Sec-Butylamine: A Closer Look at an Understated Industrial Workhorse
Historical Development
Sec-butylamine doesn’t turn heads like some fancier molecules, but it’s been quietly fueling industrial chemistry for decades. Its story began in the early 1900s as chemical processing matured and demand for versatile amines increased. As industries searched for more efficient ways to build molecules—especially those needed in rubber, pharmaceuticals, and agrochemicals—sec-butylamine showed up as a handy building block. Researchers and plant managers experimented with countless routes to make sure production could scale, costs could drop, and purity standards would keep getting tighter. The amine’s journey isn’t flashy, but its impact stretches across lab benches and factory floors worldwide.
Product Overview
Sec-butylamine steps into the spotlight every day across the chemical industry. Its unique, branched-but-sturdy molecular structure makes it more than just another small amine. Rather than serving as a niche specialty, it provides a foundation for synthesis, especially where primary amines with secondary carbon centers are needed. The stubby, four-carbon skeleton makes this molecule less volatile than some lighter amines, while its moderate reactivity offers chemists plenty of control when building more complex molecules. Over the years, producers have refined it to deliver a colorless, straightforward liquid that handles predictably even in large-scale batch operations.
Physical & Chemical Properties
In handling sec-butylamine, I find its physical properties more user-friendly than certain more hazardous amines. It comes as a clear to slightly yellow liquid, carrying a strong—often somewhat unpleasant—amine odor. Boiling just north of 63°C, it doesn’t flash off as quickly as the lighter methylamines, yet it remains very flammable. With a molecular weight of 73.14 g/mol, storage and transfer equipment built for amines can usually manage sec-butylamine without special design. In water, it dissolves reasonably well, and its basicity often plays a central role, reacting vigorously with acids to form salts, but showing less tendency to cling to glassware and surfaces than sticky, higher-molecular-weight amines.
Technical Specifications & Labeling
Producers mark sec-butylamine with labels that signal hazard: flammability, potential toxicity, and the need for careful handling. The typical industrial-grade material offers purity above 99 percent, as even small impurities can impact the performance in pharmaceuticals or pesticide intermediates. Many operations check for water content and non-volatile residues before accepting a batch, since handling inconsistencies or container leaks can quickly affect downstream yields or product stability.
Preparation Method
One of the better-known ways to make sec-butylamine involves the reductive amination of butanone using ammonia and hydrogen gas with a nickel or platinum catalyst. This process turns a common ketone and simple ammonia into sec-butylamine with surprisingly high selectivity—assuming a good catalyst and well-tuned conditions. Labs sometimes use alkyl halides and ammonia under pressure, but fouling from byproducts often makes that path less favored at scale. Over decades, refiners have optimized temperature, pressure, and flow rates to feed the world’s appetite with the highest efficiency possible.
Chemical Reactions & Modifications
In my own lab and process work, sec-butylamine’s versatility makes it a true workhorse. Its primary amine group steps into acylation, alkylation, and condensation reactions without too much fuss, making a fit partner for building all sorts of intermediates. Its secondary carbon offers just enough steric hindrance to make selectivity tweaks possible, especially when compared to isopropylamine or n-butylamine. Through salt formation or N-derivatization, you can tailor this amine to diverse downstream uses—from herbicides to chiral auxiliaries in pharmaceuticals.
Synonyms & Product Names
In the chemical world, more names are usually a sign of a product’s reach. Sec-butylamine goes by names like 2-butylamine and (sec-butyl)amine. Depending on the supplier or regional naming conventions, you may also see sec-butylamine listed as SBA or with its IUPAC designation. These alternative labels reflect the broad adoption across continents and production traditions.
Safety & Operational Standards
No matter how much experience you have in a chemical plant, sec-butylamine demands respect. Its vapor can irritate the eyes and respiratory tract, and skin contact leaves unmistakable burns. Flammability sits center stage in risk assessments. Always keep sources of ignition far away and make sure ventilation systems run properly. Storage in tightly closed metal drums with grounding wires eliminates many headaches, as does using minimal open handling and making spill kits standard. Best practice means wearing gloves, goggles, and flame-resistant lab coats every time you approach the amine storage area. Regulatory bodies like OSHA set strict exposure limits, while safety officers enforce rigorous training and monitoring routines.
