Quinoline: Why This Old Chemical Still Matters in Science and Industry
Historical Development
My first brush with quinoline took me back to the dusty pages of chemistry’s long history, where stories of tar distillation and accidental discoveries fill textbooks and tales told in college labs. Back in the 19th century, coal tar seemed to be more nuisance than treasure—black, sticky, and smelly. Chemists soon realized these ugly leftovers packed quite a punch, unlocking all kinds of ring-shaped molecules. Quinoline came out of that bubbling tar, and scientists like Friedlieb Runge and Charles Gerhardt put their names in the books for figuring out how to tease it apart from the mess. By the end of the century, quinoline took on a practical life, turning from an academic curiosity into a building block for new medicines and dyes. This transition from unwanted waste to crucial chemical reminds me that unexpected value often hides in the places folks least expect. History shows how much persistence and a knack for seeing opportunity matter in changing raw materials into tools for progress.
Product Overview
Quinoline stands out as an aromatic heterocycle with a flair for versatility. Its two fused rings—one benzene, the other laden with nitrogen—combine to handle a surprising array of challenges. The chemical sits as a colorless to pale yellow oil that sometimes takes on a greenish tint in open air. In a lab, opening a bottle of quinoline delivers a sweet, somewhat pungent odor that lingers, never letting you forget its presence. Production hasn’t drifted far from its roots, with industry still relying on processes tied to the original coal tar heritage, though now pushed along with plenty of synthetic muscle. To me, quinoline’s draw comes from how this one molecule pops up across so many areas, from the production of antiseptics to the world of fluorescent dyes, and even in the development of drugs that save lives.
Physical & Chemical Properties
Getting your hands on quinoline means dealing with a liquid that floats above water, evaporates more quickly than you might expect, and dissolves with ease in organic solvents. The granules or oil show a boiling point around 237 degrees Celsius and a melting point just above freezing, making storage and handling manageable but never absent of challenge. Nitrogen in the ring system gives quinoline its special chemistry, allowing it to act both as a base and blend into complex reaction schemes. Stability holds up pretty well, but exposure to air and light can spark slow oxidation—little hints of yellow turning deeper over time signal that you need to think about storage. As far as chemical relatives go, quinoline has a whole clan: isoquinoline acts as a key cousin, and its methylated or chlorinated offshoots bring different tweaks to medicinal and industrial applications.
Technical Specifications & Labeling
On the technical side, quinoline often lands in bottles stamped with purity percentages and grades. Analytical labs call for the reagent to meet strict standards, while bigger operations working on bulk dyes or intermediates tend to relax the reins a little, tolerating minor impurities. Metrics like specific gravity, refractive index, and water content give buyers confidence in the consistency batch to batch. Labeling for hazards can’t be skipped; quinoline brings enough risk that all containers need those big pictograms seen in any chemical storeroom, serving as a reminder not to handle this aromatic with bare hands or without a working fume hood. Over the years, industry has moved toward clearer, safer warning messages—something everyone who’s spent time around organic solvents can appreciate.
Preparation Method
Lab traditions for making quinoline bring together skill, patience, and mastery of high heat. The classic approach runs through the Skraup synthesis, a reaction that never wins awards for prettiness but gets the job done. Mixing aniline, glycerol, sulfuric acid, and an oxidizer like nitrobenzene inside a rugged flask produces quinoline in fair yields. What stands out in my experience is how raw and aggressive the process feels—boiling, churning, and producing noxious fumes. Scale-up on the industrial side relies on similar reaction schemes, but with controls to manage the hazards. Synthetic tricks keep chipping away at cost and efficiency, but the basics haven’t shifted much in over a century, a testament to the stubborn effectiveness of tried-and-true chemistry.
Chemical Reactions & Modifications
Quinoline doesn’t just sit on the shelf collecting dust; its chemical reactivity keeps researchers and industrial chemists experimenting. The nitrogen atom sets up easy modifications—both for adding new groups at specific spots on the ring and for swapping in heavier elements or sidechains to adjust performance. Hydrogenation produces tetrahydroquinoline, a molecule valued for both pharmaceutical work and new catalysts. N-alkylation and halogenation crank up the options even more, widening the toolbox for building new compounds. Experience tells me that quinoline’s structure invites creativity, and the variety of possible transformation paths means that the chemical can evolve with research needs and technological advances.
