What Potassium Pivalate Says About Chemical Progress and Practical Science
Historical Development
Potassium pivalate didn’t rise out of a vacuum. Its history mirrors the larger story of modern organic chemistry—where practicality steers the ship more than theory. The birth of this compound goes back to the mid-20th century, a period marked by exhaustive exploration of carboxylate salts and their potential in synthesis. Researchers first turned their gaze to pivalic acid derivatives as they hunted for reagents with both shielding abilities and improved solubility. That particular time in chemistry was about breaking new ground, and potassium pivalate’s ability to prop up reactions as a stable, easily handled salt gave the field fresh options. This wasn’t about glamour or headlines but about solving day-to-day problems in the lab: finding ways to coax a stubborn reaction forward or keep byproducts at bay. The routine, almost humble, development drew little public attention, yet shaped the toolkit that academic and industrial chemists still rely on.
Product Overview
Potassium pivalate comes across as an understated performer—a white, crystalline solid that rarely takes center stage. Its main value is not in being flashy but in doing its job without fuss or drama. The compound showcases a formula of C5H9KO2, a simple marriage of pivalic acid and potassium. Some see it as just a supporting player, but anyone who’s mixed it into a flask knows how it changes the course of a reaction: it dissolves in water with minimum protest, stands up to heat, and won’t pick needless fights with other functional groups. Industry veterans keep it around for its reliability. Whether in a small university research group or on the floor of a bustling production plant, its general dependability keeps projects on schedule and budgets under control.
Physical and Chemical Properties
Potassium pivalate proves the point that not every chemical needs to be hazardous or exotic to make a difference. It appears as a fine, odorless powder, carrying a slightly sweet note thanks to the pivaloyl origin. Its melting point rests safely above room temperature, avoiding the issues of liquid handling or atmospheric loss. In practice, its high solubility in water and many polar organic solvents keeps processes straightforward and scale-up friendly. Its chemical footprint is unassuming: a carboxylate salt, wide open to straight-chain and branched molecule synthesis. The tert-butyl group at its core fends off unwanted transformations, and in that way, potassium pivalate gives chemists room to breathe. Time and again, it avoids side-reactions that often plague unshielded carboxylates.
Technical Specifications and Labeling
People forget how crucial clear labeling and reliable specifications are until the day something goes wrong. For potassium pivalate, consistent appearance and content matter more than glossy brochures. You look for a uniform white powder, almost chalky to the touch. Purity runs above 98 percent for most research and manufacturing needs. Any residual water content, frequently listed under loss on drying, stays under tight control—excess moisture turns even the best batch unpredictable. Contaminants such as other potassium salts or by-products throw off reaction yields and force reruns, wasting both time and money. Experts pay close attention to what’s written on the drum as well as what’s left unsaid; slight deviations from spec spell bigger headaches downstream.
Preparation Method
Making potassium pivalate is a lesson in straightforward chemistry and controlled conditions. The process usually pairs pivalic acid with potassium hydroxide or carbonate, often in a simple aqueous or alcoholic medium. The reaction moves quickly, with carbon dioxide bubbling off when carbonate enters the picture. There’s something almost elegant in the simplicity: combine acid and base, collect the white solid, wash, and dry. Industry prep methods keep environmental concerns in mind, capturing released gases and recycling water whenever possible. Small labs might whip up a beakerful in an hour, while commercial producers scale up to reactor systems that churn out kilograms per batch, always keeping yields and purity in focus. Long-term users know that the time invested in a careful, controlled reaction pays off in smooth performance later.
Chemical Reactions and Modifications
Many of potassium pivalate’s strengths show up in the realm of organic synthesis. Experienced chemists reach for it during esterifications, as a base or coupling reagent, appreciating how the bulky tert-butyl group keeps unwanted reactions in check. In transition metal catalysis, the compound stands up well under both heat and pressure, opening doors to cross-coupling reactions previously blocked by more reactive, less tolerant salts. Friends in pharmaceutical synthesis describe it as a peacekeeper: reactive enough to do its job, inert enough not to cause trouble. Modifications start with the tert-butyl group, which blocks nucleophilic attack and protects sensitive moieties during multi-step syntheses. This resilience under harsh reaction conditions often proves its value, cutting out steps and eliminating the need for excessive purification.
