Examining Sebacic Acid: A Longstanding Chemical With Modern Influence
Tracing the Roots of Sebacic Acid
Sebacic acid goes way back in the history of industrial chemistry. Before industries boomed across Europe and Asia, people extracted sebacic acid mostly from castor oil. The name itself comes from the Latin word for tallow, reflecting its greasy origin. Scientists in the 19th century recognized the substance’s versatility, especially once they realized they could produce it from plant sources rather than relying on less sustainable animal fats. Over the decades, searing demand for novel polymers and plasticizers brought this compound out of the shadows and into busy factory floors, especially once nylon production exploded in the 20th century. Sebacic acid, once a laboratory curiosity, found permanent roles beyond industrial paints, unfamiliar lubricants, and obscure plastic goods. It has become part of the conversation for anyone who cares about how raw materials shape the world.
The Nature and Behavior of Sebacic Acid
In practical terms, sebacic acid takes the shape of a white, powdery solid. Its chemical formula is C10H18O4. It doesn’t dissolve in water the way salt or sugar does, but strong polar solvents like alcohol break it down just fine. From experience, you can tell by its faint, waxy smell that it comes from natural fats. This dicarboxylic acid gives you a clue by its structure: two acid groups at opposite ends of a ten-carbon chain. That stretch of carbons—the “aliphatic” chain—accounts for why it bends and flexes easily in polymer chains. This flexibility, turned into a clear benefit, enables manufacturers to build plastics that don’t crack in cold temperatures, making it indispensable for synthetic rubbers, tough coatings, and even some biodegradable plastics. Melting starts around 132 degrees Celsius, which places it comfortably into most industrial uses where high heat isn’t an issue.
Technical Standards and On-the-Ground Realities
Chemical quality often becomes a deal-breaker. Sebacic acid’s purity is more than just a number; it influences downstream products, especially in cosmetics and medical devices. Analyzing its technical specs—like acid value, ash content, and color—reminds me that small mistakes ripple far. Cheap grades with higher moisture or yellowing degrade product appearance and stability, a lesson I learned years ago working around polymers exposed to low-grade additives. Don’t overlook labeling rules, either. The rules keep tightening; regulators in Europe and Asia set strict guidelines for batch testing, safety data sheets, and impurity profiles, forcing producers to adapt. Skipping these steps just to get the product out the door invites a headache nobody needs—especially as more customers demand clarity and traceability with every shipment.
Crafting Sebacic Acid: From Oil to Acid
The bulk of global sebacic acid starts its life in castor beans. Processing castor oil to sebacic acid might sound straightforward, but the chemistry reveals otherwise. The route runs through saponification, hydrolysis, and then cracking the ricinoleic acid apart to get those carboxyl groups in the right place. Factories in India and China have pretty much perfected the trick, squeezing out decent yields while handling caustic soda, heat, and strong acids. Lab-scale routes play with other vegetable oils, though costs and scalability hold back most alternatives. There’s a push for green chemistry approaches—less waste, milder reactions—with academic groups experimenting on catalyst systems to cut pollution from waste streams. Nobody has landed a breakthrough to unseat the castor oil workhorse, but economic incentives and tighter emission controls keep the race going.
Chemical Tinkering and Derivatives That Matter
Sebacic acid sits at a crossroads for chemical modification. Mix it with simple alcohols under heat, you produce sebacates—esters that show up in plasticizers and lubricants with better environmental profiles than many phthalate competitors. Toss it into a polycondensation reaction, you get nylon 6,10. That grade of nylon resists oils and wears down less than its shorter-chain siblings. Scientists keep fiddling with its backbone, grafting on side chains or coupling with polyols to chase better flexibility, weather resistance, or even antimicrobial properties for niche markets. From firsthand experience working with research chemists, the challenge usually involves keeping costs down and process steps efficient, since every reaction step multiplies the expense and potential regulatory scrutiny.
Sebacic Acid’s Aliases and Naming Conventions
Ask for sebacic acid in different countries and you get a parade of names: decanedioic acid, 1,8-octanedicarboxylic acid, or sometimes just its salt form, sebacate. Industry veterans still use old trade names that pop up in legacy literature, though regulatory harmonization means product labels often carry the IUPAC tag. Tracking synonyms becomes a chore, especially for import/export, leading distributors to double-check documentation—nobody wants customs held up over a naming slip-up. I’ve seen old shipments delayed because outdated synonyms crept into the paperwork; it underscores the value of staying organized and current with global databases like ChemIDplus or the ECHA registry.
