Sodium Carbonate: The Story Behind the White Powder Shaping Modern Life
Tracing the Arc of Sodium Carbonate in Human History
Sodium carbonate, better known to many folks as soda ash or washing soda, has woven itself into daily routines and global industries all the way back to ancient times. People living along the banks of the Nile noticed crusty deposits left behind from evaporating lakes and realized washing with those mineral-rich ashes worked wonders on clothes and household tools. Egyptian artisans mixed this simple white powder into the famous glassware and faience beads we still marvel at centuries later. Production hit its stride in Europe only after inventors like Nicolas Leblanc and Ernest Solvay worked out reliable ways to mass-produce soda ash in the eighteenth and nineteenth centuries. Back then, pollution and caustic byproducts kicked up plenty of concern in industrializing communities. The shift from burning plants and seaweeds to chemical manufacture lowered costs and opened the gates for soda ash to move from the hands of ancient glassmakers to factories shaping everything from soap to textiles.
Physical Characteristics and What They Mean for Everyday Use
At first glance, sodium carbonate doesn’t seem spectacular—a fine, white, odorless powder, sometimes crunchy or crystalline if moisture gets in. But this stuff packs serious punch where it counts. Drop it in water and you get a basic, alkaline solution, which feels slippery between your fingers. This odd soap-like feel is the clue that soda ash has started breaking down fats and oils, just the thing needed for laundry and cleaning. Crystals tend to tug in moisture over time, so tight containers help keep the powder loose and easy to handle. One of the oddities about sodium carbonate is its willingness to dissolve, giving you close to 30 grams per 100 mL at room temp, which is plenty strong for most jobs. It won’t burn up in a regular house fire either—it takes about 850°C before you’ll start to see this compound melt. That kind of stability is no small thing.
Inside the Chemistry and Label on Sodium Carbonate Products
On the back of a carton or drum, you might spot “Na2CO3” as the chemical name. Other times, the packaging calls it “soda ash” or “washing soda”—they’re all talking about the same substance, just with different grades or purity for the tasks at hand. High-purity stuff heads to the glass and chemical plants, sometimes stamped as “dense soda ash,” while light grades offer easier handling for cleaners. On paper, a manufacturer will often share sodium carbonate’s formula, purity percentage, trace impurity levels, moisture content, and perhaps pH in water solution. Plenty of places rely on international standards like ASTM or ISO to keep batches reliable and safe, but even in a well-regulated shop, somebody should make sure labeling gives you pure product without mystery additions.
The Path to Soda Ash: Making Sodium Carbonate
Not so long ago, factories relied on the Leblanc process, which cooked up sodium carbonate using salt, limestone, and sulfuric acid. This method worked, but pollution and noxious leftovers plagued European towns and poisoned waterways. The Solvay process soon knocked Leblanc’s out as the new champion—mixing salt with ammonia and carbon dioxide, then bubbling the result through brine tanks until a slurry of sodium bicarbonate formed, ready to bake into sodium carbonate. This approach slashed waste, squeezed out more yield, and gave industry a clean way to spike production. Natural soda deposits, like trona beds in Wyoming, let modern miners skip the chemistry and pull up nearly-pure sodium carbonate, ready to process. Both approaches still play a role, but customers want both low cost and fewer environmental hang-ups, so the push continues to refine extraction and cut energy use.
The Chemistry That Powers Industries
Sodium carbonate stands as the go-to ingredient for a raft of chemical reactions that drive tens of billions in global trade. When you add acid, you get fizz and carbon dioxide that’s essential for leavening baked goods, cleaning up pH in water, or bubbling into soft drinks. Glassmakers rely on sodium carbonate to lower the melting point of their sand, making the difference between lumpy batch and smooth pane. Mix it with silicates, and all sorts of modern windows, screens, and bottles start to take shape. Soap manufacturers find sodium carbonate breaks up grease and softens water, helping detergents do their job and saving folks from scrubbing for hours. Even labs reach for it to buffer solutions or tinker with chemical balances. None of these reactions would take place quite as efficiently without the ready power of soda ash.
