Polypropylene: Cornerstone of Modern Manufacturing
Roots and Revolution
The story of polypropylene stretches back to the breakthroughs of the mid-20th century. Once chemists Karl Ziegler and Giulio Natta found a way to wrangle the unruly double bonds of propylene gas into straight-laced, repeating chains, they set the stage for a shift in both industry and daily life. Until then, folks leaned heavily on older plastics like polyethylene or PVC for tough, moldable materials, but polypropylene unlocked new territory. It arrived just as consumer culture bloomed, giving rise to lighter car parts, durable packaging, rugged textiles, and containers that could take a beating without turning brittle. Ziegler and Natta bagged the Nobel Prize for good reason, as what started with a beaker and some clever chemistry now lines store shelves by the ton.
What Sets Polypropylene Apart
Polypropylene stands out for its toughness and resistance to fatigue. Push, bend, or tap it—this plastic bounces back when others would crack. In my experience with everything from food storage to power tool handles, it's the stuff that sticks around long after other gear gives out. The material keeps its shape well even after hundreds of cycles through the dishwasher or a season rattling around in the trunk, thanks to its semi-crystalline structure. In contrast to polystyrene, which shatters if dropped, polypropylene’s ductility makes it a workhorse in more demanding jobs.
Properties That Matter
Polypropylene is a thermoplastic, melting somewhere between 130 and 170 degrees Celsius. That means you can heat and reshape it multiple times, which supports recycling efforts, though only a small share of plastics actually wind up recycled these days. The material shrugs off most acids, bases, and organic solvents; that's why chemical drums and lab beakers often use it. Compared to polycarbonate, it's lighter and less prone to stress cracking, though it softens at lower temperatures. Its low density results in lighter products—crucial for reducing shipping costs or slashing fuel consumption. Additives—fibers, pigments, stabilizers—expand its range, but even the plain stuff has a natural resilience that’s tough to beat.
Technical Details and Labeling
Truth be told, most people never check the recycling triangles stamped on their yogurt cup or medical syringe, but that little “PP” tells a story of utility. Polypropylene products carry the resin identification code number 5. Detailed specs like melt flow index or tensile strength guide industrial buyers, but for the rest of us, safe use comes down to basic labeling—microwave, freezer, or dishwasher safe. Labels don’t tell the full tale, though; behind the scenes, standards like ISO 1873 or ASTM D4101 make sure manufacturers stick to the right blend, so the parts don’t snap at the first sign of trouble.
How Polypropylene Comes Together
The process that takes light, pungent propylene gas and transforms it into reliable household goods starts on a massive scale at refineries. Propylene, a byproduct of oil cracking or gas refining, reacts with catalysts—often titanium-based—to produce long chains, each link a propylene molecule oriented just right. Factories spin out two main flavors: isotactic polypropylene, where every methyl group lines up like soldiers, and atactic polypropylene, with a more jumbled structure and less practical use. Real magic happens in polymerization reactors, which run under tight temperature and pressure controls to get the chain length and consistency just right.
Chemistry In Action: Modifying the Basics
The path from propylene to polypropylene doesn’t stop at base resin. Chemists tweak the material to create copolymers and blends tuned for impact resistance or clarity. Ethylene-propylene copolymers, for example, stretch flexibility and boost performance in products expected to take a hit—think automotive bumpers or football helmets. Grafting, cross-linking, or adding flame retardants as needed shapes the plastic’s final properties. Each blend sidesteps certain weaknesses of untreated polypropylene, and that’s why you see it in places as varied as lab gear and garden furniture.
Other Names in the Toolbox
Polypropylene goes by names such as polypropene or simply “PP” in trade and science circles. Major producers might slap their own trademarks on a batch, but at the end of the day, the product remains much the same: a tough, versatile resin recognized by the number 5 inside the recycling logo. In the field, folks might talk about random or block copolymer types, distinguishing blends by toughness or clarity, but this variety just underscores its adaptability.