Keeping People Safe and Processes Smooth
Nobody in the factory wants a surprise, and with sebacic acid, standard good practice rules keep everyone safe. Dust can irritate lungs and eyes, so basic PPE—gloves, goggles, and dust masks—stay mandatory during handling. Spills don’t explode or corrode like strong acids, but cleaning up quickly means less risk of slips and dust clouds. Storage away from water and strong oxidizers keeps it stable, which saves facility managers headaches down the line. Industry standards, like those from OSHA or REACH, expect manufacturers to train staff and keep audits up to date. Twenty years in specialty chemicals taught me that real-world discipline beats paperwork—spend the time teaching people on the floor, and accidents don’t need to become memorable stories.
Sebacic Acid’s Roles and Everyday Impact
This compound backs up more industries than many realize. Adhesives, plasticizers, and coatings tap into its flexible backbone—they stick, bend, and stretch better because of it. Nylon made with sebacic acid blends shows up in toothbrush fibers, bristles, and even medical sutures, where surgeons trust the material’s toughness and inertness. It doesn’t stop at polymers; engine oils depend on sebacates for better flow at low temperatures. Some cosmetic creams and ointments use it for texture or stability, banking on its plant-based credentials amid a wave of “clean beauty” trends. Animal feed additives, corrosion inhibitors, and biodegradable plastics round out the impact zones. Every time demand surges—whether for electric vehicles, solar panels, or sustainable packaging—the ripple spreads back to sebacic acid producers, sparking upgrades in their plants and supply chains.
Where Research and Breakthroughs Are Heading
Sebacic acid research doesn’t slow down. Scientists keep testing new catalysts and bioprocessing routes to make cleaner, faster, and cheaper synthesis a reality. I see excitement gathering around enzymatic processes, engineered microbes, and low-temperature routes, partly because old petrochemical methods can’t compete with sustainability targets. Industry-university collaborations dig into how slight tweaks in molecular structure could unlock better flame resistance, antimicrobial coatings, and even new battery electrolytes. The march toward bio-based and compostable polymers keeps the pressure on, especially as single-use plastics face regulatory crack-downs.
Toxicity, Safety, and Public Health Confidence
Compared with many organic acids, sebacic acid rarely causes alarm. Animal testing points to low oral toxicity and minor skin or eye irritation risk, which gives regulators peace of mind when approving it for food-contact plastics and cosmetics. That said, long-term studies on chronic exposure or environmental fate still draw scientific attention, as governments grow stricter about what substances drift into water or soil. Wastewater from older production sites can build up organic load if not handled right. The best producers invest in closed-loop systems and real environmental monitoring to spot risks early. Giving communities and workers confidence matters more than ever in the chemical sector—transparency on safety and environmental impact signals that lessons from the past won’t get swept aside.
What To Expect for Sebacic Acid’s Future
Looking ahead, everything points to rising demand and new use cases. Battery tech, medical polymers, and green lubricants lean heavily on this acid’s adaptability. Supply chains continue wrestling with swings in castor oil harvests, political issues in producing countries, and global climate disruptions. More producers invest in digital tracking, cleaner reactors, and circular approaches to cut waste and emissions. In conversations with sustainability teams, the word is clear: those who move slow will lose out as industry standards shape-shift each year. Expect more interest from bio-innovators, designers of green products, and even policy-makers, all chasing reliable, plant-based routes to tough, flexible materials without the toxic legacy of yesterday’s chemistry.
What is sebacic acid used for?
From Castor Oil to Daily Life
Sebacic acid hardly earns headlines, yet it quietly shapes many modern comforts. This powdery white material traces its origins to castor oil. Old-timers in industrial chemistry will remember long mornings refining castor beans, getting hands oily in the process. Many probably never saw the end product. Missing those steps hides the true reach of this underappreciated acid.
Nylons, Plastics, and What’s Inside Your Yogurt Lid
If you’ve ever popped open a container of yogurt or twisted the cap on a new bottle of water, you’ve gotten close to the work of sebacic acid. Nylon 6,10—one of the most versatile plastics around—relies on sebacic acid. This helps make plastics that bend just enough for convenience without snapping. Durable, light gear for campers and hikers, medical syringes, and car parts don’t just materialize. Producers trust this unique acid to help those products flex, last, and function.