Synonyms & Product Names
Reading through catalogs, you’ll see quinoline under several names—one chemist’s quinoline is another’s 1-aza-naphthalene or benzopyridine. These synonyms pop up in patents, research papers, and on bottles in university storerooms. Even small changes or substitutions (like turning it into 2-methylquinoline or its isomers) generate fresh trade names as companies seek to carve out niches. This name soup sometimes confuses newcomers, but with some practice, experienced chemists learn to decode the lingo and find what matches their needs.
Safety & Operational Standards
Working with quinoline always means treating it with respect. The molecule crosses skin easily and can cause irritation; breathing the vapors or handling spills leaves anyone with direct, unpleasant reminders of its toxicity. Regulatory bodies around the globe lay down rules, covering everything from workplace exposure limits to requirements for spill containment and disposal. Personal experience tells the same story—rubber gloves, tight-sealing containers, and eye protection aren’t up for debate. Organizations investing in safety training and clear operational standards prevent mishaps and protect both experienced professionals and newcomers trying to learn the ropes. Rushing things, skipping recommended procedures, or underestimating hazards risk costly and painful incidents.
Application Area
Quinoline earns its keep across a surprising range of fields. Pharmaceutical chemists lean on it as a building block, with antimalarials like chloroquine and antileishmanial agents showcasing just how many lives a single molecular scaffold can touch. Dye makers hold quinoline-based pigments in high regard for the deep, vibrant colors they produce on cloth, paper, and even under fluorescent lights. In the laboratory, the molecule helps drive research in catalysis, analytical chemistry, and molecular electronics. Agrochemical firms use derivatives in pesticides and herbicides, swinging the pendulum from scrappy tar byproduct to high-value specialty chemical in only a few steps. My own work with heterocyclic chemistry regularly runs into quinoline’s fingerprints, whether prepping standards or diving into new reaction pathways for academic projects.
Research & Development
Quinoline hasn’t faded as a subject for cutting-edge research. Scientists keep hunting for safer, greener ways to make and modify the structure, hoping to replace rough old acid mixes with cleaner and more energy-efficient options. Medicinal chemists look for untapped opportunities: finding fresh quinoline analogues that break through in tough therapeutic areas, including cancer or antibiotic-resistant infections. The molecule’s participation in organic electronics and material science has grown—conductive polymers, organic LEDs, and stronger resins all count quinoline among their family trees. From my perspective, what stands out is how questions about quinoline keep evolving as technology advances and regulations tighten the grip, pushing both industry and researchers to innovate.
Toxicity Research
Discussions on quinoline’s dark side don’t get skipped by anyone paying attention to chemical safety. Animal studies, lab tests, and years of industrial monitoring have shown that quinoline has the potential to harm more than bacteria. Chronic exposure links up with organ toxicity and potential carcinogenicity, a verdict reached by organizations following detailed reviews. The risks push regulators to enforce controlled ventilation, careful waste disposal, and periodic health monitoring for workers. Community experience in sites with heavy quinoline production paints a clear picture—companies and local authorities bear a heavy responsibility to keep emissions and contamination under tight control. Safer alternatives and improved formulations remain an urgent R&D priority, and the push for open toxicological databases supports better public understanding and oversight.
Future Prospects
Quinoline’s future depends on innovation balancing usefulness and safety. At the heart of green chemistry lies a drive to design cleaner, more sustainable methods—switching from harsh acids and oxidizers to milder, catalyst-driven transformations. Regulatory reform, stricter product stewardship, and community pressure create momentum for companies to debut safer derivatives and improved formulations. Academic labs get crowded with projects targeting eco-friendlier synthesis and predictive modeling of quinoline toxicity. Opportunities in electronics and photonics tease scientists with promises of new applications, while pharmaceutical discovery keeps turning to quinoline’s core structure for inspiration in treatments yet to see the light of day. As regulatory scrutiny grows, scientists and firms face pressure to innovate responsibly, putting safety and environmental health right beside commercial value. This balance will shape how quinoline continues to matter for the next generation of breakthroughs and the lives they touch.
What is Quinoline used for?
