Potassium Methoxide: Deep Dive into a Key Chemical in Modern Synthesis
Looking Back: Origins and Historical Development
Potassium methoxide, known to chemists as potassium methylate, has found its place in laboratories since the middle of the twentieth century. The growing demand for efficient alkali catalysts in organic transformations, especially in biodiesel production and fine chemical synthesis, brought potassium methoxide into the spotlight. In the early days, researchers couldn't get enough of the milder and safer alternatives to sodium-based reagents; potassium methoxide stood out as a powerful base that offered a blend of reactivity and solubility. Scientists with backgrounds in alkali metal chemistry can recall how the development of anhydrous preparations expanded the reaction toolkit. As the chemical industry moved away from hazardous solid bases, potassium methoxide slowly took over tasks where fast, controlled deprotonation and methylation counted most. Over decades, handling techniques matured and manufacturing scaled, yet the heart of its application remains rooted in those simple early experiments that pointed to its strengths.
Product Overview and Identity
In academic papers, potassium methoxide sometimes pops up under names like potassium methylate or methanolate. These synonyms speak to a substance that's remarkably straightforward at its core: the potassium salt of methanol, with the chemical formula CH3OK. Unlike bulk industrial commodities, potassium methoxide goes out mostly to those who need a punchy base or methylating agent—think organic chemists, pharmaceutical developers, and producers working in biodiesel plants. You can spot it offered as a solid, a powder, or in solution, often dissolved in methanol. It's one of those chemicals that likes to hide under multiple labels, but with properties and hazards that never stray. Wise chemists keep it on hand for reactions where you need clean, strong base conditions, but never lose respect for its potent chemistry.
Physical and Chemical Properties
Potassium methoxide is usually a white to off-white crystalline solid, though anyone who's left a jar open will quickly see it absorb water and methanol vapor from the air until it cakes together. It's hygroscopic, pulling moisture fast, and contact with water releases methanol and caustic potassium hydroxide. In organic solvents like methanol, it dissolves well, kicking out a clear, colorless solution. Those solutions tend to be highly alkaline, with a sharp, characteristic odor pointing to escaped methanol. Potassium methoxide packs real power as a nucleophile and base. Unlike sodium methoxide, potassium's larger ionic radius slightly tweaks solubility and reactivity, which shows up in fine-tuned reaction outcomes. This difference matters to folks trying to squeeze higher yields out of methylations and transesterifications where subtle changes mean the difference between profit and loss.
Technical Specifications and Labeling
Chemicals like potassium methoxide ride on quality. Purity hits above 98% for lab and industry use, usually highlighted directly on the bottle or drum. Labels matter here, since incorrect markings can lead to accidents or ruined batches. Real chemists learn to check not just the purity but also to cross-reference batch analysis to avoid hidden water content or organic impurities that could spoil a reaction. Potassium methoxide in solution poses sampling challenges, with methanol content and base strength usually specified in molarity (mol/L). Transport and storage labels carry hazard symbols pointing to flammability and corrosiveness. It's not just bureaucratic red tape—missing those warnings costs hands and eyes. Each batch leaving a supplier needs to tell a clear story about its contents, dangers, and how it should be handled once it hits a lab or factory floor.
Preparation Methods: From Lab to Plant
The most direct route to potassium methoxide runs through the reaction of potassium metal with dry methanol. Potassium chunks dropped into methanol react quickly, fizzing up hydrogen and yielding the methoxide as a strong solution. The reaction chugs away exothermically, and old-timers remember the hazards—hydrogen gas with a flare for explosions if not vented well. In industrial settings, process engineers stick to tried-and-true methods, using controlled feeds and robust mixers, because a slip means catastrophic fires or chemical burns. Other prep options start with potassium hydroxide, mixing it with methanol and removing the water generated, but purists prefer direct synthesis from potassium metal to avoid lurking water. After production, manufacturers dry and store the product under inert atmospheres—think nitrogen or argon—to keep it from grabbing carbon dioxide or water, which both degrade its usefulness fast.
