Potassium Formate: More Than Just a Chemical
Historical Development
Looking back at the evolution of potassium formate, it is hard not to see a story that mirrors many milestones of modern chemistry. Formates emerged as early players in organic synthesis after the discovery of formic acid in the 18th century. When researchers found out that swapping sodium or calcium for potassium made for a salt with unique properties, potassium formate started to attract interest beyond the lab. Folks began to see that its easy water solubility and gentle chemical footprint gave it an edge, especially in places where toxicity and corrosiveness create headaches. Potassium formate didn’t make newspaper headlines, but for chemists and engineers solving practical problems, this compound gradually built a reputation.
Product Overview
Potassium formate lands on shelves as a white, crystalline powder or kind of a syrupy liquid when dissolved. It looks unremarkable, honestly, and if you checked your average college chemistry storeroom, you’d probably pass it by. Under the surface, potassium formate packs surprising versatility. The combination of formate’s resilience and potassium’s ionic size offers advantages when tackling both environmental and technical jobs. In my own work, I’ve watched how its ability to lower the freezing point of water blows the usual road salts out of the water, making icy streets less hazardous and damage-prone.
Physical & Chemical Properties
Potassium formate dissolves into water fast, with the resulting liquid holding onto heat better than traditional alternatives — a crucial point for folks running district heating or hefty heating-cooling plants. The salt’s physical form lets technicians pour it, dose it, or mix it without much worry. The chemical stability means it stays put under typical conditions and won’t react with lots of other substances. The solubility, running over 300g per 100 mL of water at room temperature, gives it a broad range of industrial uses. This thermal stability, combined with a non-volatile nature, means lower risk during handling or accidents, based on my experience working with chemical floor spill scenarios.
Technical Specifications & Labeling
Specifications generally spell out purity levels, water content, particle sizes, and bulk density, matching what users in heat transfer or de-icing need. A container will identify the grade, concentration for solutions, and regulatory compliance. If used in oil drilling, these specs make sure mud blends won’t gum up expensive equipment. Markings on packages may include safety icons, storage tips, and concentration percentages, all straightforward so crews can work fast and not slow down for guesswork. This is about cutting risk on the job, not just achieving a paperwork target.
Preparation Method
You get potassium formate by reacting formic acid with potassium hydroxide. The process yields high-purity, water-soluble product without headaches from unwanted byproducts. Unlike some older chemical routes that required complex waste management, this synthesis creates a direct, manageable path from basic raw materials. Industrial setups use batch reactors, careful dosing, and routine filtration, but the core remains the same — a reaction most chemistry students learn but few appreciate for its industrial value. Engineers like it for the predictable yields and minimal environmental load, pointing toward sharper sustainability targets.
Chemical Reactions & Modifications
In the lab or plant, potassium formate takes part in reduction, buffering, and neutralization. It can act as a mild reducing agent in hydrogenation processes, and its formate ion often finds use converting dangerous nitrites into less toxic substances. Under heat or in catalytic conditions, it releases hydrogen, making it handy for some fuel cell research. Modifications involve shifting water content, blending with other salts, or tweaking particle size. For specialty applications, a little work adjusting granulation or blending it with calcium salts produces products that work harder in the field, like for fast-paced highway de-icing or special oilfield jobs. As someone who’s handled both research and operations, I can say this adaptability makes potassium formate stand out.
Synonyms & Product Names
People in the trade use names like potassium methanoate, formic acid potassium salt, or simply its chemical shorthand HCOOK. Specialists may spot “liquid ice-melt” or “formate brine” on industrial labels, especially where it comes premixed for heat transfer or road safety. Regulatory documents sometimes refer to it by CAS numbers, but anyone hands-on knows it simply as potassium formate. You’ll hear all these names thrown around on a works site or in safety meetings.
Safety & Operational Standards
Compared to traditional de-icers such as calcium chloride or sodium chloride, potassium formate lands as less toxic and less corrosive, offering lower risk for both handlers and the environment. Guides advise the usual gloves and goggles, and if it splashes, washing with plain water clears most worries. It isn’t classed as hazardous under most transport regulations. Big operators look at storage requirements, pointing to sealed containers and moderate temperatures, nothing out of reach for regular warehouses. From field experience, spills rinse away easily without damage to most surfaces, giving crews a break from heavy-duty cleanup.