Application Area
The practical uses of sec-butylamine stretch across the wildest corners of industrial chemistry. You’ll spot it at work producing rubber accelerators that keep tire manufacturing rolling, in the synthesis of pesticides like butralin, and as a core ingredient in making APIs for pharmaceuticals. Its ability to react cleanly and rapidly lets process chemists trim steps from synthetic routes, saving both raw materials and reactor time. I’ve seen it adopted as a preferred amine catalyst in polymerizations where cost and reproducibility matter. In water treatment, its derivatives sometimes help scramble pathogens in municipal systems. The amine’s broad reactivity gives it a foothold far beyond any one sector.
Research & Development
R&D labs continue to find clever ways to push sec-butylamine into new territory. Green chemistry initiatives have focused on reducing waste through in situ generation and recycling excess byproducts. In pharmaceutical development, medicinal chemists turn to sec-butylamine as a scaffold for novel molecules, fine-tuning activity and solubility by strategic substitution. Analytical labs keep a close eye on trace impurities using state-of-the-art chromatography, since many active pharmaceutical ingredients need exacting purity standards. The future likely holds both improved catalytic methodologies and hybrid applications in material science.
Toxicity Research
Investigating the risks that sec-butylamine brings, animal studies have flagged moderate oral and inhalation toxicity. Direct exposure to skin or eyes brings pain and real injury, while repeated inhalation sometimes triggers headaches or even central nervous system effects in people working near open containers or spills. Regulatory assessments often flag environmental concerns, as the compound can harm aquatic life in high concentrations. Researchers have combed through animal models to map long-term risks, and most protocols urge keeping exposure well below set thresholds. Proper labeling and training continue to save lives across the supply chain.
Future Prospects
As chemical manufacturing grows more environmentally conscious and globalized, sec-butylamine’s role will likely shift. Producers aim to squeeze more out of existing resources by improving catalysts and introducing waste-minimization processes, making sec-butylamine both greener and cheaper to produce. Its presence in pharma and agrochemicals looks unlikely to fade, but expanded regulatory scrutiny will push for even cleaner synthesis at every step. Industry veterans keep their eyes open for safer alternatives, yet most alternatives struggle to match the cost-effectiveness or reactivity that sec-butylamine brings to the lab bench. For all its simplicity, sec-butylamine stays essential for countless synthetic challenges, quietly powering the machinery of modern chemistry.
What is sec-butylamine used for?
A Simple Chemical With Big Uses
Sec-butylamine has a long and rather underrated role in the world of specialty chemicals. People walking by a busy chemical plant don’t realize the stuff getting poured into big mixing tanks often ends up affecting daily life. I remember a friend who worked in agriculture telling me about the endless search for better herbicides. He explained how sec-butylamine keeps showing up whenever scientists need a starting point for designing molecules that mess with weeds but not with crops. It’s like one of those unglamorous but reliable utility players on a sports team that helps put the stars in the right spot to win.
Helping Crops Grow
For the most part, sec-butylamine hides behind the scenes in agriculture. It often forms a key part of the chemical recipe for making powerful herbicides and fungicides. Without compounds like these, the job of growing healthy crops grows a lot tougher. Weeds run wild, disease takes hold, and suddenly food prices edge up. The structure of sec-butylamine allows scientists to attach it to other pieces, shaping what eventually ends up in that bottle of weedkiller in your neighbor’s garage. I’ve seen how frustrated farmers get when the weeds take over, and it’s clear that until we find perfect alternatives, these industrial ingredients keep supermarket shelves full and prices steady. According to the Food and Agriculture Organization, efficient weed management can boost crop yield by as much as 40%.
