-cost grades of printer and newsprint paper. So-called vat dyes, which produce the deepest blues and blacks, simply cannot be run without a reliable supply. Beyond dyes, mineral processors look to the compound to separate ores, especially those that need delicate treatment before extracting valuable metals. On the water treatment side, some engineers rely on sodium dithionite to remove heavy metals or unwanted chlorates, though more often, it serves as a finishing step rather than a core solution. Occasionally, the food industry used it to bleach certain products, but regulatory pushback and better alternatives have pushed that trade to the margins.
Where Research Points Next
The science behind sodium dithionite actually energizes quite a bit of research. Academic groups keep tinkering with its structure for use as a reducing agent in organic synthesis—especially in pharmaceutical and specialty chemical laboratories. Some labs test encapsulating the powder in polymers or mixing it with other agents to prevent breakdown before use. Environmental scientists explore how to use this reducing power to remediate groundwater, treating sites contaminated with heavy metals or chlorinated compounds, where other agents either cost too much or don’t reach deep enough into the ground. Papers keep appearing that document how changing production conditions or stabilizing additives can minimize dust, odor, and risk—making safer workplaces and more reliable final products.
Toxicity and Worker Health
Fresh out of college, I saw a chemical burns case after an untrained employee accidentally inhaled dust while cleaning out a shipment truck. Many think a little dust can't harm, but sodium dithionite can irritate the lungs and cause headaches, nausea, and on rare occasions, more serious respiratory stress. Chronic exposure doesn’t lead to long-term toxicity in the ways that heavy metals do, but short-term accidents make employers rethink training and gear. Toxicity studies on aquatic systems matter just as much—runoff, even in small quantities, can affect streams and rivers, though it tends to break down rapidly. Its byproducts, mostly sulfate ions, are safer but still need monitoring if discharged in large quantities. Research suggests keeping limits tight in both human and environmental exposure, with fast cleanup systems and constant air monitoring in warehouse and production settings.
Looking Forward: The Road Ahead for Sodium Dithionite
Anyone who tracks trends in manufacturing senses a slow shift. Environmental rules keep getting stricter. Factories invest more in recovery systems and try to find methods to recycle leftover solutions and filter off solid residues. Companies chase new production methods that use less energy, produce fewer emissions, and make safer workplaces. Research into new, less hazardous reducing agents could chip away at sodium dithionite’s slice of the market, but right now, no stand-in offers the same balance of speed, cost, and strength. I expect big companies and small labs alike to adapt new safeguards before giving up the compound, combining automation, better ventilation, and constant monitoring to keep sodium dithionite’s benefits within reach without sacrificing worker health or environmental safety. As old industries fade and new ones emerge, the story of sodium dithionite will evolve alongside, adapting to demands few could predict during its early days in smoky dye works.
What is Sodium Dithionite used for?
A Substance With a Job to Do
Sodium dithionite doesn’t show up much in everyday conversations, but its work touches every closet, kitchen, and stack of office paper. Factories put it to use for a simple reason: it gets things clean and bright. I’ve worked in a textile town, and the smell from those plants sticks in your mind almost as much as the visuals—rows of cloth dunked in baths, pulled from vats, then set out to dry. Much of that cloth passes through a sodium dithionite wash. This compound strips away color from raw fabrics, turning off-white cotton into a blank page for new shades and prints.
Sodium Dithionite and Textile Life
Most jeans and T-shirts start out yellowish or grayish. Sodium dithionite steps in to bleach those fabrics, particularly in the process called vat dyeing. Indigo, the classic dye for blue jeans, works with it too. The chemical reduces indigo so fibers can grab it evenly. Companies care about sodium dithionite because it lets them deliver sharp, consistent colors and bright whites at scale.
Paper and Pulp Cleaning
Factories making paper chase brightness with the same sort of hunger as textile folks. Natural pulp looks dingy after coming off the first rollers. Adding a sodium dithionite solution to the mass lightens it up, cutting out unwanted pigments and contaminants. This isn’t about vanity—it prevents books, magazines, and boxes from looking dirty. People expect their paper goods to look fresh. The bleaching step powered by sodium dithionite keeps up with that demand.
