Potassium Hydrogen Sulfate: A Close Look at a Chemical Powerhouse
Historical Development
Potassium hydrogen sulfate’s story stretches back hundreds of years. In the early days of chemistry, mineral acids like sulfuric were some of the first to be harnessed, and salts born from these acids, including potassium hydrogen sulfate, quickly found a place in the toolkit. Back when glass manufacturing and early alchemical processes drove the demand for reagents, chemists experimented in cramped labs, using primitive glassware and open flames, not surrounded by modern safety gear. The simple act of heating potassium bitartrate with potassium sulfate became a stepping stone in the industrial chapter of chemical history, showing ingenuity long before computers and automated reactors. The evolution from a mysterious byproduct to an essential intermediate demonstrates how the exploration of everyday compounds can transform entire industries.
Product Overview
Potassium hydrogen sulfate, often labeled as potassium bisulfate or monopotassium sulfate, pops up in a surprising range of workflows. This white, crystalline solid doesn’t turn heads on the shelf, but in the laboratory or factory it delivers pure function—a standard for pH calibration, a catalyst in organic transformations, and a way to acidify cleaning and etching baths. If you’ve worked in a chemistry lab or dabbled in ceramics and fertilizers, you probably brushed sleeves with it more than once. Its price and availability reflect consistent industrial demand, and its utility in processing rare earth elements, refining sugar, and preparing analytic samples speaks to a compound that has long punched above its weight for such a simple formula.
Physical & Chemical Properties
Long, needle-like crystals and a clear, colorless form mark potassium hydrogen sulfate at a glance. It dissolves easily in water, releasing heat as it goes, and brings strong acidity to the solution, enough to challenge glass over time. If you leave a batch of this salt exposed to moist air, it slowly picks up water, becoming sticky and clumped. The sharp, sour taste makes it unpalatable in every sense, and even a whiff of dust stings the nose. Melt it, and you’ll see decomposition into potassium pyrosulfate with the evolution of sulfur oxides—one hell of a reminder to keep fume hoods in top shape. As a monovalent salt with the formula KHSO4, it shows a unique balance between stability and chemical drive, handing off protons to water with an eager force chemists value for digestion procedures and other acid-driven steps.
Technical Specifications & Labeling
Purity levels matter a lot, no matter whether the package heads to the lab for reagent work or to a plant for industrial use. Most suppliers offer grades ranging from technical for industrial-scale usage up to analytical and trace analysis levels. The appearance—free-flowing, white, largely free of visible impurities—tells you whether the production batch met basic quality expectations. Solubility checks are routine, as is the measurement of acidity through titration. Proper labeling means clear hazard pictograms, statements about skin, eye, and respiratory irritation, and instructions that advise users to avoid ingesting or inhaling the dust. Chemists quickly recognize the importance of clear labeling: one contaminated scoop or careless substitution can mess up results and trigger a chain reaction of costly troubleshooting.
Preparation Method
Most industrial potassium hydrogen sulfate comes from mixing potassium sulfate with excess sulfuric acid. Heat the mixture above 200°C, remove water, and potassium hydrogen sulfate crystallizes out. On a smaller scale, the method may switch: adding potassium carbonate or potassium hydroxide to sulfuric acid in a careful, slow pour, since the exothermic reaction can get away from you if you push too fast. The simplicity appeals to those who appreciate straightforward chemistry, but it also means oversight is critical. Minor fluctuations in temperature or ratios can tip yields off balance and bring impurities along for the ride. Whether you’re aiming for food-safe specifications or high-purity analytical salt, every ounce of quality starts with consistent preparation and strict attention to process control.
Chemical Reactions & Modifications
Throw potassium hydrogen sulfate into hot water and it splits apart, releasing potassium ions and hydrogen sulfate anions that make an acidic solution, handy for acid digestion and certain titrations. Heat it above 200°C, and it condenses to potassium pyrosulfate, a more complex salt used as a powerful oxidizer in specialty organic and inorganic syntheses. Mix it with bases such as sodium hydroxide and you get double replacement reactions—one route to other potassium salts, or, with careful handling, to sulfuric acid recovery. The salt’s driving force as an acid makes it versatile in wet-ashing protocols, where dissolving tough, organic-rich samples needs a steady stream of protons under heat. That’s chemistry coming full circle: simple reagents, reliable transformations, and few surprises as long as you know what to expect.
