Sodium Benzotriazole: More Than Just a Rust Protector
Looking Back: The History Behind Sodium Benzotriazole
Before anyone cared about sodium benzotriazole, engineers just fought corrosion with brute force. As industry took off in the 20th century—metal pipes running everything from city water to oil refineries—the need to slow down rust became critical. Chemists started noticing that triazole compounds stopped copper from turning green and corroding away, and sodium benzotriazole grew out of this search for a decent, real-world answer. Its practical benefits caught people's attention, building on work by chemists in the 1950s and 60s. They didn't want pipes to fall apart or electrical contacts to fail, so they looked for something easy to use and reliable. Sodium benzotriazole fit the bill. It began to show up in everything from cooling water systems to industrial detergents. The push came not from a sudden discovery, but from decades of trial and error out of simple necessity: industrial assets are expensive, and corrosion eats up equipment, money, and time.
What Sodium Benzotriazole Brings to the Table
Sodium benzotriazole usually appears as a yellowish to off-white powder or granule. The stuff dissolves pretty well in water, which matters when you mix it into cooling towers or open recirculating systems. Unlike some of the harsh chemicals people first threw at rust, this compound leaves copper and its alloys looking like they should, without pitting or unsightly tarnish. That slippery, faintly bitter smell that sits on the powder hints at the heterocyclic nitrogen backbone that gives it real chemical muscle. Its usefulness depends a lot on this stable structure, which resists breaking down in neutral and slightly alkaline conditions—just the way most municipal and industrial water systems are set up. The physical form—from fine powder to larger granules—makes handling easier, but if you get it on your hands, you’ll notice that chalky texture and the persistent, mild irritation if you forget gloves. Over the past decade, I’ve watched maintenance teams trust sodium benzotriazole because it lives up to the promise: metals don’t corrode as fast, and maintenance budgets get some breathing room.
Understanding Its Chemistry and Technical Specs
A lot of chemistry revolves around understanding the bonds and stability of the product. Sodium benzotriazole consists of a benzene ring fused to a triazole, balanced with sodium to improve water solubility. The compound generally lands near 58-60 percent active content for industrial-grade material. The ability to maintain that balance between solubility and reactivity means it holds up during storage and transport, which prevents nasty surprises at the worksite. Workers recognize bottles labeled with its various synonyms—BTA-Na, sodium BTA, or sodium 1H-benzotriazolate—but they understand it all refers to the same core structure. Most labeling standards require hazard pictograms, but the real risk comes from dust inhalation or prolonged skin contact, not acute toxicity. On the technical front, purity often reaches above 98 percent, but trace impurities don’t usually mess with its performance unless you get careless with batch handling. It survives most basic and neutral process waters, but tends to lose effectiveness in acidic environments, where breakdown speeds up and protection wanes.
How Industry Produces and Modifies This Compound
The most common preparation takes 1H-benzotriazole, itself made from o-phenylenediamine and sodium nitrite, then neutralizes it with sodium hydroxide. That simple one-pot approach keeps production scalable—factories don’t want fancy steps that slow output or pile up waste. Over the years, tweaks in purification have reduced color bodies and improved flow, so users see fewer surprises in application. Chemical modifications mostly go after improving the interaction with specific metals or compatibility with other corrosion inhibitors, like tolyltriazole. Occasionally, researchers graft polar groups onto the ring or complex it with transition metal ions to target newer applications in electronics or aerospace. Still, the core process hasn’t shifted much: reliable synthesis, straightforward byproducts, and a product that rinses out well during cleaning cycles.
Field Use: From Heavy Industry to Household Cleaners
Factories running vast heat exchangers depend on sodium benzotriazole to slow down copper corrosion, since every day spent de-scaling or swapping out corroded parts means lost output and big bills. Municipal water departments lean on the compound for the same reason—old cities run copper and brass pipes everywhere, and nobody wants drinking water that tastes metallic or turns green at the faucet. What surprised me most came from talking to cleaning product developers. Dishwashing detergents sneak in small amounts of sodium benzotriazole to stop cutlery and cookware from picking up spots. It also finds a place in anti-freeze for engines, electronic contact cleaners, and even in artist-grade metal conservation. Over the years, its role has shifted from just a miracle anti-corrosive to a quiet workhorse hiding in plain sight across products that need metal to last the distance.