Cleanup Crew in Chemicals
Sodium dithionite pops up where cleaning and decolorizing need some muscle. Chemical labs and big processing facilities use it to mop up polluted water streams, pulling out heavy metals and dyes. I came across a water treatment plant experiment trying to remove chromium from run-off. They used sodium dithionite to change chromium’s form, making it easier and safer to catch. This compound draws attention because it reacts fast and breaks down easily, so plants aren’t left with new waste headaches.
Food Processing: The Limits and the Laws
Some places once relied on sodium dithionite for “refining” certain foods—like making sugar whiter or decolorizing gelatin. But food scientists found that some breakdown products might cause trouble for health, so regulators cracked down. These days, food use is tightly controlled or outright banned in most countries, including the U.S and across the European Union. That’s not just bureaucracy—food safety watchdogs want to keep risky chemicals out of meals.
Sodium Dithionite in Mining and Beyond
Mining communities sometimes lean on sodium dithionite to help recover precious metals. Gold refining may use it to strip away impurities, proving once again that chemical know-how makes tough tasks possible. You also find small operations using it to process minerals for local industries.
Health, Safety, and Moving Forward
Working with sodium dithionite isn’t without challenges. It throws off sulfur dioxide gas, which can irritate the nose and throat if you inhale it too much. Anyone handling big batches inside factories must follow safety steps—ventilation, gloves, protective gear. Mistakes or short-cuts risk sickness. Public health agencies monitor how much ends up in air and water, trying to lower any impact on folks living near production plants.
Sodium dithionite may not be a household name, yet modern life leans on its strong reducing power. By keeping a close watch on its uses and side effects, industries can keep benefits while cutting down on accidents and pollution. This chemical keeps factories humming but calls for real responsibility at every step.
Is Sodium Dithionite hazardous or toxic?
What Sodium Dithionite Is and Where You See It
Sodium dithionite has a place in everyday products: papermaking, textile dyes, and even some water treatment processes. I’ve seen it change the way denim looks in jeans, turning them from that bright blue to the faded shade many people like. Despite its usefulness, stories about chemicals like this always spark the question—do they come with health risks?
How People Are Exposed
I grew up near a plant that made industrial bleach. We used to hear stories about workers who forgot their respirators and got dizzy or developed rashes after handling certain powders. Sodium dithionite is no stranger to such tales. Breathing in its dust or getting it on your skin can bring on irritation. Touch a lot of it, and it burns. Swallow it, and you’ll likely feel sick fast—nausea, vomiting, or worse.
Breaking Down the Hazards
Handling sodium dithionite without respect leads to trouble. As a strong reducing agent, it reacts fiercely when it meets acid or water. Those reactions can make sulfur dioxide gas, which has its own health effects. Sulfur dioxide stings the eyes and nose, and heavy exposure can leave someone coughing or wheezing for hours. The Occupational Safety and Health Administration lists sulfur dioxide as a regulated chemical, signaling researchers already recognize real risks with exposure.
Spontaneous combustion stories float around some workplaces. Sodium dithionite powder in a damp spot turns hot on its own, sometimes catching fire. Fires in chemical warehouses often start this way, and the smoke that comes off burns the lungs. Firefighters carry extra gear for exactly these scenarios, and chemical safety training makes all the difference.
What the Science Says
Toxicology reports from the World Health Organization and the US EPA show that sodium dithionite doesn’t stick around in the body or the environment. It breaks down fast with air and water. Chronic toxicity seems low, and cancer links remain unproven. Still, enough acute poisonings get reported every year that workplace exposure limits exist. Wearing gloves, goggles, and masks—plus having proper ventilation—cuts risks to almost nothing, provided people actually follow safety plans.
Health and the Big Picture
People sometimes think of chemical hazards as an all-or-nothing problem. It’s not that simple. Risks pile up in unsafe conditions: poor ventilation, sloppy storage, or lack of training. I’ve walked through old factories where barrels sat unlabeled or open, and the air scratched at your throat. That’s where accidents start. Sodium dithionite will never be as infamous as asbestos or lead, but it still wins respect because of how it behaves under the wrong conditions.
