Looking Deeper into Propionic Anhydride: More Than a Chemical on the Shelf
A Walk Through History
People have tinkered with acids and anhydrides for centuries, but propionic anhydride rode the wave of industrial chemistry in the late 1800s. I remember poring over old papers describing early syntheses, which always seemed to turn up after propionic acid itself caught attention around 1844. Growing interest in organic synthesis and pharmaceutical manufacturing pushed researchers to search for efficient ways to link molecules, and anhydrides like this one became go-to reagents in labs that needed to make amides, esters, or introduce the ‘propionyl’ group into more complex products. As industry demanded slicker extraction processes, propionic anhydride found routines in both small labs and chemical plants.
What Propionic Anhydride Brings to the Table
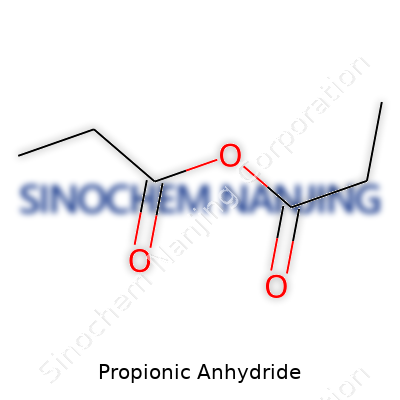
Propionic anhydride stands out as a clear, colorless liquid with a sharp, biting odor—it’s not the sort of thing you forget after an afternoon in the lab. Its boiling point hovers around 167 degrees Celsius, and its density settles a little lighter than water. Chemistry-minded folks respect that this anhydride reacts vigorously with water, making its handling a serious matter. Storing and moving this chemical means keeping an eye on moisture because a drop in the wrong place triggers a hissy hydrolysis, releasing propionic acid. With a formula of C6H10O3, two propionyl (C2H5CO–) groups join up to make it more reactive than basic carboxylic acids.
Labeling and Handling: Getting Practical
In any lab or warehouse, proper labeling has saved more than a few fingers and noses. Clear hazard symbols announce its corrosive properties. I’ve seen labs refuse deliveries if containers lacked the correct GHS pictograms and signal words. Spill response drills often circle back to propionic anhydride, since it can damage skin, eyes, and lungs quicker than people realize. Beyond the hazard warnings, temperature control during storage matters. Leaky caps or cracked drums make a mess not just for those nearby, but for air filtration systems too.
How Chemists Prepare Propionic Anhydride
Most commercial batches start with propionic acid and acetic anhydride, or sometimes phosphorus pentoxide as a dehydrating agent. These routes focus on removing water without introducing too many waste byproducts. I remember troubleshooting a reaction that kept stalling, only to find a leaky condenser letting in the very water we were trying to keep out. Alternative preparations involve reacting propionyl chloride with propionate salts, but cost and availability of raw materials push producers to stick with the acid-anhydride combo. Each method introduces its own quirks—byproducts, reaction times, and purification logistics all weigh in on the final price and purity.
Chemical Reactions and Real-World Modifications
Put propionic anhydride in a flask with alcohols, and esters form with brisk efficiency. Add it to amines, and you get amides—a staple step in medicinal chemistry and peptide synthesis. The substance also acts as a propionylating agent in the creation of fragrance molecules, pharmaceutical intermediates, and specialty plastics. I’ve witnessed many creative uses, such as protecting groups in multi-step organic syntheses. But one careless pipetting, and the fumes force a hasty exit—it’s unforgiving about ventilation. It doesn’t just stop at lab scale; chemical engineers build reactors with corrosion-resistant linings to withstand its bite.
Alternate Names and Industry Lingo
Chemists love a good shorthand. Ask around, and you’ll hear propionic anhydride called ‘propanoic anhydride’, ‘dipropionyl oxide’, or simply ‘PPA’. These aren’t just linguistic quirks—shipment records, regulatory filings, and safety documents need to match up, or supply chains grind to a halt. I recall a customs hang-up because someone used ‘dipropionyl oxide’ on a shipment manifest instead of the IUPAC-approved term. Mislabeling wastes time and sometimes forces inventory to sit, unresolved, in a bonded warehouse.
