Pyrrole: More Than a Five-Membered Ring
Looking Back: Pyrrole’s Story
Take a stroll through the history of organic chemistry and soon enough, you’ll meet pyrrole. Chemists isolated this distinct-smelling compound from coal tar in the mid-1800s. They scratched their heads at its odd structure, a five-membered ring with one nitrogen, but recognized its potential almost at once. Pyrrole soon joined the pages of chemical exploration alongside the likes of benzene, indole, and furan. Over the next decades, researchers unraveled more about its behavior, wove it into dye chemistry, and found its skeleton in the core of hemoglobin and chlorophyll. Pyrrole’s journey tracks the progress of chemistry itself — from crude smelly extracts to fine-tuned syntheses, and finally to recognition as a central building block for life and materials science.
Pyrrole — The Basics
Pyrrole presents as a pale yellow to colorless liquid with a mild odor, though it quickly darkens when left out in light or air. Its molecular formula, C4H5N, only hints at its true character. The nitrogen atom, embedded in the five-membered aromatic ring, causes this modest-looking molecule to punch above its weight in reactivity and biological relevance. Pyrrole’s boiling point hovers below 131°C, and its volatility poses handling challenges, especially because exposure to oxygen leads to rapid polymerization or degradation. Many chemists recall their first whiff of this compound and the stubborn stains it leaves on glassware — stories shared in university labs the world over.
Properties that Matter
There’s much to say about pyrrole’s properties, but few things stand out like its aromaticity and reactivity. Aromaticity makes it more stable than typical amines, but not immune to attack. Pyrrole dissolves easily in organic solvents, less so in water, with a pKa for its conjugate acid pushing towards the lower end, showing it doesn’t act as a typical basic amine. On the technical side, its reactivity comes both from the electron-rich nitrogen and the delocalized electrons sweeping around the ring. Pyrrole’s liquid nature below room temperature and sensitivity to air make fresh samples necessary for reliable results.
Labels and Technical Details: Why Do They Matter?
Every lab worker remembers poring over bottle labels, and with pyrrole, attention to technical details is more than red tape. Storage recommendations call for inert atmosphere and minimal light, described clearly by suppliers — not just for shelf life but to avoid dangerous polymers or impurities. Purity often hits 98% or higher, but any deviation can change experimental outcomes in organic synthesis. Accurate labeling, such as precise CAS numbers or hazard statements, underpins safe and reproducible work. As someone who once grappled with degraded pyrrole in a grad school experiment, I know the cost of cutting corners on technical specifications.
Bringing Pyrrole to Life: How It’s Made
The earliest prep methods involved distilling coal tar or processing bone oil, both smelly and inefficient affairs. Over time, chemists turned to more efficient syntheses, like the Paal-Knorr and Knorr routes, where 1,4-diketones or alpha-aminoketones react with ammonia or amines. Here, control over temperature, reactant purity, and exclusion of oxygen define the success or mess of an organic reaction. Industrial-scale efforts center on safer, higher-yielding methods, often involving commercial synthesis from furan derivatives using ammonia sources and acid catalysts.
How Pyrrole Reacts and Adapts
Reactivity stands as pyrrole’s calling card. Its ring, both exposed and electron-rich, suffers nucleophilic and electrophilic attack with surprising ease — more so than benzene. Classic reactions include halogenation, acylation, and alkylation, where the 2- and 5-positions become prime targets. Oxidation can prompt ring opening, while metal complexes often grab the nitrogen’s lone pair. Modification doesn’t stop at simple reactions; many push the limits further, using pyrrole as a key step in assembling complex natural products, pharmaceuticals, or conductive polymers. In my own synthetic work, the challenges of controlling substitution patterns often led to new tricks with protecting groups and milder reagents.
What’s in a Name?
Chemists sometimes call pyrrole by its older names — azole, pyrroline, or tetrahydropyrrole, though the last refers to its saturated cousin. Synonyms pop up in literature, usually tracing back to translations or old German and French texts, where pyrrolen and pyrrol form part of dye chemistry lore. The world over, pyrrole keeps its identity anchored by CAS number 109-97-7, making sure everyone talks about the same thing, whether they’re building basic research or launching commercial applications.