Known By Many Names: Synonyms Reflecting Its Many Roles
Depending on the job and the country, folks might call sodium carbonate by another name. Soda ash dominates in glassmaking and big chemical plants. Washing soda turns up in laundries and home cleaning aisles. Sometimes, chemists reach for “disodium carbonate” on their spice racks of reagents. Even “soda crystals” and “sal soda” make an appearance for crafts and old-school homemakers. Rarely does another basic chemical get so many labels, but the flexibility of sodium carbonate means every trade claims a bit of the name for itself.
Safety, Handling, and What Caution Looks Like
Despite its humble reputation, sodium carbonate needs respect. Dry powder floating in the air scratches the eyes and dries out skin for folks not careful with gloves and goggles. Wet solutions burn a bit stronger, especially in higher concentrations, so rinsing spills and keeping soda ash away from acids and organics matters at work and at home. Regulations from the Occupational Safety and Health Administration and their European equivalents set up exposure limits for airborne dust and keep production floors on alert for safe handling. Every plant and shipping warehouse I’ve seen puts sodium carbonate high and dry, away from food and incompatible chemicals, with material safety data sheets filled out and easily found for all workers. There’s wisdom in giving even common chemicals the full attention they deserve.
Laundry Rooms to Glass Plants: Where Sodium Carbonate Shows Up
Walk through a supermarket, and sodium carbonate pops up in powder detergents for laundry and dishes. Look at the industrial supply catalog, and soda ash heads off to glass production, treating city water, scrubbing flue gases at power plants, and even processing pulp for paper or aluminum. Pool owners swear by it for adjusting pH, and artists work it into the process of dyeing fabric or crafting ceramics. Soda ash landed a starring role in the mid-century boom of synthetic indigo dye, helping clothe the world in denim blues. The stuff slips quietly into foods as a dough conditioner, though food-grade purity and careful regulation keep it away from unintentional overuse. All across these applications, sodium carbonate’s twin strengths—ripping into grease and keeping pH in the safe range—prove themselves again and again.
The Ongoing Push for Better Soda Ash: Research and Fine-Tuning
Every big industrial process has its critics, and sodium carbonate production is no different. Researchers now track emissions and waste even closer, mapping pollution from mining or chemical reactors and weighing whether emerging alternatives, like electrochemical synthesis, can match or beat the old standards. Teams at national labs and universities focus on slashing energy bills by lowering reaction temperatures or recycling process heat. Folks look for greener ammonia sources to cut down on the carbon footprint tied to the Solvay process. Some trial projects explore using captured carbon dioxide from factories, offering an air-cleaning twist to a classic production method. Dealing with brine leftovers and waste salts drives new engineering, pushing the soda ash supply chain closer toward a sustainable cycle.
Diving Into Toxicity and Human Health
For a chemical that cleans so much, sodium carbonate comes across as fairly mild on the toxicity front. In reasonable doses, it doesn’t build up in the body or linger in the food chain, though accidents involving high concentrations prompt quick doctor visits for eye or skin burns. Swallowing a mouthful isn’t wise and does prompt a call to poison control, but moderate environmental releases rarely trigger big alarms. Chronic exposure to dust in a poorly-ventilated plant leads to more serious issues, and regulators keep a close eye on workplace air monitoring. The biggest challenge lies not in acute poisoning, but in balancing widespread use with common-sense hygiene and clean site management to keep both workers and the public out of trouble.
What’s Next for Sodium Carbonate?
As global industry steps up to face tighter emission rules and greater environmental expectations, the future of sodium carbonate will depend on how manufacturers clean up old processes and embrace innovation. Recycling, better energy use, and closed-loop systems offer hope for curbing pollution and shrinking the impact of this essential compound. New glass formulations, battery tech, and experimental water treatment methods could spark fresh waves of demand, all while keeping costs within reach. As someone who’s seen the waves of change hit every corner of chemical and manufacturing plants, I see in sodium carbonate both a reminder of industrial history and a challenge to rethink what a humble white powder can accomplish when steered with care and vision.
What is sodium carbonate used for?
A Household Helper With Industrial Muscle
Sodium carbonate, better known in the home as washing soda, proves its value in all sorts of places. For generations, people have tossed it into laundry loads to tackle stubborn stains and soften hard water. I grew up watching my grandmother scoop the same powder that filled the bottom of our pool skimmer into her mop bucket. She always claimed floors stayed cleaner and colors brighter, especially in our town where the tap water threatened to crust everything with limescale.