Chemical Reactions and Modifications
If you hand potassium methoxide to a synthetic chemist, expect reactions galore. In organic labs, it's a mainstay in methylation reactions, nucleophilic substitutions, and as a base to generate enolates or drive transesterifications. The formation of methyl esters—especially in biodiesel production—takes full advantage of its power to convert triglycerides and methanol into fatty acid methyl esters, which go straight into fuel tanks. In medicinal chemistry, potassium methoxide unlocks routes to complex molecules by cleanly deprotonating sensitive intermediates or adding methyl groups where they're desired. Unlike sodium analogs, potassium methoxide sometimes delivers snappier or more selective reactions because potassium's larger size and different solvation play subtle roles in transition states. On top of this, clever minds have tried tweaking the basicity or solubility by forming mixtures or double salts, though simple, pure potassium methoxide remains king for most bread-and-butter applications in synthesis.
Application Area: Where It Really Matters
Biodiesel production probably claims the lion’s share of industrial potassium methoxide use; plant managers rely on its speed and reliability to convert vegetable oils and animal fats into useful fuel in transesterification reactors. Pharmaceutical labs keep it nearby to support routes that hinge on clean methylations and efficient enolate formations. Agrochemical makers appreciate how potassium methoxide steps in for both routine and tricky base-mediated transformations. Academic teams and industrial researchers both value how easily it slips between roles as a strong base or nucleophile. Those in the field realize that potassium methoxide shortens synthetic timelines or helps avoid toxic byproducts linked to weaker bases or mineral acids. Its reach goes deep into flavor and fragrance chemistry, fine chemical manufacture, and specialty polymer work.
Research and Development: Pushing Boundaries and Safety
Researchers have been on a hunt to make potassium methoxide prep safer, less hazardous, and more sustainable. Process engineers are trying to scale up production methods that cut down risks tied to handling metallic potassium. Some innovators are exploring solid-supported forms or non-methanol solvents that could both reduce flammable risk and address sustainability concerns. Analytical chemists in my network focus on tracking contaminants and designing titration protocols that confirm base strength down to fractions of a percent, improving consistency and batch reproducibility. Others shape protocols that minimize methanol evaporation and exposure, dialing in new reactor designs or encapsulated products for safer, easier handling. Research teams looking to cut down the carbon footprint of alkali base production find themselves reviewing potassium methoxide's upstream impacts and exploring recycling routes for waste streams created during synthesis. Every gain in safety or green chemistry quickly turns into a competitive advantage for companies and a reduced burden on chemical handlers down the line.
Toxicity Research: Human and Environmental Impact
Anyone working with potassium methoxide knows firsthand its hazards. Brief skin contact leads to severe burns, and eye splashes run a high risk of permanent injury. Inhalation or accidental ingestion spell disaster, combining the corrosive power of strong base with the internal toxicity of methanol. Toxicologists warn that exposure—especially in combination with methanol—demands fast first aid and specialist treatment. Environmental teams flag runoff or spills as a real concern. Once released, potassium methoxide hydrolyzes fast to potassium hydroxide and methanol, both of which pack their own toxic punches. Methanol leaches into water systems, endangering aquatic life and risk of fire, while potassium hydroxide makes soil and water alkaline, scrambling ecosystems. Regulations set tight controls on how plants store, transport, and neutralize waste, but the margin for error stays small. Training operators and investing in robust containment and ventilation end up paying for themselves through avoided incidents.
Future Prospects: Where It’s All Headed
Looking at the years ahead, the need for efficient, green catalysts shows no sign of slowing down. Potassium methoxide stands to gain ground wherever cleaner methylations and faster transesterifications drive profit and sustainability. Pushes to upcycle waste oils, develop greener biodiesel processes, and trim the ecological footprint in chemical manufacturing all keep potassium methoxide near the front of the line. Researchers targeting safer operations may turn to encapsulated formats or solid-supported variants that slash spillage risk and fire hazard, which would mark a big win for chemistry workplaces. Demand for digital monitoring tools—autotitration, in-line purity checks, real-time safety sensors—will only rise as plants seek faultless product and ironclad compliance. The biggest gains will likely come from steps that let companies deliver the same reliable chemistry with less environmental cost and fewer human hazards, keeping potassium methoxide not only indispensable, but safer to handle and less likely to make headlines for the wrong reasons.
What is Potassium Methoxide used for?