Regulation, Safety, and Day-to-Day Handling
On a busy factory floor or in a municipal pump house, the most important thing about sodium benzotriazole is handling it with care. The material, while not highly toxic on casual contact, irritates eyes and skin, especially if fine dust floats around in confined areas. Workers following proper procedure—goggles, gloves, and dust masks—go home without issue, but stories circulate about careless use leading to red, itchy hands or mild headaches. Regulatory agencies keep an eye on its use, especially in water treatment where trace chemicals can show up downstream, but its long track record and low environmental persistence in controlled doses quiet most concerns. The bigger challenge arrives if large quantities spill—cleanup means containment, not just rinsing away toxins. International shipping standards require clear hazard labeling and checked inventory, reflecting bitter experiences where lack of oversight led to confusion or damage. Training crews about these routines keeps accidents low, but real-world experience says the biggest risk comes from skipping steps or underestimating powdered chemicals' ability to get everywhere.
What's Happening in Lab Research and Health Studies
Labs around the world keep sodium benzotriazole under the microscope. Researchers work on tweaking the structure to squeeze out better performance, higher selectivity, or lower costs. One persistent thread focuses on the breakdown products under sunlight and biological action, asking: does this compound add to environmental burdens in sensitive waterways? Most studies show that sodium benzotriazole breaks down slower than some alternatives, raising questions among environmental scientists. Toxicity research doesn't set off huge alarms, though. Rodent studies and cell tests mostly point to mild irritation or metabolic stress at high doses, which is rare in real-world use given the dilution in water systems. Regulators ask for monitoring in drinking water, but measured values usually stay well under limits set by public health groups. University teams aim for smart modifications—maybe adding biodegradable side chains or combining it with other inhibitors for cleaner breakdown. The work never really ends because industrial needs shift and everyone wants to reduce both cost and environmental footprint.
What the Future Holds
Shifting regulations and growing climate concerns will change how sodium benzotriazole shows up in products. Companies keep hunting for green alternatives, but most substitutes fail to match its low cost and high reliability for copper and brass. My experience tracking new product launches in the sustainability sector suggests most breakthroughs land as “blends” where sodium benzotriazole works with organic acids or natural polymers, carving out new niches in high-performance lubricants and electronics. The real pressure likely comes from water treatment: governments everywhere scrutinize even trace chemicals, spurring interest in variants that break down faster but deliver lasting metal protection. The next big steps probably revolve around integrating digital dosing systems, smarter sensors tracking corrosion in real time, and ongoing studies to show the compound’s life-cycle impact is manageable. Chemistry isn’t static, and as demands for clean water, longer-lasting infrastructure, and safer workplaces keep rising, sodium benzotriazole will either adapt or get replaced piece by piece. For now, its mix of chemical cleverness and practical reliability keeps it on the shelf not just because it works, but because after decades of trying, the world hasn't found anything better for the job.
What is Sodium Benzotriazole used for?
Keeping Metal Safe from Corrosion
Sodium benzotriazole protects metal, plain and simple. Anyone who’s worked with machines, pipes, or cars knows what rust and green corrosion can do. This compound acts as a shield, keeping that expensive copper or brass from turning ugly or breaking down. Most plumbing systems, especially ones moving water nonstop, face a constant battle against corrosion. Sodium benzotriazole steps in as a guard, sticking to the metal and making it tougher for water and oxygen to trigger that damaging process.
Making Antifreeze and Coolants Last Longer
Engine coolants and industrial antifreeze would struggle without solid corrosion control. Sodium benzotriazole improves the lifespan of these fluids. It latches onto copper and other parts inside engines, cooling towers, and heavy machinery, building a layer that keeps the metal from wearing down. I’ve seen what coolant sludge can do in a radiator, so using a proven inhibitor pays off—reducing breakdowns and saving on replacement parts.