Quinoline in Medicine and Pharmaceuticals
Quinoline stands out as a compound with a long history in medicine. Most folks recognize its name from the antimalarial drug chloroquine. That’s only the tip of the iceberg. Many doctors and researchers have trusted quinoline-based compounds to fight everything from malaria to certain bacterial infections. The structure of quinoline forms the backbone of several drugs in the pharmaceutical world. This versatility comes from its unique chemical features, allowing scientists to tweak its structure to target specific illnesses.
Years ago, my uncle told me stories of taking quinine, a relative found in tonic water, back when malaria was a greater threat in rural areas. It worked because the quinoline ring structure disrupts the life cycle of the malaria parasite. Modern medicines have evolved, but quinoline still plays a giant role in synthesizing new, more effective drugs.
Industrial Applications
The story doesn’t end in hospitals or clinics. Manufacturing relies just as much on this compound. Producing dyes, for example, often starts with quinoline derivatives. Colors used for clothing and labelling products get their hues from these essential building blocks. Quinoline also helps in making resins and polymers that show up in everyday products—paint, coatings, and adhesives.
During my early days at a chemistry lab, I watched how quinoline-derived compounds acted as intermediates, connecting raw ingredients to the finished product. Without such compounds, the manufacturing process either slows down or grows more expensive. Factories looking to improve efficiency keep looking for ways to make use of quinoline's chemical reactivity.
Agrochemicals and Pesticides
No one wants bugs ruining a whole season’s work. Quinoline steps up here as well. Pesticide and herbicide producers count on quinoline compounds to keep crops healthy and profitable. The chemical structure, once tweaked, can target fungus, bacteria, or insects. Companies use these compounds because they hit the mark with fewer side effects compared to some older chemicals.
Farmers have started to worry about pesticide residues in the food supply and soil. Research keeps looking for quinoline-based solutions that break down faster and do less harm to the environment. One key challenge is striking a balance between getting rid of pests and keeping fields safe for future planting.
Environmental and Safety Concerns
Handling quinoline calls for caution. It offers plenty of value but brings some risks. Studies show that breathing in large amounts or handling it without proper protection may harm health. Short-term symptoms can appear after exposure—headaches, nausea, and dizziness top the list. Chemists working with quinoline follow strict safety rules, using proper ventilation and gloves even for quick tasks.
Companies and regulators need to keep working together to track quinoline's effect on water and soil. New technology may cut emissions or create safer forms for use in medicine or farming. Clear labeling and strong regulations help keep both workers and the public safe.
Looking Ahead
Quinoline proves its value in fields ranging from healthcare to industry and farming. Ongoing research tries to make safer derivatives, sometimes using artificial intelligence to spot promising new chemicals. By investing in cleaner processes and stricter safety, society can get the best from quinoline without losing sight of the risks.
What are the safety precautions when handling Quinoline?
Working with Chemical Hazards Up Close
Quinoline might sound like an old chemistry textbook footnote, but for a lot of folks—especially in labs and chemical plants—it’s a daily reality. I’ve found that just hearing about its toxicity doesn’t hit home until you catch a whiff of that strong, sharp odor or you learn about its irritant kick. Many overlook how a lapse in safety routines can really set back not just the work but everyone’s health.
The Hazards Go Beyond the Book
Quinoline doesn’t give warnings before causing harm. Inhaling the vapors, even briefly, can leave you with headaches, dizziness, or a sore throat. The liquid burns skin and eyes. Spend enough time around it without precautions, and the risk grows for liver damage and even cancer—quinoline shows up on lists of possible human carcinogens. Once, a co-worker forgot goggles, and that trip to the clinic stayed with us all.
Gear Up for the Real Risk
Personal protective equipment saves a lot of grief. Nitrile gloves, goggles, and a sturdy lab coat aren’t extras—they’re essentials. If I had a dollar for every time someone tried to “just do a quick transfer” without gloves, I could retire early. The stuff seeps through thin skin like clockwork. Goggles shield eyes from splashes, and a lab coat keeps shirts and arms covered, blocking direct skin contact.
Ventilation Means Breathing Easy
Quinoline vapors need to go somewhere safe. Fume hoods cut down exposure fast, and I’ve seen some labs get creative with airflow when resources get tight. Just cracking a window doesn’t cut it. When you smell quinoline, you’re already breathing more than recommended. Proper fume extraction ensures the air stays crisp, even after a spill or splash. Some labs invest in local exhaust systems for extra control; they keep the risk right where it belongs—out of your lungs.
