Sec-Butyl Alcohol: A Down-to-Earth Look at an Industry Workhorse
Looking at the History of Sec-Butyl Alcohol
Sec-butyl alcohol, known to folks working in both labs and industry by its formal name 2-butanol, hasn’t always gotten the spotlight like ethanol or isopropanol. Still, its history tells us plenty about industrial progress. For much of the last century, chemical plants produced it as a side product during the manufacture of methyl ethyl ketone and butane derivatives. Early research pointed toward its use in solvents and chemical synthesis as demand grew for versatile alcohols that could go beyond cleaning and sanitizing. Factories in North America and Europe found plenty of uses for it as economies boomed after both world wars, especially as new plastics and synthetic rubbers rolled off the assembly lines. Sec-butyl alcohol’s evolution has never been flashy but the story sits at the intersection of chemistry, commodity markets, and real-world needs.
Getting a Sense of the Product
In everyday terms, sec-butyl alcohol looks like a clear, colorless liquid that packs a distinctive odor, typical of higher alcohols. Industrial suppliers ship it in steel drums, tank trucks, and railcars. Many people who’ve worked in paints, coatings, or chemical manufacturing remember the sharp, somewhat sweet smell that comes from freshly opened containers. Technicians and chemists value it for its ability to dissolve greases, oils, resins, and synthetic compounds where simpler alcohols don’t cut it. Affordable production makes it an easy choice for many midsized operations, even though its reputation doesn’t match pure ethanol when it comes to food, drugs, or beverage uses.
Physical and Chemical Properties: More Than Just a Number
A casual observer might read off the boiling point, density, and flash point from the label, but there’s a lot to unpack behind those numbers. Sec-butyl alcohol boils at about 100 degrees Celsius, which fits right between lighter alcohols and heavier ones. That boiling point means it works well in both open vats and closed systems. Its lower solubility in water compared to ethanol sets limits but also brings advantages in extractions and separations. Folks dealing with flammable liquids know to watch the flash point—the risk deserves respect, and the liquid will ignite if careless hands treat it like water. On the chemistry front, sec-butyl alcohol features a secondary carbon attached to its hydroxyl group, making it distinctly different in reactivity from its isomers like n-butanol or tert-butanol, which goes a long way in deciding how chemists use it.
In the Factory and on the Label
Labels on sec-butyl alcohol containers list purity, water content, color, and acidity. Quality inspectors use sophisticated chromatography and spectroscopic techniques to nail down composition and spot any contamination. In the best labs I’ve seen, QC steps move fast—one batch hits the market only when numbers line up and the documentation passes muster. Packaging isn’t just for show. Workers and supervisors keep their eyes on drum condition and seals, since leaks mean exposure and lost product. Most sites adopt the international GHS system, using diamond-shaped labels and signal words that instruct anyone handling the liquid exactly what they’re up against. During loading and unloading, operators rely on both training and muscle memory to avoid spills and inhalation. Looking at these real-world operations, sharp paperwork and safer labels probably save more headaches than any new rule.
How Sec-Butyl Alcohol Gets Made
Most chemical workers learn early on that sec-butyl alcohol typically comes from hydration of butenes (straight from petroleum cracking) or by catalytic hydrogenation of methyl ethyl ketone. In industrial plants, butenes react with water over acidic catalysts, and process tweaks control which isomer becomes dominant—this matters because primary, secondary, and tertiary butanols have different practical uses. The industry favors continuous processes, where raw materials and energy drive steady output. In smaller batches, labs stick to the old playbook: start with methyl ethyl ketone and reduce it with a solid catalyst under hydrogen gas. It amazes me how little the fundamentals change, even as controls, instrumentation, and sensors improve year by year. Environmental footprints, by-products, and catalyst recycling shape real-world practices these days more than ever before.