Synonyms & Product Names
Potassium hydrogen sulfate pops up under several names: potassium bisulfate, monopotassium sulfate, acid potassium sulfate, and even potassium acid sulfate. Each reflects different traditions or technical standards, and a chemist’s preference usually depends on childhood textbooks or regulatory paperwork. International regulatory bodies—REACH, OSHA, GHS—list it among controlled substances, each with regulations affecting trade, labeling, and disposal. But wherever you spot it, whether abbreviated as KHSO4 or not, it’s the same hard-working acid salt, recognized by properties rather than name alone.
Safety & Operational Standards
Experience teaches that thorough respect for safety demands more than just gloves and goggles; protocols need rigorous planning, and everyone using potassium hydrogen sulfate should recognize its ability to irritate eyes, skin, and respiratory tracts. Inhaling even small amounts of dust brings burning sensations, coughing fits, or worse if chronic exposure sets in. Splash it on unprotected skin and the burn soon follows. Local exhaust ventilation, dust masks, and chemical-resistant gloves have saved more than a few careless workers from trips to the safety shower. Emergency procedures—eye wash stations, spill control, and neutralization chemicals—should always be within arm’s reach. The industry’s push for GHS-aligned labeling and risk communication reflects lessons learned from accidents of decades past, and it’s a push that continues shaping daily work.
Application Area
Potassium hydrogen sulfate’s reach spans laboratories, fertilizer plants, food-processing lines, and even the crafts world. Analytical chemists use it for sample digestion, where its potent acidity breaks down tough organic matrices—making light work of soils, plants, and food products. Metallurgists depend on its ability to dissolve and refine rare earths and precious metals, providing a cleaner route to high-value elements. Sugar refineries count on it for cleaning and pH adjustment, and its role in glass manufacture gives artisans a reliable flux for specialty compositions. In fertilizer manufacture, the carefully measured addition of potassium hydrogen sulfate boosts potassium availability in acidic soils, supporting the world’s agricultural backbone. Each year, stack after stack of bags moves across continents to supply these spinoffs—and that’s only the business end, before you reach the niche uses in ceramics glazing and specialty cleaners. The breadth of application proves its resilience in face of changing industry trends.
Research & Development
New uses for potassium hydrogen sulfate keep emerging. Recently, researchers began exploring it as a starting point for green chemistry syntheses, emphasizing reduced energy consumption and waste generation. Electrochemical studies explore its behavior in ionic liquids and superacid mixtures—seeking new routes to catalysis or energy storage. Analytical chemists won’t give it up, relying on its clean, sharp effects for sample prep, but the biggest gains may come in materials science. Modifications of the basic salt, sometimes by doping with trace metals, suggest possible uses in non-traditional batteries and as supports for heterogeneous catalysis. Multinational collaborations open new avenues by blending traditional wet chemistry with modern, automated analytics, squeezing better yields and fewer emissions from age-old processes. The challenge lies in convincing conservative industries to adopt what looks, at first glance, like small tweaks with uncertain payback.
Toxicity Research
Compared to heavy-metal salts, potassium hydrogen sulfate does not raise alarms—at least not for acute toxicity. Studies show low oral and environmental toxicity in mammals, birds, and aquatic life at typical exposure levels, though intense eye and skin irritation keeps the risk profile elevated for direct contact. Chronic exposure can still bring complications, primarily from acid burns or airway inflammation. Research into long-term, low-level environmental release shows only mild effects at the concentrations found downstream from fertilizer runoff, but scientists track rising cumulative use, knowing well that today’s small numbers can add up to tomorrow’s persistent problem. The push for better industrial hygiene standards, personal protective equipment, and spill containment measures only grows as more factories, farms, and labs boost consumption to meet global demand.
Future Prospects
Each shift in the chemical industry—more sustainable practices, tighter regulations, demand spikes from new tech—throws potassium hydrogen sulfate onto a moving target. The push for “green” fertilizers and smarter crop management calls for lower-impact, more precisely tailored blends, and this salt fits comfortably in those plans thanks to its high potassium content and acidic release. Organic synthesis may unlock new potential if researchers keep refining eco-friendly protocols. As energy storage tech matures, electrochemical applications could bring the salt into design discussions around safer, cheaper batteries. One persistent challenge: the need for tighter quality controls to limit contaminant buildup, especially in food and pharmaceutical segments. If research keeps expanding into hybrid materials and greener process engineering, potassium hydrogen sulfate’s next hundred years could look very different from its last, spurred onward by the push for practical, affordable chemistry that meets ever-evolving global needs.