A Builder in the Lab
Chemists call sec-butylamine an “intermediate.” What this means in plain English is it acts as a building block in the lab. Need to make a medicine for high blood pressure? This chemical might be on the recipe card. Need to whip up artificial flavorings that make snacks taste like grilled steak? It could find its way in there, too. Pharmaceuticals often demand precision, purity, and reliability, and sec-butylamine checks those boxes for thousands of researchers. The American Chemical Society lists it as crucial for several pharmaceutical syntheses, especially for drugs where a well-placed amine group tips the scales from inactive to effective.
Beyond the Obvious
Plenty of folks don’t realize sec-butylamine helps out in the production of rubber. The tires under your car, the soles on running shoes, hoses, seals, and various bits of industrial gear often use additives to protect them from falling apart too soon. Sec-butylamine finds work here too. It can show up in what chemists call accelerators—the chemicals that help rubber harden at just the right speed and toughness. Without those, tires wear out sooner and more of them end up in landfills. Anyone who’s changed a flat on the shoulder during a rainstorm appreciates the small victories of modern rubber science. The European Tyre and Rubber Manufacturers’ Association points out the value of carefully sourced additives in improving both performance and lifespan.
Risks, Responsibility, and Looking Ahead
Handling sec-butylamine at scale comes with some baggage. It can irritate the skin, it smells harsh, and it’s not something you want dumped into streams. Companies and lab workers treat it with respect. Smart policies, good training, and strict monitoring keep the risks manageable. There’s always a hunt for safer or greener alternatives, but until those become reliable, sec-butylamine keeps rolling out of factories for jobs that touch nearly everyone. People don’t talk about it much, but as chemical processes keep evolving, so does the demand for builders like this one.
What is the chemical formula of sec-butylamine?
Understanding the Structure: More Than a Formula
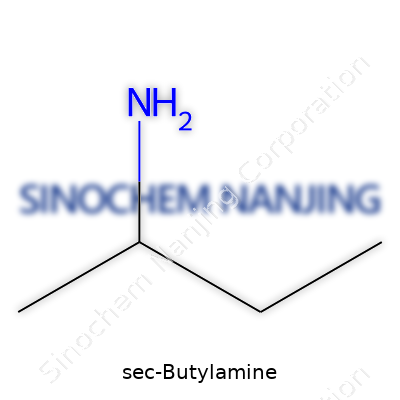
Sec-butylamine brings together carbon, hydrogen, and nitrogen. Its formula, C4H11N, points to its backbone: four carbon atoms, eleven hydrogen atoms, and a single nitrogen. In practical terms, this means you get a straightforward amine, hooked onto a four-carbon chain that bends in just the right way to stand apart from its more familiar cousin, n-butylamine. That branching may seem like a small structural tweak, but it influences important behaviors—boiling point, smell, how it interacts with other chemicals, and how people handle it in a lab or an industrial setting.
Amines: Unassuming but Unforgettable
Most folks outside of chemistry might never notice a bottle of sec-butylamine. Inside chemical plants and college labs, things look different. This amine appears as a colorless liquid with a pungent, fish-like odor. That sharp scent comes from the nitrogen atom, which can sneak into the air and catch your attention right away—anyone who has opened a bottle of amine in a chemistry lab won’t forget it.
People often ask why chemical structure matters so much. Having spent hours measuring out stinky liquids and running reactions, I learned quickly that a minor tweak to a molecule’s shape can change its use. Sec-butylamine, being a secondary butyl group attached to an amine, provides just enough difference to carve out a niche. It dissolves well in water, yet cuts through non-polar solvents too. That flexibility gives it an edge as an intermediate for making things like pharmaceuticals, pesticides, and even rubber chemicals.
Why Sec-Butylamine’s Formula Matters in Real Life
Sec-butylamine’s formula doesn’t just sit on paper. With C4H11N, chemists know exactly what they’re working with and how it’ll react. In pharma, that might mean tweaking painkillers or anti-cancer agents. In agriculture, you might be talking about herbicides or fungicides that keep food supplies healthy. Its formula guides companies making dyes, resins, and coatings.
Years ago, in a drug development setting, small changes to an amine chain determined whether an experiment led to success or a dead end. The formula spelled out instructions for what could be stable, what might react too quickly, or what might stay safe to handle. Regulations strongly consider the effects even small molecules like sec-butylamine can have on human health and the environment, so understanding its formula stays central to safety. Reliable production, safe storage, and effective transportation all rely on people knowing those details.