Biodiesel: Cleaner Fuel, Simple Chemistry
Potassium methoxide shows up a lot in biodiesel facilities. Factories trust it to drive the chemical reaction that turns vegetable oils and animal fats into biodiesel. This isn’t just some lab project; it’s a process woven into efforts to make cleaner-burning fuels. In my time volunteering at a community co-op, I helped with small-scale biodiesel production. We needed something to break the natural bonds in waste cooking oil so it could become a usable fuel for our delivery van. Potassium methoxide did the heavy lifting every time. Without it, the oil just stayed oil. The process looks simple: the chemical acts as a catalyst, helping methanol and oil blend into usable fuel and a byproduct called glycerol.
Pharmaceuticals: Behind the Scenes in Medicine
It’s easy to take everyday medications for granted, but before pills end up in your bathroom cabinet, chemists depend on compounds like potassium methoxide to build them. In some drug factories, this chemical helps form key ingredients for diabetes, heart disease, or antibiotic drugs. Skilled operators need to respect its strength: a splash or inhalation in the lab causes real harm. That’s why trusted pharmaceutical manufacturers build strict safety routines around its use. The fact that many modern medicines depend on smooth chemical reactions shows potassium methoxide isn’t just for factories—it’s tangled up in healthcare and lives.
Other Uses: Paints, Agrochemicals, and Everyday Chemicals
Potassium methoxide slips into all sorts of products. Paint makers use it in the background to pull off those chemical transformations that keep finishes smooth and tough. The story’s the same in agricultural chemicals—farmers often count on fertilizers or pesticides tweaked with help from this compound. Even some cleaning products start off with a chemical chain that needed potassium methoxide somewhere in the process.
Risks, Safety, and What Could Change
Working with potassium methoxide means facing hazards. It reacts sharply with water, so even a humid room spells trouble. Touching or breathing the fumes can cause burns or lung damage. At the co-op, training and protective gear kept us safe, but not everyone has access to the same resources. With accidents in poorly equipped places, health and environmental risks creep in quickly. Studies from the American Chemical Society remind us: the more widespread the chemical, the easier it becomes to overlook safety.
Regulations keep a tight grip. In the US, OSHA sets rules about handling and storage—proper gloves and goggles, ventilation, and chemical containment. Still, mistakes and shortcuts happen, especially where corners get cut for profit or speed. Cleaner, safer alternatives keep getting tested in the lab. One hope: green catalysts that don’t stir up the same hazards or create as much waste. While some researchers have found promising leads, nothing yet matches the reliability of potassium methoxide for big production runs.
The Bottom Line
Walking down a city street, you’d never know that something as reactive and hazardous as potassium methoxide sits behind cleaner fuels, medications, crop yields, and coatings on benches and bus stops. Factory workers, chemists, and even hobbyists take the risks seriously. They push for better safety, smarter regulation, and chemistry that does the job without putting lives or the environment on the line.
What are the safety precautions when handling Potassium Methoxide?
Why Potassium Methoxide Demands Respect
Potassium methoxide shows up in a lab or factory, usually because someone needs a strong base or catalyst. I remember the sharp, bitter scent the first time I heard a chemist crack open a bottle. What always hits me is the power packed into this white powder. Touching it with bare skin burns, and breathing it in can knock you sideways. It doesn't take a veteran chemist to know that every decision matters with a substance this reactive.
Handling Personal Protection
Full coverage saves skin and lungs. Use chemical-resistant gloves—nitrile or neoprene beats latex every day of the week. Go for a face shield on top of your goggles. A lab coat keeps your everyday clothes clean, but if you have access, splash-resistant aprons and arm guards matter more. Potassium methoxide powder goes airborne easy. Stick with a respirator fitted with particulate filters instead of trusting a dust mask.
Safe Workspace Setup
Some chemists cut corners, thinking a fume hood doesn’t matter unless you’re working with really nasty stuff. That’s risky thinking. Potassium methoxide interacts with moisture in the air and can release methanol, which is flammable and toxic. Use the hood. Surfaces catch fire more often due to spills mixing with stray drops. Line your work area with absorbent pads or trays, and double-check there’s a fire extinguisher nearby—one rated for chemical fires, not just kitchen grease blazes.