Chemical Reactions and What Chemists Actually Do With It
Chemists who reach for sec-butyl alcohol do so because its secondary structure opens up paths to oxidation, etherification, and esterification. In many organic syntheses, converting it to sec-butyl chloride or sec-butyl acetate builds building blocks for pharmaceuticals, flavors, and specialty chemicals. Oxidizing sec-butyl alcohol forms methyl ethyl ketone, an industrial solvent with a big footprint in coatings and adhesives. Reacting it under acidic conditions with strong acids makes sec-butyl ethers—these compounds sneak into gasoline blends to improve combustion and reduce emissions. Lab workers use it as a reagent, knowing it behaves differently from its primary or tertiary cousins: better selectivity in some reactions, higher yields in others, all depending on the pathway’s quirks. Its versatility keeps sec-butyl alcohol appearing in undergraduate chem labs as much as full-scale production sites.
Names, Synonyms, and What’s On the Label
This liquid has gone by plenty of names: 2-butanol, sec-butanol, SBA, and secondary butyl alcohol all show up in literature. Certain industry catalogs itemize it under the CAS number 78-92-2. Troubleshooting in the field, I’ve learned to double-check synonyms before starting any process or ordering chemicals, avoiding costly mix-ups with n-butanol or isobutanol. Many companies keep the “sec-” prefix to steer clear of confusion; regulated documents nod toward “2-butanol” pretty consistently these days. Older textbooks might reference “methyl ethyl carbinol”—a name you don’t hear as often, but it pops up in historical contexts and patent literature.
Staying Safe and Keeping Operations Smooth
Working safely with sec-butyl alcohol means respecting both toxicity and flammability. Old-timers in the industry tell stories of skin irritation and headaches from sloppy handling. Proper ventilation, gloves, and splash goggles beat cautionary lectures every time. Industrial plants post clear standard operating procedures, detailing spill control, fire prevention, and cleanup. Fire teams train for alcohol fires, keeping foam extinguishers, water spray, and personal protective equipment close by. Supervisors often walk new hires through the difference between inhalation and skin contact risks, learning from minor mishaps instead of waiting for major ones. From my own time in the lab, I know even the best operators slip up, so routine safety drills and access to safety data sheets prevent a lot of grief. Regulatory regimes, such as OSHA in the United States and REACH rules in Europe, push plants to stricter controls and regular site audits, which—as frustrating as they appear—raise everyone’s baseline safety.
Where Sec-Butyl Alcohol Lands in the Real World
Manufacturers use sec-butyl alcohol as more than just a solvent. In pharmaceuticals, it sneaks into synthesis steps, helping to build active ingredients that treat infections, pain, or metabolic disorders. Paint and coating firms mix it into formulas for better drying times and finish quality—every hard hat walking the shop floor knows the difference a good solvent makes. Petroleum refineries blend sec-butyl alcohol derivatives into fuels, aiming for lower emissions and smoother engine running. I’ve seen plants push research into surfactants, flavors, and fragrances—sec-butyl alcohol’s molecular shape works in surprising places. Water treatment, though not a classic application, occasionally benefits when specialty chemicals derived from sec-butyl alcohol boost coagulation or remove trace contaminants. It’s never just about the liquid itself—its real benefit shines in what it builds, what it dissolves, and how it keeps big machines running or keeps products shelf-stable.
Research, Ongoing Questions, and What Scientists Want to Know
Innovation moves forward in fits and starts, and sec-butyl alcohol hasn’t escaped this pattern. Academic labs and corporate research teams run pilot studies on using sec-butyl alcohol as a starting point for biodegradable plastics or eco-friendly solvents with lower toxicity and environmental impact. Some researchers target process efficiencies: How can catalysts improve selectivity and yield, while using greener processes? Alternative feedstocks—think bio-based butenes or even fermentation routes—grab headlines as the petrochemical world seeks new footing in a climate-conscious era. I know graduate students who dig deep into the subtle ways sec-butyl alcohol’s structure influences reaction mechanisms, and their work sometimes rewrites accepted wisdom from older reference books. Research dollars have chased recycling processes, energy minimization, and ways to produce less waste. A lot depends on the broader global shift toward circular economies, but the research door stays open for anyone with new questions or creative approaches.