From Formula to Safer Work and Better Solutions
Efforts to handle chemicals like sec-butylamine keep evolving. Safety data sheets, smarter storage solutions, and new reaction techniques play a part. I remember updating a storage protocol after seeing how quickly fumes could escape a loose cap. There’s always a push for better ventilation, tighter labeling, and education of young chemists so nobody takes shortcuts. Routine monitoring and strong workplace training mean spills get cleaned up fast and exposures drop. Those steps add up to a safer environment and more reliable chemical production.
Paying attention to formulas like C4H11N stands for more than memorizing letters and numbers. It’s about equipping everyone from bench chemists to regulatory officials with facts that shape decisions about health, productivity, and progress in every field that depends on chemical science.
Is sec-butylamine hazardous or toxic?
Understanding What Sec-Butylamine Brings to the Table
Sec-butylamine often gets used in making pharmaceuticals, rubber chemicals, and pesticides. It looks like a simple colorless liquid and doesn't sound flashy, but its presence in so many products calls for a close look at its safety. The first clue is in its smell: it gives off an odor that hits the nose and lingers, which usually means strong chemical activity.
Human Health Risks from Handling Sec-Butylamine
Folks working around sec-butylamine won't forget its impact—after a whiff, irritation creeps into the nose and throat fast. Breathing in the vapor can lead to headaches, nausea, or even confusion. In one case, I remember a plant worker telling me just being near an open drum made his eyes burn and his chest feel tight. He learned real quick the value of a good respirator.
Its liquid form doesn’t spare the skin either. A splash can cause redness and sometimes blisters, almost like a mild acid burn. Even with gloves, there’s a strong incentive to stay focused because a small mistake can lead to hours of stinging discomfort. Eyes exposed to the liquid react even more harshly, sometimes leading to serious injury if not washed out immediately. The need for eye-wash stations right where sec-butylamine gets used isn't just safety theater; it’s practical protection.
Beyond the Shop Floor—Wider Impacts
Sec-butylamine doesn't threaten just the people handling it. Spills or poor storage let vapors escape, putting building occupants at risk, and run-off risks reaching groundwater. The U.S. EPA says that sec-butylamine is classified as a hazardous air pollutant. Long-term exposure matters, too. A 2021 review in the journal "Critical Reviews in Toxicology" points out that repeated low-dose inhalation may sensitize the lungs, adding to the risk of asthma-like symptoms.
Problems don’t end at the workplace door. Sec-butylamine breaks down quickly in the air but can stick around in soil and water under certain conditions. While no big community poisoning case has popped up—in part because most people never get near it outside industry—the environmental risk climbs quickly with poor waste storage or illegal dumping.
Safer Solutions Are Possible
People don't get stuck with unsafe chemicals if companies and regulators stay focused. Strong ventilation, sealed transfer systems, and meaningful personal protective equipment bring down the health risks right away. For example, lab-scale work can happen in chemical fume hoods, while factories do well to seal production lines and use leak detectors. Rushing or skipping inspections always comes back to bite—leaks rarely fix themselves.
Substitutes exist for certain pesticide and pharmaceutical setups, but replacing sec-butylamine across the board isn't practical overnight. Still, enforcing strict storage laws and making sure spills get cleaned up quickly both keep risks lower for workers and the wider environment.
Why Care? Responsibility and Trust
Worrying about sec-butylamine may seem like fussing over nothing for folks who never walk into a plant, but it all boils down to trust in the products we use and the places we live. A little planning and honesty from chemical users build trust with workers and neighbors, and nobody wants a reputation for ignoring health risks in search of profits. After seeing how often “minor” mishaps spiral out of control, I take chemical safety personally—if not for myself, then for the people who come next. That approach, driven by direct experience and the evidence in scientific reviews, always pays off in the long run.
What are the storage requirements for sec-butylamine?
