Propionic Acid: The Hidden Workhorse in Industry and Daily Life
The Road from Discovery to Broader Use
Chemistry can feel remote for most people, but the story of propionic acid reflects the way discoveries in dusty labs eventually shape everyday systems. Back in 1844, Johann Gottlieb discovered this colorless, pungent carboxylic acid during the redistillation of sugar. At the time, chemists were on a quest to unlock the grouping and logic of organic molecules, and propionic acid’s simple structure—just three carbon atoms—fit perfectly between acetic and butyric acids. Still, the world didn’t immediately rush to embrace it. Its value grew over decades as industries and scientists found that this little molecule, with its strong smell and antimicrobial punch, bridged gaps between food preservation, animal feed, plastics, and pharmaceuticals. Its history started quietly, but its legacy stands on sturdy ground now, with millions of tons transported around the globe.
Understanding What Propionic Acid Really Is
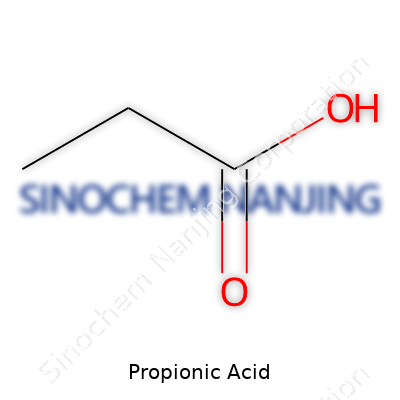
Propionic acid, often called propanoic acid, sticks out in the carboxylic acid family. It flows as a clear, corrosive liquid with a sharp, vinegar-like scent that tells you it’s potent. Chemically, the formula C2H5COOH hints at its three-carbons-plus-a-carboxyl-group backbone. While the smell can clear a small room, many industrial users seek it for precisely that strength. It dissolves well in water, ethanol, and ether, which means it mixes easily for a range of jobs. Its boiling point hovers around 141°C, and it starts to melt at -21°C, allowing straightforward storage in temperate climates.
Labeling Requirements and Specifications Call for Clarity
Regulations have shaped the way propionic acid travels through the supply chain. Labeling specifies its CAS number and purity, often above 99% for food and pharmaceutical applications. Labels identify hazard classes because leaking vapors or spilled acid can burn skin or lungs. A clear, bold warning about its corrosiveness stands on every drum; if handled in a lab or factory, training and protective equipment matter as much as the contents themselves. States and countries have their own rules, but the pattern stays the same: inform people before they encounter the acid so risk drops dramatically.
How Industry Actually Makes It
Factories don’t dream up propionic acid from thin air. The main method starts with hydrocarboxylation, a process combining ethylene and carbon monoxide using water and a catalyst, often nickel carbonyl. Sometimes chemical companies rely on fermenting specific bacteria with glucose or glycerol, especially when markets press for sustainable feedstocks. On the edge of new trends, some research groups use engineered microbes to improve yields or cut energy spending. But even the oldest pipeline-driven plants keep churning out most of the world’s supply. There’s rarely a shortage, though geopolitical swings sometimes shake logistics and prices for customers around the world.
Chemical Reactions Push Its Use Further
Propionic acid stands ready for a range of chemical reactions. Its main job is as a building block: turning into esters for artificial flavors and fragrances, anhydrides for plastics, or salts like calcium propionate for food preservation. The acid reacts with alcohols to make fruity-smelling esters that end up in everything from perfumes to bakery products. In other cases, it presses into action as an intermediate for herbicides, certain pharmaceuticals, or rubber auxiliaries. Skilled chemists have coaxed it toward modified molecules that show promise for greener agriculture or longer-lasting coatings.
Multiple Names, Same Powerful Molecule
Some call it propanoic acid, others stick to the classic “propionic acid.” Walk into a research lab and you’ll hear “ethylformic acid” sometimes, or see abbreviations like PA. Label-makers print the international identifier “CAS 79-09-4,” while European buyers might refer to it as E280—especially in bakery or dairy settings. Across these synonyms, the science remains the same, but the language shifts depending on the industry, making knowledge-sharing between sectors a challenge for those without a good translation guide.