Untangling Toxicity—What’s at Stake?
On the factory floor or in a chemistry lab, questions about toxicity come up fast. Sec-butyl alcohol hits the central nervous system, causing headaches, dizziness, and—at high enough levels—serious fainting spells. Animal studies pick up liver and kidney strain, so long-term exposure brings worries about organ damage. Regulatory agencies flag sec-butyl alcohol as a moderate health hazard. Not all solvents offer clear substitutes, which puts pressure on both management and health professionals to balance process needs with worker safety. Air monitoring, biological testing, and exposure limits form the nuts and bolts of responsible handling. Hospitals and clinics report few incidents involving sec-butyl alcohol compared to more notorious chemicals, partly thanks to industry vigilance and careful training. In discussions with industrial hygienists, I hear growing interest in lowering exposure even further, calling for engineering controls and substitution as technology develops.
Looking Ahead: Where Sec-Butyl Alcohol Could Go Next
Sec-butyl alcohol doesn’t make headlines, but that’s never meant the story is over. Global pushes for safer and greener chemicals point toward new methods for making and using sec-butyl alcohol with a smaller environmental toll. Demand in electronics, high-performance coatings, and even bioprocessing could carve new niches if research lines up. Investors bet on supply chains that depend less on fossil fuels, creating new opportunities for sec-butyl alcohol produced from sustainable feedstocks. Digital monitoring and automation in chemical plants will upgrade efficiency, cut energy use, and improve safety in ways that touch this alcohol as much as any commodity. People who work directly with sec-butyl alcohol—engineers, chemists, technicians—see the value in getting the details right, knowing that every gallon used supports bigger projects in industry, research, and manufacturing. As new markets develop and safety standards rise, sec-butyl alcohol stays relevant by adapting with the times.
What is sec-butyl alcohol used for?
Where Sec-Butyl Alcohol Shows Up
Most people walk past shelves of cleaning supplies or paint thinners without thinking much about ingredients. Sec-butyl alcohol, though, isn’t just a technical name tucked away on safety sheets. This chemical steps onto the stage in ways that touch daily life, sometimes without fanfare, sometimes with an unmistakable whiff you recognize right away.
A Working-Solvent in Industry
I’ve spent time volunteering in community art studios and one thing you notice: artists and carpenters both value solvents that break things down, evaporate fast, and don’t leave sticky residues. Sec-butyl alcohol plays this very role. Paint factories use it to dissolve dyes and resins, giving paints the right texture and finish. You find it behind the scenes in cleaning formulations. Industrial shops lean on it when stripping grease from metal before coating or welding. The U.S. National Institutes of Health points out its place in adhesives and coatings, as well as uses in the making of plastics. You don’t see it, but you sure notice how well things blend or how surfaces shine after using products made with it.
Making Perfumes and Flavors More Lively
Sec-butyl alcohol works as a building block for making flavors and fragrances. Beyond just making a room smell nice, perfumes rely on solvents like this to suspend scents and help them spread on the skin. Food chemists tap it for its ability to help manufacture artificial flavors. The U.S. Food and Drug Administration recognizes it as safe when handled properly and used within limits. Still, flavor houses and labs need to watch their handling carefully; a slip-up with quantities or ventilation can create headaches for workers and neighbors.
Fuel Additive and the Push for Cleaner Combustion
Anyone who’s worked near small engines or cars running on alternative fuels knows fuel blending is a careful science. Sec-butyl alcohol ends up in fuel additives, helping gasoline burn cleaner. The goal: reduce knocking and squeeze out a tiny bit more efficiency. The U.S. Environmental Protection Agency monitors and sets up limits for concentrations in commercial fuels. I’ve seen how local hardware store owners and auto shops lean on these improvements: less smoke, engines last longer, air in the neighborhood smells a bit fresher.