Storage and Spills—No Corners to Cut
Label every bottle with clear, bold writing. Store quinoline away from heat sources and sunlight, in sealed containers, ideally in a ventilated, fire-resistant cabinet. A small leak or open container fills the room with fumes, and open flames invite disaster. Treat spills fast: soak up with absorbent pads, scoop into a sealed bag, and take it to hazardous waste disposal. Once, a rushed cleanup meant someone emptied a bucket down the drain—let’s just say that pipe repair took longer than planned.
Training and Routine Checks Keep People Safe
Training never stops with quinoline. Old hands know the rhythms, but new staff need clear guidance. Regular drills make safety routines second nature, and knowing the location of eyewash stations and showers goes a long way in emergencies. I always encourage asking questions—even if it means running late. One smart question about storage once uncovered a crack in a bottle that could have caused real trouble.
Looking Out for Each Other
Using quinoline safely isn’t just about rules—it's about a culture of care. Watch for the signs of overexposure in those around you. Share tips, swap stories about close calls, and double-check each other’s gear. The small, everyday actions—gloves on before the bottle opens, goggles up at every station—build habits that last a career and keep everyone healthy. Safety with quinoline grows out of paying attention not just to the rules, but to each other.
Is Quinoline toxic or hazardous?
Quinoline in the Real World
Quinoline pops up in a lot more places than most realize, from certain dyes and resins to medicines and even some pesticides. It's one of those chemicals that quietly finds its way into everyday objects and processes, especially across industries that touch everything from healthcare to textiles. Any chemical with that wide a footprint deserves closer scrutiny, especially on the health and safety front.
Understanding Toxicity Concerns
The talk about quinoline’s toxicity isn’t just fearmongering. Several credible studies show real risk for both short-term exposure to vapors and long-term buildup. People breathing in quinoline at work or through contaminated air sometimes struggle with respiratory irritation, headaches, and even dizziness. Skin contact isn’t harmless, either. Rashes and other reactions aren’t that rare for those handling quinoline regularly without strong gloves and proper barriers.
Digging into the deeper science behind quinoline, researchers point to possible links with liver and kidney damage. I once toured a chemical plant where safety officers stressed how the stuff lingers in the environment, sticking to soil and finding its way into groundwater. A few years back, a study in the journal “Chemosphere” called out quinoline’s persistence in water and the difficulty in breaking it down. This all raises tough questions about safe handling, not just at work but in terms of public health.
Possible Cancer Risk
Probably the biggest red flag with quinoline is its possible role as a carcinogen. The International Agency for Research on Cancer lists it as a "possible human carcinogen" based on data from animal studies, where exposure over time led to the development of tumors. That’s a serious classification; even if it’s not a known cancer-causer in people, common sense says caution makes sense. It only takes one high-profile case for trust to evaporate in an industry or in how products are regulated.
From my view, risks stack up most for people handling quinoline day after day, often in jobs where masks and fume hoods aren’t used consistently. That said, environmental leaks and water contamination could put entire communities in harm’s way if companies cut corners or oversight falls short.
Safe Handling and Solutions
Avoiding unnecessary health risks means taking prevention seriously right from the ground up. Facilities using quinoline have to focus on strong ventilation, full-face respirators, and real training for everyone involved. Disposal is a sticking point. Dumping quinoline into the regular waste stream isn’t just bad for rivers or soil—regulators impose serious fines for it. More than that, once water picks up contaminants like quinoline, treatment gets expensive and not all municipal systems catch it completely.
Improvement also comes through technology. I spent some time with environmental engineers who handle hazardous waste. They swear by closed-loop systems that keep contaminants contained. Targeted filtration and advanced chemical treatments help, but best results come from starting with less hazardous chemicals in the first place, or minimizing how often quinoline runs through a process.
Looking Forward
Public pressure keeps growing for more transparency about chemicals like quinoline. Supply chain managers, lab scientists, and neighbors near factories all want to know what’s being released and what risks linger. Regulations can move slowly, but big companies now face tough questions not just about their products, but about the full journey from raw materials to finished goods. For anyone working around chemicals like quinoline, real-world vigilance and steady improvements in safety offer the best path forward.
What are the physical and chemical properties of Quinoline?