Keeping Workers and Communities Safe
Workplaces that use propionic acid take safety as seriously as the acid takes its corrosive work. Direct skin contact burns; inhaled vapors irritate airways and eyes; spilled liquid damages surfaces and requires special cleaning. Regulatory agencies like OSHA in the United States or the European Chemicals Agency require clear ventilation, designated storage spaces, eye wash stations, and gloves or aprons where exposure risk runs high. Factory and warehouse staff walk through drills so that everyone knows how to contain a spill before it spreads. Attention to these standards not only keeps accidents rare, it protects community water systems from acid leaks during storms or transport accidents. For smaller businesses or farms using the acid as a preservative, the message stays the same: don’t ignore protective measures, even if quantities seem small.
How Propionic Acid Shapes Modern Industry
Hidden in bread, animal feed, and even sandwich wrappers, propionic acid touches more lives than most people imagine. Bakers use calcium propionate to halt mold growth in loaves stacked on grocery store shelves. Cattle feeders rely on it to keep silage from spoiling, stretching feed stocks between harvests and droughts. In chemical manufacturing, it holds its own as an intermediate for plastics and synthetic rubbers that form parts of tires, insulation, or packaging films. Artists and hobbyists may not know that the sharp, starchy scent in glue or solvent-based products owes its bite to propionic acid. Food safety departments list it in the “generally recognized as safe” category for almost all uses, at least at the concentrations found in consumer goods.
What Researchers Are Learning
Scientists across food, health, and materials science continue to pull propionic acid under the microscope. Microbiologists look at how propionic acid acts as both a natural and synthetic preservative, exploring ways to slow bacterial growth in food storage—an effort to cut down on waste and foodborne illness. In medicine, researchers dig into how naturally-produced propionic acid in the human gut links with inflammation, mental health, and immune function, sparking new interest in prebiotics and diet-driven wellness programs. Chemical engineers test greener production methods, swapping out fossil-derived feedstocks for agricultural waste streams or direct fermentation from food processing leftovers. Some studies argue that propionic acid can improve shelf-life for animal vaccines, while others look toward safer derivatives for use in organic farming.
Toxicity: Balancing Benefit and Risk
Propionic acid features a low acute toxicity profile, at least by industrial chemical standards. Small, naturally-occurring amounts show up in healthy human digestion. In agriculture or food preservation, the permitted doses sit far below levels causing harm. Problems rise in concentrated form: direct ingestion, inhalation, or skin exposure at industrial strengths can damage tissue, burn lungs, or poison animals and humans if not care is not observed. Long-term animal studies show hardly any cancer risk or lasting genetic damage at low concentrations, but some researchers remain focused on the impacts of chronic workplace exposure, especially in older factories without modern ventilation or handling gear. For now, as long as strictly enforced safety standards persist, risk for consumers looks negligible.
Where Propionic Acid Heads Next
Faced with rising food waste, shifting climate, and urban growth, industries search for ways to keep edible products safe longer without packing aisles with artificial chemicals. Propionic acid, especially from biogenic sources, stands to gain from this trend. Improved fermentation methods could trim the carbon footprint, turning agricultural byproducts into preservatives instead of landfill contributions. Plastic companies eye bio-based propionic acid as a way to ease off fossil fuels. Farms in dry regions increasingly rely on silage inoculants driven by this pure acid, keeping animal herds fed during tough seasons. With regulatory committees promoting evidence-based approvals and researchers fine-tuning risk profiles, its future looks more secure than ever. If sustainable sourcing, transparent labeling, and worker safety keep pace with technology, propionic acid’s understated role in fresh food, safe plastics, and resilient farming beds will only grow.
What is Propionic Acid used for?
What Propionic Acid Means in Daily Life
Walk through any bakery aisle, and you’re likely to find bread that tastes fresh long after it’s left the oven. That’s thanks, at least in part, to propionic acid. This simple molecule, which has a sharp, almost vinegar-like smell, gets added to foods because it keeps mold and bacteria from turning loaf into science experiment a week before payday. Food safety matters more than most people realize until they open a moldy sandwich at work.