Keeping Safe and Following Protocols
Pyrrole isn’t a stranger to risk. It’s flammable, irritates eyes and skin, and decomposes in air — not something to leave uncapped. Good ventilation, gloves, and eye protection stay standard for any real lab. Storage under inert gases, like argon or nitrogen, cuts risk and preserves product quality. Training in handling hazardous organics pays off quickly during synthesis or large-scale work, and written protocols set industry standards. I once watched a poorly capped container of pyrrole stain a fume hood with gummy, brown residue — a sure sign that even trace exposures to air bring real consequences. Strict adherence to safety rules not only protects people but keeps research trustworthy.
Where Pyrrole Shows Up
You find pyrrole and its family everywhere from pharmaceuticals to polymers. The backbone of many drugs builds off this five-membered ring, taking advantage of its ability to interact with biological systems, especially enzymes and receptors. Porphyrins — which feature four linked pyrrole rings — show up in heme, chlorophyll, and vitamin B12, touching everything from blood oxygen to plant energy capture. Industrial uses branch into polymers like polypyrrole, a conductive material used in sensors, batteries, and antistatic coatings. Dyes and pigments gain color fastness and intensity from pyrrole derivatives. Medical tests and imaging harness these properties for new diagnostics or treatment strategies. In my collaborations with materials scientists, pyrrole chemistry frequently offered the path to smarter, more responsive coatings and devices.
R&D: Digging Deeper
Pyrrole research stays vibrant. New discovery often springs from its use in natural product synthesis, where it mimics biological frameworks or offers shortcuts to complex targets. Synthetic chemists tinker with greener, higher-yielding prep methods to match stricter environmental standards. In materials science, work continues into tuning polypyrrole’s conductivity, flexibility, and environmental resistance, often guided by advanced spectroscopy, modeling, and clever chemical modification. Drug development draws heavy inspiration from naturally occurring and modified pyrrole compounds, tackling challenges in antibacterial or anticancer therapy. Each breakthrough, whether in generating enantiopure derivatives or multistep total synthesis, drives the field forward. Working at the intersection of organic and polymer chemistry, I’ve seen firsthand how tweaks in pyrrole structure spark new lines of inquiry.
Toxicity: Facts and Fears
Toxicity data on pyrrole has grown as demand for safety increases. Short-term exposure to vapors irritates the respiratory tract, while chronic contact damages the liver and kidneys, based on animal studies and reported incidents in workers. Metabolism leads to further oxidation, raising concern about the reactive breakdown products. Long stretches in the lab remind me that complacency around even “routine” chemicals can backfire, so regular air monitoring and biological surveillance become crucial where handling volumes rise. Regulatory limits and labeling requirements have tightened, rightly focusing both on acute hazards and the risk of longer-term exposure.
Eyes on the Future
Pyrrole stands at the edge of old chemistry and new materials. Demand for conductive polymers, smart sensors, and drug discovery keeps driving research into safer, easier, and more sustainable derivatizations. The challenge comes from balancing application growth against environmental and safety considerations. Green chemistry advocates press for renewable feedstocks and less hazardous prep, hoping to move away from toxic starting materials or byproducts. Advances in catalysis promise improved yields and selectivity, especially for sophisticated functionalized products that once took too many steps. Education remains as important as technology, making sure workers, students, and industry take the lessons of history to create safer, smarter futures for this quietly influential molecule. In talking with colleagues across academia and industry, it’s clear pyrrole’s story keeps growing, carrying lessons that cut across science and society alike.
What is Pyrrole used for?
What Makes Pyrrole Unique?
Pyrrole is one of those chemical building blocks that goes unnoticed outside scientific circles, but chances are, its presence has already shaped your daily life. The molecule, with a five-membered ring of four carbon atoms and one nitrogen, has a reputation for versatility. Working in a university chemistry lab, I learned how one simple structure could transform into a diverse set of useful products. This versatility put pyrrole at the core of several break-throughs across science and industry.
From Lab Benches to Everyday Products
Synthetic chemists spotted the value of pyrrole over a century ago. They began using it as a precursor for producing drugs, dyes, and specialty plastics. Pyrrole-based compounds ended up in pharmaceuticals like anti-seizure medications or antibiotics, often as the backbone that holds the active pieces together. While that might sound abstract, the impact turns concrete with the rise of new treatments for chronic conditions — our healthcare progress leans on the chemistry rooted in pyrrole.