Risks of Storage and Spills
In school, I saw a jar of potassium methoxide left open for about twenty minutes. Hours later, a white crust formed along the rim. Exposure to air means it starts to degrade, forming potassium hydroxide and methanol. Both burn and corrode. Screw caps tight, stash containers in a cool, dry cabinet, and label everything in clear, waterproof ink. Heat and sunlight turn sealed jars into pressure traps. Old jars? Dispose of them using a hazardous materials service—never dump leftovers down the drain.
Coping with Exposure
Even with proper gear, accidents happen. Splashes burn fast. If potassium methoxide touches the skin, run—don’t walk—to the eyewash or safety shower. Douse the area fast, peel off affected clothing, and don’t rub the injury. Medical help can’t wait. Inhaling dust means headache, dizziness, or trouble breathing will come quick. Get out to clean air and let emergency services know what you breathed. Methanol poisoning can take a while to show up, so insist on a checkup.
Why Training and Culture Save Lives
Watching a seasoned technician run through the safety process, you see confidence comes from practice. Ongoing training beats posted rules. Shortcuts in real life mean injuries down the road. Team culture changes when people refuse to rush. Supervisors modeling safe practice mean coworkers look out for one another and catch mistakes before they turn serious.
Better Solutions for Safer Workplaces
Switching to less hazardous alternatives sometimes makes sense. Sodium methoxide, for example, carries some risk but not as much. If production requires potassium methoxide, safer work practices—like smaller batch sizes, automated handling, and remote mixing—cut down spill potential. Well-maintained spill kits and a strong relationship with emergency responders finish the job. The power and danger of potassium methoxide both demand constant respect and a refusal to get comfortable.
How should Potassium Methoxide be stored?
Packed with Hazards
Anyone who has ever handled potassium methoxide knows it takes more respect than most folks realize. This chemical looks like a run-of-the-mill white powder, but mix it with air or a little bit of humidity and things get risky. Ignoring the rules just invites accidents that can hurt people and ruin equipment. I once watched a colleague clean up a tiny spill with wet rags, and the smoky mess that followed taught me to read up on the real dangers. Skin burns. Fumes that sting the lungs. Even the stubborn labels on the drums feel like a warning from someone who’s had a long shift and enough close calls.
Tight Control Over Moisture and Air
Potassium methoxide wants to react with just about anything. Water turns it into methanol and strong caustic potash, both of which aren’t great for human health. For this reason, storage always starts with the right containers. I’ve seen companies skip the basics and use makeshift bins, but corrosion sets in fast and you start to smell trouble. Stainless steel, with sturdy seals that keep out damp air, works best in my experience. Forget open shelves or rusty cabinets—this stuff belongs in closed, labeled drums, with gaskets that haven’t gone soft after a few seasons. Even simple humidity from a sloppy warehouse floor can creep into a poor seal and set off a slow, dangerous change inside.
Temperature Control: The Cheap Insurance
Potassium methoxide does its best work in the shade. Exposing it to warm rooms or direct sunlight hurries up its natural urge to break down, raise pressure inside drums, or turn into a fire hazard. Some labs invest in climate-controlled storage for good reason. I’ve watched summer heat push tanks to distortion, making the opening of a drum feel like opening a soda bottle that’s been shaken one too many times. Cool, stable temperatures keep the risk lower and stretch out shelf life. I tell anyone who will listen: a well-ventilated, air-conditioned spot is worth every penny come audit day.
Standing Orders for Safety
Training matters even more than locks. Folks should know not to poke around containers without gloves and goggles, not just from reading a manual but from hands-on walk-throughs that show what can go wrong. Good labeling—big, bold, and specific—keeps new staff from reaching for the wrong tool or mixing up supplies. Spill kits need to sit right where leaks might happen, not in some drawer across the building. Fume extraction systems, even simple ones hooked to storage cabinets, cut down on the vapor exposure that creeps up in busy workspaces.
Trained Eyes Beat Luck Every Time
I’ve learned that a regular walk-around, checking seals, labels, and drum shapes, catches more problems than any spreadsheet. Leaks and bulges mean disaster if ignored. Mixing potassium methoxide with acids or even strong oxidizers sets off reactions faster than most would expect—I saw a drum top pop once from a mix-up and have never opened one casually since. Small changes—using color-coded tags, designating storage zones, holding safety reviews—can save real money and real lives. Even experienced hands pick up new habits after seeing just one incident up close.