Staying Safe and Playing by the Rules
Production and use of polypropylene run up against health, fire, and environmental standards. I’ve seen first-hand how a haphazard approach in the shop—melting it over a bare flame or recycling contaminated batches—leads to trouble. The industry follows standards from groups like the FDA or EU REACH when using polypropylene for food contact, toys, or medical gear. Workers follow ventilation and dust control measures during manufacture to cut the risk of static sparks or inhalation. Compared with older materials like PVC, polypropylene merely melts and drips rather than producing clouds of toxic smoke, but that’s no reason to relax. Safe operations mean lower emissions and less chance of workplace injury.
Everyday Uses and Surprising Reach
If a product bends and bounces back, odds are polypropylene had a hand in making it. Food containers, car interiors, home appliances, reusable shopping bags, outdoor furniture—they all tap into the material’s durability. Nonwoven fabrics spun from its fibers show up in diapers and surgical masks. Carpeting owes its stain resistance to polypropylene’s water-shedding properties, and battery cases exploit its balance of toughness and chemical inertness. The plastic’s light weight pairs with good performance under mechanical load, which is why automakers use it to shave pounds from cars and trucks.
New Directions in Research
Scientists work to push polypropylene into niches once out of reach. Research centers and startups are tuning its microstructure for 3D printing or developing blends for medical implants that reduce rejection and degrade cleanly in the body. The race to find better antioxidants, UV stabilizers, or flame retarding treatments keeps chemists busy across universities and private labs. Antibacterial grades are gaining traction, especially after COVID-19 highlighted the need for safer hospital equipment. Recent academic journals teem with investigations into thermal stability, recyclability, and novel copolymers designed for niche engineering challenges.
Toxicity and Environmental Burden
Concerns about microplastics are on the rise, and polypropylene sits squarely in the conversation. Studies in environmental science warn of fibers and particles finishing up in oceans, rivers, and fish guts. While it doesn’t leach hormone-altering additives like BPA, the sheer volume produced worldwide means even low-toxicity plastics can add up to a problem. Animals digesting debris or inhaling particles might face chronic issues, though hard toxicity data for regular use in food and drink containers remains reassuringly low. A key issue is not acute poisoning but long-term accumulation and disruption to ecosystems. On the human health front, burning polypropylene in the open releases pollutants, so proper end-of-life handling matters more than ever.
What’s Next for Polypropylene
Future progress pivots on better recycling, smart blends, and widening bio-based supply chains. The current global appetite for disposable packaging and medical supplies won’t vanish overnight, so researchers and manufacturers lean toward mechanical and chemical recycling to cut landfill waste. Early signs suggest enzymes or novel catalysts could break down polypropylene at lower environmental cost, but those technologies still wait for prime time. On the production side, using propylene from plant sources might reduce carbon footprints, while clever design—keep a product in use longer, use less plastic in each piece—offers practical ways to reduce impact without losing the strengths that have kept polypropylene in demand since the 1950s.
What are the main uses of polypropylene?
Everyday Objects You Don’t Think Twice About
People often pick up containers, snack on chips, even tug at the handle of a shopping bag. Polypropylene plays a part in all these moments. This plastic shows up in food packaging, yogurt cups, medicine bottles, and bottle caps, shaping much of what sits on kitchen shelves. Its toughness keeps food fresh longer and helps medicines last the journey from factory to home. From my own kitchen drawers, I’ve found polypropylene lids and utensils surviving years of dishwasher cycles without damage.
Why the Textile World Relies On It
Wrap yourself in a fleece blanket or wipe down with a cleaning cloth—polypropylene fibers make these comforts possible. The fibers feel light, resist moisture, and dry quickly. They shrug off stains—meaning a spilled drink won’t ruin a rug made from this plastic. I learned this during a winter camping trip, where my gear stayed dry and warm thanks to the woven fabrics. Polypropylene’s role in nonwoven medical masks, gowns, and sterilization wraps took on fresh importance in hospitals. These items protect workers and patients, showing just how much material science steers health and safety.