Synonyms and Product Names
People in the chemical business know language can twist and turn, and potassium pivalate picks up a few aliases along the way. You might see it listed as potassium trimethylacetate or trimethylacetic acid potassium salt—names that all reflect its structural roots in the easily recognizable pivalic acid skeleton. Some trade catalogs shave down the details to just “K-Piv” or “pot. pivalate,” especially in informal lab settings. Awareness of alternate names matters. Miss a synonym on a materials request, and you’ll have weeks of delay and confused communication. Accurate, up-to-date cataloging bridges these language gaps and keeps projects moving.
Safety and Operational Standards
Anyone handling chemicals long enough learns that safety starts before the first scoop or pour. Potassium pivalate offers a margin of comfort compared to more hazardous salts, but nobody benefits from careless habits. Good ventilation, gloves, and tight-sealing containers help avoid accidental inhalation or moisture uptake. The powder’s tendency to cake and clump if exposed to humid air means regular lab practice calls for quick transfers and diligent sealing. Eye and skin contact merits the same caution you’d give to any fine particulate—immediate washing and disciplined habits prevent slip-ups that could lead to irritation. Reputable operations keep storage away from strong acids, oxidizers, or sources of ignition, erring on the side of order. Waste management rarely challenges anyone—routine disposal follows established pathways for non-hazardous organic salts.
Application Area
Potassium pivalate doesn’t headline many product brochures, but its fingerprints show up everywhere. The pharmaceutical industry values its ability to support the synthesis of active pharmaceutical ingredients, particularly where selectivity and clean separation matter most. Chemists tackling new polymers, or fine-tuning catalysts, recognize its stabilizing abilities, helping to build robust manufacturing pathways. It makes life easier in academic research settings too, letting teams focus on molecular innovation rather than wrangling dirty side-products. Its gentle handling encourages broader adoption in educational labs, serving as a bridge between theory-heavy lessons and hands-on experimentation. You don’t have to look hard to find someone in industrial fine chemicals nodding in recognition at the mention of potassium pivalate—it quietly anchors many behind-the-scenes processes that shape everyday goods.
Research and Development
Chemists are always hunting for smarter, faster, greener workflows. Potassium pivalate has earned steady attention in journals reporting new catalytic cycles and step-saving organic transformations. After years working with various carboxylate salts, many researchers turn back to potassium pivalate for its resilience and selectively bulk. Groups developing new metal-catalyzed cross-coupling protocols highlight how it withstands diverse metals and ligands, helping open routes that old salts couldn’t support. As research shifts towards sustainability, teams are dissecting its ability to operate in aqueous or mixed media without pushing by-products or lingering residues. In my own collaborations, switching to potassium pivalate from less robust salts often improved batch-to-batch reproducibility, especially in temperature-sensitive reactions. The research future circles around cleaner synthesis, waste reduction, and closer collaboration between academic and process chemists, all areas where potassium pivalate’s record inspires quiet confidence.
Toxicity Research
Talk about toxicity and the conversation shifts towards prevention and common sense rather than fear mongering. Animal studies and industrial hygiene data indicate potassium pivalate falls into a relatively low-risk bracket for both acute and chronic exposure, particularly when standard handling protocols prevail. Long-term studies don’t flag major health impacts at occupational exposure levels. That said, no one wants powder in their eyes or clouding up a workspace, so modern labs and plants enforce routine best practices: dust control, ventilation, and prompt clean-up. As part of continuous improvement, researchers still check for subtle bioaccumulation or breakdown product concerns, keeping up with new analytical techniques and regulatory updates. Thoughtful operators pay just as much attention to these findings as to up-front yields or product stability.
Future Prospects
Looking down the road, potassium pivalate seems unlikely to fade into obscurity. A growing number of synthetic challenges involve complex, sensitive molecules that demand robust supporting reagents. I see continued use in designing greener processes, from water-based synthesis to energy-efficient batch reactions. Automation and flow chemistry both benefit from its stability and predictability, making it a logical choice as new reactors and protocols emerge. As process optimization projects highlight environmental impact and resource management, potassium pivalate’s easy recyclability and minimal hazard rating become even more attractive. The next generations of chemists—those who blend practical skills with digital, data-driven design—stand to gain from building on experiences rooted in simple, effective compounds. Potassium pivalate’s story isn’t flashy, but its steady presence keeps science moving ahead in labs big and small.
What is Potassium Pivalate used for?
Getting to Know Potassium Pivalate
Potassium pivalate sits in a strange spot. You don’t hear politicians talking about it, and nobody’s starting heated debates at dinner parties about it. Yet, in the labs and process rooms where science shapes our medicines, potassium pivalate pulls plenty of weight. Here’s a chemical that, to most people, looks like another obscure powder. To researchers in pharmaceuticals, it turns into something much more useful.