Essential in Chemical Cleaning Solutions
Industrial cleaning often demands tough chemicals that can easily damage the very surfaces meant to stay clean. Adding sodium benzotriazole makes a big difference: it allows cleaners to do their job on deposits, but it keeps the metal intact. Factories that run large-scale water treatment turn to these formulations, protecting their investments and keeping systems running efficiently. I’ve heard from maintenance teams how skipping solutions with proper inhibitors often wrecks pipes or pumps down the line.
Important for Electronics and Manufacturing
Printed circuit boards show up everywhere, from everyday gadgets to medical devices. Sodium benzotriazole helps keep contact points free from corrosion as these parts pass through cleaning and soldering steps. It creates a barrier that halts oxidation, giving electronics a longer, more reliable life. Manufacturers aiming for top quality and fewer returns trust these additives.
Helping Preserve Art and Cultural Heritage
Museum conservators have a surprising use for sodium benzotriazole. When old coins or bronze sculptures start to show that distinctive green or blue corrosion, specialists use this chemical to stabilize and slow the process. I know preservation experts value every tool that helps precious items survive another generation.
Looking at Safety and Responsibility
Like any chemical, care matters. Sodium benzotriazole finds its way into waterways if factories don’t manage their wastewater, and research has shown potential harm to aquatic life. Responsible companies now invest in water treatment systems that remove or neutralize these chemicals before discharge. Finding safer alternatives is part of the ongoing effort, but so far, few substitutes match its effectiveness. Proper handling, reuse where possible, and modern filtration methods help keep its benefits without causing unnecessary harm.
What Could Change?
Awareness makes all the difference. The more maintenance teams and manufacturers learn about the risks, the better choices they make. Regulatory agencies and watchdog groups push for regulations and research into greener alternatives. This pressure nudges industry forward, spurring innovation. Until replacements match the qualities of sodium benzotriazole, it remains a critical tool—one best used with respect for safety and the environment.
Is Sodium Benzotriazole safe to handle?
Getting Hands-On with Sodium Benzotriazole
Sodium Benzotriazole shows up in all sorts of industries. Maintenance workers find it in cooling systems, shipping companies rely on it to protect metals from corrosion during long voyages, and even folks in automotive garages get a whiff of it when treating metal parts. So, a natural question pops up: How safe is it to handle this stuff, really?
The Chemistry Underneath
Sodium Benzotriazole stands out for its corrosion-inhibiting power. Add it to water systems or corrosion-sensitive environments, and it keeps rust at bay, especially on copper and its alloys. That does not make it completely harmless. Reading the safety data sheets, a few red flags come up—irritation, toxicity if enough ends up in the body, and the usual risk of careless exposure piling up over a career.
Straight Talk on Exposure
There’s no sugarcoating the facts here. If powder or solution lands on your skin, you might notice reddening or dryness. Breathe in the dust, and the respiratory system may react—sneezing, coughing, and irritation. Splash some into your eyes, and they sting. Anyone who has worked with chemical additives in tight spaces knows how quickly things escalate without decent ventilation and basic protective wear.
Documented Risks and Questions That Matter
Research has not tied Sodium Benzotriazole to serious, long-term health conditions like cancer, though the data on long-term human exposure still leaves a few gaps. There’s good animal data showing that if swallowed in large doses, problems pop up in the liver and blood. The main concern for most workplaces sits with short-term irritant effects. Some studies hint that Sodium Benzotriazole can disrupt aquatic ecosystems, especially if factory waste meets river water without proper treatment. Friends in wastewater management bring up these concerns every year during audits.
Practical Ways to Stay Safe
Treating this chemical with respect pays off. Most safety professionals push for gloves, safety glasses, and decent masks in any space where dust or spray could linger. Installing proper extraction fans in tight workshops helps. Simple habits make a difference—washing hands before lunch breaks, keeping work jackets in the shop instead of bringing them home, and storing chemicals in labelled, sealed containers.
Training counts just as much as gear. Seeing a buddy suffer a skin rash because someone left a solution spill on a worktable makes the importance hit home. Quick access to safety showers and eye-wash stations goes beyond just following codes—it genuinely keeps injuries low. Factories and labs that keep their spaces clean and organized avoid a lot of problems.