Why Sec-butylamine Storage Really Matters
Storing hazardous chemicals isn’t just a box to check—it’s a real concern for anyone handling these substances daily. Sec-butylamine, widely used in pharmaceuticals and organic synthesis, brings big risks with it. If you’ve ever opened a storage cabinet and caught a whiff of something sharp, you know why attention to detail matters with chemicals like this one.
Straightforward Rules, Serious Outcomes
Sec-butylamine goes by the formula C4H11N, and it comes with a stinging, ammonia-like odor. The vapors sting the nose and eyes. For this reason, it calls for locked cabinets, decent airflow, and a clear “no casual access” policy. A university lab I worked with treated amines seriously. Every time someone left the cap loose, the chemical smell spread fast. Everybody nearby felt their eyes water—no fun, and a reminder that vapors move quickly.
Flammability is where sec-butylamine shows its teeth. Flash points run low—around -1°C (30°F)—so even a mild breeze won’t keep you safe if there’s an open flame or stray spark nearby. Metal containers with good seals and flame-proof storage rooms save trouble. Nobody wants to deal with an amine fire. It’s not just paperwork headaches. Insurance jumps, regulatory bodies call in, and people might end up in the ER.
Site Choices: Shelves Won’t Cut It
A shelf might work for books, but sec-butylamine deserves sealed ventilated cabinets. Some labs skimp on this, stacking flammable liquids alongside acids or oxidizers. I’ve seen seasoned researchers walk over, spot this mistake, and start separating bottles right away. You don’t want a spill mixing sec-butylamine with acid. That reaction gets violent and makes a mess. Good practice involves boxing similar chemicals together and logbook records. Clean labeling stops mix-ups and accidental contact.
Humidity and sunlight also play a role. Sun baking through a window will warm this chemical up and that speeds up vapor production. Humidity may corrode metal containers over time. The fix—solid, opaque storage drums, checked for rust or cracks every couple of weeks. Small problems turn big fast in warm or wet conditions.
Safety Lives in the Details
Spill containment often gets overlooked. Absorbent mats or trays under containers trap drips or leaks. Emergency eyewash and showers shouldn’t be afterthoughts. Workers in a nearby plant shared stories about a forgotten shower during an accident, costing minutes when time mattered most. Small extras like these can be a gamechanger.
Personal protective equipment counts, too. Nitrile gloves, splash goggles, and lab coats aren’t just for show. The few times people ignore these rules, skin burns or eye damage become real risks.
Solutions Aren’t Exotic—They’re Just Discipline
Training does more than a long list of safety rules. Educating staff, running drills, and sharing real-life mishaps drive the point home far better than any printed sign. Regular inventory checks spot small leaks before they grow. Disposal bins for used gloves or paper towels need frequent emptying to reduce fume build-up. Good ventilation, tight storage, and strict labeling are habits, not just guidelines.
At the end of the day, sec-butylamine storage takes attention, some sweat, and a no-shortcuts mindset. Out of sight never means out of mind when you’re keeping people safe around chemicals that bite back.
What are the physical properties of sec-butylamine?
A Closer Look at a Common Amine
Sec-butylamine isn’t a headline-grabber, yet anybody who’s worked in a chemical lab might recall the first time they opened a bottle of it. You notice a strong odor right away—it’s pungent, a sign of its volatility. Sec-butylamine comes as a clear, colorless liquid under normal temperatures, with a boiling point sitting around 63 to 64°C. It evaporates quickly, so you want decent ventilation or a snug fume hood.
Working with sec-butylamine, you get a sense of how easy it mixes into things. It dissolves well in water thanks to the small, soluble amine group, and it handles common organic solvents like acetone and ethanol just fine. In practice, chemists find this matters when they prepare solutions or need to rinse glassware. Left unsealed or poorly stored, you’ll pick up the smell outside the storage cabinet by the end of the shift, driving home the importance of proper containment. The quick evaporation is handy for processes needing a fast-drying amine, but you have to watch for loss through evaporation during weighing or transferring.