Eco-Friendly Moves
Modern companies hunt for greener building blocks as the plastic waste piles up. Sebacic acid, drawn from a renewable source, beats petroleum products on sustainability. Some specialty bioplastics, used in everything from compostable cutlery to automotive interiors, feature sebacic acid. This isn’t some utopian experiment; it’s a tool that can work with our waste crisis. Many European auto manufacturers have started adopting these plastics. They’re responding to real pressure—regulatory and from buyers—who want products that don’t just break down into microplastics.
Personal Care and Pharmaceuticals
If you rub on skin cream or lip balm, chances are you’re getting help from sebacic acid or its derivatives. This acid softens and stabilizes cosmetic products. The same compound finds its way into the world of pharmaceuticals. Suppliers use it to help control the release of drugs—so people don’t have to remember eight pills a day. It acts predictably, which matters most to doctors treating complex diseases. Long-acting injectables, wound dressings, and some transdermal patches all owe their effectiveness in part to this flexible acid.
Lubricants, Adhesives, and Everyday Reliability
Few people think about what keeps machinery from grinding to a halt. Lubricants—down to the special greases protecting gears in freezing cold—get a boost from sebacic acid. Its stability and low reactivity keep engines humming in the Arctic and extend shelf life in suburban garages. Industries also count on it in adhesives and coatings, keeping paint bright over time and glue sticky, even in tough environments.
Growing Demand, Scarcity, and Smarter Sourcing
As demand grows, supply tightens. Supply chains still depend on castor bean crops, mostly grown in India and Brazil. Droughts or disrupted harvests send shockwaves through prices. To counteract the uncertainty, several labs have started testing bioengineered yeast that could make sebacic acid in big fermentation tanks. It’s a gamble that needs time and investment but could be a game changer for stability and sustainability.
Looking Ahead
Sebacic acid, while not famous, shapes personal comfort, health, and green ambitions. Research and smart policies can secure a supply that lets us keep reaping these quiet benefits without undercutting environmental gains. If green chemistry matters, then so does every link in the supply chain, right down to this humble acid.
Is sebacic acid safe for skin contact?
The Lowdown on Sebacic Acid
Sebacic acid has a bit of a reputation in chemical and manufacturing circles. Sourced mainly from castor oil, this white, powdery substance finds its way into plenty of places—from lubricants to some cosmetic products. People start to wonder: can it safely touch your skin, or does it pose risks?
Why People Care About Sebacic Acid in Skin Products
Seeing a chemical name on a skincare label can make alarm bells ring. With so many allergies and skin conditions, people want products they can trust. Sebacic acid doesn't sound soothing—most folks associate ‘acid’ with something harsh. Toss in the fact that many can’t pronounce the name, and it ends up on the “double-check” list. I always read labels during family shopping trips, so I get the anxiety when an ingredient seems out of place.
How Sebacic Acid Interacts With Skin
Sebacic acid acts as a dicarboxylic acid. Chemically, it helps balance pH and sometimes stabilizes formulas in skincare. Because it’s derived naturally, some companies use it as a “clean” alternative for certain product roles. Still, natural doesn’t automatically mean safe.
For most healthy adults, touching sebacic acid in diluted form doesn’t usually cause problems. Well-formulated products take skin sensitivity into account, and sebacic acid sits at low percentages like 1-2% in over-the-counter creams. The European Chemicals Agency lists it as having low skin irritation risk, provided it’s used in normal amounts. My own experience trying a sample moisturizer gave no stinging or redness, and that’s backed up by studies where volunteers reported little to no discomfort at typical concentrations. If someone already deals with eczema or allergies, caution is wise—patch testing pays off.
What About Inhalation and Accidental Exposure?
Industrial workers have different worries. Pure sebacic acid dust in large amounts can irritate eyes, airways, and skin. It makes sense: nobody wants a chemical loafing around on raw hands all day. Wearing gloves and handling powder in ventilated spaces gets recommended under workplace safety rules. The risk drops off in finished products, where the acid gets well-blended and diluted. Safety data sheets encourage washing after direct contact just to be thorough.