Cleaning Power That Goes Beyond the Sink
I noticed early that cleaning products line store shelves with “soda” on the label. Sodium carbonate sits at the core of many of them, including laundry detergents, automatic dishwasher tablets, and even some scouring powders. Its high pH dissolves grease and neutralizes acids, loosening everyday gunk. In my own kitchen, a little goes a long way to lift baked-on leftovers from pans and clear cloudiness off glassware.
Essential for Water Softening and Pool Care
Unlike baking soda, sodium carbonate carries a stronger punch in shifting pH levels. Pool owners lean on it to keep their swimming water balanced. In municipal water systems, it helps prevent pipes from clogging up with mineral deposits. Its role stretches to places most people never see, like the boilers in factories or steam engines, where it helps fight the corrosion that can grind machinery to a halt. Without simple substances like this one, industrial equipment wouldn’t last half as long.
Supporting Glass and Paper Industries
Glass manufacturing relies on sodium carbonate to lower the melting point of silica. This trick saves a lot of energy—one reason windows, bottles, and mirrors don’t cost a fortune. Paper mills depend on it too, breaking down wood fibers and helping bleach the pulp. These aren’t just recipes for factory workers. They shape everything from the cereal box on your table to the windshield in your car.
Food Uses and Health Considerations
Food-grade sodium carbonate stirs into certain Asian noodles or pretzels to create that classic taste and chewy texture. Bakers and cooks need to handle it with care, since it’s a strong alkaline. Eating large amounts isn’t safe, but tiny doses punch up recipes and clean produce. Regulations help keep these uses in check, and food manufacturers list it clearly so people know exactly what lands in their meals.
Environmental Impact and Responsible Use
As cleaning agents go, sodium carbonate earns a reputation for being less toxic than a lot of alternatives. It breaks down in water, avoiding the lingering harm caused by heavy-duty solvents or phosphates. The catch lies in how it’s sourced and disposed of—mining trona ore and releasing waste can pollute air and water. Recycling and greener production methods are catching on in big industry, which points to a future where every load of laundry or batch of glass comes with a lighter footprint.
Smart Solutions Moving Forward
Simple fixes unlock a lot of good. Swapping in sodium carbonate at home means fewer harsh chemicals down the drain. Communities that invest in local recycling for products and packaging linked with soda ash help close the loop. I’ve watched neighbors band together to clean parks and river banks after noticing white powdery residue from runoff—proof that small changes and shared knowledge add up. Industry can pitch in by tightening environmental controls and offering customers more choices for safe, eco-friendly products. That way, the benefits of sodium carbonate can stick around for years without leaving a mess behind.
Is sodium carbonate safe for cleaning?
What Is Sodium Carbonate?
Sodium carbonate, known as washing soda, shows up in a lot of cleaning conversations. It looks like a white powder, something you’d spot next to boxes of baking soda at the grocery store or in an old-school laundry room. Most people recognize it for its ability to tackle greasy messes and tough stains, particularly on laundry and hard surfaces.
Why Do People Use It?
Many look for safer, less complicated ways to clean kitchens, bathrooms, and laundry. Sodium carbonate stands out because it lifts grime without much scrubbing. Hard water makes soap less effective, and this is where sodium carbonate earns its keep by softening water and making detergent work better. I’ve seen it cut through greasy pans and lift dirt out of tiles that detergent alone couldn’t touch.
Safety: Should You Worry?
Sodium carbonate is not toxic like bleach or ammonia, and it doesn’t release strong fumes. Still, it isn’t mild either. It’s an alkaline powder, so it feels slippery on wet fingers, sometimes irritating skin after heavy contact. If it gets in your eye, it stings and needs a good rinse with cool water.
Most research, including guidance from the Centers for Disease Control and Prevention, boils it down to common sense: don’t eat it, don’t get it in your eyes, and don’t leave it on your skin for long. One glove is all it takes to avoid most problems. Anyone with asthma or sensitive breathing might notice a tickle in the nose if they handle a lot of powder, especially in tight spaces, so go easy and keep the windows open if you’re pouring heaps into a washing machine or soaking a floor.
Is It Green?
Much fuss about green cleaning skips the fact that even natural stuff can irritate or pollute. Sodium carbonate comes from the earth—either mined as trona ore or produced with salt and limestone. It doesn’t linger in the air like synthetic sprays or leave behind microplastics. Most wastewater plants break it down, and it won’t build up in rivers. Still, pouring buckets into your garden is not a great idea, because high doses could hurt plants.