Solutions Worth Repeating
If companies put money into sealed, stainless storage and push clear safety training, a lot of the accidents fade into memory. Keeping the chemical away from damp, heat, and confusion limits accidents that become news stories for all the wrong reasons. It doesn’t take fancy upgrades—just careful, honest work, a bit of pride in the routine, and a policy of sharing lessons learned before the next shift clocks in.
What is the chemical formula of Potassium Methoxide?
The Formula and What It Means
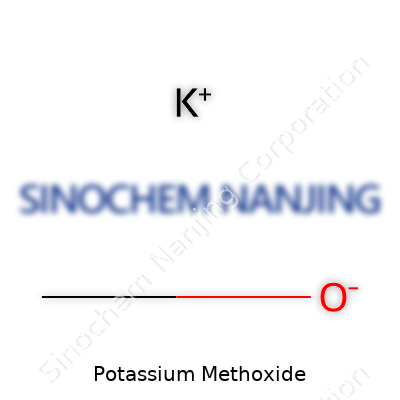
Potassium methoxide shows up on the blackboard as CH3OK. For folks who spend their days in a lab, that arrangement isn’t just a string of letters—it shares a story about how potassium, a reactive metal, comes together with the methoxy group drawn from methanol. Chemically, you start with methanol and a strong potassium base; out comes potassium methoxide and hydrogen gas. So, the straightforward formula, CH3OK, covers a lot more than meets the eye. It marks this compound as an organopotassium reagent, and that little K tacked on the end brings in a whole new set of chemical behaviors compared to something like sodium or lithium methoxide.
Why It Matters in Industry and Labs
Synthetic routes in organic chemistry depend on solid bases, and potassium methoxide fills that need. It reacts fast, acts strong, and keeps things moving in biodiesel production, pharmaceuticals, and various lab syntheses. Anyone mixing up biodiesel knows that choosing the right base speeds transesterification. Potassium methoxide, thanks to that potassium atom, creates cleaner separations. Facts back this up—potassium methoxide often shows higher conversion rates than sodium methoxide, translating into more fuel per input and less waste down the drain.
In research, it’s often the little differences that tip the scales. Potassium methoxide has a higher solubility in methanol compared to its sodium cousin. This means more reagent dissolved in the same amount of liquid, so reactions run quicker or produce purer products. From personal experience in the lab, seeing a reaction finish hours sooner because a more soluble base has been used is worth its weight in gold when deadlines pile up.
Handling and Safety Concerns
Chemistry brings risks as well as rewards. Potassium methoxide throws off hydrogen gas during preparation—that gas doesn’t just float away. Hydrogen is flammable, and it only takes a small spark to set off an explosion. Direct contact with the powder or solutions can also burn skin and eyes. So, anyone reading up on chemical preparation needs to put safety at the top of their list. Gloves, goggles, and fume hoods aren’t negotiable in this setting.
Companies and universities run regular safety briefings for just this reason. In graduate school, stories about someone skipping gloves or eye protection always get told more than once. The point sticks: better to take five extra minutes and avoid catastrophe. Proper chemical storage and handling procedures stop most emergencies before they start.
Issues and Smarter Choices
Access to reliable sources of potassium methoxide sometimes stalls smaller biodiesel startups or labs, especially in developing regions. One fix could involve fostering partnerships between institutions and established suppliers, lowering bulk costs and building better safety networks. There’s also room for more open-source chemical safety manuals, since rules set by major journals like the Journal of Chemical Education don’t always make it to every classroom or workplace. Sharing real-world safety practice and up-to-date handling tips, not just bland lists, will keep more people both productive and safe.
In the end, the formula CH3OK brings together science, safety, industry, and people. It’s more than textbook trivia—the story of potassium methoxide connects labs with the global drive toward more efficient, responsible chemistry.
Is Potassium Methoxide soluble in water?
Potassium Methoxide: Beyond the Lab Bench
Potassium methoxide shows up in textbooks and industrial recipes, usually described as a strong base and a useful chemical for synthesis. I ran into it back in graduate school, where it helped speed along a range of organic reactions, especially those creating biodiesel. Its punchy basic nature makes it handy, but also a bit unpredictable compared to sodium methoxide, its close cousin.
What Happens in Water?