Without preservatives like propionic acid, bread would spoil in a few days, and stores would waste massive amounts of food. According to the United States Department of Agriculture, Americans throw away about one-third of their bread because of spoilage. This hits family budgets and contributes to food waste—a problem bigger than an old loaf sitting in the cupboard.
A Tool for Farmers and Cattle Ranchers
Out on the farm, propionic acid shows up in places people don’t often see. Farmers spray it on animal feed and silage so mold won’t set in before winter. Just a few decades ago, spoiled silage meant sick cows and lower milk yields. A fresh pile of grain or hay, preserved with propionic acid, keeps cattle healthy and farm operations steady.
Many farmers worry about heavy use of preservatives, but research from the Food and Agriculture Organization confirms that small, regulated doses keep feed safe without harming livestock. Fewer animals risk illness, and farmers cut their losses during tough years.
Other Everyday Uses Beyond Food
In factories, propionic acid takes on a new job. It works as a building block for products like herbicides, plastics, and even some medicines. The idea sounds distant, but most households contain at least something made with plastic, and weeds will show up in gardens whether or not crops grow nearby.
Manufacturers rely on this acid for one simple reason: it helps prevent breakdown from microbes, so chemicals and plastics last longer. With more plastics comes more responsibility, though, so manufacturers need to use safe handling and proper storage to protect both workers and communities.
Potential Risks and Smarter Choices
Everything in life comes with trade-offs. The same propionic acid that keeps bread fresh can irritate skin or eyes if touched directly. Workers handling bulk supply wear gloves and eye protection for a reason. Also, some parents worry about feeding kids too many additives. While global health organizations—including the World Health Organization—agree that propionic acid sits well within safe limits as a food additive, it makes sense to pay attention to labels and enjoy more fresh foods alongside processed ones.
Scientists have kept a close eye on propionic acid for years. Large reviews of toxicology and patient reports support its safety in approved doses. Still, conversations about additives benefit from transparency and care. Support for more studies keeps the science honest and public trust higher.
Better Choices for the Future
Propionic acid supports farmers, bakers, and manufacturers every day. Real progress comes from looking out for safe ways to use these ingredients while limiting unnecessary exposure. Choosing whole foods where possible, making room for education in science classes, and checking product labels go a long way. With smart regulation and community awareness, everyone benefits from the balance between freshness, health, and responsible production.
Is Propionic Acid safe for human consumption?
The Role of Propionic Acid in Food
Bread, cheese, and a big chunk of processed foods rely on preservatives. Propionic acid fits right in with this crowd. It helps stop mold and some bacteria, especially in baked goods. Food companies like it because it keeps products looking and tasting fresh. I’ve looked through ingredient labels at the grocery store and seen it plenty of times, usually listed as E280 or “calcium propionate.”
Is Propionic Acid Safe?
It catches attention, because nobody wants to eat something harmful. The US Food and Drug Administration classifies propionic acid as “Generally Recognized as Safe.” The European Food Safety Authority landed on a similar position. Between animal studies and decades’ worth of people eating bread with these preservatives, researchers found that it passes through our body quickly. It breaks down to regular body compounds, mostly carbon dioxide and water.
A study published in the Journal of Food Science pointed out that even high doses in animal studies didn’t produce toxic effects. Most people eat nowhere close to those high doses. The estimated daily exposure stays below the Acceptable Daily Intake set by international health agencies.
Concerns and Controversies
Old stories about preservatives seem to come around every few years. Some worry about links to gut health or allergic reactions. Several articles pop up connecting additives and children’s behavior, but researchers ruled out a strong connection for propionic acid at the levels found in food. My own household deals with food allergies, so I dig into additive safety more than most. The bulk of credible evidence doesn’t warn off the average person from eating foods with this preservative.
What about sensitive groups? Some people do feel best when they skip processed foods — and sometimes, for people with preexisting gut conditions, any preservative can be irritating. If that’s you, listening to your body makes sense. Most of us don’t have to worry.
The Bigger Picture
Eating real, minimally processed food helps avoid many food additives, not just this one. It makes sense to keep asking, “Do I need this in my diet?” For folks interested in food safety, propionic acid doesn’t fall into the same league of risk as things like trans fats or heavy metals. It helps keep bread from turning green and fuzzy.