Key Uses in Pharmaceuticals
Potassium pivalate takes on an unflashy but critical role in drug synthesis. It isn’t medicine, but chemists turn to it as a building block or a “reagent.” Think of it like one of those underappreciated kitchen tools that rarely gets a headline, but can make or break a recipe. Chemists use potassium pivalate to help with “esterification”—a process that links molecules together in a way that keeps them strong and stable.
I remember watching a team at a pharmaceutical plant rely on potassium pivalate while preparing a batch of antibiotics. The compound served as a smart buffer to keep things in the right chemical state—almost like a referee making sure both sides play fair during the reaction. Without it, the whole process could veer off course, leading to weaker or even unsafe results.
Role in Organic Synthesis
It doesn’t stop in the world of medicine. Potassium pivalate helps chemists build molecules in research labs, often during the preparation of “pivalate esters.” These esters might sound niche, but they show up when scientists want to protect sensitive parts of molecules from reacting too early. This makes the compound a trusted sidekick in synthetic chemistry, where every little tweak can lead to a whole new class of materials or therapies.
Colleagues often find potassium pivalate reliable because it works well at room temperature, and it doesn’t leave behind sticky messes that gum up sensitive equipment. I talked with a chemist who valued its predictability—few surprises, low risk of wild byproducts, and reactions clean up pretty easily.
Food Additive Talk—A Word of Caution
You may come across references to potassium pivalate in the world of food and preservatives. That’s not where most of the action happens, thankfully. Safety data on potassium pivalate for food use stays sparse, and there’s no widespread approval as an additive by big agencies like the FDA or EFSA. Whenever conversations wade into “should we use this in food,” experience pushes me to lean on peer-reviewed safety profiles and regulatory green lights. There’s just too much at stake to gamble on chemicals that haven’t been fully vetted for human consumption.
Challenges and Solutions
If you look at lessons learned in the lab, one sticky point with potassium pivalate comes during disposal. Being potassium-based, disposal down drains can harm aquatic environments if not supervised with care. Labs tackle this with closed-system waste processes. Proper training and oversight keep things above board and the environment protected.
Sourcing can also pose issues, especially in regions where supply chains still lag. In my own experience, planning ahead and building relationships with reputable chemical suppliers cuts down on the risk of delays or subpar batches.
Why It Matters
Potassium pivalate stands as one of those behind-the-scenes players making modern chemistry safer and more reliable. Knowing where it fits—and where it doesn’t—helps chemists, researchers, and production leads trust what winds up in our medicine cabinets. Each step in the supply chain, from careful selection of chemicals like this one, to environmentally-sound disposal, shapes outcomes for real people. That’s a responsibility any industry can’t afford to ignore.
What is the chemical formula of Potassium Pivalate?
Potassium Pivalate: More Than Just a Formula
Potassium pivalate doesn’t usually make headlines, yet it finds its way into serious research, pharma, and industrial work. Folks in a lab, or those working with certain organic reactions, see real value in chemicals like this, whether purifying new compounds or catalyzing specific reactions. Getting to know its structure makes using it a lot easier and safer for everyone involved.
Getting Down to the Chemistry
Potassium pivalate carries the chemical formula C5H9KO2. This is the potassium salt of pivalic acid. Sometimes you’ll spot it written as potassium trimethylacetate. Its backbone centers on a three-pronged cluster of methyl groups huddled around a central carbon. That cluster isn’t just for show. It creates what chemists call a “steric barrier,” making the molecule much less reactive than many other carboxylate salts.
It comes together like this: pivalic acid contributes five carbons, nine hydrogens, and two oxygens. Once potassium takes the place of the acid’s hydrogen atom in the carboxyl group, you get a stable salt. Those extra methyl groups make it chunky, as molecules go, helping it resist unwanted side reactions. I remember working with pivalate salts in a university synthesis project. Their bulkiness actually saved us from product breakdown, more than once.
Real-World Impact
Many who work on developing active pharmaceutical ingredients reach for potassium pivalate. The compound can help control the reactivity of other chemicals so that reactions don’t get out of hand. With industrial-scale batches, that stability translates to fewer side products and higher yields—which keeps costs down and quality up. Plus, the potassium ion offers decent water solubility, so dissolving this compound in standard lab solvents takes no special effort. That practical solubility allows better mixing and more predictable outcomes when scaling up processes.