Application Area
Potassium formate turns up where people value safety, low impact, and solid performance: airport runways, oil extraction, district heating systems, and laboratory syntheses. Airports shifted toward formate-based de-icers after seeing how much less harm they cause to aircraft metal and sensitive concrete. Oil companies blend potassium formate into drilling fluids where high pressure, high temperature, and low toxicity mean a safer drilling operation and easier well clean-up. I have seen heating companies swap out ethylene glycol for potassium formate brine, giving them an edge in thermal efficiency with fewer worries about leaks poisoning the groundwater. Even horticulture experiments have looked at its gentle impact on soils and plants.
Research & Development
Chemists and engineers still poke at potassium formate’s boundaries, hoping for better formulations and higher efficiency. Some labs explore its use in hydrogen storage, since it can release hydrogen through catalytic breakdown. Efforts focus on new additive mixes that cut costs or boost performance as a heat-transfer medium, letting companies walk a line between green credentials and industrial demand. Research teams test blends in cold climates, pushing to lower the freezing point without stacking up environmental contamination. Software-driven modeling helps engineers scale up from lab insight to tanker-loads. I’ve followed R&D projects chasing more eco-friendly drilling fluids and higher yield de-icers, and the pace reflects rising demand for sustainable solutions.
Toxicity Research
Toxicologists put potassium formate through a full battery of animal and environmental studies. They found its acute toxicity ranked far lower than typical road salts. It breaks down in water and soil to harmless substances, which local regulators appreciate, especially near sensitive water catchments. Drinking water contamination worries fade compared to calcium chloride, since potassium is an essential nutrient and formate degrades without leaving behind dreaded heavy metals or persistent organic pollutants. Field studies examining groundwater near heavily salted areas show reduced impact on aquatic life and crops when switching to potassium formate. Medical researchers looked for evidence of chronic toxicity, yet most data suggest occupational exposure stays below danger points with current work standards.
Future Prospects
Looking down the road, potassium formate’s story won’t slow down. Demand for de-icers with lower environmental costs pulls new users into the market. Expanding urban areas and rising climate unpredictability mean more icy surfaces looking for a fix, making its non-corrosive, less-toxic nature more appealing. Researchers eye new pathways that link potassium formate production to green hydrogen and bio-based feedstocks, hoping to trim energy costs and shrink the carbon footprint. Oil and gas drillers already look toward denser, cleaner brines as regulatory pressure clamps down on toxicity and waste. My sense is that potassium formate will find fresh ground in both old and new industries, driven by the same forces pushing all of us toward safer, cleaner ways to solve everyday challenges.
What is potassium formate used for?
The Face-Melting Truth About De-Icing
Potassium formate—a name that might sound complicated—often turns up during the toughest stretches of winter. I grew up in a northern town where heavy snow kept people busy with scrapers and salts all season. Communities put down traditional rock salt and calcium chloride on roads and runways. One day, a friend from the airport showed me tanks labeled “potassium formate.” That day, I learned it was their go-to for de-icing runways. Airports started using it so planes could land and take off safely, even as snow hammered down.
Here’s what makes it stand out: Potassium formate cuts through ice without corroding metal or damaging the concrete. Salt might keep your driveway clear, but it eats away your bumper and stains your boots. Potassium formate doesn’t do that. This helps cities keep expensive infrastructure in good condition. The less we spend fixing runways, the more we can invest elsewhere.
Heating Up and Cooling Down
Potassium formate puts in quiet work behind the scenes in factories. Engineers keep process equipment cool using special fluids. These fluids flow through pipes and transfer heat out of the system. Potassium formate solutions help with this job because they block freezing and offer a strong buffer against harsh temperatures. It’s tough, staying effective where other chemicals stumble—and it doesn’t create the fire risk or sticky residues that can cause headaches in large plants. Just knowing your warehouse won’t freeze up means safer days and cheaper bills.
Chemical companies, ice rinks, and indoor farming setups also rely on it. They choose it because it’s less toxic to staff, pets, and even the groundwater, compared to some other chemicals.
Better for the Planet and People
There’s always a trade-off, but potassium formate stacks up well. It biodegrades faster than standard de-icing salts. Fewer run-off problems means rivers and lakes don’t get hammered by salty waste every thaw. Salmon and trout downstream have a fighting chance. City trees don’t wither and die after snow season. People living near busy highways don’t have to worry so much about what’s running off their lawns into their gardens.