Working with Propionic Anhydride: Safety and Standards
Long sleeves, splash goggles, and chemical-resistant gloves all find regular use around propionic anhydride. People learn quickly not to get casual with the stuff. Proper fume extraction, emergency eyewash stations, and training drills anchor safe work habits. The Occupational Safety and Health Administration has exposure limits; persistent vapor can irritate respiratory passages, and spills eat through rubber and even some plastics. Disposing of leftovers usually means careful neutralization—diluting leftovers in a controlled way before joining them with the broader waste stream. The hard truth: familiarity doesn’t breed safety. Each mishap or near-miss only hardens respect for the chemical.
Propionic Anhydride Across Industries
Pharmaceutical manufacturers count on propionic anhydride for key intermediates. It builds blocks for anti-inflammatory drugs and antibiotics. In the fragrance trade, it shows up as both a reagent and an ingredient for certain smelly molecules. The chemical’s knack for building esters and amides turns it into a fixture in research reactors. People engineering cellulose plastics and modifying starches also draw from PPA stocks, tapping its reactivity to dial in product properties. I’ve seen it spec’d for synthesizing agricultural protective agents, though environmental rules continue to tighten due to the potential for misuse.
Modern Research and Innovation
Academic and private labs push the boundaries of what’s possible with propionic anhydride. Improved synthesis routes keep trickling in—methods designed to cut out hazardous byproducts or use renewable starting materials get the most attention. Analytical chemists plow into new detection and quantification tools, driven by environmental controls and workplace safety needs. The controlled substance regulations mean researchers sometimes have to justify and log their stocks with extra scrutiny. Safer derivatives and less aggressive analogs stay on the drawing board in many chemical R&D departments, but the classic anhydride remains the popular choice when reactivity counts.
What We Know About Toxicity
Toxicology studies paint a clear picture: target organs include skin, eyes, and lungs, with acute exposure producing irritant effects. Animal studies report that significant inhalation exposure causes respiratory distress, while ingestion or uncontrolled skin contact adds risk of burns or systemic toxicity. Chronic long-term exposure hasn’t been linked to carcinogenicity, but the immediate corrosive risk trumps most other concerns. Organizations like the National Institute for Occupational Safety and Health review new studies every few years, and workplace exposure limits occasionally get tighter as fresh data roll in. People working with propionic anhydride keep up with these findings both for personal welfare and to satisfy inspectors.
Where the Future Might Lead
Demand for propionic anhydride doesn’t look set to vanish. Tightening controls over synthetic drug manufacturing in some regions steer bulk sales into permitted, regulated industry sectors. Sustainability plays a bigger role, with process engineers under pressure to find greener, safer production methods. Labs value purity, consistency, and reliability; regulatory agencies want less impact at every step from plant to end-product. New research papers hint at bio-based feedstocks, innovative recycling techniques, and improved emission controls to make large-scale use less risky. Those who work with this anhydride know the dance between practicality and responsibility—it’s not just about making chemistry happen, but doing it without leaving a legacy of accidents or contamination for someone else to clean up.
What is Propionic Anhydride used for?
What It Does and Why People Care
Curiosity about chemicals can usually lead down two paths: the science behind them, and the impact they have on everyday life. Propionic anhydride floats beneath the radar for most, but it plays an underrated role in how some products are made. It acts as an important building block, shaping ingredients behind many familiar items. Sometimes, the story of what goes into a product gets lost, but learning the role of propionic anhydride shows how small things drive bigger changes.
The Backbone for Better Chemistry
In industry, propionic anhydride matters most for the things it helps create. This compound enters the scene during the making of specialty chemicals, like propionates—these show up in food preservatives, certain plastics, and pharmaceuticals. I remember first reading an ingredient label on bread as a teen and tripping over "calcium propionate." It keeps mold away, helping bread last longer without crumbling into stale paste. Without propionic anhydride, manufacturers face more hurdles, especially when trying to keep food fresh on store shelves.