What is Potassium Hydrogen Sulfate used for?
More Than Just a Chemical Formula
Potassium hydrogen sulfate doesn’t show up on headlines about new tech, drug breakthroughs, or even hot debates about fertilizers. Still, this low-profile compound drives work in classrooms, factories, and even in environmental labs. Most people meet this stuff first in high school chemistry when learning about acids and the magic of turning rocks into metals.
Role in Everyday Lab Work
If you’ve ever sat in a lab trying to break apart minerals and wondered why the teacher reached for potassium hydrogen sulfate, here’s the trick: it’s strong enough to help dissolve tough minerals, transforming solid rock samples into a liquid form so scientists can test what’s inside. For example, soil specialists use it to figure out which metals are stuck in a rock or dirt sample.
I remember my earliest experiments with it, crushing a mineral, adding the salt, and watching in awe as everything broke apart in the heat. It’s moments like that which make chemistry feel less like memorizing and more like solving puzzles. Without this compound, breaking down tough geologic material takes way longer or gets messy with stronger, less safe acids.
Supporting the Food Industry
Some folks think chemistry has nothing to do with what’s on the dinner table. Potassium hydrogen sulfate challenges that idea. It helps food producers analyze fats and oils by getting rid of water in lab samples, making tests more accurate. The details behind cooking oils—whether that bottle of olive oil from the store contains the right amount of good fats—often comes down to quality control using compounds like this one.
Keeps the Paper Mills Running Smoothly
It’s easy to forget how much science shapes paper. Potassium hydrogen sulfate helps get rid of certain unwanted minerals during pulp preparation. Cleaner pulp gives a finer texture and a better print, even on receipts or old-school notebooks. When I walked through a mill for the first time, seeing the giant vats churn out watery pulp, it felt surreal that something as simple as this compound could matter so much to something we use every day.
Chemistry Classroom Mainstay
Students start with this chemical because it handles like a solid, not a dangerous liquid. Safe and predictable, it turns into a strong acid only after heating. There’s no drama, just reliable results. Kids grind it, heat it, and watch glassware cloud up with dogged persistence as potassium hydrogen sulfate breaks bonds and releases what was locked away inside the sample.
Health and Safety
Safety gets overlooked until something goes wrong. Potassium hydrogen sulfate isn’t as scary as concentrated acids, but breathing in its dust or handling it carelessly isn’t smart. My early days in the lab drilled respect for goggles and gloves—habits that stick for life. Keeping chemicals in their place, away from food or bare hands, should always come first.
Pushing for Greener Options
Some industries keep looking for ways to cut down on chemical use, especially anything that creates waste. Potassium hydrogen sulfate produces less mess than strong acids, and it’s easier to handle in smaller, safer lab settings. Switching to smarter workflow tools or recycling chemical leftovers both help cut the impact a little at a time—small changes with big results.
Is Potassium Hydrogen Sulfate hazardous or toxic?
Everyday Encounters with Chemicals
Most people never buy potassium hydrogen sulfate on purpose. This white, powdery chemical shows up in certain labs and factories, but it doesn’t sit on grocery shelves or get dropped into soda for fun. Still, its harsh properties and the growing attention to lab safety make it worth talking about. After working with dozens of substances over the years, I’ve learned that “hazardous” can mean several things—skin irritation, toxic fumes, long-term health effects. Potassium hydrogen sulfate checks a few of those boxes.
What Happens After Contact
Potassium hydrogen sulfate can burn skin and eyes. If you breathe it in, the respiratory tract gets irritated. Ingesting it brings cramping, vomiting, and worse. These reactions stem from its strong acidity; it forms sulfuric acid on contact with water, which explains its caustic punch. The World Health Organization’s chemical safety data points out the way this acid eats away at tissue, which makes personal protective gear and eye shields more than cosmetic extras.
How Much Exposure Is Dangerous?