Keeping Accountability in Mind
Regulators like OSHA and the European Chemicals Agency keep Sodium Benzotriazole on their radar. There’s a push for better workplace labeling and tracking, plus more frequent audits by health and safety inspectors. Workers have the right to know the real risks—and speak up if practices fall short. Employers hold the responsibility to provide up-to-date training, usable PPE, and open reporting culture.
Everyone handles risk differently, but practical, real-world experience says one thing loud and clear—safety slips happen when people get too comfortable, or when a job site skimps on safety gear and airflow. Sticking to clear safety routines, and keeping up with changing science and workplace laws, keeps people safe on the job.
What industries commonly use Sodium Benzotriazole?
Protecting Metals: The Invisible Shield
Factories and tradespeople have long looked for answers to one stubborn problem: metal corrosion. Take a walk through any plant that processes steel, copper, or their alloys, and you’ll find maintenance schedules jammed with tasks meant to slow down rust and tarnish. Sodium benzotriazole steps in like an invisible shield. It reacts at the surface of metal, building a layer that blocks out air and moisture—the main triggers for corrosion. The metal fabrication world leans on this compound to stretch the lifespan of pipes, pumps, sheets, and heavy equipment.
Power Plants and Water Treatment: Keeping It Running Smoothly
Running a steam turbine or cooling tower doesn’t leave much room for error. A small flaw in a heat exchanger pipe can lead to shutdowns, leaks, and expensive downtime. Sodium benzotriazole helps power plants and municipal water facilities shield their copper and brass components. Water by itself isn’t always the enemy; trace minerals and chemicals swirl around, corroding the surfaces they touch. Since sodium benzotriazole forms a bond at a molecular level, it gives engineers enough confidence to keep systems running with less frequent overhauls. Data from industry reports show that using this chemical reduces unscheduled repairs and maintenance costs, helping clean water and energy keep flowing.
Automotive And Aerospace: More Than Just A Shiny Finish
If you look under the hood of a car or peek inside an airplane fuselage, you will spot copper wiring, brass connectors, and intricate metal parts. Sodium benzotriazole shows up in the manufacturing process, coating these metals to hold back corrosion. Over years of road salt, humidity, and engine heat, these parts stay reliable. In my early days working with a car restoration shop, we noticed that parts pre-treated with corrosion inhibitors like benzotriazole aged slower and saved customers money on replacements. The story repeats in aviation, where safety rules demand lasting integrity for every critical part.
Electronics: Quietly Guarding Circuit Boards
While consumers race after smaller, more powerful devices, makers of electronics worry about the chaotic world inside every gadget. Copper traces on circuit boards need protection from moisture and residue, which can short out connections. Sodium benzotriazole acts as a low-key defender here, lending reliability to laptops, phones, and medical devices we depend on each day. This chemical doesn’t call attention to itself, but its role supports the global electronics sector worth trillions.
Challenges And Smart Solutions for the Future
Sodium benzotriazole brings big benefits, but questions keep popping up about safety and runoff. Wastewater from metal processing and car washes can carry traces into rivers and groundwater. Research from environmental science journals highlights the persistence of this compound in nature. Industry leaders now look for new filtration tech, tighter waste handling rules, and even safer cousins of benzotriazole to swap in. Sustainable chemistry and regulatory oversight can push this story in a healthier direction.
Looking Ahead
Sodium benzotriazole isn’t a magic cure, but it does a job that few other chemicals can match. Whether keeping the lights on, planes in the sky, or laptops humming, this compound holds industries together where rust and failure once crept in. The real challenge sits in using it wisely, balancing performance with the health of communities and the environment.
How should Sodium Benzotriazole be stored?
The Real World Context of Handling Chemicals at Work
Keeping chemicals like sodium benzotriazole in a shop, plant, or lab challenges everyone involved. Having spent time in facilities where corrosion inhibitors and other specialty chemicals come through the door almost weekly, I’ve seen more chemical mishaps than anyone wants to admit. It’s one thing to read a product safety sheet and something else to watch bags break open because someone stacked them wrong or didn’t check the temperature before unloading a pallet.