Smarter Cleaning, Smaller Risks
Experience shows that a little goes a long way. I add a scoop to laundry for muddy work clothes or to unclog clogged drains, never using more than the label suggests. For scrubbing, dissolving a couple spoons in a gallon of hot water takes care of most chores. Every product label says keep out of reach of children, and sodium carbonate follows that rule—no different from dishwasher powder or even table salt.
Better Practices
Always store cleaning powders away from food, label containers, and rinse surfaces with clean water after use. People with eczema or very sensitive skin have better luck wearing gloves. For parents or pet owners, keeping cleaning agents where little hands or paws can't wander is a must. If you try to make cleaning solutions at home, measure carefully and don’t mix with other cleaners. Mixing it with acids like vinegar can mean extra fizz and mess, which isn’t dangerous but just wastes time.
What Works Best?
Use sodium carbonate on tough surfaces like tiles, sinks, or heavily soiled clothes. Skip it for natural wood, delicate fabrics, or anything with paint that scratches easily. Households trusting old remedies often see great results for pennies on the dollar—but only when used right. Common sense goes further than any warning label.
What is the difference between sodium carbonate and baking soda?
Clearing Up the Confusion
Standing in a grocery aisle, folks might think washing soda and baking soda are basically twins. Both show up as white powders, both come from similar chemical families, and both end up in all sorts of household projects. Look closer, though, and the differences begin to matter a lot, especially in the kitchen or laundry room.
Understanding the Basics
Sodium carbonate gets labeled as washing soda. Its chemical tag is Na2CO3. On the other hand, baking soda goes by sodium bicarbonate or NaHCO3. The difference sits right in their chemistry. Sodium carbonate has a stronger alkaline kick. You touch it with wet hands—skin feels slick, maybe even irritated. That strong pH means it breaks down greasy stains and softens water better than its distant cousin, making it a favorite in cleaning, detergent boosters, and even glass-making.
Baking soda brings a gentler approach. That’s the powder people reach for to calm an acid stomach, freshen the fridge, or help cakes rise perfectly. Its pH fits safely with foods. Any baker can tell you one wrong scoop of washing soda instead of baking soda will ruin a batch of cookies in seconds and make them taste bitter or soapy.
Uses Around Home and Beyond
Baking soda finds its way into everything from toothpaste to bath bombs. Science backs it up: safe for cleaning a child's high chair or mixing into homemade deodorant because its mildness works for personal use. The powder’s a superstar when you want to tackle funky odors, since it actually neutralizes acids behind sour smells. Medical studies suggest rinsing with a little baking soda mixed in water can help people tackle mouth sores and heartburn, though it shouldn’t replace prescribed medication.
Sodium carbonate handles tougher jobs. Drop it in a laundry load fighting grass stains and it changes the water chemistry—those minerals in hard water get tamed, and soap can get down to business. It’s also used in pool maintenance to keep water in balance. In glass factories, sodium carbonate helps sand melt at lower temperatures, so production runs smoother and costs drop.
Safe Handling Counts
Home users who treat every white powder the same get burned, sometimes literally. Baking soda feels gentle enough to brush teeth or mix into batters. Sodium carbonate, with its punchy alkalinity, irritates skin and eyes if not handled with gloves. Swallowing it would be a trip to the emergency room, not a solution for indigestion. Poison control centers often warn users that mixing up the two can mean more than a ruined recipe; it can send someone seeking emergency care.
How to Choose the Right One
Read the label closely before tossing anything into your recipe or wash cycle. Baking soda sits with baking ingredients for a reason—it keeps things safe and edible. Sodium carbonate belongs with the cleaning agents and laundry boosters. Food manufacturers and nutrition experts keep pushing the message: each one fits a different job. It pays to buy the right box for what you actually want to accomplish at home.
Finding Smarter Solutions
The number of accidental poisonings involving household chemicals highlights an ongoing concern. Educators, product designers, and policymakers have work to do, making sure packaging and public awareness keep these two powders from being swapped by mistake. Clearer warning labels and more distinct packaging color schemes offer a practical starting point—and could prevent both ruined dinners and avoidable trips to the hospital.
How should sodium carbonate be stored?