Another place where it shapes outcomes is the pharmaceutical industry. Companies tap propionic anhydride for specific drugs, including painkillers and antibiotics, because it helps create reliable, stable formulations. Getting consistent quality in medicines isn’t a minor detail. Precision at the chemical stage can decide if a drug works the way it should.
The Hidden Side: Regulation and Risk
Whenever a chemical gets mass-produced, there’s a flip side. The same properties that make propionic anhydride valuable in legal manufacturing also draw attention from regulators. Some criminal groups target it to make illegal drugs, which is why authorities keep it tightly controlled.
Over the past decade, law enforcement agencies across the world have caught shipments intended for use in narcotics production. That's a detail most people miss, tucked away behind headlines about drug seizures. I have seen the effects of poorly monitored chemicals in the community—the cost shows up in emergency rooms, addiction clinics, and public spending on law enforcement. Strong regulations protect not only the supply chain for legitimate businesses but also the well-being of families who would otherwise deal with the consequences later.
Toward Safer, Smarter Practices
It feels easy to overlook mundane chemicals, but the truth shows up when things go wrong. Transparency in sourcing and processing, clear labeling, and robust oversight all keep the line between legitimate and illegal use. Companies investing in safety audits and government agencies expanding real-time tracking can stop misuse at the source. Educating staff in chemical plants about risks helps close loopholes, tightening the gaps that traffickers look for.
Manufacturers large and small stand to benefit from understanding what their supply chains touch. End-users—whether that’s consumers picking up a loaf of bread or patients filling a prescription—rely on these safeguards, often without knowing it. Ongoing research into chemical alternatives is picking up steam, too, offering hope that industry can lean on safer compounds for the same work. I’ve met scientists driven by the urge to find cleaner, less divertible substitutes. They don’t just focus on the bottom line; they focus on public good.
Why Paying Attention Matters
Ignoring the details around propionic anhydride exposes weak links in a chain that shouldn't break. Smart policy and tech solutions paired with old-fashioned vigilance in manufacturing can shape a future where this chemical continues to serve its practical uses without fueling black markets. The way we manage these quietly critical compounds can say a lot about the health of our industries and the priorities of our society.
Is Propionic Anhydride hazardous or toxic?
Looking Past the Lab Coat: Why Propionic Anhydride Matters
Propionic anhydride comes up in chemical supply circles more often than most people realize. It pops up in pharmaceutical manufacturing, crop protection, and even in the plastics world. Most people never handle it directly, but for those who do, knowing its risks isn’t optional. In my time covering science and health stories, I’ve learned that substances like this one demand respect because too many workers have paid the price for treating chemicals as harmless.
Toxicity and Risks: More Than a Lab Note
Propionic anhydride can trigger some nasty effects. Breathing in its vapors irritates your eyes, nose, and lung lining. Splash it on bare skin, and you might feel the burn almost immediately. Swallow a little—accidentally or otherwise—and stomach pain follows quickly. Labs report headaches, nausea, and even respiratory distress from overexposure. These aren’t just lab manual warnings—they’ve shown up in incident reports and emergency room stats too.
Manufacturers post strict warnings for good reason. Propionic anhydride reacts aggressively with moisture, which includes the water in your skin and lungs. Industrial accident files document injuries from careless handling, and not just in older, poorly ventilated spaces. This is one chemical where a fast response means less injury, and complacency isn’t just dangerous—it’s often expensive.
Environmental and Social Concerns
Propionic anhydride’s hazards extend beyond the site floor. Spills enter local waterways or soil and affect nearby communities. Reports from farming towns show crops wilting and aquatic life suffering after improper disposals or leaks. Regulators in the US and Europe track this chemical closely, and police monitor its movement because criminal organizations sometimes use it to manufacture illegal drugs.
There’s another layer to the discussion—workers in low-safeguard environments. It’s easy to take for granted the safety standards in wealthier countries, but some factories overseas cut corners. Workers in those places sometimes don’t even get gloves or masks. That sort of situation puts real faces behind the chemical’s sterile hazard lists.