The Stuff You Notice First
Quinoline stands out before you even get it under a microscope. Pour a little into a glass vial and you’ll spot a pale yellow liquid that pushes out a sharp, fish-like smell—the odor gets stronger if it’s not sealed tight. If you spill it, your nose picks up the sting long before your skin does. At room temperature, quinoline flows like water, but try and freeze it, and you'll need to cool down to about -15 degrees Celsius. Crank the heat up to 237 degrees, and it hits its boiling point, turning into vapor with a noticeable puff of that signature scent.
Getting to Know Its Makeup
This molecule feels simple on paper: C9H7N. It stacks a benzene ring onto a pyridine ring, with nitrogen tucked into one side. That N atom mixes up the chemistry. You don’t get the same reactions you’d expect from a plain hydrocarbon ring. Quinoline doesn’t dissolve well in water; try it, and you’ll just see oily beads swimming on the surface. Switch to alcohols or ether, and the stuff mixes right in. Scientists and industrial users rely on this for extractions and distillations.
Why It Reacts the Way It Does
This isn’t a molecule that sits quietly. Quinoline resists acids and bases much more than a regular benzene—thanks to the nitrogen. Expose it to strong oxidizers, and it won’t go out without a fight. With some heat and an acid catalyst, it likes to add things to its rings, sometimes making new drugs or dyes. Chemists grab onto this trait—they’ve turned quinoline into anti-malarials, antibiotics, and dyes. We even see it show up in specialty lubricants and resins.
Where It Fits in Real Life
Walk through a coal processing plant, and you’ll run into plenty of quinoline. It pops up in coal tar, which gets refined into tons of everyday items. For a stretch, people thought of quinoline as just another waste chemical. Now, with smart chemistry, it plays a piece in medicines we can’t do without. In the lab, it’s a clue for other chemicals—a marker, a reactant, a starting block. Environmental experts watch it closely because runoff from coal facilities, or accidental spills, can put quinoline into streams and groundwater. The chemical breaks down slowly, and the residue stays on people’s minds for good reason. It can hurt aquatic life.
Staying Safe with Quinoline
I’ve seen colleagues forget their gloves for just a minute handling quinoline, end up with headaches and a sick stomach. Breathing even a low dose for a few hours feels brutal. The vapor gets into your lungs, your blood. Regulations spell out safety steps: ventilate the workspace, seal containers right, pull on the protective gear. The chemical’s not considered a major carcinogen like benzene, but nobody suggests chucking safety for the sake of convenience. Respect for quinoline goes a long way in the workplace and outside, where its residue might reach soil or rivers. We need better clean-up techniques and catchment options at plant sites. Squeezing research dollars into green chemistry pays off big whenever another “dirty” byproduct lands in modern medicine or tech.
Everything about quinoline, from the way it pours out of a bottle to the way it links up in reactions, reminds me that chemistry in the real world stays messy, layered, and loaded with opportunity—if we approach it with knowledge and respect.
How should Quinoline be stored and disposed of?
Quinoline—What Makes It Tricky to Handle
Quinoline isn’t just another lab chemical you stash on a dusty shelf; it’s got a bigger presence than folks outside of chemical plants realize. Used to make dyes, pesticides, and even certain medicines, quinoline shows up in labs and factories more than most would expect. But this means mistakes with storage or disposal quickly spiral into environmental trouble—plus serious risks for anyone in the vicinity.
Storing Quinoline Right
Stuffing quinoline next to anything flammable, acidic, or under direct sunlight is asking for leaks or explosions. Metal drums with tight, corrosion-resistant seals work much better than open shelves or cheap containers. During my work in industrial safety, I saw a barrel stored in a poorly ventilated corner corrode shockingly fast, leading to fumes and a near cleanup disaster.
Never trust the smell test. Quinoline's vapors can mess with lungs long before you notice any odor, and they build up if storage space doesn’t have proper ventilation. Simple fixes—a vent fan, a chemical fume hood, or investing in purpose-built storage cabinets—cut risks down to size. Good labels do as much as any lock: nothing beats bold hazard warnings and clear spill instructions fixed directly on each container.
Temperature swings cause more than sweat. If a day gets too hot, sealed drums might swell until they burst. A cool, dry place below room temperature keeps quinoline stable and stops vapor pressure from creeping up. Even the best-sealed room needs regular checks. Ignoring leaks or trying to mop up tiny spills with paper towels just spreads contamination. Training workers to recognize problems and log every bottle's movement builds in accountability. Trust comes from having clear rules about who checks what, how often, and using strict inventory logs so nothing disappears unnoticed.