Sturdy Parts Behind The Scenes
Dining outside or at a busy event, plastic chairs and tables hold up to rough handling. Polypropylene stands up to bumps, sunlight, and rain. In the car, dashboards, bumpers, and battery cases use this stuff because of their strength and low weight. Saving weight in vehicles translates to better fuel efficiency—a fact the auto industry takes seriously as gas prices change and emission rules tighten.
Saving Resources And Protecting The Environment
Polypropylene uses less material to make strong, lasting products. This lowers the load on raw materials and cuts costs. It’s also one of the few plastics that can be recycled into new items, from storage bins to outdoor furniture. Not all recycling bins accept it in every city, but when it gets another life, it keeps waste out of landfills and oceans. With pollution in the headlines, every step toward recycling helps.
Medicine, Machines, and the Modern World
Hospitals depend on safe, sterile gear. Polypropylene fits clean environments—syringes, pill bottles, and lab equipment rely on it because it blocks bacteria and doesn’t react with most chemicals. In labs where safety means everything, I’ve seen polypropylene containers shield sensitive ingredients. In plumbing, random house repairs, or job sites, the material shows up as pipes, fittings, and insulation wraps.
Choosing Smarter Products
Why care what a cup, mask, or rug is made of? Polypropylene proves that the right material creates better products, saves energy, and solves problems big and small. As everyday life grows more complicated, paying attention to these choices lets us pick products that work better and last longer. Solutions to plastic waste start with smart design and good recycling habits. Small changes here ripple through homes, hospitals, and the wider world.
Is polypropylene safe for food contact?
Understanding Polypropylene in the Kitchen
Plastic has invaded just about every spot in our kitchens. Polypropylene, or PP, shows up in yogurt cups, microwaveable containers, reusable water bottles, and even in the stack of takeaway boxes waiting in the cupboard. Questions about safety pop up with every new documentary or food trend. Years ago, I remember my mother using whatever plastic tub was close by for leftovers, washing and reusing containers without giving it much thought. That’s changed, and more people look for the little recycling symbol on the bottom—and want to know what it actually means.
Why Polypropylene Gets the Green Light
Polypropylene, marked with recycling code ‘5’, stands out for its resistance to heat, moisture, and most chemicals. This stability is why lunchboxes and microwavable containers often use it. Official agencies like the U.S. Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) both sign off on polypropylene for direct food contact. They examine whether substances from plastics might seep into food and check for any negative health effects. According to these bodies, polypropylene does not leach significant amounts of chemicals known to cause concern. That’s a big reason you see it everywhere, from restaurant packaging to toddler snack cups.
Concerns About Plastics and Long-Term Exposure
Lately, concern about all plastics has shot up—microplastics, hormone disruptors, chemical residues. Some people lump all plastics into the same bucket of suspicion. A 2019 study found low migration levels from polypropylene into food, mostly under extreme conditions. Unlike polycarbonate, which leaches bisphenol A (BPA), polypropylene does not rely on BPA, which matters to people worried about hormone disruption.
Research so far shows that under normal household use, like reheating leftovers or storing dry foods, polypropylene holds up. The usual advice circles back to common sense: follow the instructions, don’t nuke the container for too long, and toss out old containers that show cracks or stains. In my house, I try to use glass for heating but still grab polypropylene for lunches and picnics, trusting its track record and approval from food safety authorities.
Safer Habits for Using Plastic Containers
Some real-life habits cut risks even further. Scrubbing plastic too hard or putting it in a super-hot dishwasher can roughen the surface and, over time, create more leaching. I learned that after noticing the cloudy, scratched bottoms of containers I kept using for years. Replacing those with new ones or swapping in glass for certain foods offers peace of mind.
Recycling also comes into play. Polypropylene can get recycled in a growing number of communities. By choosing containers that last and recycling when possible, it’s possible to cut down on single-use plastics and keep lunch from adding to the landfill.
Looking Ahead
No solution is perfect. The bigger picture involves finding better materials, less wasteful packaging, and stronger recycling systems. For now, polypropylene stands as one of the safer bets for the lunchbox or the fridge, especially with a little care and attention. Transitions to plant-based plastics hold promise but, as of now, polypropylene balances practicality, safety assessments, and widespread availability.