Solutions That Make a Difference
Training stands at the center of most long-term solutions. No one wants to work in fear, but skipping safety refreshers because of cost risks far more—both in human terms and financial penalties. Automated spill detection tools bring another layer of protection, especially in places where staffing is stretched thin.
Regulatory oversight remains essential. Chemists and plant managers advocate for transparent record-keeping and spot inspections. Countries sharing intelligence clamp down on illegal diversion of propionic anhydride for controlled substance synthesis. This kind of cooperation keeps both workplaces and communities safer.
I’ve spoken with people who’ve been through chemical accidents. They’d rather see the industry err on the side of caution than wind up in the hospital, or see their friends in trouble. Respect the risks. Use the tools. Speak up about shortcuts. Propionic anhydride lasts in the headlines because its impact—when misused—gets personal in a hurry.
What are the safe storage and handling requirements for Propionic Anhydride?
Packing a Punch: What Works and What Risks Remain
Propionic anhydride shows up in all kinds of industrial labs and chemical plants. It's got value as a building block, but the risks stick around if the right safety steps fall through the cracks. Like many acylating agents, this chemical cuts deep with corrosivity—just a little bit on skin or eyes delivers pain and possible injury. Inhalation irritates the lungs badly, and stories from those working in chemical warehouses confirm that the fumes start up nose and throat discomfort almost instantly.
Anyone who has spent time in a storage facility or chemical distribution center knows the rules change as soon as propionic anhydride rolls through the back door. It gives off a strong smell, similar to vinegar but sharper. That’s the first warning sign. The second: even quick contact with moisture (including the air) unleashes fumes. In warm weather, pressure in tightly closed drums can rise, making leaks more likely.
Smart Practices for Storage
Some of the biggest risks show up before the containers even get opened. Anyone in charge of storing propionic anhydride makes sure the warehouse stays cool, dry, with solid ventilation. The drums—usually steel or high-density polyethylene—never go near water, acids, or strong oxidizers. Pallet racks and floors keep clean and dry, free from old spills or residue. Most places keep a chemical spill kit right by the storage area, with plenty of absorbent material, neutralizer, and eye wash bottles good and close.
Staff in these facilities confirm that smaller containers work much better for day-to-day use. Nobody wants to wrestle with a rusty, half-ton drum when a smaller, labeled bottle will do for a few liters at a time. Clear labels don’t just satisfy the paperwork—they warn new hires and old hands alike why this contents demand gloves, goggles, and respiratory masks.
Handling Precautions: More Than a Checklist
Chemical-resistant gloves (think butyl rubber or neoprene) make a concrete difference, since nitrile doesn’t always cut it. Splash goggles protect eyes; full face shields beat safety glasses if volume or splash risk runs high. Vent hoods and local exhaust fans capture lingering fumes, and everyone I’ve known who’s worked with anhydrides spends at least half the prep time double-checking that the vents run. Lab coats and closed shoes feel like common sense, but every veteran has a story about new staffers who needed a stern reminder after a close shave.
The consequences of getting this wrong go far. Propionic anhydride belongs on the DEA’s list of watched chemicals because of its history as a precursor for illicit drugs. Recordkeeping matters almost as much as physical safety—every bottle goes accounted for by weight and destination. Unsupervised access invites legal trouble fast, with law enforcement scrutiny adding to everyday hazards.
Solutions That Stick
What keeps facilities safe often comes down to culture. Training must happen often, not just at hiring. Managers encourage staff to speak up about odd smells, makeshift storage, or missing PPE. The right spill and fire plans get reviewed regularly, and drills help staff react without hesitation.
Inspections from safety teams don't only check paperwork. They shovel through clutter, test air for vapors, and demand clean, organized workspaces. Keeping up strong supplier relationships helps, too—getting fresh, sealed drums reduces the risk of leaky, aged containers.
Handling propionic anhydride builds a healthy respect for clear rules and teamwork. Veterans remind newcomers that confidence comes from repetition, not shortcuts. Working around this chemical means focusing on safety every single time, and not trusting luck to solve the problems smart prep and communication can prevent.