Straight to the Core: What the Numbers Mean
On the safety sheet, sec-butylamine earns a low flash point—just about -18°C. That puts it squarely in flammable territory. I learned early on that even a small spill near a heat source can mean real trouble. One colleague once dripped some on their gloves, and in minutes, the fumes were noticeable. Anyone handling it should pay attention and wear gloves that hold up against amines.
As for density, sec-butylamine clocks in at roughly 0.74 g/cm³ at room temperature, making it lighter than water. Spill it in a sink full of water, and watch the droplets float. People rarely think about this property, yet in accident scenarios or waste disposal, floating chemicals can spread out faster, creating cleanup headaches.
Sec-butylamine's refractive index, around 1.4, gives a clue toward its purity and concentration—useful for analytical folks. This trait, paired with its miscibility, helps researchers track how reactions are progressing in the flask or in industrial reactors.
The Risks and Responsible Use
Fast evaporation paired with low ignition point increases risk, especially in labs with less airflow. Safer storage uses tightly sealed containers, kept away from heat. In production facilities, even a slight increase in room temperature can boost vapor concentration. Regulations require explosion-proof equipment for transferring and storing this stuff, not just as a formality, but out of hard lessons learned from accidents.
Sec-butylamine’s amine group makes it basic (alkaline), stinging the eyes on contact and irritating skin. I once heard a story from a plant operator who learned the hard way that even small spills can burn when not cleaned quickly. Emergency showers and eye wash stations come in handy for a reason, but training keeps people prepared.
Lessons for the Lab and Factory Floor
The physical properties of sec-butylamine—its volatility, solubility, and lightness—turn into strengths or hazards depending on the situation. It gets used in making pharmaceuticals and rubber chemicals precisely for this quick reactivity and easy handling. The strong odor isn’t just a nuisance; it’s a warning sign. Pay attention to the material, store it right, and respect the risks it carries. People who handle chemicals like sec-butylamine every day build habits around these facts. Clear information and good equipment matter as much as experience for keeping things safe and productive.
| Names | |
| Preferred IUPAC name | butan-2-amine |
| Other names |
1-Methylpropylamine sec-Butan-1-amine 2-Aminobutane |
| Pronunciation | /ˌsɛkˈbjuːtɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 13952-84-6 |
| Beilstein Reference | 1740724 |
| ChEBI | CHEBI:16040 |
| ChEMBL | CHEMBL15369 |
| ChemSpider | 14347 |
| DrugBank | DB13836 |
| ECHA InfoCard | ECHA InfoCard: 100.003.712 |
| EC Number | 202-753-3 |
| Gmelin Reference | 8186 |
| KEGG | C01112 |
| MeSH | D013479 |
| PubChem CID | 6361 |
| RTECS number | EJ4200000 |
| UNII | J85U2FYS9T |
| UN number | UN2330 |
| Properties | |
| Chemical formula | C4H11N |
| Molar mass | 73.14 g/mol |
| Appearance | Colorless liquid |
| Odor | Ammonia-like |
| Density | 0.741 g/mL at 25 °C (lit.) |
| Solubility in water | miscible |
| log P | 0.62 |
| Vapor pressure | 15.4 kPa (20 °C) |
| Acidity (pKa) | 10.73 |
| Basicity (pKb) | 3.37 |
| Magnetic susceptibility (χ) | -7.97×10^-6 cm³/mol |
| Refractive index (nD) | 1.396 |
| Viscosity | 0.39 mPa·s (at 20 °C) |
| Dipole moment | 4.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 222.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -34.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3884.7 kJ/mol |
| Pharmacology | |
| ATC code | C01CA14 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H302, H311, H314, H331 |
| Precautionary statements | P210, P233, P280, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 3-3-2-A |
| Flash point | 34 °C |
| Autoignition temperature | 285 °C |
| Explosive limits | 1.2% - 13% |
| Lethal dose or concentration | LD50 oral rat 650 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 616 mg/kg |
| NIOSH | SEB |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sec-Butylamine: 5 ppm (18 mg/m³) |
| REL (Recommended) | 20-25°C |
| IDLH (Immediate danger) | 400 ppm |
| Related compounds | |
| Related compounds |
n-Butylamine Isobutylamine tert-Butylamine |