Handling Risks and Staying Smart About Exposure
People working with sec-butyl alcohol know it comes with responsibility. The CDC lists its vapor as an irritant. In poorly ventilated spaces, headaches and dizziness show up in minutes. Companies now set up improved exhaust systems and personal protective equipment. This matters, because plenty of workers—painters, machinists, lab techs—spend years breathing the same air. I know a few who’ve developed sensitivities simply from repeated skin contact. Smart practices, like gloves and face shields, make all the difference.
Looking Forward: Safe Use and Honest Labels
Sec-butyl alcohol isn’t a household name, but it leaves its mark on everything from cars to art supplies. Factories shifting toward safer alternatives and clear ingredient labeling help protect workers and customers. Smaller firms track chemical sourcing more closely, responding to demand for cleaner products and more transparency. People deserve to know what’s in the goods they buy. As tools and knowledge grow, the hope is for safe handling to be the norm, not the exception.
Is sec-butyl alcohol safe to handle?
What You’re Working With
Sec-butyl alcohol pops up in labs, factories, and some niche products. This clear liquid doesn’t get the same fame as rubbing alcohol, but it holds a place in chemical processing. Working with any alcohol in an industrial setting means thinking about more than wiping a kitchen counter.
Personal Experience With Handling Chemicals
The first time I handled an industrial alcohol, I felt confident. I’d seen alcohol products all my life, and figured how risky could it really be? Turns out, a single headache when the spill wasn’t cleaned fast enough drove the message home. Experience has taught me that familiarity with a product breeds shortcuts—until the side effects show up. Sec-butyl alcohol has a strong smell and can cause dizziness, nausea, and irritation if you’re not careful. Even PPE veterans let their guard down until that stinging eye or scratchy throat reminds them why this stuff stays locked up when not in use.
Hazards And Risks
Sec-butyl alcohol won’t attack your skin at the first touch, but prolonged contact means dryness and irritation. Splashing in the eyes quickly goes from discomfort to pain. Because the fumes can sneak into your lungs, areas without steady airflow become a problem: headaches, drowsiness, or worse. It’s flammable, so even a minor spark or open flame spells trouble. Government health agencies grade this alcohol as a hazardous material, and that means the rules around it are built from experience—not guesswork.
What The Research Says
Agencies like OSHA and NIOSH set exposure limits well below the level where symptoms crop up. The Centers for Disease Control warn about the vapors: overexposure can slow your reflexes and, in bad situations, affect breathing. Animal tests point to the fact that large doses throw the body off balance, even though most accidental exposures at work don’t reach that level. The truth is, someone careless enough to skip gloves or goggles sets themselves up for headaches today and more serious problems tomorrow.
How To Stay Safe
Lab workers know the drill: barriers, shields, gloves, goggles, and fans keep you out of trouble. It’s not just labs, though—maintenance staff and operators need the same backup. Eyewash stations and clear labels on containers matter just as much as good instructions. Emergency protocols can’t just live in the safety manual; they need teeth on the job. In jobs I’ve held, regular safety walks and peer checks made sure no one treated sec-butyl alcohol like ordinary rubbing alcohol.
Training staves off accidents more than fancy equipment. Everyone who works with chemicals should see what real exposure looks like—images, stories, case studies. Chemical risk doesn’t sound thrilling, but practical reminders drive home that a quick shortcut can mean hours in a medical bay.
Better Practices, Fewer Problems
Following rules isn’t about pleasing inspectors. I’ve known colleagues who shrugged off “one quick pour” without gloves just to hit a deadline, only to regret it later. Clean storage, tight sealing, and quick spill cleanup shave risk down. Every time a new worker joins the team, someone has to walk them through the hands-on safety steps. That personal touch sticks longer than a poster in the break room.