Where can I purchase Propionic Anhydride?
A Chemical With Strings Attached
People look up where to get propionic anhydride for plenty of reasons. Some folks in research or industry need it for legitimate work. But there’s a hard edge to this topic. This chemical sits in a group watched closely by regulators, and for good reason. Propionic anhydride can help make things the world doesn’t need more of—specifically, illegal drugs like fentanyl. This link to crime and health risk changed the way honest businesses and curious tinkerers can get near it.
Strict Laws and Safety Stand Tall
Governments do not take these risks lightly. The US Drug Enforcement Administration lists propionic anhydride as a List I chemical. That label means only certain authorized companies can legally sell or buy it, and the paperwork piles up fast. Even if you run a real lab, paperwork like end-user declarations and business verification come before anyone ships you a bottle. On top of this, state laws can throw in extra steps or bans.
Across the Atlantic, the European Union tracks and controls this substance under their own rules. Most other countries use similar stances—if you’re not a business with the right paperwork, forget about making an order. Gray market sellers and overseas stores advertising propionic anhydride almost always break local laws, and buying from them brings risks nobody wants. Seized shipments, fines, or prosecution can all follow an illegal purchase.
No Room for “Just Browsing”
In the chemical trade, background checks happen as a matter of course. Reputable suppliers—Sigma-Aldrich, Fisher Scientific—won’t look twice at private buyers. Orders line up with registered labs, pharmaceutical producers, or companies in plastics, dyes, or agrochemicals. If you lack a legitimate business history and clear explanation of use, these companies hit “cancel” fast.
Why This Matters
The stakes run deeper than bureaucracy. Propionic anhydride itself eats away at skin and eyes on contact, and vapors can choke you. Poor storage or handling causes leaks, injuries, or worse. Retailers know direct sales to the public would invite disaster, so strict policies protect both staff and the wider community. The same safety-first mindset shapes rules for ammonia, red phosphorus, and other watched chemicals.
Add the danger of illegal drug manufacture, and it’s clear why this is not a chemical found at corner stores or hobby shops. Some buyers with a chemistry passion have good intentions, but a few bad actors have spoiled it for everyone else.
Building Solutions Rather Than Shortcuts
Some folks go online hoping for loopholes or gray-market routes, but this only lands them in more trouble. If you work in a field that could use propionic anhydride—organic chemistry research, pharmaceuticals—start with clear communication and compliance. Build trust with approved chemical vendors, maintain up-to-date licenses, and run background checks regularly. If you’re curious about chemical synthesis but do not work for a registered lab, universities and open science programs help future professionals gain skills without crossing legal lines.
Asking about propionic anhydride’s sale means entering a complex world where public safety always comes first. The best bet: respect the red tape, work above board, and shape a culture that values safety every step of the way.
What precautions should be taken in case of a Propionic Anhydride spill or exposure?
Understanding the Risks
Propionic anhydride doesn’t appear in the news often, but people handling it up close know it holds real hazards. This chemical, found in the production of pharmaceuticals and plastics, releases fumes that bite at your eyes and lungs. A splash burns the skin, while breathing it in stings your throat. Over the years, I’ve met folks in labs and in transport jobs who’ve learned the hard way that one missed step in protocol puts them or their coworkers at risk.
With reports from the Centers for Disease Control and Prevention showing that exposure causes severe skin and respiratory irritation, these risks are no exaggeration. There’s nothing vague about the shortness of breath or severe eye pain someone feels if exposed unprotected. Fatalities from chemical exposure stand out in occupational safety statistics. Safety matters far beyond a checkbox on a training sheet.
Practical Steps for Prevention
Every time propionic anhydride hits a shop floor, warehouse, or laboratory, the first thing on my mind is ventilation. A tight space holds in vapors, so working outside or in a fume hood cuts down risk straight away. Mechanical ventilation systems are a must in enclosed areas. Fresh air isn’t just comforting—it helps keep exposure low and stops invisible vapors from gathering into a dangerous cloud.