Why Moisture and Sunlight Matter
Sodium benzotriazole helps stop rust and corrosion in water systems, engines, and heat exchangers. It comes as a yellowish powder or granules. Tossing a sack into a broom closet or leaving a drum outside sets up all the ingredients for trouble. This stuff clumps up if it gets damp and breaks down under strong light. Once that happens, people end up tossing out expensive product or dealing with less protection against rust in their machines—or both.
A dry, well-ventilated place, away from sunlight, keeps sodium benzotriazole in usable shape. You don’t need fancy custom cabinets, but an unused shelf in the breakroom doesn’t cut it either. The aim is to find a spot indoors where humidity stays low and the direct sun doesn’t cook supplies. We had a corner in our storehouse lined with wooden pallets, always labeled, with no roof leaks overhead.
Heat: The Hidden Threat
Heat changes things fast. One summer, the thermometer inside our maintenance shed hit ninety-five. The crew noticed the corrosion inhibitor in open drums turned hard around the edges. Some production lines missed their dosing targets for days. Turns out, high heat not only dries the product but can make it react with even minute traces of water or air.
Storing chemicals like this below eighty degrees always made a difference for us—especially when long-term storage ran into months rather than weeks. If air conditioning isn’t possible, windows can be covered and fans used for air movement. No storage next to boilers or machinery that heats up.
Separate and Label—For More Than Just Regulations
Mixing up containers or storing sodium benzotriazole right next to strong acids, food ingredients, or flammable supplies can send you scrambling in a hurry when leaks or spills happen. I’ve seen the fallout: chemical burns, ruined inventory, and compliance officers with clipboards. Shelving with clear labels, dividers, and locked doors saves time, money, and maybe a trip to the ER.
Personal Responsibility and Simple Habits
Everyone gets busy or distracted, some more often than others. In my experience, posting a plain checklist by the storage area helps. Even better—walk-through checks at the start of every shift. Cracked lids or broken bags get handled before anyone gets hurt. Regular staff refresher on the handling, protective gear like gloves and goggles, and simple cleanup supplies within arm's reach close the loop.
Using sodium benzotriazole safely means more than following a rulebook. It’s about building daily respect for the materials—proven, tough, and surprisingly vulnerable stuff that can save or ruin expensive equipment. Everyone who touches a storage shelf or supply bin can shape how safe—or risky—the story ends.
What are the chemical properties of Sodium Benzotriazole?
A Closer Look at Its Composition
Sodium benzotriazole stands out for more than one reason, but its defining trait lies in how it combines the reactivity of sodium salts with the unique protective role of benzotriazole rings. It's a white to off-white crystalline powder, water-soluble, and packs a distinctive, mildly aromatic odor. Beyond what you see or smell, the real action comes down to its molecular structure—a sodium ion paired with a benzotriazole anion, C6H4N3Na. This structure might seem simple on paper, but it creates a versatile corrosion inhibitor that manufacturers and engineers count on every day.
Stability and Reactivity: Daily Importance
Sodium benzotriazole doesn't break apart easily. It keeps its cool under normal temperatures and pressures. It doesn't burn or explode, so workers aren’t losing sleep over fire hazards in the lab or on the production floor. Left on a shelf, it can last for quite some time without decomposing. Chemically, the benzotriazole ring itself proves tough—it doesn’t react with oxygen or water at room temperature. Drop it in water, and it dissolves smoothly, which makes it convenient for practical mixing into solutions.
Corrosion Resistance Secrets
Corrosion eats up metal faster than most folks realize. Water, salt, and oxygen work over time to turn valuable metal into rusty scrap. Sodium benzotriazole offers something vital: it forms an invisible film on metal surfaces—especially copper and its alloys—blocking out corrosive elements. This protective shield stands up under a wide range of pH levels, so it's useful in harsh industrial setups or everyday plumbing.
I remember working on a water circulation system where copper pipes spent years in tough chemical environments. Ordinary inhibitors fizzled out, but adding sodium benzotriazole kept those pipes clean. No fancy equipment needed, and maintenance costs dropped. This personal experience mirrors what many industrial reports and peer-reviewed studies show—sodium benzotriazole goes beyond standard protectants, making it one of the top choices for metal longevity.