Some chemicals turn deadly with the tiniest whiff. Potassium hydrogen sulfate works more like an instant irritant. It won’t attack the nervous system the way cyanide does. People who work in chemical plants or academic labs tend to watch out for skin and breathing exposure. Everyone I’ve spoken to in technical settings remembers getting a good safety briefing. A splash in the eye will land someone at the emergency station. The Centers for Disease Control and Prevention mentions that industrial workers must rinse immediately with water in case of contact.
Concerns Beyond the Lab
Industrial waste is another worry. Cities with lots of food-processing plants or chemical manufacturers sometimes report accidental releases of acids. Spills, even small ones, can destroy aquatic life and corrode pipes. Potassium hydrogen sulfate breaks down into sulfate and potassium ions, both found in nature, but the acidic byproducts can kill fish or damage crops if they aren't neutralized first. According to environmental agencies, regular monitoring and water treatment are the go-to fixes—these prevent contamination and keep rivers and soil from turning acidic.
Managing the Danger
Chemistry teachers and plant operators don’t trust luck. They train for spills and buy splash-proof goggles. I’ve seen strict rules enforced: never open large containers near drains, never store acids next to bases, always label every jar. Washing a chemical off right away makes all the difference. Emergency stations must stand at arm’s reach, not halfway down the hall. The National Institutes of Health classifies potassium hydrogen sulfate as hazardous, but not as a rare poison—just as a tough acid that’s easy to respect with the right habits.
Better Safety and Smarter Use
Every new worker hears the same warning: don’t underestimate a chemical just because it seems common. Potassium hydrogen sulfate pops up as a reagent or cleaning agent in labs, food processing, and wastewater treatment. These days, safety data sheets come loaded with detailed instructions, and workers keep up with the latest guidance. Smart handling and quick cleanup lower the risk. Wearing the right gloves and using chemical hoods stop accidents before they take hold. Responsible disposal and treatment keep the environment healthy, and everyone a little safer.
What is the chemical formula of Potassium Hydrogen Sulfate?
Real Science, Real Life
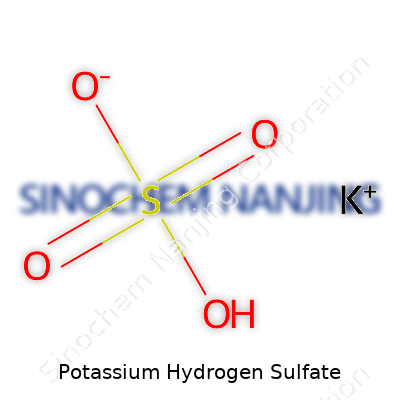
Potassium hydrogen sulfate balances on the edge of school chemistry classes and real-world industry. The compound’s formula, KHSO4, looks simple at first glance. Potassium (K), hydrogen (H), sulfur (S), and oxygen (O): each one has a distinct role, and the arrangement brings more meaning than a worksheet could show. In labs across the world, those four elements come together to serve more than textbook equations.
Everyday Impact: Why Knowing This Matters
In high school, most students memorize chemical formulas just to pass a test. Later on, the same formulas reappear in mining, food processing, and even sunscreen. I remember my early days in a university laboratory—sodium chloride and potassium chloride were everywhere, but potassium hydrogen sulfate came with stricter safety checks. One slip could throw off the pH. Its acid salt properties let it play referee between acids and bases, which turns out to be essential in several industries.
Beyond the Classroom
This compound doesn’t limit itself to the chemistry lab. Factories that manufacture fertilizers use KHSO4 to add potassium in a controlled, soluble form. The wine industry relies on potassium hydrogen sulfate when controlling acidity during production. Even the paper and dye industries turn to it for specific chemical reactions. Whenever I talk with folks running small food processing plants, they emphasize that KHSO4 lets them adjust tartness without heaps of additives. That’s a behind-the-scenes application many consumers never see.
Environmental and Health Factors
Chemical formulas like KHSO4 serve as shorthand for much bigger conversations about safety and environmental responsibility. Improper handling can release sulfur oxides or create waste that challenges water treatment operations. To protect workers, businesses use clear labeling, proper instructions, and gear—compliance isn’t just about regulations, it’s about keeping people healthy on the job. Schools train students to respect chemicals early on, making safety habits part of daily routines. That investment pays off for communities downstream, where runoff management keeps drinking water safe.