Sec-butyl alcohol doesn’t belong on the danger shelf next to the deadliest chemicals, but it isn’t soap and water either. Handling it with care means fewer sick days, lower stress, and more trust on the team. Simple actions—ventilation, proper gear, real training—offer better protection than any lucky streak.
What are the physical properties of sec-butyl alcohol?
What Sec-Butyl Alcohol Really Looks Like
Sec-butyl alcohol isn’t some mysterious chemical. If you’ve worked in a lab or spent time reading chemical safety data, you might have handled a clear, colorless liquid with a faint smell that blends a little sweetness with the punch you expect from alcohols. This smell isn’t just a warning; it’s a clue about how easily sec-butyl alcohol evaporates. Its boiling point lands just over 99°C, so a little heat in the workplace and the fumes are in the air. Back in college, during an experiment, I spilled a few drops and the scent filled the room right away. It doesn’t hang around on the skin, though—the rapid evaporation shows just how volatile it can be.
The Feel and Flow: Texture in the Lab
Pour sec-butyl alcohol between your fingers and you’ll notice it’s not sticky, just slick and free-flowing. With a density lower than water—just about 0.81 grams per cubic centimeter—anyone can watch it sit right on top when it’s poured in. This difference matters during chemical separations or solvents in extractions where layer formation tells you what’s going on. Its index of refraction, clocking in around 1.395, shows that light bends less through this alcohol compared to heavier liquids.
Water Matters: Mixing and Solubility
Sec-butyl alcohol plays well with water, but only in small doses. Try to dump a ton of it into water, and you’ll run into separation. The two phases are easy to see. Around 12 grams dissolve in 100 grams of water, which is serviceable for making standard chemical solutions. Toss it with ether or many of the usual lab solvents, and you’ll see it blends right in. This partial solubility in water matters for dilution in pharmaceuticals and industrial cleaning—places where balancing cleanup with safe handling stands front and center.
Flammability: A Real Concern
If you’re storing sec-butyl alcohol, don’t ignore its flash point. It lights up at a pretty modest temperature—about 35°C. I remember working at a summer internship where careless storage led to a scary near-miss because someone forgot how quickly vapors could travel toward a heat source. Its vapor pressure, measured at 17 mmHg at 20°C, pushes a steady stream of vapor into the air even at room temperature, underlining why proper ventilation really matters. The liquid will burn with a blue flame, so the danger isn’t always obvious in bright light. Fire suppression standards treat it with as much respect as gasoline.
What That Means for Daily Work
Anyone using sec-butyl alcohol in the lab or an industrial site must respect its volatility and flammability. Adequate ventilation turns into a daily requirement. Containers stay sealed, and chemical handling goes hand-in-hand with fire safety. I’ve seen too many students assume alcohols only burn and evaporate—nothing more. In reality, sec-butyl alcohol challenges you to think about temperature management, material compatibility, and the ever-present risk from an open flame or unguarded heat source. Even on a small scale, all these physical properties impact safe and effective handling, from the simple stuff like cleaning glassware to bigger jobs like fuel blending and intermediate synthesis.
How should sec-butyl alcohol be stored?
Why Storage Standards Matter
Any technician who's lifted a drum of sec-butyl alcohol will tell you this chemical brings both utility and headaches. Used in making perfumes, pharmaceuticals, and solvents, this clear, flammable liquid won't pose a hazard just sitting in a bottle—unless that bottle sits on the wrong shelf, in the wrong room, or under the wrong conditions. In my early days working in a small lab, a careless mistake with solvent storage set off a fire alarm—an incident no one in the building forgot. It's these close calls that sharpen your focus on safety.
Understanding the Risks
Sec-butyl alcohol, or 2-butanol, gives off vapors that catch fire at fairly low temperatures. Those vapors can sneak into the air if a container isn’t tight or gets left open. OSHA lists the flash point at about 24°C (75°F). A hot summer day is all it takes for dangerous vapors to build up. If someone gets a whiff of that distinct, slightly sweet smell, you know it's time for better ventilation—or a check for a leaky cap.