Below the surface, every chemical used in manufacturing needs oversight for both safety and environmental reasons. A salt like potassium pivalate rates as low-to-moderate risk, mainly because both potassium and pivalic acid have well-studied effects on living systems. Still, high-purity chemicals should stay stored in dry, airtight conditions. Even small amounts of moisture can lead to clumping or breakdown, which in turn can mess with reaction consistency. In my experience, failing to seal up containers after a long day of labwork often meant tossing out whole batches the following week. No one likes losing money or time over something so preventable.
Solutions to Common Issues
One hurdle for anyone new to handling potassium pivalate is waste management. Even though this salt seems fairly benign, best practice means collecting and neutralizing residues rather than sending them down the drain. Training new entrants and experienced personnel alike on these routines keeps operations both safe and environmentally sound.
Quality practice also means sourcing potassium pivalate from suppliers who can trace their batches and produce (or provide) certificates of analysis. That way, labs and manufacturing sites sidestep contamination headaches. Using poorly characterized chemicals brings the risk of false results, wasted effort, and even fines if a regulatory body comes knocking. For those working in regulated industries like pharmaceuticals, those headaches turn into major compliance risks.
In the end, knowing the formula for potassium pivalate and what that structure means in practice lets chemists, engineers, and safety officers put both knowledge and respect to work, every day.
How should Potassium Pivalate be stored?
Understanding the Stakes with Lab Chemicals
Potassium pivalate doesn’t carry a famous name, but it shows up all over chemistry labs, especially where precise organic syntheses happen. Some view chemicals as bottles on a shelf, yet that attitude invites accidents and wasted resources. There’s a reason every safety sheet includes storage instructions—the wrong move can turn a safe environment into a risky one. When I started working in a research lab, it took only one mislabeled jar spilling its fine white powder over a damp benchtop for everyone nearby to scramble. Cleaning up wasn’t just about sweeping the mess. We had to assess air quality and make sure nobody inhaled the dust. Mistakes stick in the mind, and storage habits tighten up after you’ve seen one go wrong.
Choosing the Right Spot for Potassium Pivalate
Potassium pivalate draws moisture from the air easily. Let it sit out with the cap loose or in a humid storeroom, and you open yourself to clumping, contamination, and poor reaction results. I always put it in tightly sealed amber glass bottles, keeping it away from sunlight, high heat, and any busy workspace. Most lab veterans know that dry and cool means a room close to 20°C, free from HVAC vents. Silica gel packs inside the storage cabinet help keep everything drier. Once, our team had to toss out stock because it absorbed water from the air and clumped, ruining months of planning. It’s not just about tidiness—this is about research budgets and safety.
Labeling Isn’t Optional
On tough days, it feels tempting to skip a step, but failing to label storage containers wrecks inventory control. Without knowing exactly what’s in a jar or when it was opened, even experienced chemists struggle to maintain discipline in the lab. I’ve seen labels fade or fall off, leaving behind mystery powders. Clear, permanent labeling solves half the risk before it arrives. Names, concentration, date of receipt, and hazard info: that’s what I always include, and it’s saved us from confusion when multiple teams share the same chemical library.
Minimizing Cross-Contamination
In any shared lab, powders migrate with surprising ease. A single scoop left unwashed, or a cap left ajar during experiments, can let other chemicals creep in. This turns reliable potassium pivalate into a source of failed reactions. Setting up a bench routine helps: fresh spatulas, sealed containers, and a policy to close lids as soon as you’re done. These small habits build real consistency and stop cross-contamination before it ruins more expensive work. Nobody wants to rerun a reaction just because of careless handling at the shelf.
Adopting Best Practices as Daily Routine
Caring about storage isn’t about rules for rules’ sake. It’s about safeguarding people, the science, and every dollar spent on reagents. Keep potassium pivalate tightly sealed and in a dry, cool, and dark place. Build labeling into your routine. Wash the tools, and never assume the next person knows what you know. Problems start small: a little clumping here, a faded label there. Address these head on, and your lab runs smoother—there’s more trust, fewer accidents, and less wasted effort.
Is Potassium Pivalate hazardous or toxic?
The Basics of Potassium Pivalate
Potassium pivalate is a potassium salt derived from pivalic acid. It pops up most in research labs and chemical syntheses, not in everyday products or grocery shelves. Its chemical structure puts it on lists of special-use chemicals, especially in select pharmaceutical or organic chemistry processes. Safety always comes up as a concern, especially for people handling it in labs, so accurate information means a lot to scientists, technicians, and others who might interact with it, even if only by accident.