It wasn't just about medicine. Textile dye industries tapped into pyrrole because of its affinity for creating stable, vivid pigments. Colorfastness improved washing after washing. Even artists who paint with tough acrylics may unknowingly rely on pyrrole pigments that stay bright for years, resisting the wear of sunlight.
Cutting-Edge Electronics and Emerging Tech
Pyrrole’s story doesn’t end with old-school chemistry. Around the turn of the millennium, research labs got excited about conductive polymers. When linked together, pyrrole units create polypyrrole, a dark, flexible material carrying electric charges. In a world eager for lightweight and adaptable electronics, polypyrrole found a home. Companies now develop sensors, flexible screens, antistatic coatings, and even lightweight batteries using this technology. Wearable tech and energy storage devices push for higher performance, and it’s no exaggeration to say that advances in this field owe plenty to pyrrole-based chemistry.
The Challenge: Green Chemistry and Safety
There’s no straight line from lab success to a marketable product, though. Pyrrole and its derivatives, while useful, carry some risks. Its volatile fumes mean safe handling is crucial in industrial settings. Regulations keep workers safe and protect communities near chemical plants, but enforcement varies worldwide. Green chemistry has stepped up in the past decade, promoting processes that reduce waste and switch to renewable resources. Researchers are experimenting with biosynthetic methods—using engineered bacteria to make pyrrole compounds—cutting down fossil fuel reliance and toxic byproducts.
Pyrrole Points to a Sustainable Future
Sustainable innovation thrives on knowledge. Scientists and engineers learn from small wins and setbacks, using them to develop cleaner production. I’ve seen chemical companies offer training on safer pyrrole handling and partner with academic groups researching greener synthesis. These partnerships help set higher safety standards and share technical advances quickly. Public awareness about what goes into everyday products pushes the industry to keep getting better.
Looking Forward
Pyrrole’s quiet influence spans medicine, color, electronics, and beyond. It demonstrates how a single molecule, introduced into a creative system, can alter the progress of technology and health. Responsible innovation and honest dialogue about safety ensure society continues to benefit from chemicals like pyrrole without losing sight of environmental and ethical concerns.
Is Pyrrole toxic or hazardous?
Understanding Pyrrole and Its Uses
Pyrrole shows up in many science and manufacturing labs thanks to its role in making dyes, pharmaceuticals, and advanced polymers. A sharp, sweet odor comes off pyrrole, and the liquid evaporates easily at room temperature. This basic chemical structure, five-membered and nitrogen-packed, forms the building block of molecules inside our bodies—things like vitamin B12 and hemoglobin. Yet in its pure, synthetic form, working with pyrrole takes some care.
What Makes Pyrrole a Concern?
For people spending long hours in chemical labs, hearing that pyrrole is “hazardous” suggests a practical question: can it hurt me? Inhaling pyrrole vapors irritates the nose, throat, and lungs, sometimes causing dizziness or headache. I remember the sting from a quick whiff near an open bottle during college lab—definitely not pleasant. Skin contact can trigger redness or rashes. The solvent properties of pyrrole mean that it soaks in through skin and can damage tissue with repeated exposure.
Animal studies point to a deeper issue. When rats or mice inhale or swallow enough pyrrole, their liver shows early signs of damage. Because the body’s natural enzymes break down pyrrole, harmful byproducts build up in the liver, creating toxicity. Nobody in their right mind messes around with high concentrations at home, but chemistry students and industry workers should know the risk doesn’t only come from single big spills—the danger can add up over time.
Another thing to watch for: pyrrole can catch fire if exposed to open flames or high heat. Its vapors travel and ignite well beyond the liquid source. Some accidents come from folks forgetting that those vapors drift through the air—one spark and the whole bench can light up. In storage, containers must stay tightly sealed and away from oxidizers or ignition sources.
Facts about Regulation and Safe Levels
Government safety bodies such as OSHA in the US or the European Chemicals Agency flag pyrrole as hazardous. Safety data sheets recommend personal protective equipment: goggles, strong gloves, and tight lab coats. Fume hoods go from “nice-to-have” to “absolutely required.” Official exposure limits still lack clear science for humans because most data comes from animal models, so most labs treat it with the kind of respect saved for aggressive irritants or suspected toxins.