Seeking out trustworthy information and developing mindful everyday habits matters far more than simply avoiding plastics altogether. Kitchens keep changing, and the tools we use to store food should keep up with what science really tells us.
How does polypropylene compare to other plastics in terms of strength?
A Straightforward Look at Polypropylene
Polypropylene often pops up in lunchboxes, sports gear, battery cases, food packaging, and car interiors. As someone who has worked in environments ranging from industrial floors to community recycling drives, I’ve seen companies and households rely on this plastic for both rugged and everyday uses. Polypropylene’s reputation for balancing durability, chemical resistance, and light weight stacks up against other plastics in noticeable ways.
How Polypropylene Handles Force
Some people ask about “strength” in the sense of withstanding weight and impact. Polypropylene bends and flexes without breaking in many settings. Try folding a reusable shopping bag or those hinged food containers—pressure creates a bend, not a crack. Polypropylene’s structure forms long molecular chains that give it this flexibility. PVC and polystyrene, by comparison, break far more easily under stress. If you drop a polystyrene cup on concrete, it’ll shatter. The same cup made from polypropylene usually shrugs off the impact.
Polypropylene gives decent performance at pressures up to about 5,000 psi. By contrast, ABS (used in Lego bricks or power tool shells) can take closer to 7,500 psi, so engineers reach for ABS when a part needs to resist deformation under heavy loads. Polycarbonate blows both out of the water for brute strength, staying clear and tough in eyeglass lenses and bulletproof windows. You could whack a polycarbonate panel with a hammer and barely leave a mark. Polypropylene can’t match that, but it beats polycarbonate in resisting many solvents, making it a practical champion for chemical containers.
Everyday Resilience
I’ve watched polypropylene bounce back from the sort of wear that ruins other plastics. Long exposure to water, many detergents, and even some acids rarely affect it. Polycarbonate and PVC will cloud or crack from chemical attacks that leave polypropylene untouched. Plumbers use polypropylene pipes for hot and cold water because these pipes don’t snap after years of pressure cycles.
Direct sunlight is a tough enemy. Polypropylene weakens and turns brittle outdoors due to UV rays, unless it gets special additives. High-density polyethylene, in contrast, can take more sun exposure before failing. In areas with punishing heat and sunlight—like farming in Texas or roofing jobs in Arizona—people look at UV resistance first. Polypropylene handles indoor and shaded uses best.
Practical Facts and Solutions
Value drives many choices in manufacturing. Polypropylene costs less than ABS and polycarbonate, so cost-sensitive industries use it for food tubs, caps, carpets, and even car battery shells. Its toughness gets a boost from adding glass fibers or minerals. This composite approach gives the material a big jump in both stiffness and breaking strength, rivaling even much more expensive plastics without losing chemical resistance. Thanks to this adaptability, engineers don’t always turn to the “strongest” option—they optimize for cost, process, and function.
Recycling presents another consideration. Polypropylene carries recycling code #5 and faces more barriers in recycling streams than PET or HDPE. About 1% of polypropylene packaging was recycled in the United States each year by 2021, according to the EPA. Some new programs look promising, with improved sorting and chemical recycling technologies aiming to keep more polypropylene out of landfills. Consumers can help by rinsing and sorting plastics—that one step directly increases the value and reusability of the material.
Can polypropylene be recycled?
The Trouble with Recycling Polypropylene
You’ll spot polypropylene everywhere—food containers, takeout boxes, yogurt tubs, bottle caps. Number 5 on the recycling triangle signals its presence. People toss these items in blue bins hoping they’ll get a new life, but the truth isn't that simple. Polypropylene can be recycled, but the journey has a lot of bumps that most folks don’t see.
Why Polypropylene Gets Left Behind
Curbside recycling programs in big cities collect plenty of polypropylene. Sorting facilities then scramble to separate it from different plastics. Unlike PET (think water bottles) or HDPE (milk jugs), polypropylene doesn’t always find a buyer. Markets for recycled polypropylene shift quite a bit, and some places don’t bother to process it at all. This plastic also turns up dirty after holding greasy foods or dairy, making cleaning and reprocessing expensive and complicated.