Is Potassium Pivalate Toxic?
Toxicity depends on several things—how much enters the body, how long the exposure lasts, the route it takes, and the person's own sensitivity. Scientific literature on potassium pivalate’s toxicity remains limited compared to well-known chemicals. Here’s what can be pulled together from what experts and sources say: animal studies and predictive tools point to low acute toxicity. A person or animal would have to consume a rather hefty dose before experiencing real danger. So, compared with notorious chemicals or strong pesticides, potassium pivalate sits on the milder end.
Even so, this doesn’t give license to ignore safe handling. Dust or powder forms, once airborne, can irritate the skin, eyes, or respiratory passages. Those who have ever worked with chemical powders know fine particles can linger and find their way into the most surprising places.
Environmental and Health Considerations
Looking at the environment, potassium ions exist all throughout soil and water with no issue. Pivalate, on the other hand, breaks down pretty steadily in nature, so this compound likely poses minimal long-term environmental threat, based on its chemical cousins’ behavior. Still, spilling a large quantity near a waterway or storm drain creates an unpredictable scenario. No one wants a substance with an uncertain toxicity profile washing downstream. That's a lesson many research labs and factories learn the hard way, after a bit of complacency trips up the safety record.
Why Chemical Safety Still Matters
Having handled chemical reagents behind a fume cupboard, complacency can sneak in if a substance isn’t flagged as “dangerous” in bold red letters. Potassium pivalate often doesn’t carry major hazard pictograms in material safety data sheets, but that doesn’t mean gloves, goggles, and a dust mask can be skipped. Even benign-sounding chemicals can become hazardous once mixed, heated, or handled in large volumes. Lab stories are full of minor chemical burns and wheezing from forgotten dust just because someone thought something “wasn’t that bad.”
One important idea that pops up in lab safety training: treat every chemical like it carries risk until proven otherwise through careful investigation. This is about more than obeying rules. Chemical exposure can add up or surprise you in untested situations. The best labs keep fresh safety sheets and make sure training covers not just the big scary chemicals but the unfamiliar ones too.
Moving Toward Safer Labs
Solutions exist for managing risks, even for lesser-known chemicals. Basic protective equipment forms the bedrock. Ventilation and spill kits matter just as much. Labs doing risk assessments every few months do much better in catching problems before they happen. Asking questions and sharing information becomes even more important when dealing with chemicals that don’t have long sectioned Wikipedia pages or buzzy media coverage.
It helps to stay connected with trustworthy sources: peer-reviewed research, current safety data from suppliers, local regulatory guides. Open discussion between team members or workers keeps everyone sharper than just reading a manual once a year. Modern science moves fast, but chemical safety is built on steady habits and a willingness to double-check.
What are the physical properties of Potassium Pivalate?
What You See, What You Get
Potassium pivalate doesn't draw much attention sitting on a lab shelf. Usually, you find it as a white, fine powder or sometimes in a granular form. It doesn’t emit any unusual smell, so the usual “sniff test” chemists sometimes try on organics won’t give you clues here. Light bounces off it in a dull way, meaning you won’t spot any sparkle like you would with sodium chloride crystals. This matters if you're handling it in bulk, since dust management becomes an issue—too much fine powder in the air isn't healthy for lungs or lab gear.
What Happens to It With Heat and Water
Heating potassium pivalate leads to a melting point just above 300°C—a solid performer compared to less stable salts. This tells you it won’t fall apart in most day-to-day lab work or process steps. It’ll take a strong oven or a dedicated furnace before you see any changes. The substance doesn’t burn, nor does it create a fire hazard, but that much heat might bring on a decomposition odor and noxious fumes. Most safety data sheets point out that good ventilation and protective gear matter, especially under high heat.
In water, potassium pivalate dissolves easily. Stir it into a beaker, you get a clear solution. It doesn’t form any fancy crystals or separate out into layers; what you see is what you get. If you try stirring it into alcohol or organic solvents, you probably won’t see much dissolve—the potassium ion likes water much more than oil. Solubility plays a big role in how chemists use it to adjust pH or as a base in reactions, since cleanup becomes a rinse-down-the-drain job, instead of scraping stubborn residues from glassware.