Some workers ignore simple safety because they think small quantities won't matter. But the unpredictable nature of chemical accidents has taught a different lesson: there’s no magic volume where care stops being important. Even a liter spilled or vaporized in a confined space can do real harm. In a university lab, a cracked jar of sec-butyl alcohol forced an evacuation. The problem wasn't the size of the container, but a lack of preparation.
Practical Storage Steps
A locked chemical cabinet rated for flammable substances should be the first line of defense. These cabinets do two things right: they keep vapors contained if a leak happens and shield contents from flames if a fire breaks out elsewhere. Cabinets away from direct sunlight or heating vents make a difference, because heat speeds up evaporation. Avoid storing near oxidizers like hydrogen peroxide or acids—unexpected reactions can ruin more than equipment.
Never use glass containers with questionable integrity; plastic or metal makes more sense only if it’s built to stand up to organic solvents. I’ve seen too many makeshift substitutes crack or warp, leaving puddles that put everyone on edge. Manufacturers print expiration dates and recommendations for a reason, and those must guide your choices. Important: keep lids tight, check seals, and write opening dates clearly. A shelf check every few weeks can prevent bad surprises.
Good Habits Make the Difference
Proper ventilation reduces risk from invisible fumes. At work, I saw that a small lab with a working fume hood had fewer headaches and near-misses than larger spaces without one. Clean up spills right away, never with a random rag—absorbent pads and plenty of fresh air protect your skin and your lungs.
Some folks skip the paperwork, but updated storage logs tell the real story. If something goes wrong, those records help trace what happened. A good log lists container sizes, purchase dates, and planned disposal times. This stops old stock from lurking in the back, waiting to become the next problem.
At the end of the day, sec-butyl alcohol gives as much trouble as it’s allowed to. Thoughtful storage doesn't demand new technology or endless rules, just steady attention to the basics—solid cabinets, fresh air, and honest tracking. That’s how trouble gets stopped before it starts.
What are the potential health effects of sec-butyl alcohol exposure?
Getting Real About What Sec-Butyl Alcohol Does Inside the Body
At first glance, sec-butyl alcohol may seem like just another chemical in a massive industrial toolkit. Most folks never give it a second thought, unless they spot it listed on a workplace safety sheet. People use it in everything from making paints to producing cleaning agents, and it keeps manufacturers busy day and night. But once you get past the technical lingo, the risks make you stop and think about what really matters—basic health and safety.
What Happens After You Breathe or Touch It
Breathing vapors from sec-butyl alcohol doesn’t just irritate the nose. You may feel dizzy, develop a headache, or even struggle to keep your balance. After years on job sites, I learned to trust what my own body tells me. If a coworker feels suddenly lightheaded after opening a drum marked “BUTANOL,” it’s rarely a coincidence. Some folks feel nauseous. Others end up coughing for hours. High concentrations can cloud thinking and slow reflexes. Just last month, a report showed temporary central nervous system symptoms in workers exposed to high levels in a poorly ventilated distillation area.
Touching sec-butyl alcohol isn’t harmless, either. Everyone’s skin handles chemicals a little differently, but one thing stays the same: red, itchy patches don’t leave you guessing long. Most symptoms show up quickly, including burning sensations on the skin. Open wounds or raw spots only make things worse, giving chemicals a fast track straight into the bloodstream.
Long-Term Worries Are Tough to Ignore
People want answers about long-term risks. Some animal studies link repeated exposure to kidney and liver changes. These studies can’t always tell the full story for humans, but they do raise red flags worth respecting. Agencies like the National Institute for Occupational Safety and Health (NIOSH) suggest keeping levels well below those that trigger symptoms in animals. That recommendation came after researchers saw changes at just 100 parts per million.