Adding potassium methoxide to water isn’t like mixing table salt in a glass. The powdered or sometimes liquid chemical interacts violently. You won’t just see it dissolve; instead, you’ll see a rapid, almost explosive fizz. Potassium methoxide reacts with water in a flash, breaking apart into methanol and potassium hydroxide, both potent and hazardous in their own right. This reaction releases heat, sometimes enough that those of us working with it need to use ice or heavy gloves to keep things safe.
Solubility and Chemistry Lessons
Chemists discuss solubility as the ability of a substance to dissolve and create a uniform solution. In the classic sense, potassium methoxide never really gets that chance. Water tears the molecule in half faster than you can stir. What ends up in solution isn’t potassium methoxide anymore, but a mix of potassium hydroxide and methanol. The hoped-for chemical is gone; only its reactive pieces remain. Someone working in industry or with a keen interest in clean, controlled reactions soon learns water always wins this fight.
Why It Matters Outside the Lab
People making biodiesel pay special attention to this. If water sneaks into their process, their potassium methoxide stops doing its job, and the spent solution turns slippery and caustic from all the potassium hydroxide. Even a small amount of water in the feedstock makes things unpredictable, leading to less yield and more waste. Homebrewers and technicians often keep everything dry to avoid surprises. The result? Better quality biofuels, safer processes, and less corrosive equipment. It’s messy to fix a process gone wrong by a tiny drop of water.
Safety and Solutions
The dangers with potassium methoxide aren’t only chemical. Breathing in fumes from the quick reaction or letting splashed solution touch skin can cause serious burns. Methanol vapors pose their own issues, including toxicity. Handling solutions means using good ventilation, gloves, and goggles every step of the way. I’ve known more than one colleague who came away with a story about cleaning up a spill that boiled on contact with a little water—never twice from that same mistake.
Labs and industries solve these problems by using dry solvents whenever possible. Keeping containers closed tight, adding moisture scavengers, and training staff on the risks all help. If water exposure happens, neutralizing spills right away and ventilating spaces make a difference. The lesson from both personal and industrial experience: treat potassium methoxide with respect and keep it bone-dry if you want it to play by the rules.
| Names | |
| Preferred IUPAC name | Potassium methanolate |
| Other names |
Methoxypotassium Potassium methylate Potassium methanolate Methanol, potassium salt Potassium methyl oxide |
| Pronunciation | /poʊˈtæsiəm məˈθɒksaɪd/ |
| Identifiers | |
| CAS Number | 865-33-8 |
| Beilstein Reference | Beilstein Reference: 3565006 |
| ChEBI | CHEBI:41247 |
| ChEMBL | CHEMBL54937 |
| ChemSpider | 14043 |
| DrugBank | DB11233 |
| ECHA InfoCard | 100.029.179 |
| EC Number | 246-917-9 |
| Gmelin Reference | Gmelin Reference: 16824 |
| KEGG | C14295 |
| MeSH | D017794 |
| PubChem CID | 9004 |
| RTECS number | OG6600000 |
| UNII | W5W4G630CS |
| UN number | UN 2014 |
| CompTox Dashboard (EPA) | DTXSID0066065 |
| Properties | |
| Chemical formula | KMethO |
| Molar mass | 68.13 g/mol |
| Appearance | White to yellowish solid |
| Odor | Alcohol-like |
| Density | 1.2 g/cm³ |
| Solubility in water | Very soluble |
| log P | -0.24 |
| Acidity (pKa) | 14.18 |
| Basicity (pKb) | pKb = 0.5 |
| Magnetic susceptibility (χ) | +14.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.332 |
| Viscosity | Viscosity: 1.0 mPa·s (at 20 °C, 25% solution in methanol) |
| Dipole moment | 1.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 91.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -409.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -567 kJ/mol |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H260, H314 |
| Precautionary statements | P210, P222, P223, P231, P232, P261, P264, P280, P301+P330+P331, P303+P361+P353, P305+P351+P338, P310, P321, P370+P378, P402+P404, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Autoignition temperature | > 430°C |
| Lethal dose or concentration | LD50 Oral Rat 430 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 430 mg/kg |
| NIOSH | SN 3850000 |
| PEL (Permissible) | PEL for Potassium Methoxide: Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of Potassium Methoxide is: "0.5 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium methoxide Lithium methoxide Potassium ethoxide |