Reducing Exposure and Risk
Good practice—skip shortcuts. Open your pyrrole bottles only under a working fume hood. I’ve seen folks try to pipette or dilute it at an open bench after a late-night study session, thinking it’s fine because “I’m only using a drop or two.” Next thing you notice: watery eyes, hacking cough, and lingering headaches. Always store pyrrole in amber bottles away from chemicals that can react violently.
Educators should train students and employees on the latest hazards. Spills need immediate cleanup with absorbent materials that don’t generate heat. For accidental skin contact, wash thoroughly and don’t hesitate to visit occupational health if redness or pain lingers. In a fire risk situation, only use extinguishers suitable for chemical solvents like CO2 or dry chemical units—not water since pyrrole floats and spreads, making the situation worse.
Closing Thoughts on Handling Chemical Hazards
Modern labs keep most toxic exposures in check with the right gear and attitude. Pyrrole can be managed safely with clear respect for its health risks and flammability. No shortcut justifies risking a lifelong injury or illness. Reading the real-world stories behind the safety data, anyone who works with pyrrole walks away understanding that knowledge—along with quality gloves and working hoods—keeps you out of harm’s way.
What is the chemical formula of Pyrrole?
The Formula Unpacked
Pyrrole carries the chemical formula C4H5N. That stands for four carbon atoms, five hydrogen atoms, and a single nitrogen atom forming a five-membered ring. I remember my first encounter with this molecule during an organic chemistry course—the distinctive smell, sort of nutty but also mildly sour, still sticks with me. That ring structure isn’t just a neat piece of trivia; it gives pyrrole its signature behavior, reactivity, and place in both science and everyday life.
What Pyrrole Means Beyond the Lab
Pyrrole isn’t an obscure lab chemical. Its core ring appears everywhere, from medicine to the pigments coloring your morning eggs. Hemoglobin—the stuff that carries oxygen in blood—contains porphyrins built from linked pyrrole rings. So does chlorophyll, the pigment that drives photosynthesis in plants. Without these, most of the world’s living systems would grind to a halt.
Because of its structure, pyrrole serves as both a building block and an inspiration for new molecules. Pharmaceutical chemistry leans on it heavily. Anti-inflammatory drugs and antibiotics often use a pyrrole ring. Industry turns to pyrrole for things like conductive polymers—the kinds of materials that go into organic LED screens. Early on, chemists realized pyrrole’s versatility, and they’ve kept pushing the boundaries since.
The Value in Getting the Formula Right
Chemicals might look simple on a chalkboard, but the stakes shoot up outside the classroom. Misunderstanding a formula can have consequences, even outside direct exposure. Once, a friend accidentally calculated a reaction yield based on an incorrect structure, and the project nearly collapsed over wasted time and raw materials. A single digit off in C4H5N and you’re not working with pyrrole at all.
Collecting knowledge about molecules like pyrrole also matters for safety. Pyrrole smells pleasant, but it’s not exactly harmless. Inhaling enough vapor can make you dizzy and repeated contact can cause more harm. Fact-based chemical knowledge isn’t just for labs; it belongs in classrooms, factories, and even in home workshops. Ensuring that workers and hobbyists are informed protects health, budgets, and the environment.
How We Support Safe and Smart Chemistry
Teaching chemical formulas is a good first step, but context closes the loop. Making sure the formula for pyrrole sticks in memory happens through hands-on discussion, lab practice, and real-world stories. Broad access to reliable safety data and educational resources strengthens the whole system.
For anyone handling chemicals, strong habits make all the difference. Proper labeling, careful storage, and actually reading up on safety sheets turn a recipe for disaster into just another day on the job. Industry has a responsibility to update training and make sure every canister in the back room is actually what the label claims.
Looking Forward
Pyrrole’s C4H5N skeleton links student memories to breakthroughs in medicine and technology. That small ring isn’t small in impact. Keeping chemical know-how strong doesn’t just support researchers; it shapes the tools, treatments, and technologies we’ll all depend on tomorrow.
How should Pyrrole be stored?