Why Storage Isn’t Just About Shelf Space
I’ve worked in a few labs, and one lesson sticks—how you store chemicals changes everything. Sodium carbonate, or washing soda, turns up all over the place, from glassmaking to fixing pool pH. Tossing it on any shelf and calling it safe just leads to headaches down the line. Someone once left an open container near the sink at my first job. Next thing you know, it turned gritty and clumped, grabbing moisture from the air. The cleanup wasn’t pretty.
Moisture: Sodium Carbonate’s Silent Enemy
Sodium carbonate pulls in moisture like a sponge. Sitting out in the open, it goes from powder to a hard lump so fast. If it cakes up, measuring out accurate amounts becomes tough, whether in an industrial batch or a simple high school experiment. In wet form, it can corrode containers. Keeping it dry isn’t just some fancy precaution—it’s the only way to keep it useful. Lining up facts, sodium carbonate will absorb about 15% of its weight in water from humid air over a week.
Sealed Containers Win Every Time
Practical experience always points to simple solutions. Use containers that seal tight, preferably glass or heavy plastic with solid lids. Over years in storage rooms, metal tins never did well. Sodium carbonate can react with certain metals, especially aluminum, and cause pitting. Commercial suppliers pack it in thick plastic bags and then box it—there’s a good reason for that. Transparent containers let you see clumping before it becomes a mess, so you catch problems early.
Storing Away from Acids and Water
One mistake that crops up all the time is stacking sodium carbonate near acids or cleaning supplies. If vapors get in, or a bottle tips, you have a chemical reaction that produces carbon dioxide—and if you’re unlucky, a foaming spill. Separation keeps things simple. Dry shelves above ground are best, far from sinks and chemical baths. In a pinch, sturdy plastic bins tucked away from damp walls make a good backup.
Label Clearly—Don’t Guess
A faded label risks disaster. I’ve grabbed unmarked containers only to discover they held something completely different. That’s how one ends up pouring sodium carbonate into the wrong solution, wrecking an afternoon’s work. Clear, bold labelling with date and name lowers error and keeps everyone safe, including those with less experience.
Potential Solutions for Widespread Issues
Problems with improper storage happen because people don’t know what sodium carbonate does over time. More training on chemical basics in workplaces and schools could stop a lot of mistakes. For places with heavy humidity—like coastal warehouses or school labs—low-cost silica gel packs inside containers can trap moisture. Regularly checking stock and discarding any that shows signs of clumping or contamination does more to save money than pretending shelf life is endless. Keeping storage spaces cool, dry, and ventilated makes a bigger difference than any expensive storage gadget. Sometimes, common sense wins over fancy systems every time.
Putting Safety on the Front Shelf
From the smallest classroom to the largest factory, good sodium carbonate storage stops waste, prevents accidents, and keeps processes moving. Rushed or sloppy practices create problems that some planning solves easily. In my own work, a sealed tub with a label saves more time and stress than any cleanup ever could. The point: treat storage as seriously as handling, and you won’t be left with a brick of ruined powder when you reach for it.
Is sodium carbonate harmful to humans or pets?
Looking at Everyday Exposure
Sodium carbonate shows up in surprising places at home. I spot it under the sink in laundry detergents and in some dishwasher powders. It’s also tucked into swimming pool chemicals and sometimes used as a food additive. Because it’s not a stranger in daily life, the natural question comes up: does it pose a threat to me or the dog napping under the table?
Direct Contact Isn’t Harmless
My hands felt dry and itchy after I once tried to unclog a drain with a powder cleaner that listed sodium carbonate. Skin contact, especially if you’re cleaning often, can lead to dryness and irritation. For people with allergies or skin conditions, these effects hit faster. Pets lick their paws or fur after walking through laundry spills—dogs and cats absorb compounds faster from these behaviors. Even though sodium carbonate doesn’t cause instant, deep burns, pets or kids rubbing it in their eyes or swallowing some by accident can lead to real discomfort.
Eating or Drinking It Isn’t Safe
While sodium carbonate isn’t the poison that something like sodium cyanide is, consuming enough brings health concerns. Even a small amount snuck into a curious pet’s mouth triggers gagging, drooling, and vomiting. Higher doses can lead to mouth and throat burns, stomach pain, or—in rare cases—dangerous changes in body chemistry. In humans, swallowing it by mistake often means nausea, stomach upset, or worse if a lot goes down.