Instead of fighting over individual preservatives, focusing on eating a balanced diet with plenty of whole foods tends to work better. Plenty of traditional breads skip preservatives by baking fresh each day, but a lot of people want loaves that last longer. Preservatives like propionic acid make that possible.
Where Do We Go From Here?
Food safety depends on ongoing research. Regulators keep an eye out for new data. If scientists saw real harm, groups like the FDA or EFSA would take action. For now, staying informed and reading ingredient lists helps people make choices. If propionic acid or any food additive raises concerns, there are plenty of preservative-free options at farmers’ markets, bakeries, and health food stores. Knowledge gives us power over what ends up on our plates.
What industries commonly use Propionic Acid?
Food Preservation and Safety
Walk into any bakery or sandwich shop, and it’s likely you’re seeing propionic acid at work behind the scenes. Bread and other baked goods tend to mold quickly without some help, and this acid stands out for stopping unwanted fungal activity. The food industry relies on its ability to slow spoilage. Many families count on bread, tortillas, and cheese that keep for more than a day or two, especially with busy schedules and tight food budgets. Regulatory agencies like the FDA approve propionic acid’s use, and global demand hasn’t wavered as people look for longer shelf lives and reduced waste at home and in stores.
Animal Feed Protection
Propionic acid supports farmers, especially those who manage cattle and dairy operations. Grain silage and livestock feed often get too moist, which creates a breeding ground for mold and bacteria. Without some intervention, entire stores of feed could end up in the compost heap. Adding propionic acid to animal feed helps prevent this waste, cuts back on feed-borne illnesses, and protects the animals’ health. Healthy herds mean better productivity—less wasted input and steadier food production over time. Many ranchers choose to pay a bit more for protected feed stored over the winter, rather than risk the costs of mold-related sickness and nutrient loss.
Pharmaceuticals: A Building Block
Medicine makers, both large and small, use propionic acid to create certain active ingredients. It acts as a raw material in the manufacture of drugs for pain relief and inflammation, such as ibuprofen. Chemical processes inside manufacturing plants rely on simple compounds like this one to build more complex molecules. High purity and tight controls around contamination are essential, and many companies dedicate entire teams to monitoring quality along each step of production.
Manufacturing and Plastics
Propionic acid also finds its way into the chemical industry as a starting material for plasticizers, cellulose-based plastics, herbicides, and even artificial flavors. Industrial chemists value its versatility. For example, propionic acid plays a part in producing propionates, used in solvents and coatings that help paints and finishes stand up to daily wear and tear. Beyond coatings, some plastic products in homes and vehicles get their durability thanks to intermediate chemicals made from this acid.
Wider Industrial Uses
Outside the fields and bakeries, propionic acid supports rubber production and specialty lubricants required for machines that run non-stop. It serves a function in selecting specialty solvents for use in cleaning and degreasing, especially in settings that need to balance cleaning power with worker safety. Even niche uses, such as in the making of esters for perfume production, lean on the predictable reactions possible with propionic acid.
Room for Safer Alternatives
No chemical comes without questions. Some critics worry about residues or long-term exposure, especially in food and the environment. Broader transparency in sourcing and processing, as well as more research into green chemistry, could nudge these industries to create even safer options. Businesses that invest in tracking how much and how often they use such preservatives build trust with customers and regulators.
Takeaway for Everyday Life
Propionic acid quietly keeps food fresh, feed safe, and manufacturing efficient. It’s often overlooked, but for many industries—all the way from the farm to the pharmacy—its good track record and adaptability keep people coming back to it, even as technology moves ahead.
What are the storage and handling requirements for Propionic Acid?
Why Storage and Handling Matter
Propionic acid holds value in many industries, from preserving animal feed to acting as a food additive and playing a role in manufacturing. It packs a punch with its distinct, pungent odor and corrosive nature. People often overlook how easily strong chemicals like this can cause harm without proper respect. Too many accidents trace back to skipped steps or underestimating the risks. I once toured a feed mill where an open Propionic acid drum left fumes in the air. One worker came away with a burned arm and permanent scarring. That’s not a risk worth taking.