Is Sebacic Acid Toxic?
Toxicity stands low. A toxicologist would point out you’d have to eat or inhale large quantities to reach dangerous levels. Animal tests using higher doses didn’t show major organ trouble. The U.S. Food and Drug Administration even labels certain derivatives as “Generally Recognized as Safe” in food. That doesn’t translate directly to skincare, but it helps show the margin of safety is generous at low concentrations.
Better Safe Than Sorry
Everyone’s skin reacts a bit differently. Children, those with broken skin, or people prone to allergies should check with a doctor before slathering on something new. Reading product labels and patch testing lets people avoid surprises. Scientists constantly review data and update safety standards, which builds confidence in regulations set by agencies like the FDA or Health Canada.
What’s Next for Consumer Safety?
If skin contact ever leads to burning, peeling, or swelling, toss the product and speak with a healthcare professional. Watch out for new research, especially since formulas can change as companies update their ingredient lists. Asking questions—whether in a drugstore or at a dermatologist appointment—keeps consumers safe and informed.
What is the chemical formula of sebacic acid?
Understanding the Formula: C10H18O4
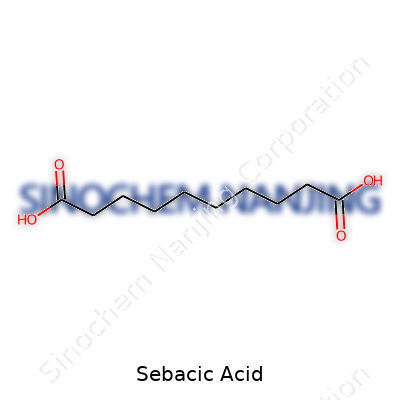
It’s a mouthful at first glance—C10H18O4. Sebacic acid comes with this simple but powerful formula. It’s built from ten carbon atoms, eighteen hydrogen atoms, and four oxygen atoms. That combination gives sebacic acid its unique character and its spot in countless manufacturing processes. I remember my college days wondering why we even needed to care about the makeup of dicarboxylic acids like sebacic acid. Years later, walking through an industrial facility, I understood better. Every number in C10H18O4 lines up with real-world applications and production challenges.
Why This Formula Matters
On the surface, the formula points to a dicarboxylic acid. Those two -COOH groups at each end let the acid cross-link with other compounds and form all kinds of polymers. Nylon 6,10 sure wouldn’t look the same without the long-chain backbone that sebacic acid provides. Out in the garage, the glossy finish on some old tools comes from lubricants where sebacic acid plays a deciding role. It’s all chemistry put to practical use.
Bigger Picture: Health and Environment
For folks working in industries, sebacic acid’s biological edge can’t be ignored. The cosmetic jars lining store shelves or the nail polishes at the drugstore—many depend on this acid for safe, skin-friendly formulas. It’s a compound that allows safer substitutes for more toxic additives without sacrificing quality or texture. The formula gives it moderate solubility and low toxicity, and I’ve seen safety data sheets repeatedly assure operators about sebacic acid’s low hazard profile. That helps keep both the workforce and eventual consumers safer.
Supply Chain and Sustainable Sourcing
I once talked with a supplier from Kerala, India, who pointed out that castor oil stands as the main source of sebacic acid today. The chemical formula connects to plant-based supply, which sets off a series of wins for farmers and manufacturers trying to cut away from petroleum-dependence. India now handles most of the world’s production, turning castor beans into a long-chain acid thanks to a process called alkaline cleavage. This stepwise transformation covers many chemical processes, each taking cues from the properties embedded in C10H18O4.
Challenges and Real-World Solutions
Supply doesn’t always keep up with demand. Each year, industries need more biologically sourced ingredients. That tension challenges growth in the synthetic fibers market or the push for “green” plasticizers in packaging and automotive parts. As a writer covering industry for years, I’ve watched companies innovate: integrating closed-loop systems, using waste from castor oil to feed back into energy production, and partnering directly with farmers to stabilize sourcing. The answer rests in both the reliable chemistry of sebacic acid and the creative ways people can rethink production systems. Companies, scientists, and farmers each play a part in turning a simple chemical formula into better materials, better products, and fewer environmental trade-offs.