Interaction With Other Chemicals
Sodium benzotriazole doesn’t shy away from complex environments. It stands up to acids and alkalis—rare for many organic compounds. I found that out myself during a job dealing with cooling towers. The system’s chemicals fluctuated often, but sodium benzotriazole held steady, keeping its protective power. It does have its limits: strong oxidizing agents can break it down faster. So, it pays to keep strong bleach or peroxide out of the solution if corrosion resistance remains a priority.
Environmental and Safety Aspects
Safe handling stays crucial, since sodium benzotriazole’s breakdown products can impact aquatic life if those chemicals get into natural waterways. Responsible use and proper disposal protect workers and the environment. Water treatment facilities in many countries use closely monitored doses, and disposal goes through approved channels. The chemical doesn't bioaccumulate, but a little forethought in management ensures its benefits outweigh possible risks.
Looking Toward Better Practices
Engineering teams and manufacturers have plenty of room to further lower environmental risks. Closed recycling systems capture water used for metal rinsing, and safer substitutions continue to emerge for projects where that’s possible. Still, the strong track record of sodium benzotriazole means it isn’t fading from industry use anytime soon.
Understanding what makes sodium benzotriazole tick helps professionals make smart choices in designing processes and protecting valuable equipment. Practical chemistry always rewards those willing to dig deeper than the label.
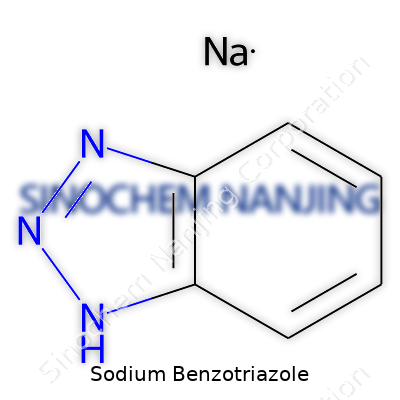
| Names | |
| Preferred IUPAC name | sodium 1H-1,2,3-benzotriazol-1-ide |
| Other names |
1,2,3-Benzotriazole sodium salt Benzotriazole sodium Sodium 1,2,3-benzotriazolate Sodium benzotriazolide Sodium BTA |
| Pronunciation | /ˌsəʊdiəm ˌbɛnzoʊˈtraɪ.əˌzoʊl/ |
| Identifiers | |
| CAS Number | 15217-42-2 |
| Beilstein Reference | 3588504 |
| ChEBI | CHEBI:91205 |
| ChEMBL | CHEMBL135251 |
| ChemSpider | 185693 |
| DrugBank | DB16664 |
| ECHA InfoCard | 100.028.722 |
| EC Number | EC 246-599-9 |
| Gmelin Reference | 62238 |
| KEGG | C14391 |
| MeSH | D014601 |
| PubChem CID | 12406 |
| RTECS number | DM1225000 |
| UNII | T4T28ZC49G |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | EPA CompTox Dashboard ID for Sodium Benzotriazole: **DTXSID3027277** |
| Properties | |
| Chemical formula | C6H4N3Na |
| Molar mass | 181.17 g/mol |
| Appearance | Light yellow to off-white powder |
| Odor | Odorless |
| Density | 1.36 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -1.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | pKa ≈ 8.2 |
| Basicity (pKb) | 11.8 |
| Magnetic susceptibility (χ) | -86.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.658 |
| Viscosity | 15 - 40 mPa.s |
| Dipole moment | 2.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 155.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -220.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -270.8 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause an allergic skin reaction. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P330, P501 |
| Flash point | >100°C (212°F) |
| Autoignition temperature | > 204°C (399°F) |
| Lethal dose or concentration | LD50 oral rat 720 mg/kg |
| LD50 (median dose) | LD50 (median dose): 825 mg/kg (rat, oral) |
| NIOSH | LW5425000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50 mg/kg |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
Benzotriazole 1H-Benzotriazole Methylbenzotriazole Copper benzotriazole Benzotriazole sodium salt Tolyltriazole Sodium tolyltriazole |