Inhalation Risks
Powdered sodium carbonate blows easily into the air. One time I opened a bag a bit too vigorously, and the dust caught my throat. I spent the next hour coughing. For people or animals with asthma, breathing powdered chemical dust triggers wheezing and shortness of breath. With daily use, even low-level exposure increases the chances of irritation of the nose and lungs.
Environmental Effects in the Home
I worry about where that mop bucket water ends up. Sodium carbonate changes the pH of surface water, which isn’t a big deal in my city with strong sewer treatment, but is more of an issue near natural streams or private septic systems. Small pets sometimes drink from puddles in garages or near laundry rooms, sipping in chemical residues without owners noticing.
Safer Use and Simple Precautions
Wearing gloves during cleaning makes a big difference for my skin. I keep powders and liquids in locked cabinets, well above four-legged reach. After using anything with sodium carbonate, I mop up spills right away. I learned to keep detergent containers tightly closed. A quick rinse fixes most accidental skin contact. If the dog ever licks up a spill or if a kid eats some, a call to a vet or poison control is always smart. Most households use sodium carbonate safely every week by following simple habits—store it right, clean up messes, and keep curious creatures busy elsewhere.
Learning from Real Incidents and Research
The CDC and poison control centers track reports tied to household chemical exposures. The numbers stay low compared to dangerous household products, but real cases do involve skin rashes, stomach upset, or eye injuries. Studies point out that sodium carbonate ranks lower on the hazard ladder compared to bleach or ammonia. But vigilance matters. As someone who’s wiped up more than one soapy splash and scolded the cat for popping open detergent pods, I keep respecting household chemicals—even the everyday ones.
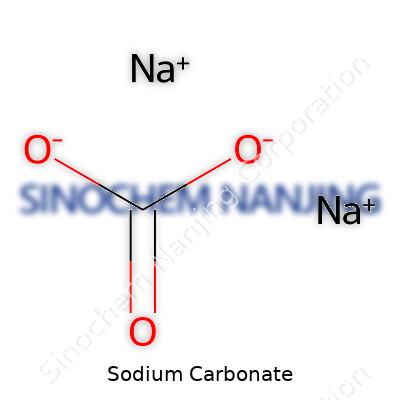
| Names | |
| Preferred IUPAC name | Sodium carbonate |
| Other names |
Soda ash Washing soda Sal soda Soda crystals |
| Pronunciation | /ˈsəʊ.di.əm ˈkɑː.bə.neɪt/ |
| Identifiers | |
| CAS Number | 497-19-8 |
| 3D model (JSmol) | `data:model/vnd.jmol;content=Na2O3C` |
| Beilstein Reference | 3531273 |
| ChEBI | CHEBI:29377 |
| ChEMBL | CHEMBL1201180 |
| ChemSpider | 72848 |
| DrugBank | DB06738 |
| ECHA InfoCard | 100.011.261 |
| EC Number | 207-838-8 |
| Gmelin Reference | ''Na-C-1'' |
| KEGG | C07244 |
| MeSH | D012592 |
| PubChem CID | 10340 |
| RTECS number | VZ4050000 |
| UNII | 45SY345LBS |
| UN number | UN3077 |
| Properties | |
| Chemical formula | Na2CO3 |
| Molar mass | 105.99 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.532 g/cm³ |
| Solubility in water | 33.2 g/100 mL (20 °C) |
| log P | -6.19 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 10.33 |
| Basicity (pKb) | 3.67 |
| Magnetic susceptibility (χ) | +1100.0e-6 cm³/mol |
| Refractive index (nD) | 1.378 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 135.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –1130.7 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1131 kJ/mol |
| Pharmacology | |
| ATC code | A06AD Sodium carbonate |
| Hazards | |
| Main hazards | Causes serious eye irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | Causes serious eye irritation. |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-0-2-N |
| Lethal dose or concentration | LD50 Oral Rat: 4090 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 4090 mg/kg |
| NIOSH | SN8225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Sodium Carbonate: "15 mg/m³ (total dust), 5 mg/m³ (respirable fraction) |
| REL (Recommended) | 20 mg/kg bw |
| Related compounds | |
| Related compounds |
Potassium carbonate Calcium carbonate Sodium bicarbonate Sodium hydroxide Sodium sulfate |