Shaping the Future With Science
The story of sebacic acid doesn’t stay confined to a test tube. The formula serves as a foundation, but the choices people make around that chemistry decide its broader impact. Through steady investment in plant-based chemistry, increased transparency along supply chains, and smarter recycling initiatives, the journey of sebacic acid can lead to improvements not just in products, but in the economic opportunities for those growing the raw ingredients and the sustainability practices businesses champion moving forward.
With the chemical formula C10H18O4, sebacic acid threads its way through science labs, factories, homes, and fields—filling roles big and small, and showing just how much careful chemistry shapes everyday life.
How is sebacic acid produced?
Understanding the Journey from Castor Oil to Sebacic Acid
Sebacic acid comes from castor oil, a pale yellow plant oil pressed from the seeds of the castor plant. People often know castor oil for its use in cosmetics or as a medicinal product, not realizing that it’s a backbone for certain industrial chemicals. Most of the world’s supply comes from India and China, where castor beans grow well in subtropical climates.
The Manufacturing Process
The process starts with the pressing of castor seeds to get crude castor oil. Once you have that, the oil heads into industrial reactors to extract ricinoleic acid. Here’s the crucial part—through a reaction called “alkaline hydrolysis,” manufacturers break down the castor oil using a strong base such as sodium hydroxide. This step splits the ricinoleic acid and gives a mixture of sebacic acid and a byproduct called octanol.
After hydrolysis, the resulting mixture gets acidified with mineral acid, releasing crude sebacic acid. The crude material doesn’t look pure; it still holds onto color and impurities. Factories then use purification methods like solvent extraction and crystallization. Even with big stainless steel machines turning, there’s something familiar in the process—it feels a lot like refining sugar or salt for the kitchen, just on a much larger scale.
Challenges and Risks in Production
Castor oil isn’t always easy to work with. It’s sticky, has a strong odor, and at the source, the beans contain ricin, a dangerous toxin. The whole industry has to put serious attention into handling raw materials safely. Skilled workers in the field know that mistakes can bring safety risks—not just for people in the plant but for the communities nearby. Factories often locate in specific zones, but leaks and spills can still happen if caution gets tossed aside, raising environmental issues. Water contamination from these plants has made headlines before, especially when waste disposal falls short of regulations.
Traditional sebacic acid production also eats up a lot of energy. Most plants use high heat and strong chemicals, so the carbon footprint stacks up. Experts have criticized the industry for lagging in the adoption of clean technologies. While government regulations have tightened, actual improvements often depend on the willingness of business owners to change equipment and processes, costing money up front.
Why Sebacic Acid Matters
Products made from sebacic acid end up all over the place—in nylon for toothbrush bristles or engine parts, in cosmetics, even in biodegradable plastics. Curious minds might not notice, but nylon 6,10, a common plastic, gets its strength and flexibility from this simple molecule. Without it, plenty of everyday goods would start to look very different.
Cleaner Paths for the Future
Lately, some companies have started moving toward “greener” methods. One promising idea centers on enzymatic processes, using natural proteins to break down castor oil instead of harsh chemicals. Others test new catalysts that work at lower temperatures, cutting down on fossil fuel use. The environmental impact hasn’t vanished, but there’s a push for better transparency and accountability. Audits, updated factory designs, and strong local oversight can help reduce risks. If manufacturers keep pressure on for sustainable innovation, clean sebacic acid might soon become the norm rather than the exception.
What industries commonly use sebacic acid?
Everyday Products Stand on the Shoulders of Chemistry
Even if you’ve never given sebacic acid a thought, you’ve probably used something it helped create. Step into a hardware store, a pharmacy, your own garage, and you'll likely meet something that got its start with this versatile compound. The strongest impression comes from synthetic polymers—paints, lubricants, adhesives—where chemistry and practical life intersect. Age-old wisdom from the plastics industry makes one thing clear: you only get lasting results with dependable building blocks. Synthetic fibers and plastics lean on sebacic acid because it offers flexibility, strength, and resistance against water and heat. This blend suits the production of nylon 6,10 better than many others, which in turn delivers durable bristles in toothbrushes and robust gears in power tools. From camping gear to the zipper on your favorite jacket, this chemistry supports quality and reliability.