As someone who’s watched a lot of recycling bins sorted at local facilities, I’ve seen how often polypropylene piles up with everything else that processors reject. Some of it gets incinerated. Some ends up in landfill. Tossing clean containers in the right bin only works if there’s a company ready to take them. Many towns skip it and focus on easier materials instead.
Change Starts with Better Infrastructure
Manufacturers want cheap recycled material that works like the fresh stuff. Polypropylene can lose strength each time people recycle it, so it rarely returns as new yogurt tubs or takeout boxes. It usually becomes molded items like storage bins, car battery cases, or landscaping tools. Chemical recycling, which can “unmake” plastic and turn it back into oil or monomer, offers hope but remains expensive and rare.
Some companies are stepping up. Consumer brands now invest in sorting robotics and build supply chains for clean polypropylene. A few towns have joined big efforts, adding new sorting equipment that separates polypropylene from the recycling stream. Groups like The Recycling Partnership have helped ditch the guesswork, putting clearer labels on packaging and standardizing bins and rules. These moves mean more polypropylene could soon find its place in the recycling loop.
Everyday Choices Matter, But Policy Drives Real Change
Households can make a difference by emptying and cleaning containers before recycling. Still, this tackles just part of the problem. Polypropylene recycling will take off only if state and local governments support new equipment and strong end markets for recycled products. Extended producer responsibility laws, which force companies to help pay for recycling what they sell, push brands to design better packaging and close the loop. Europe already moves faster here than the US, setting ambitious targets and backing them up.
Simple education helps too. Most people can’t tell what’s recyclable and what’s not unless the rules stay clear and consistent. Local governments and companies must give honest info. I’ve given recycling workshops where attendees act shocked once they learn so much gets tossed out. It’s discouraging, but it sparks good questions and fuels real change.
Looking at the Road Ahead
Polypropylene recycling faces hurdles, but it’s not doomed. As public pressure grows, companies and lawmakers respond. Smarter machines, better policy, and consumer effort team up to keep more plastics in use and out of the environment. With the right groundwork, polypropylene’s story could shift from “wishcycling” to real reuse.
What are the temperature limits of polypropylene?
The Everyday Plastic That Doesn't Love the Heat
Polypropylene often shows up in my daily life—food containers, takeout cups, even medical tools I’ve handled over the years. People trust it. Still, the truth about its temperature range sometimes gets glossed over. Most folks know it’s tough, lightweight, and popular because it resists chemicals and water. Far fewer realize it’s not invincible once the heat rises or drops past certain points.
Hot Drinks? Think Again
I remember the first time I tried reheating takeout noodles inside a polypropylene tub. The container never melted, so I figured it must be fine. After a few uses, though, lids started warping, and corners turned soft. Turns out, polypropylene typically holds its shape up to about 100°C (212°F). Some lab data show it can handle brief touches with temperatures just above this, inching toward 120°C (248°F). Keep it at that level too long, though, and the material weakens, bends, and starts to lose integrity.
The higher end—where microwaves or steam come in—brings risk. Food tastes off, flavors seep into plastic, and, in worse cases, micro-particles might sneak into the dinner. European food safety agencies have looked closely at this. Heat-tolerant plastics must go through migration testing to limit the risk of unwanted chemicals drifting into food. Even then, most experts recommend swapping out old, scratched containers for new ones if you regularly heat them.
Cold Snap: How Polypropylene Reacts
Winter groceries sit just fine in polypropylene packaging in the freezer at home. Drop the temperature way down, though, closer to -20°C (-4°F), and it starts to stiffen. Shoving a container out of a deep freeze sometimes snaps the lid or sends hairline cracks through the plastic. Polypropylene doesn’t crumble easily, but it loses flexibility as the chill sets in. Anyone who’s seen a cracked lunchbox after a week at the very back of a chest freezer knows the feeling.