Right Conditions for Storage
Nothing about Propionic acid makes careless storage safe. Tanks or containers built with stainless steel or high-density polyethylene keep the acid from eating through. I’ve seen containers labeled “chemical-resistant” crumble over time when cheap plastic replaced the right material. Leaks mean trouble fast. Those working with this acid avoid storing near heat sources or in direct sunlight, since both will boost pressure inside containers and raise the chance of rupture. Ventilation matters just as much. A poorly ventilated shed traps fumes, making it uncomfortable at best, dangerous at worst.
Temperature control deserves real attention. Propionic acid keeps best in a cool, dry spot. Humid environments lead to rising pressure in sealed drums, sometimes causing swelling or even bursts. Spills in a moisture-rich room release vapor clouds, which put workers at risk for burns and breathing problems. Warehouses with stable climate control lower these dangers, so I always recommend spot checks—no matter how new the building seems.
Protecting Workers and the Community
Respiratory masks, safety goggles, gloves, and long sleeves save skin and lungs from lasting problems. Direct acid contact delivers both pain and permanent damage faster than many realize. My neighbor, who managed a dairy operation, always kept an eyewash station close by and made it routine for staff to check their gear. That planning paid off more than once when spills left workers scrambling.
Warehouse managers do well by training every new staff member on spill response instead of only relying on a “safety officer.” Good habits build a safety-minded culture that protects families, too—any incident inside can mean danger outside if acid vapors or liquid escape a facility.
Fire and Spill Response
Propionic acid burns easily, so open flames, smoking, or sparks near storage areas are bad news. Carbon dioxide or dry chemical extinguishers offer a reliable line of defense if a fire starts, but water can spread acid further. Local emergency plans serve well when written with fire departments in mind. Early notification limits the spread of harm, both chemical and physical.
Careful spill cleanup uses nonreactive absorbents, not sawdust or regular sand. Neutralizing agents, like sodium bicarbonate, help minimize lingering fumes without producing even more heat. Waste disposal rules always come into play here. Rushing to toss neutralized residue in standard dumpsters threatens both site staff and nearby communities. Licensed hazardous waste handlers know the drill.
Looking Ahead: Simple Actions Make a Difference
Propionic acid doesn’t require new technology or expensive solutions to store safely. Ordinary awareness, solid protective gear, and clear communication keep injuries low. Regulatory agencies offer regular updates and guidance. Reviewing protocols once a year and after every small incident turns mistakes into better habits. That’s where safety becomes second nature.
What concentrations and packaging sizes are available for Propionic Acid?
Real-World Uses Meet Real-World Packaging
My first experience with propionic acid involved a food preservation project. I remember standing in a warehouse, staring at steel drums labeled “Propionic Acid 99%.” It occurred to me that the way propionic acid gets packaged and its concentration could easily decide if it’s heading for a feed mill, a chemical plant, or even a bakery. Rather than one-size-fits-all, distribution matches practical, on-the-job use.
Common Concentrations That Matter on the Ground
Industrial players and labs both stick to one core concentration: nearly pure, 99% propionic acid. This purity handles diverse jobs, from fungal control in stored grain to intermediate chemical synthesis. Pharmaceuticals or food often require similar grades—sometimes slightly diluted for easier handling, but 99% covers most bases. Small differences exist; for instance, some importers offer 90% or 96% for specific applications that don’t require absolute strength, yet in most cases, suppliers default to 99% for assurance and consistency.
If you work in food safety, animal nutrition, or chemical manufacturing, the 99% label probably feels familiar. For people who want diluted solutions, like farmers spraying silage, suppliers often sell diluted blends—often between 10% and 30%. The difference isn’t just academic; lower concentrations reduce handling risks and improve safety when direct application is required. Labs needing precise measurements might even go for ampoules or pre-mixed solutions, although these are rare compared to bulk shipments.
Packaging Sizes: Bulk, Mid-Range, and Small Quantity
My background in manufacturing gave me plenty of reasons to respect container choice. Most of the world’s propionic acid moves in 200-liter drums or 1000-liter IBC totes. These fairly standardized sizes work for bulk handlers. You just wheel them onto a pellet, pump what you need, and keep the workflow moving. Places like feed mills, bakeries, or chemical processors won’t manage without these industrial packages.