Automotive Needs Meet Chemical Ingenuity
Anyone who’s changed engine oil or fixed a noisy door hinge knows the frustration of worn-out lubricants. Most automotive greases used to break down under pressure or high temperatures. Sebacic acid changed that game. Its derivatives form the backbone of synthetic lubricants and hydraulic fluids, especially for cars and trucks that need resilience under extreme heat or freezing winters. It’s not just about making oil last longer; better lubricants protect engines and moving parts, saving car owners money on repairs and replacement. According to industry data, cars using synthetic lubricants made with sebacic acid derivatives require fewer oil changes and tend to show less corrosion inside the engine. These practical benefits make a big difference, especially as more automakers push for higher efficiency and cleaner emissions.
Changing the Face of Cosmetics and Pharmaceuticals
Many would not expect to find an industrial chemical in their lip balm or medicated ointment. Yet sebacic acid often acts as a key ingredient in creams, lotions, and pharmaceuticals. It offers a unique mix—it’s gentle and safe, and it creates stable, smooth emulsions. This stability lets cosmetic scientists develop products that don’t break down or separate on the shelf, giving customers real value. In the pharmaceutical world, sebacic acid-based polymers provide a slow-release mechanism for medications, especially in biodegradable sutures and implants. This leads to fewer hospital visits for patients and fewer complications after surgery, according to clinical studies.
Resins and Coatings: Durability That Lasts
Sometimes, what matters in a product is what you can’t see: the protective layer between the outside world and what’s underneath. House paint, industrial coatings, waterproof fabrics—these depend on protective resins formed with sebacic acid. Paints stick better and last longer because sebacic acid delivers the toughness that shields against weather, sunlight, and everyday wear. Factory floors, ship hulls, and household furniture all benefit from the improved chemical resistance and flexibility. Manufacturers report fewer callbacks and less maintenance, which saves money for both business owners and consumers.
Looking Forward: Sustainability and Supply
People often overlook sourcing and production. Most sebacic acid comes from castor oil, which means it relies on a renewable plant source. In a world where supply chains and environmental impact matter more than ever, this sets sebacic acid apart from petrochemical alternatives. Some chemists are already testing ways to increase the yield from castor seeds without heavy chemical processing. Reducing environmental impact strengthens the appeal for industries facing pressure to cut carbon emissions and waste.
Thinking Practically About Innovation
Stronger, longer-lasting, more effective—that’s what manufacturers chase, and sebacic acid delivers. Success boils down to understanding what materials bring both in performance and in real cost and impact. Innovation in the lab finds its way into homes, cars, and even medicine cabinets, powered quietly by the workhorses of industrial chemistry.
| Names | |
| Preferred IUPAC name | decanedioic acid |
| Other names |
1,8-Octanedioic acid Decanedioic acid Octane-1,8-dioic acid Sebacinsäure |
| Pronunciation | /siːˈbæsɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 111-20-6 |
| Beilstein Reference | Beilstein Reference: 1721186 |
| ChEBI | CHEBI:30780 |
| ChEMBL | CHEMBL1387 |
| ChemSpider | 5466 |
| DrugBank | DB02720 |
| ECHA InfoCard | 100.009.235 |
| EC Number | 204-669-1 |
| Gmelin Reference | 74152 |
| KEGG | C00554 |
| MeSH | D010423 |
| PubChem CID | 9148 |
| RTECS number | VS7875000 |
| UNII | V7T9C5J7HQ |
| UN number | UN1875 |
| CompTox Dashboard (EPA) | DTXSID8035072 |
| Properties | |
| Chemical formula | C10H18O4 |
| Molar mass | 202.25 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.21 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.93 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 4.72 |
| Basicity (pKb) | pKb ≈ 11.51 |
| Magnetic susceptibility (χ) | -63.4·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.419 |
| Viscosity | 1.7 mPa·s (at 150 °C) |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 256.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1335.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5736.6 kJ/mol |
| Pharmacology | |
| ATC code | D11AX10 |
| Hazards | |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| Flash point | 230 °C |
| Autoignition temperature | > 355°C |
| Lethal dose or concentration | LD50 oral rat 16000 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Sebacic Acid: "8000 mg/kg (oral, rat) |
| NIOSH | SEB3500000 |
| PEL (Permissible) | PEL: 5 mg/m³ |
| REL (Recommended) | 500 mg/m³ |
| Related compounds | |
| Related compounds |
Suberic acid Azelaic acid Dodecanedioic acid |