Building Trust with Science
Trust in chemical information builds over time. Open, clear labeling and reliable sourcing give scientists, teachers, and workers confidence in what they use. Potassium hydrogen sulfate with the formula KHSO4 needs a traceable supply chain—any impurity changes its reactivity. Once, during a project at a small lab, a mislabeled bottle led to hours wasted troubleshooting. Transparent documentation from reputable suppliers avoided issues in future batches, underscoring how trust in chemistry comes from experience and integrity.
Solutions and Progress
Clear communication about the properties and safe use of chemicals such as potassium hydrogen sulfate bridges the gap between scientific discovery and public trust. Educational programs help demystify formulas, showing students and workers how elements shaped our world—long before they filled a bottle on a factory shelf. With consistent monitoring and respect for environmental guidelines, the risks tied to KHSO4 decrease. That makes chemistry safer for everyone, from curious students to career professionals shaping tomorrow’s technology.
How should Potassium Hydrogen Sulfate be stored?
Understanding the Risks Up Close
Potassium hydrogen sulfate often lands in chemistry labs, classroom experiments, and various industrial mixing rooms. People usually treat it like another simple white powder, but experience quickly shows that ignoring the risks brings headaches and sometimes much worse. I remember grabbing a dusty, unlabeled jar in an old stockroom—turns out the cap had corroded from humidity and a light trace had etched nearby metal. It taught me that this isn’t an ordinary shelf item.
Choosing a Storage Spot That Makes Sense
Humid air doesn't get along with potassium hydrogen sulfate. Storing it in a dry place really cuts down on the chances of it clumping together or corroding nearby surfaces. Avoid rooms prone to leaks or wild swings in humidity. People sometimes shove containers at the back of a chemical closet, but airflow around the shelf helps more than packing it deep out of reach. Direct sunlight also heats things unevenly and can degrade packaging. So a cool, dim shelf works best—think simple, sturdy cabinets that leave room for some air and have a working lock.
Picking the Right Container
Plastic usually stands up to the acid nature of the powder better than metal. I’ve seen glass work, but once a glass jar chipped at the rim and dumped more on the bench than anyone needs. Screw-top HDPE bottles with clear labeling give the best shot at avoiding leaks or confusion. Labels need to show not just the name but hazard warnings and the date it entered storage.
What Goes Around: Keeping Chemicals Separated
This compound doesn’t just react with water. It can throw off sulfur dioxide or hydrogen sulfide if it tangles with the wrong materials. That means keeping it well away from any strong bases, cyanides, and certainly oxidizers. Boxes of old sodium hydroxide pellets nearby raise a risk; a small spill can mean a chemical cloud. Separate shelves or cabinets marked for acids and kept dry cut down on these dangers. Don’t trust random order—separation saves headaches.
Limiting Doorway Traffic and Access
Plenty of near-misses in labs come down to untrained hands reaching for things they barely recognize. Restricting access to trained workers makes sense, especially for students or new staff. Training doesn’t just mean going through a slide deck—real safety involves practice and respect. Checklists on the inside of cabinet doors remind regulars what matters, like keeping lids tight and checking for worn labels.
Regular Checks and Cleanups
Monthly rounds looking for crusty spills, damaged containers, or expired stock stop small problems from turning big. I once found a pool of unidentified powder under some bins—everyone assumed it was harmless until safety goggles fogged up from the acid fumes. Using a damp paper towel (never dry sweeping, which can spread dust) handles little spots, and any big incident calls for proper gear and disposal know-how.
Fast Facts and Smarter Storage
Potassium hydrogen sulfate causes burns, corrodes some surfaces, and spoils if left in damp places. Safe storage centers on dry, cool cabinets, sturdy plastic bottles with tight seals, good labels, and clear separation from incompatible supplies. No shortcuts pay off here—taking time to get this right keeps both people and workplaces safer.
What are the safety precautions for handling Potassium Hydrogen Sulfate?