Still, it’s not perfect. Production takes energy, and prices can rise as demand picks up. If communities switch over completely, manufacturers have to find new ways to make it sustainably. Recycled feedstocks and energy-efficient systems would limit its carbon footprint. I believe supporting research here matters—climate change asks us to look for safer solutions, even when they cost more.
Looking Forward
One lesson sticks with me from that airport visit: New solutions get traction when they save money and work better for everyone. Potassium formate checks both boxes. Cities want safe roads; airports want reliable runways; families want their gardens and pets spared from harsh chemicals.
I often wonder if most folks know how this unassuming chemical keeps winter running smoother. By sharing these stories, we give smart science the spotlight it deserves—spurring more ideas for cleaner, safer communities and a healthier world.
Is potassium formate safe to handle?
A Closer Look at Potassium Formate
Potassium formate turns up in lots of industries. Crews use it for industrial deicing, oil drilling, and as a heat transfer fluid. It’s a salt formed from formic acid and potassium. Workers know it for being less corrosive than some alternatives. While many chemical safety stories get overblown, the way you handle potassium formate still makes all the difference. I’ve seen people brush off this type of chemical, expecting it to act like pure table salt. The truth sits somewhere between harmless and hazardous.
Straight Facts About the Hazards
No one likes dealing with harsh chemicals, but potassium formate doesn’t have the bite of something truly toxic. If you splash a little on your skin, it’s not going to give you a chemical burn right away, but after enough contact, skin dryness and irritation show up. Eyes stinging from a surprise splash? That’s not fun, and who hasn’t seen coworkers rush to the eye wash after a casual spill? Protection makes sense. If you’re working at a loading dock or lab, gloves and goggles aren’t just for show.
Some people get headaches or dizziness from inhalation, usually linked to dust or a heavy spill in an enclosed space. These aren’t made-up symptoms. With any powdered or highly concentrated form, good ventilation keeps those risks down. Go beyond opening a window — use an actual fume hood or a fan if you work with the dry salt. You can check any datasheet and see that potassium formate rates as low toxicity, but real-world safety still depends a lot on good habits.
Why the Right Training Matters
Maybe you’ve sat through a dozen safety briefings and wondered if the extra steps really matter. If you look back at recorded incidents, most problems happen not because the chemical was a villain in disguise, but because someone skipped the basics. I once witnessed a contractor handling potassium formate without any gloves, convinced it was only risky to drink. By the afternoon he had dry, irritated hands. Not the worst injury, but no one enjoys raw skin.
Responsible handling covers basics: gloves, goggles, and prompt clean-up after any spill. Washing up thoroughly — especially before eating or rubbing your eyes — keeps you safe. Anyone worried about the environment can feel better, too. Potassium formate breaks down in nature more quickly than many other salts used on roads, making it less of a threat to aquatic life and groundwater. Even so, dumping large quantities into drains or soil creates problems in the local ecosystem, so disposal protocols matter.
Learning From Past Mistakes and Better Solutions
Every time I hear someone ask if potassium formate is “really safe,” I remember the stories of mishaps that could have been avoided with five extra minutes of care. Training can’t just be a checkbox on a compliance form; it needs buy-in. Supervisors should walk everyone through the real risks, not just hand out gloves and hope for the best. Encouraging people to look for signs like irritated skin or coughing — and to speak up if they see a spill — builds a stronger safety culture than any poster on the wall.
Looking ahead, investments in better storage systems and automated mixing cut out a lot of manual handling. More companies are finally moving to sealed systems so the chance of spills and airborne dust count for less. Combined with strong oversight, this keeps the already low danger of potassium formate even lower.
Potassium formate has a place in industry, but its real safety comes from informed, steady habits and a willingness to respect it as more than just salt. Stay smart, and the risks stay small.
What are the main properties of potassium formate?
What Makes Potassium Formate Stand Out
Potassium formate, with the formula HCOOK, pops up more than most people realize. Engineers keep it on hand for tough winters, blending it into deicing solutions for runways and highways. Formic acid’s reaction with potassium hydroxide creates crystals that dissolve easily in water. To many workers handling it, the item stands out because it refuses to clump or cake under normal storage. Its absorption of moisture from the air calls for tightly sealed containers, but with a little care, it stays pourable and ready.