What Pyrrole Brings Into the Lab
Pyrrole comes out of the bottle looking pretty harmless. Plenty of us have cracked one open in an undergrad lab and gotten a dose of that sweet, spicy odor before reading up on what it could do. It’s used for lots of things—polymers, pharmaceuticals, dyes. Still, it doesn’t take long before someone asks, “where’s the best place for this bottle to go?” It’s not a question to gloss over. Pyrrole, left out in the open or handled with a lazy hand, can ruin experiments, damage health, and start fires.
Why Pyrrole Goes Bad
This chemical is stubbornly reactive. Light and air push pyrrole to form a tarry mess. So, that brand-new clear liquid starts turning brown and thick in days if left uncapped or near open windows. The issue isn’t just appearance. Contaminated or oxidized pyrrole spoils reactions and wastes money. There’s also fire risk. With a flash point near freezing, you can’t ignore it.
How To Store Pyrrole Like You Mean It
Few lab tasks frustrate me more than chasing after ruined chemicals because someone skipped basic storage steps. Pyrrole should stay in tightly sealed amber bottles, away from bright lights or heat. The amber glass isn’t just tradition. It really makes a difference in slowing down light-triggered decomposition. Store it in a flammable chemical cabinet with strong ventilation. No shortcut matches dedicated flammable storage for keeping vapors contained and temperatures steady.
Some large-scale users go further and blanket their pyrrole bottles with an inert gas before closing them. Argon or nitrogen can push away oxygen and moisture inside the container. I’ve watched folks in busy research centers swear by the practice. It isn’t overkill—oxygen-triggered oxidation sneaks up even at room temperature. I once had a sample set aside for a week develop visible deposits even with the cap on. Not worth the risk, if you ask me.
Keeping People Safe
Lab training manuals never mention the sickly feeling pyrrole fumes produce until you experience it for yourself. Storing this liquid with other volatile organics crammed side-by-side doesn’t make the lab safer. I always separate containers in ways that make sense—pyrrole away from acids, strong bases, or oxidizers. Mixing mistakes or leaks produce hazardous reactions or poisonous gases. Labels should stay readable even after years, and spill kits need to stay close.
Smarter Storage, Fewer Problems
It’s not enough just to shove chemicals on a shelf after buying them. Chemical suppliers like Sigma-Aldrich caution about pyrrole’s shelf life; once opened, the countdown really speeds up. Smaller bottles mean less headspace for air and less loss to spoilage. Ordering only as much as needed for the next batch or semester keeps the lab stock fresh and safer. In my own practice, tracking open dates on each bottle helped cut down on wasted product and improved experiment consistency.
Real Solutions Come From Real Care
Every lab worker who’s walked in on a sticky, leaking container of pyrrole knows that safe storage isn’t about being fussy or following pointless steps. It’s about health, valid data, and well-spent resources. Proper storage for pyrrole is just good science and, frankly, respectful of everyone who shares the workspace. It saves money, time, and peace of mind, all at once.
What are the physical properties of Pyrrole?
Pyrrole in Everyday Science
Think about the kinds of molecules chemists get excited about, and pyrrole’s name pops up more times than you’d expect. This five-membered ring, holding a single nitrogen atom, packs more punch than its structure suggests. In my college days, the sharp, unique scent of pyrrole in the organic lab made an impression that never quite faded. The liquid looked harmless, but it packed a volatility that students learned to respect. That sharp, somewhat fish-like odor lingers at low concentrations, a signal for careful handling before you even start reading the label.
Physical State and Appearance
Pyrrole is a colorless to faintly yellow liquid in its pure state. Give it some air or sunlight, and it darkens, picking up a brown hue quickly. That tendency to change tells you something about its sensitivity—oxidation is no small problem here. Once, I left a small sample vial exposed to the bench lights for an afternoon, and by evening, a golden liquid sat above a crust of darker resin—wasted material, lesson learned.
Boiling, Melting, and Volatility
This compound boils at 129° Celsius—relatively low compared to its aromatic cousins—so it doesn’t need much coaxing to jump into vapor form. Its melting point is well below room temperature, clocking in at -24° Celsius. Pyrrole stays liquid in most lab settings, but you won’t see it sticking around open beakers long, thanks to a noticeable vapor pressure. Even with the window open and a running hood, the odor lingers, reminding everyone that molecules this small and agile move fast through the air.