Smaller businesses, especially ones blending additives on-site or running pilot production, lean toward 25-liter jerricans. These pack sizes are manageable for limited staff in tight spaces, or for people needing to split inventory into multiple workstations. In rare cases, a supplier will offer propionic acid in 5-liter, 10-liter, or gallon containers—usually requested by specialty stores catering to craft food-makers or laboratories. I’ve even seen amber glass bottles for analytical standards in lab catalogs but these are niche and more costly per liter.
Packing It All Together: Safety and Regulation
Packing decisions are not just about convenience. Propionic acid can irritate skin and eyes, and those handling bulk shipments need protective gear and adequate ventilation. Safety sheets and local regulation steer how businesses and transporters select container types. For example, UN-approved high-density polyethylene drums resist corrosion and survive rough shipping.
As supply chains grow more transparent, traceability matters. Reputable suppliers document every batch, detailing concentration, packaging, and handling protocols. This helps companies meet food or feed safety regulations and handle product recalls quickly, if needed. It also serves as a check against counterfeit chemicals—still a genuine risk in global trade.
Smarter Choices for Buyers and Users
In practice, the most important thing isn’t just what propionic acid is used for, but how efficiently it gets from the supplier’s loading bay to its final purpose. Smaller shops often struggle to track down single 25-liter cans. Large operations want consistency, predictability, and confidence the product shows up exactly as ordered. Getting a clear answer on offered concentrations and packaging is essential, not just a fine detail. For anyone procuring chemicals, asking about safety data sheets, packaging specs, and supplier reputation makes sure the right product lands at the right time with minimal risk.
Whether the need is a thousand-liter tote or a laboratory sample bottle, concentration and packaging choices shape cost, safety, and practical daily use far more than many realize.
| Names | |
| Preferred IUPAC name | Propanoic acid |
| Other names |
Propanoic acid Ethylformic acid Methylacetic acid |
| Pronunciation | /prəˈpɪɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 79-09-4 |
| Beilstein Reference | 1105053 |
| ChEBI | CHEBI:30779 |
| ChEMBL | CHEMBL521 |
| ChemSpider | ChemSpider: 569 |
| DrugBank | DB00163 |
| ECHA InfoCard | 03e5c00b-8a0c-4eb2-b4b4-7ba55c1e9b46 |
| EC Number | 200-835-2 |
| Gmelin Reference | 657 |
| KEGG | C00163 |
| MeSH | D019311 |
| PubChem CID | 1032 |
| RTECS number | UE5950000 |
| UNII | XF06X38V1O |
| UN number | UN1844 |
| Properties | |
| Chemical formula | C3H6O2 |
| Molar mass | 74.08 g/mol |
| Appearance | Colorless liquid with a pungent, unpleasant odor. |
| Odor | Pungent |
| Density | 0.993 g/cm³ |
| Solubility in water | Miscible |
| log P | 0.33 |
| Vapor pressure | 3.99 mmHg (at 25 °C) |
| Acidity (pKa) | 4.87 |
| Basicity (pKb) | pKb = 11.33 |
| Magnetic susceptibility (χ) | -34.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.386 |
| Viscosity | 1.2 mPa·s (25 °C) |
| Dipole moment | 1.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 159.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -425.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –1536.0 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A07AA07 |
| Hazards | |
| GHS labelling | **GHS02, GHS05, GHS07** |
| Pictograms | GHS02, GHS05 |
| Signal word | Danger |
| Hazard statements | H226, H314 |
| Precautionary statements | P210, P260, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P311, P312, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-Acid |
| Flash point | 53 °C |
| Autoignition temperature | 455 °C |
| Explosive limits | 2.1–12.1% |
| Lethal dose or concentration | LD50 oral rat 2600 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2600 mg/kg (rat, oral) |
| NIOSH | WN5600000 |
| PEL (Permissible) | PEL: 10 ppm |
| REL (Recommended) | 750 mg/m³ |
| IDLH (Immediate danger) | 1500 ppm |
| Related compounds | |
| Related compounds |
Acetic acid Butyric acid Valeric acid Isobutyric acid Propionyl chloride Propionitrile Propanal |