Respecting Its Chemical Nature
Potassium hydrogen sulfate, sometimes seen in labs as KHSO4, runs as a strong acid salt. You’ll often find it playing a role in analytical chemistry and various industrial processes. I learned early on in my work that treating these types of acids without a healthy dose of respect leads to mistakes. Even if it doesn’t look threatening, those colorless crystals pack a punch—corrosive enough to irritate skin, eyes, and the respiratory system. The story always starts out the same: someone thinks gloves and goggles slow them down, but safety lapses only speed up trouble.
Personal Protective Equipment: The Non-Negotiables
Corrosive dust calls for more than rolled-up sleeves. Chemical goggles shield the eyes from splashes. I’ve seen even tiny bits of acid powder cause serious eye pain and blurred vision. Gloves—nitrile or neoprene work best—create a barrier that basic latex can’t always match. Cotton lab coats help, but anyone working with this stuff should zip on a chemical apron if there’s a spill risk. Never skip on closed-toe shoes either; drops on the feet can go unnoticed until the burn sets in.
During cleanup, the rules stay the same. If there’s dust in the air, respiratory protection becomes necessary unless strong ventilation is in place. Wearing one of those half-face elastomeric respirators with appropriate acid cartridges beats breathing trouble down the road.
Ventilation and Handling
Fresh air makes a difference. I remember one lab that skimped on fume hoods, and within minutes of opening a drum, the air stung my throat. Proper fume extraction, whether with a hood or local exhaust, keeps exposure low. Pour KHSO4 slowly, and don’t create a cloud of dust—fast motions only kick it up and spread it around.
Keep the workspace dry because this salt reacts with water, releasing sulfuric acid fumes. Control spills right away with a neutralizing agent—baking soda or lime does the trick, not water. Some places keep spill kits within arm’s reach because a neutralized mess is one you can mop up safely.
Storage Habits: Out of Sight, Never Out of Mind
Potassium hydrogen sulfate likes sealed, airtight containers—chemical-resistant types. I once saw a thin plastic bag left open, and within hours, humidity made it clump and release fumes. Store it away from bases, combustibles, and anything else that reacts violently with acids. Always keep the label clear and readable. Everyone in the lab or storage room should know exactly what’s inside each bottle; surprises rarely end well.
First Aid and Emergency Plans
Protection matters, but accidents still happen. If skin comes in contact, water flushes—quick and copious. Eyes demand the safety shower station be nearby and clear for emergency use. People sometimes hesitate to call for help fast enough. Take it from the ones who learned the hard way—doctors need full details on the chemical, so bring the label or safety data sheet along.
Training isn’t just a formality. Stay updated, take refreshers, and walk through spill drills as a group. Culture grows safest where knowledge is ongoing and everyone feels responsible for each other. Potassium hydrogen sulfate never takes a break; neither should safety.
| Names | |
| Preferred IUPAC name | Potassium hydrogensulfate |
| Other names |
Potassium bisulfate Potassium acid sulfate Potassium hydrosulfate |
| Pronunciation | /poʊˈtæsiəm haɪˈdrɑdʒən ˈsʌlfeɪt/ |
| Identifiers | |
| CAS Number | 7646-93-7 |
| Beilstein Reference | 1201393 |
| ChEBI | CHEBI:1310 |
| ChEMBL | CHEMBL1200572 |
| ChemSpider | 22860 |
| DrugBank | DB09453 |
| ECHA InfoCard | 100.029.729 |
| EC Number | 231-594-1 |
| Gmelin Reference | 56938 |
| KEGG | C14176 |
| MeSH | D011080 |
| PubChem CID | 24507 |
| RTECS number | TT2975000 |
| UNII | YG60L87T30 |
| UN number | 2460 |
| Properties | |
| Chemical formula | KHSO4 |
| Molar mass | 136.17 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.24 g/cm³ |
| Solubility in water | Very soluble |
| log P | -4.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.99 |
| Basicity (pKb) | 9.4 |
| Magnetic susceptibility (χ) | -41.0·10⁻⁶ cm³/mol |
| Dipole moment | 6.36 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 172.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1156 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1019 kJ/mol |
| Pharmacology | |
| ATC code | A12BA02 |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P280, P301+P312, P330, P305+P351+P338, P501 |
| NFPA 704 (fire diamond) | 3-0-2-Acidos |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD50 oral rat 2340 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 2340 mg/kg |
| NIOSH | NA0550000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended): 1 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium sulfate Sodium bisulfate Ammonium bisulfate |