Solubility and Interactions
Pyrrole does not mix well with water—barely dissolves, really. Try stirring it in and you get two clear layers faster than you can cap the bottle. I learned to lean on a range of organic solvents: ether, benzene, acetone, each getting the job done much better. This solubility profile hints at why pyrrole and its relatives end up in so many industrial and pharmaceutical settings. Its nonpolar backbone means it’s easy to extract and manipulate, as long as you keep moisture out of the picture.
Density and Reactions with Environment
The density sits a little lower than water, about 0.967 grams per cubic centimeter. Spill some on a surface, and it spreads quickly but doesn’t sink through most materials. Leave the cap off and air along with sunlight jump into the mix—pyrrole oxidizes, darkens, and loses its punch. Stability always comes at a premium. Proper storage makes a difference here, with amber bottles and nitrogen blankets giving longer shelf life, but I’ve seen a single careless moment turn a clear batch into useless sludge.
Practical Challenges and Solutions
In labs or industry, pyrrole’s tendency to oxidize and its stubborn, penetrating aroma can create headaches. Extra care and the right storage conditions keep those issues in line. Small lessons stick: wrap storage bottles in foil, add a touch of an antioxidant, and use nitrogen purge if you want to safeguard purity. It’s a reminder that handling unpredictable materials takes patience and planning, but the rewards—fuel-cell research, pharmaceuticals, new polymers—make the effort worth it.
Why It Matters
Mastering the details of substances like pyrrole means fewer wasted batches, safer workspaces, and more confident innovation. It’s easy to overlook these details in the rush of a busy lab, but my years working alongside determined scientists proved that getting familiar with these physical quirks set apart the successful projects from the problem-plagued ones. Pyrrole is more than just a name in a textbook; its physical properties shape how research and industry move forward, one bottle at a time.
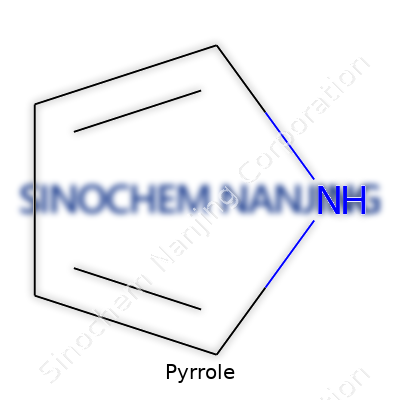
| Names | |
| Preferred IUPAC name | 1H-pyrrole |
| Other names |
1H-Pyrrole Azole Pyrrol Pyrrolidine Divinylenimine |
| Pronunciation | /ˈpɪr.oʊl/ |
| Identifiers | |
| CAS Number | 109-97-7 |
| 3D model (JSmol) | `Pyrrole|molecule|C1=CC=CN1` |
| Beilstein Reference | 63608 |
| ChEBI | CHEBI:35581 |
| ChEMBL | CHEMBL1502 |
| ChemSpider | 968 |
| DrugBank | DB04616 |
| ECHA InfoCard | 100.007.355 |
| EC Number | 203-809-9 |
| Gmelin Reference | 784 |
| KEGG | C01770 |
| MeSH | D011693 |
| PubChem CID | 8026 |
| RTECS number | UY7875000 |
| UNII | 6C7880D41X |
| UN number | UN1265 |
| CompTox Dashboard (EPA) | DTXSID1020637 |
| Properties | |
| Chemical formula | C4H5N |
| Molar mass | 67.09 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | penetrating odor |
| Density | 0.967 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.75 |
| Vapor pressure | 5 mmHg (20°C) |
| Acidity (pKa) | 17.5 |
| Basicity (pKb) | 13.6 |
| Magnetic susceptibility (χ) | -34.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.510 |
| Viscosity | 0.88 mPa·s (25 °C) |
| Dipole moment | 1.86 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 43.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 66.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1405 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D11AX15 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H301, H311, H331, H373 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 31 °C |
| Autoignition temperature | 200 °C |
| Explosive limits | 1.8% - 9.8% |
| Lethal dose or concentration | LD50 oral rat 1300 mg/kg |
| LD50 (median dose) | LD50 (median dose): 300 mg/kg (oral, rat) |
| NIOSH | SS-267 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Pyrrole: "Not established |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | 400 ppm |
| Related compounds | |
| Related compounds |
Furan Thiophene Pyridine Pyrrolidine Imidazole |