Industrial Uses and Surprising Limits
Polypropylene gets put to work in factories, hospitals, and labs. It resists sterilization routines for a while, though it still can’t beat sustained high temperatures—think steam autoclaves running above 121°C (250°F). Manufacturers use glass-filled varieties or blend in stabilizers for a slight boost, but the core issue remains: push it beyond its comfort zone too often, and it breaks down faster. If you walk through any plastics recycling center, you’ll see bins marked “PP#5” bins holding containers faded, brittle, and sometimes warped from years in the microwave or dishwasher.
Better Choices, Smarter Solutions
Whenever people ask me about heating leftovers or sterilizing lab gear, I point out the safest bet lies in knowing what the plastic can take—and where it falls short. Manufacturers can improve polypropylene with additives, mixing in chemicals or fibers, but it still won’t match true high-heat polymers, like polycarbonate or PEEK, used for industrial tasks. Home cooks get better results with glass for steaming and reheating, reducing the risk of breakdown and food safety concerns.
It really comes down to understanding the materials in your hands. Polypropylene works best at temperatures up to boiling water, but push much further, and you’re rolling the dice. Most product guides and recycling labels give hints, but listening to that creak, warp, or brittle crack saves frustration long-term. Simple changes—new containers, different packaging, or just swapping in old-school glass—go a long way toward safer, longer-lasting results.
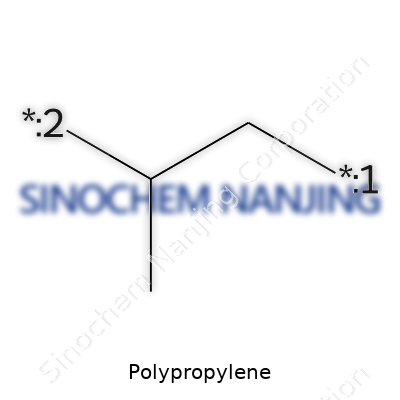
| Names | |
| Preferred IUPAC name | poly(propene) |
| Other names |
Polypropene PP Polypropylena Polipropileno |
| Pronunciation | /ˌpɒl.iˈprɒp.ɪˌliːn/ |
| Identifiers | |
| CAS Number | 9003-07-0 |
| Beilstein Reference | 1044987 |
| ChEBI | CHEBI:60828 |
| ChEMBL | CHEMBL2108751 |
| ChemSpider | 26663 |
| DrugBank | DB11140 |
| ECHA InfoCard | 03e4e4e9-aa18-405e-bc63-9ccfa42c3e41 |
| EC Number | 9003-07-0 |
| Gmelin Reference | 17521 |
| KEGG | C16297 |
| MeSH | D011115 |
| PubChem CID | 23960 |
| RTECS number | TSLPOA00XX |
| UNII | F5TD010360 |
| UN number | UN1942 |
| CompTox Dashboard (EPA) | DTXSID2020736 |
| Properties | |
| Chemical formula | (C3H6)n |
| Molar mass | 42.08 g/mol (monomer unit) |
| Appearance | White granules or powder |
| Odor | Odorless |
| Density | 0.90–0.92 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.62 |
| Vapor pressure | Very low |
| Acidity (pKa) | ~45 |
| Basicity (pKb) | > 12.5 |
| Magnetic susceptibility (χ) | –9.4×10⁻⁶ |
| Refractive index (nD) | 1.49 |
| Viscosity | Viscosity: 1.5 – 5.5 cPs |
| Dipole moment | 0.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -20.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4632 kJ/mol |
| Pharmacology | |
| ATC code | D08AX04 |
| Hazards | |
| GHS labelling | GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | No hazard statement. |
| Precautionary statements | P210, P260, P280, P314, P501 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Flash point | 260 °C |
| Autoignition temperature | 670 °F (354 °C) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): > 8,000 mg/kg |
| LD50 (median dose) | > 8,000 mg/kg (rat, oral) |
| NIOSH | 68152 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Polypropylene: "15 mg/m³ (total dust), 5 mg/m³ (respirable fraction) |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
Polyethylene Polystyrene Polyvinyl chloride Polycarbonate Polyethylene terephthalate |