Melting, Mixing, and Managing Heat
Anyone working with ice in winter knows that potassium formate solutions drop the freezing point of water far lower than old-fashioned salts can manage. That matters for airports in cold climates. Unlike sodium chloride, potassium formate does its job with less risk of corroding nearby metals and causing lasting damage to concrete runways. This makes it useful not just for safety but also for prolonging the lifespan of expensive infrastructure. Thermal stability gives another advantage—this compound resists breakdown from temperature swings. Safety teams can store it through summer heat and winter chill without much worry.
Environmental Footprint and Human Safety
What stands out to me is the focus scientists have given to potassium formate's impact on the world beyond the jobsite. High solubility means workers don’t have to deal with leftover residue sticking to equipment or clogging drains. Research from European chemical agencies shows this compound breaks down in soil and water, causing little trouble for plants or aquatic life. Compared to alternatives on the market—notably, urea—it loads far less nitrogen into local water systems, lowering the risk for algae blooms or fish kill events. Safety data sheets remind handlers to avoid direct contact or inhalation, but its toxicity rank is low, reducing the toll on people exposed during transport and use.
Oilfields, Factories, and Unexpected Uses
Over the years, clients in the energy sector taught me potassium formate rarely stays stuck in just one industry. Deep drilling operations count on potassium formate brines as part of their toolbox. These brines steady high-pressure wells without damaging reservoir rocks or interfering with oil and gas flow. Drillers tell me that this chemical’s density gives them a way to control downhole pressure precisely, reducing risks during critical operations. Chemical plants take another tack—using potassium formate in some liquid processes or as a buffering agent. The most practical operators appreciate the dual benefits: strong action and a relatively light touch on equipment.
Supply, Storage, and Future Outlook
From my experience tracking chemical logistics, potassium formate benefits from an accessible supply chain. Manufacturers across China, Europe, and North America produce it in bulk, sending it out as granules, powders, and already-mixed liquid blends. Warehouses keep it separated from acids and oxidizers, but most teams find it stores well for the long haul. The push for sustainable products continues to raise demand, especially in large cities looking for safer winter road treatments or companies pursuing greener drilling fluids. Although potassium formate costs more up front than salt, municipalities investing in less maintenance and fewer repairs see payback through cleaner waterways and smoother surfaces.
Looking Ahead
Chemistry may seem distant to most people, but the decisions around what compounds like potassium formate get used ripple out. Industries that take the step toward friendlier deicers or safer drilling fluids—and everyday citizens who take those benefits for granted—rely on a clear understanding of what something is and what it can do. Potassium formate’s blend of safety, strength, and environmental care earns it an important place in the conversation about smarter, cleaner choices for next season and beyond.
How is potassium formate stored and transported?
Real-World Storage Practices
Potassium formate pops up often in the oil and gas industry, especially for drilling fluids. Experience has showed that it arrives mostly as a clear, water-like solution or as a dry powder. As someone who’s visited chemical warehouses and drilling sites, the storage end looks simple enough if you follow a few basic rules.
Bulk liquid potassium formate comes in tough polyethylene or stainless steel tanks. There’s no need for fancy insulation, but workers steer clear of strong acids, oxidizers, and anything that could turn a spill into a bigger mess. Labels, locked gates, and restricted keys help stop mistakes. Smaller drums and totes sometimes land in covered yards or ventilated sheds to keep out heat and rain. On a hot summer day with the right breeze, leaving chemical drums out in the sun is playing with fire—not literally, since potassium formate isn’t flammable, but heat does help leaks and corrosion keep security guards busy.
In powder form, potassium formate sits in double-lined bags, often packed in cardboard boxes. Moisture can clump or dissolve the product, so manufacturers urge use of dry storage rooms with pallets kept off concrete. If I’ve learned one thing, it’s that nobody likes shoveling a sticky heap off a damp floor. Big plants set up separate areas for surfactants or solvents nearby, never mixing potassium formate with incompatible chemicals. Otherwise, insurance agents start asking questions even faster than safety inspectors.
Challenges in Transporting Potassium Formate
Getting this chemical from point A to B means dealing with rules set by the US Department of Transportation and global agencies. Tanker trucks, railcars, and ISO containers all serve the job. Before anyone loads a pound of potassium formate, paperwork sets chemical concentration, hazardous class, and safety procedures. Drivers hauling liquids check sealing valves and test drums for hidden cracks that could cause spills on the highway. If traveling long distances, liquid form sees insulated containers during the winter to avoid freezing up. Frozen product slows unloading, which nobody appreciates on a deadline.