Responsible Use and Risk Reduction
Clear labeling goes a long way. Storage rules—keeping it dry and cool—actually prevent both workplace fires and accidental exposure. Simple changes, like automatic air exhausts or spill kits, have prevented disasters in places I’ve visited. Businesses that invest in training see fewer emergency calls and safer outcomes. Regulatory bodies urge reporting of exposures, too, giving us data to see patterns in health effects.
Personal accountability closes the loop. I’ve seen warehouse workers refuse to enter storage rooms without proper gear, and that’s experience speaking: once bitten, twice shy. If you spot white powder under a workbench, treat it like the unknown it is until you know for sure. Sodium dithionite rewards careful handling. With up-to-date procedures, it doesn’t pose a daily threat—but treating it carelessly is asking for trouble that can be avoided with respect and routine diligence.
How should Sodium Dithionite be stored?
Getting Real About Chemical Safety
Years ago, my first lab job involved the drudgery of checking chemical storage rooms. Most days it felt like a chore. That changed the first time I opened a sealed drum of sodium dithionite. A slap of rotten-egg stench hit me, and every warning label made sense. Sodium dithionite might look like plain white powder, but trust me—this stuff doesn't forgive laziness.
The Way This Chemical Behaves Matters
Sodium dithionite loves to react, especially with air and water. Exposed to humidity, it decomposes, releasing sulfur dioxide, a toxic gas with a distinctive choking odor. Dumping the powder into any open bin guarantees headaches and maybe worse. Every scientist who's handled sodium dithionite can tell a story about a leaky bag or an old jar gone crumbly—or the corrosion it leaves behind.
So, storage means thinking about the enemy: moisture. Even a single drop inside a container sets off trouble. Well-sealed, airtight containers rule the day here. Think robust jars made of hard plastic or glass, always with a gasket to keep out humid air. Metal canisters corrode and rust, so those stay on the shelf for other compounds.
Keeping Air Out and Reactions Down
Sodium dithionite doesn’t just fear water; let it mingle freely with air, and slowly it turns yellow and clumpy. That’s basically wasted money and a real safety risk. Some companies store the powder under a blanket of inert gas, like nitrogen, to keep things under control. That solution works in larger operations—where the investment pays off. For smaller labs, doing everything possible to seal up the compound and keeping it snug inside desiccators often gets the job done.
Heat makes things worse. Temperatures above room level encourage the powder to break down quicker. Experienced facilities tuck sodium dithionite away in cool, dry chemical storage rooms—never on a sunny shelf near a window or above radiators. The space stays clean and uncluttered. If something spills, crews sweep it up quick, using gloves and respirators. Sensitive chemical bins marked with hazard symbols serve as a constant, silent reminder: caution matters.
Staying Ready for the Unexpected
People sometimes forget how fast a material like this can turn disaster. At a textile dye house I visited, they kept drums on pallets, away from the outside wall, right below an emergency water sprinkler. Bad placement. If that system activated, the water would hit sodium dithionite directly, triggering gas release and a clean-up nightmare. One suggestion—always double-check where these stores sit. Keep them far from sources of water and out of the way of fire suppression systems that drench overhead.
Anyone handling sodium dithionite needs to know where the nearest eye wash, emergency shower, and fresh air exit is located. Unpacking or weighing the powder involves gloves, goggles, and often a dust mask. Only work with this substance in rooms with strong ventilation or local fume extraction systems. Training pays off—staff understand risk because people take the time to teach it right.
Common-Sense Rules From Hard Experience
Smart chemical management doesn't just protect balance sheets—it saves health, lives, and property. Skip shortcuts, document every container, rotate stock so old material moves out, and never let unknown or unlabeled powder lurk in back corners. Secure labeling and up-to-date records become part of the rhythm. Simple steps, nothing fancy, but those details make all the difference in chemical storage, especially for a stubborn powder like sodium dithionite.
What is the chemical formula for Sodium Dithionite?