Personal protective equipment stands as the shield between workers and harm. Goggles that fit flush, gloves resistant to chemicals, long sleeves, and an apron made of non-porous material do more than complete a lab uniform—they help stop burns and rashes. In places with a real risk of vapors, a properly-fitted respirator becomes nonnegotiable. The Occupational Safety and Health Administration (OSHA) sets specific PPE standards for corrosive chemicals, and skipping these steps comes with real-life consequences.
I always keep an emergency eyewash and shower station in plain sight, never stashed behind boxes. The stories I’ve heard from veteran chemists and warehouse workers boil down to seconds lost if someone scrambles to find the nearest wash station after a splash. Water must flush the affected area for at least 15 minutes. Every second counts.
Handling Spills without Panic
Let’s face it: accidents happen even with good planning. During a spill, patience and process matter most. Keep others away from the area and let those trained in hazardous materials response step in. I’ve watched emergency response teams use absorbent pads and inert materials to contain and neutralize the liquid—never just a mop and bucket job. They use chemical neutralizers specific to acids, usually asking for sodium bicarbonate or another recommended agent for the situation.
Disposal turns into a legal and safety process, following state and federal rules strictly. Never dump chemical waste down a drain or into regular trash. My experience with local waste management has shown me that professional hazardous waste handlers know the ropes and keep communities safe downstream.
Leaning on Experience, Not Luck
Each step in handling propionic anhydride boils down to preparation, respect for hazards, and sticking to what has been proven to keep people safe. Nobody wants their name in a safety report for the wrong reasons. Rooting these habits into daily routines and insisting on regular training with real-life spill drills builds a team that’s ready, not lucky. New employees should get hands-on practice, and seasoned folks can always use a refresher. Safety grows from these simple but firm details—never from hoping nothing will go wrong.
If leaders listen to those working with chemicals and act on feedback, the workplace becomes safer for everyone. Simple communication, up-to-date materials safety data sheets, and honest talk about mistakes from the past help prevent new ones. Handling propionic anhydride demands the same respect today that it did when I started out, and the lessons learned along the way keep people healthy.
| Names | |
| Preferred IUPAC name | Propanoic anhydride |
| Other names |
Propanoic anhydride Propionic acid anhydride Dipropionic anhydride NSC 9272 |
| Pronunciation | /ˌproʊ.piˈɒn.ɪk ænˈhaɪ.drɛd/ |
| Identifiers | |
| CAS Number | 123-62-6 |
| Beilstein Reference | 1041659 |
| ChEBI | CHEBI:51166 |
| ChEMBL | CHEMBL1432653 |
| ChemSpider | 13740 |
| DrugBank | DB14004 |
| ECHA InfoCard | 100.004.967 |
| EC Number | 204-641-1 |
| Gmelin Reference | 3729 |
| KEGG | C18681 |
| MeSH | D011364 |
| PubChem CID | 8053 |
| RTECS number | UF9625000 |
| UNII | LBO3Q9UV7D |
| UN number | UN2491 |
| Properties | |
| Chemical formula | C6H10O3 |
| Molar mass | 150.14 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent, irritating |
| Density | 0.989 g/mL at 25 °C |
| Solubility in water | Decomposes |
| log P | 0.9 |
| Vapor pressure | 0.4 mmHg (20°C) |
| Acidity (pKa) | 9.04 |
| Basicity (pKb) | pKb: 12.68 |
| Magnetic susceptibility (χ) | -44.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.394 |
| Viscosity | 1.08 cP (20 °C) |
| Dipole moment | 2.6 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 242.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -490.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1764.9 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H226, H314, H317, H332 |
| Precautionary statements | P234, P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P311, P310, P312, P321, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 60 °C |
| Autoignition temperature | 400 °C |
| Explosive limits | 3.0% - 10.0% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1,700 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 1408 mg/kg |
| NIOSH | RN3675002 |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Acetic anhydride Butyric anhydride Propionic acid Acetic acid Isobutyric anhydride |