Tackling Disposal—Why Tossing Quinoline Down the Drain Is a Disaster
Too many people act like disposal is something only environmental officers need to care about. In the real world, one careless flush can foul up water supplies for an entire neighborhood. Quinoline causes cancer in lab rats; it seeps into groundwater fast. Dumping it in regular trash or washing it down drains skips every safeguard. I’ve seen news stories where careless disposal triggered expensive cleanups and long-term health monitoring for locals—nobody wants to be on the front page for that mess.
The safest route starts with a licensed hazardous waste handler. Waste haulers who specialize in industrial chemicals know how to pack, label, and cart off quinoline so it ends up where it can’t harm anyone. Incineration in high-temperature kilns, done at approved sites, breaks quinoline down into safer forms. Many chemical managers have to file shipping manifest records—and keep proof for years—just in case regulators ask. It’s not pure bureaucracy; it’s a line of defense.
Labs sometimes look for ways to neutralize or recycle chemicals before they leave the building. Still, nobody should attempt homegrown disposal methods without a clear green light from qualified chemists. If there’s any doubt, always treat quinoline as hazardous waste and arrange a proper pickup. Neighboring facilities sometimes join together to cut down on disposal costs—responsibility shared is responsibility enforced.
The Cost of Carelessness
No one wakes up hoping to cause a spill or land a business in legal trouble. Cutting corners to save a few bucks on storage or disposal quickly leads to expensive fines, ruins reputations, and puts real people at risk. Giving employees good gear, targeted training, and a voice in chemical safety beats every short-term fix. Once you see quinoline in the bigger picture, it’s clear: respect in handling pays back mile after mile.
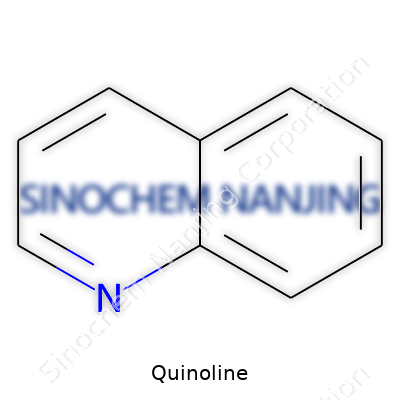
| Names | |
| Preferred IUPAC name | benzo[b]pyridine |
| Other names |
1-Benzazine 1-Azanaphthalene |
| Pronunciation | /ˈkwɪn.əˌliːn/ |
| Identifiers | |
| CAS Number | 91-22-5 |
| Beilstein Reference | 1853609 |
| ChEBI | CHEBI:17241 |
| ChEMBL | CHEMBL14 |
| ChemSpider | 493 |
| DrugBank | DB04280 |
| ECHA InfoCard | 100.015.312 |
| EC Number | 202-051-6 |
| Gmelin Reference | 3201 |
| KEGG | C08367 |
| MeSH | D011790 |
| PubChem CID | 7047 |
| RTECS number | GN7700000 |
| UNII | 7Q9187UQZO |
| UN number | 2656 |
| Properties | |
| Chemical formula | C9H7N |
| Molar mass | 129.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Strong, tarry, pungent |
| Density | 1.09 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.04 |
| Vapor pressure | 0.52 mmHg (25°C) |
| Acidity (pKa) | 4.9 |
| Basicity (pKb) | 4.85 |
| Magnetic susceptibility (χ) | -69.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.627 |
| Viscosity | 1.597 mPa·s (at 20 °C) |
| Dipole moment | 2.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 144.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 151.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5060 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | P01BA01 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | FC1=CC=NC2=CC=CC=C12 |
| Signal word | Warning |
| Hazard statements | H301, H311, H331, H373, H411 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 2, Instability: 0, Special: - |
| Flash point | 62 °C |
| Autoignition temperature | 480 °C (896 °F; 753 K) |
| Explosive limits | 1.2–7.8% |
| Lethal dose or concentration | LD50 oral rat 1,230 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,120 mg/kg (oral, rat) |
| NIOSH | QL3500000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Quinoline is "0.1 ppm (skin)". |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 900 mg/m3 |
| Related compounds | |
| Related compounds |
Isoquinoline Quinoline Yellow Quinoline alkaloid 8-Hydroxyquinoline |