Sodium Dithionite: More Than Just a Chemical Name
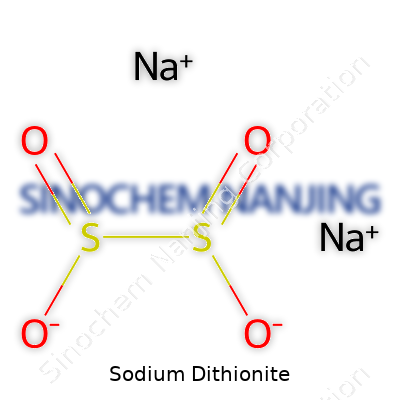
In science class, sodium dithionite’s name often pops up during lessons on industrial chemistry. The chemical formula is Na2S2O4. At first glance, those symbols and numbers might not say much, but this formula reveals a lot about the compound’s structure, reactivity, and why it matters in real-world applications.
Why Knowing the Formula Matters
Manufacturers and lab technicians rely on sodium dithionite's unique makeup for specific tasks. Every time I’ve worked with textile experts, they highlight its power as a reducing agent. This isn’t something that just happens by chance—its formula, with two sodium atoms bound to a double-hit of sulfur and oxygen, gives it just the right chemistry to strip color from fabric or paper gently, protecting fibers better than harsher bleaches. Scientists in water treatment and mining lean into its abilities as well, counting on the predictable outcomes driven by those two sodiums, two sulfurs, and four oxygens.
Real-World Impact Built on a Simple Formula
Getting the chemical formula right means more than passing a test. A misstep—using something with the wrong ratio—could waste materials, skew results, or even risk safety. About ten years ago, I helped troubleshoot for a paper mill. Their process suddenly started yielding yellowish sheets. Turns out, they were shipped an impure product that didn’t match Na2S2O4. The wrong blend stripped away less lignin, leading to the color problem and halting production. Quality control traced it back to someone misreading a shipment manifest, confusing sodium dithionite with sodium thiosulfate, which has a different formula and properties.
Production and Safety Issues Stem from Chemistry
Uses for sodium dithionite keep expanding. Environmental engineers employ it to treat groundwater contaminated with toxic metals. Given its strong reducing power, this comes at a risk—incorrect handling can release sulfur dioxide gas or cause fires if it contacts acids. The chemical formula shows two sodium atoms eager to balance charges; these atoms take up water quickly, so storage in dry, airtight containers is not negotiable. The lessons I’ve picked up working around chemical storage areas reinforce this. Someone ignoring the correct identity or handling protocols jeopardizes whole operations, not to mention worker safety.
How to Bridge Gaps in Knowledge
Misunderstandings begin with weak foundations. Too often, people skip learning or practicing how to read and interpret chemical formulas. In my experience, hands-on demonstrations—dissolving real Na2S2O4 in class, observing the reaction, connecting that bubbling action to what each atom brings—build a stronger base than memorizing lists. Professional settings benefit from refresher trainings and clear labeling. Label fatigue creeps up; periodic checks and refreshers bolster safety and reduce mix-ups. For fields that depend on sodium dithionite, this habit protects not just dollars, but lives and careers.
How is Sodium Dithionite disposed of safely?
Why Handling Sodium Dithionite Matters
Sodium dithionite gets used in a lot of industrial places—textile dyeing, paper bleaching, even groundwater cleanups. The trouble begins after it’s helped with the job at hand. This white, powdery chemical reacts fast, especially with water and oxygen. Left unchecked, it spreads harmful sulfur compounds. I remember standing in a textile workshop, watching workers shovel gray sludge into a barrel, not realizing the leftovers would foam up, heat, and send off nasty-smelling fumes five minutes later. That reaction caught everyone off guard, and it could have been worse if nobody reacted fast.
What’s at Stake
Breathing in sulfur dioxide or letting runoff seep toward a city drain can put health and neighborhoods at risk. Look at real numbers: the Environmental Protection Agency lists sodium dithionite as a hazardous waste if handled badly, mostly thanks to the fire risk and creation of corrosive gases. People on the floor don’t always have all the information. Disposing of leftover product is sometimes seen as a chore, easily forgotten. That’s how minor problems turn major.