The immune system also plays a role here. Sometimes, the very chemicals workers encounter day after day give rise to skin allergies or sensitivities. Years ago, I saw a plant worker who developed blisters and swelling after each shift; he told me each day felt worse until he changed jobs. Workers with a history of asthma should be extra careful, since vapors have triggered attacks in some people.
Cutting Exposure Is the Key
Few people outside certain industries need to worry about sec-butyl alcohol every day, but those who do deserve real, workable solutions. The fastest fix comes from using proper exhaust fans and air filtration. I’ve seen job sites where switching from a single fan to a double setup cut complaints in half. Protective gloves and goggles provide solid backup for those handling pure material. After gloves, washing up thoroughly at breaks made a big difference for teams I worked alongside in the coatings trade.
Training also goes a long way. If workers understand warning signs—dizziness, burning eyes, or skin irritation—they speak up early and avoid severe symptoms down the road. Posting clear signage with emergency steps gives peace of mind. Medical checkups every few months help spot problems before they become permanent.
The Bottom Line
Sec-butyl alcohol works as a powerful tool in industry. Its health risks stay manageable only if people respect its power and use practical methods to offset those risks. No one should learn about its dangers the hard way. It only takes a few smart steps—ventilation, protective gear, good hygiene, solid training—to protect workers and preserve health.
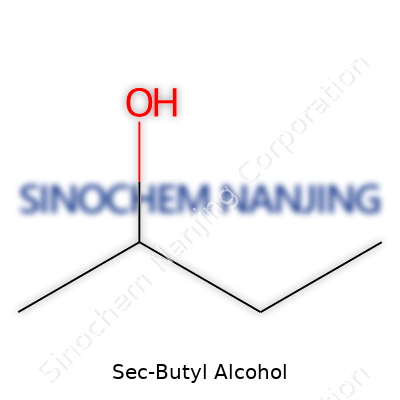
| Names | |
| Preferred IUPAC name | butan-2-ol |
| Other names |
2-Butanol sec-Butanol secondary Butyl alcohol Butan-2-ol SBA Methylethyl carbinol |
| Pronunciation | /ˌsɛkˈbjuːtɪl ˈælkəˌhɒl/ |
| Identifiers | |
| CAS Number | 78-92-2 |
| Beilstein Reference | Beilstein Reference: 1718730 |
| ChEBI | CHEBI:15602 |
| ChEMBL | CHEMBL15360 |
| ChemSpider | 6834 |
| DrugBank | DB02185 |
| ECHA InfoCard | ECHA InfoCard: 100.003.504 |
| EC Number | 200-751-6 |
| Gmelin Reference | Gmelin Reference: 83695 |
| KEGG | C02315 |
| MeSH | D013634 |
| PubChem CID | 6567 |
| RTECS number | EL5425000 |
| UNII | YQE403N1WZ |
| UN number | UN1120 |
| Properties | |
| Chemical formula | C4H10O |
| Molar mass | 74.12 g/mol |
| Appearance | Colorless liquid with a characteristic odor |
| Odor | Alcohol-like odor |
| Density | 0.81 g/cm³ |
| Solubility in water | miscible |
| log P | 0.61 |
| Vapor pressure | 10 mmHg (20°C) |
| Acidity (pKa) | 15.2 |
| Basicity (pKb) | The pKb of sec-butyl alcohol is 7.7 |
| Magnetic susceptibility (χ) | -55.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.399-1.403 |
| Viscosity | 2.6 mPa·s at 20°C |
| Dipole moment | 2.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 260.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -318.07 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2717 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H332, H319, H335 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | flash point: 22°C |
| Autoignition temperature | 401 °C |
| Explosive limits | 1.7% - 9.8% |
| Lethal dose or concentration | LD50 oral rat 2190 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat 2190 mg/kg |
| NIOSH | RT8750000 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 1400 ppm |