The Role of Structure
The chemical backbone of potassium pivalate holds together because of potassium’s charge and the bulky structure of the pivalate ion. The way the atoms are arranged sets up its properties. The compact, stable nature of the molecule means low reactivity in storage, so a bottle of potassium pivalate practically gathers dust until someone needs it. Its sturdy makeup also stops it from caking as much as highly hygroscopic salts, which would turn to a goopy mess in a damp lab. Storage becomes simple: screw the cap back on, put it on the shelf, and forget about it until the next experiment.
Why These Traits Matter in the Real World
Many labs use potassium pivalate for the strong base it brings to fine-tune chemical reactions. Because of the high melting point and easy solubility in water, batch reactions stay safer and cleanup gets easier. Sharp-minded process engineers look at the granule size as well, since it pours freely without clumping—a small but significant detail on busy days at the bench.
Packing and shipping don’t raise major red flags with this compound. The lack of odor, solid texture, and resistance to humidity allow for simple, cost-effective containers. Potassium pivalate doesn’t pose the risk of sudden decomposition or spontaneous combustion, making transport through ordinary postal services possible, a rare trait among active chemicals.
Solutions for Better Use and Safety
Handling the powder with basic dust protection gear—gloves and a face mask—eliminates most exposure risks. Laboratories looking to improve air quality use decent extraction systems so nobody breathes any suspended particles. Switching to pre-measured, sealed packets can cut down on both waste and potential human error, especially for less-experienced staff. If spills happen, the compound sweeps up without leaving a stain or strong residue, and standard washing routines suffice for cleanup.
Straightforward properties make potassium pivalate a low-maintenance, reliable chemical—one that does the job without much fuss, as long as users pay basic attention to safety and storage.
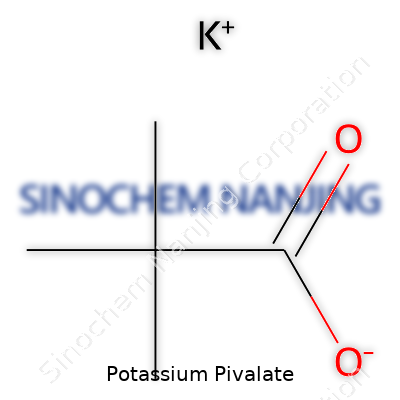
| Names | |
| Preferred IUPAC name | Potassium 2,2-dimethylpropanoate |
| Other names |
Pivalic acid potassium salt Potassium trimethylacetate Potassium 2,2-dimethylpropanoate |
| Pronunciation | /pəˈtæsiəm pɪˈveɪleɪt/ |
| Identifiers | |
| CAS Number | 4920-93-6 |
| 3D model (JSmol) | `JSmol` 3D model string for **Potassium Pivalate** (chemical formula: C5H9KO2): ``` K+ C(C)(C)C(=O)[O-] ``` This is the **SMILES** string, which can be used in JSmol or other molecule visualizers to generate a 3D model. |
| Beilstein Reference | 610126 |
| ChEBI | CHEBI:84915 |
| ChEMBL | CHEMBL3980548 |
| ChemSpider | 19011 |
| DrugBank | DB14540 |
| ECHA InfoCard | The ECHA InfoCard for Potassium Pivalate is: **03e63bea-8da0-4b69-8caf-8a918645ed90** |
| EC Number | 209-896-6 |
| Gmelin Reference | 120158 |
| KEGG | C18561 |
| MeSH | D017708 |
| PubChem CID | 86508 |
| RTECS number | TD3850000 |
| UNII | 50R5X8JP7O |
| UN number | UN2813 |
| Properties | |
| Chemical formula | K(C5H9O2) |
| Molar mass | 142.22 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.15 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -0.97 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 5.03 |
| Basicity (pKb) | pKb ≈ 9.3 |
| Magnetic susceptibility (χ) | -49.3·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.399 |
| Viscosity | 2.1 mPa·s (20°C, 20% solution) |
| Dipole moment | 1.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 165.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -575.5 kJ/mol |
| Pharmacology | |
| ATC code | A12BA02 |
| Hazards | |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319 |
| Precautionary statements | Wash face, hands and any exposed skin thoroughly after handling. IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. If eye irritation persists: Get medical advice/attention. |
| Autoignition temperature | 360 °C |
| LD50 (median dose) | LD50 (median dose) of Potassium Pivalate: **1,800 mg/kg (oral, rat)** |
| NIOSH | Not listed. |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium acetate Potassium propionate Potassium butyrate Sodium pivalate Pivalic acid |