Safe Practices in Action
Best practice starts with neutralization. I’ve learned that mixing sodium dithionite into water with stirring helps control the reaction as long as you keep things cool and add the solution to an oxidizer in small doses. Hydrogen peroxide or chlorine-based bleach finishes the reaction, turning leftover sulfur compounds harmless. Keeping this process slow is the real trick. Pouring too fast or failing to monitor heat leads to boiling chemicals or fumes nobody wants. Wastewaters should only be flushed after checking the pH and confirming no active dithionite remains, usually by test strips or a quick color reaction—the kind of thing any plant safety manager should demonstrate once a week for new staff.
Smaller labs and workshops can collect leftover sodium dithionite, label it, and hand it over to hazardous waste handlers. These professionals often stabilize the chemical, dilute it, and send it for further treatment. Trying to dump it in a regular bin creates workplace hazards and breaks regulations. Training everyone on the line, from newbies to supervisors, makes this second nature.
The Legal and Social Angle
Federal and local rules cover the disposal of hazardous chemicals, but loose enforcement sometimes breeds shortcuts. People need to see proper disposal as a core part of job safety, not paperwork. It’s easy to forget a rule if nobody checks or if managers don’t emphasize why it matters. If a warehouse, school, or business can’t handle the job properly, they ought to bring in outside help. Failing to do so risks lawsuits, fines, and, more importantly, the trust of people living nearby.
Improving Disposal Culture
At the end of the day, the safest outcomes come from speaking up and pushing for transparency. Fact sheets, training videos, and routine drills on chemical emergencies all help. Building a culture that respects the risks of chemicals pays off, especially when folks see the real-world impact beyond the lab or shop floor. Clean water, breathable air, and healthy workplaces depend on attention to these everyday details. Responsible sodium dithionite disposal means more than just following rules—it’s about doing right by coworkers and the community.
| Names | |
| Preferred IUPAC name | sodium oxidooxidosulfate(1−) |
| Other names |
Sodium hydrosulfite Sodium sulfoxylate Dithionous acid, disodium salt |
| Pronunciation | /ˈsəʊdiəm daɪˈθaɪənaɪt/ |
| Identifiers | |
| CAS Number | 7775-14-6 |
| 3D model (JSmol) | `JSML:model="sodium dithionite";data="SMILES:O=S(=O)[S-].[Na+]"` |
| Beilstein Reference | 1203672 |
| ChEBI | CHEBI:59119 |
| ChEMBL | CHEMBL1350 |
| ChemSpider | 53214 |
| DrugBank | DB14162 |
| ECHA InfoCard | 100.028.353 |
| EC Number | 015-167-00-3 |
| Gmelin Reference | 66244 |
| KEGG | C00993 |
| MeSH | Dithionites |
| PubChem CID | 24502 |
| RTECS number | WE5250000 |
| UNII | UXU6V29GYN |
| UN number | UN1384 |
| CompTox Dashboard (EPA) | DTXSID0023728 |
| Properties | |
| Chemical formula | Na2S2O4 |
| Molar mass | 174.11 g/mol |
| Appearance | White to grayish-white crystalline powder |
| Odor | Sulfurous |
| Density | 2.38 g/cm³ |
| Solubility in water | 79 g/100 mL (20 °C) |
| log P | -4.0 |
| Vapor pressure | < 0.1 hPa (20 °C) |
| Acidity (pKa) | 7.0 (for H2S2O4, dithionous acid) |
| Basicity (pKb) | 11.1 |
| Magnetic susceptibility (χ) | -60.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.334 (20 °C, 50 g/L, H2O) |
| Dipole moment | 4.6 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 125.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -587.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -587 kJ/mol |
| Pharmacology | |
| ATC code | V03AB52 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07, GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H319, H332 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P304+P340, P370+P378, P402+P404, P501 |
| NFPA 704 (fire diamond) | 2-0-1-W |
| Autoignition temperature | > 250°C |
| Lethal dose or concentration | LD50 oral rat 2,500 mg/kg |
| LD50 (median dose) | 2,500 mg/kg (rat, oral) |
| NIOSH | WX9800000 |
| PEL (Permissible) | PEL: 5 mg/m³ |
| REL (Recommended) | 120 mg/m³ |
| IDLH (Immediate danger) | IDHL: 1500 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium sulfite Sodium sulfate Sodium thiosulfate Potassium dithionite Zinc dithionite |