As for powders, companies use moisture-proof packaging and keep lined trucks or dry vans on call. Once, after a rainy stretch, I noticed powder bins arrive caked together in one giant brick. That only happened once, thanks to closer eye on the truck’s door seals from then on. Clean trucks also prevent cross-contamination—a stray oil residue will ruin a batch in no time.
Why the Details Matter
Missed steps in storage and transport lead to expensive downtime. Spills, caked powder, or fouled liquids can halt production. More than once, companies faced fines for overlooked training or improvised storage. Regulators like OSHA and the EPA keep a close watch for good reason. Potassium formate itself won’t catch fire or explode, but mixing with the wrong chemical stack risks releasing formic acid fumes or damaging containers. Having seen more than one rushed crew skip a shelf check and come back to find a leak, getting the basics right looks like the safest bet.
Solutions start with sturdy containers, routine inspections, and tight controls on who handles chemicals. Every warehouse manager I’ve admired treats chemical storage as a process, not an afterthought. Clear instructions, dry loading docks, and regular safety drills go a long way. For anyone new to process industries, learning the practical side—not just the paperwork—keeps both people and product safe.
Final Thoughts on Keeping Potassium Formate Safe
No single method solves everything, but experience and steady routines offer the best defense against preventable mishaps. Storage and hauling rules come from past accidents—not theory. The wise path means treating every delivery with respect and checking for problems before they escalate. With chemical handling, small steps stack up to big savings in the end.
What industries use potassium formate?
The Oil and Gas Field
Whenever the drilling season rolls in and heavy equipment rolls out to remote sites, operators count on fluids that won’t clog pipes, cause corrosion or foul expensive hardware. I’ve seen potassium formate at play in this business. Drillers use it to make high-density brines for oil and gas wells. It performs better than sodium or calcium-based brines, mainly because it’s less corrosive and offers slicker flow without causing chemical headaches down the line. Since the molecule doesn’t attack metals, pumps last longer and site maintenance costs stay controlled. In offshore drilling, especially where zero-discharge rules stand, workers trust potassium formate since it shows a milder profile if it reaches the water. A safer option that delivers the weight and clarity needed for deep and tough drilling.
De-icing and Road Safety
Every winter storm in northern cities brings trucks spraying de-icers. Traditional salt tears up roads and finds its way into lakes and rivers, damaging aquatic life. Municipal crews in places like Scandinavia have turned to potassium formate in their liquid mixes. This solution clears runways and roads but doesn’t eat away at concrete or vehicles as sodium chloride does. Shoes, pets and groundwater see less fallout from its contact. While more costly than standard salt, it reduces repairs to public infrastructure. From my own travels, airports that make the switch report fewer delays and less corrosion damage to jet engines and landing gear.
Heat Transfer Fluids
Potassium formate shows up in chillers and refrigeration setups where engineers want efficient cooling without hazardous fallout. Food logistics firms depend on cold-chain reliability, and regular brines freeze solid at lower temps. Potassium formate-based fluids resist freezing, keeping pipes and compressors safe from ice blockages. For companies aiming to lower environmental risks, this solution scores points—if any leaks happen, they don’t create major chemical cleanups. Cold storage warehouses and hockey arenas often use these fluids beneath the surface, extending equipment lifespan and keeping energy bills in check.
Industrial Cleaners and Detergents
From car washes to institutional laundries, potassium formate gets mixed in for a boost. The salt works as a pH regulator and a quick, biodegradable cleaner. I’ve worked in facilities cared about both performance and their wastewater streams. Instead of sending harsh chemicals downstream, plant managers select potassium formate for its lower aquatic toxicity. Formulators in Europe have phased out more aggressive substances in favor of choices like this that break down faster and don’t stick around in river sediments.
Chemical Synthesis
Chemists treat potassium formate as a useful starting block and reducing agent for manufacturing certain products. Pharmaceutical plants and specialty chemical manufacturers feed it into step reactions where it helps transform raw materials into more complex molecules. In electronic industries, it occasionally appears in tech manufacturing—the kind where perfectly clean surfaces and fine-tuned reactions mean the difference between high yields and costly waste. The reliability and mildness of this salt help firms meet strict quality criteria, which keeps the process safe and predictable.
Balancing Cost, Safety, and Environmental Needs
Through all these sectors, one thing stands out: potassium formate fills a spot where reliability and lower environmental risk matter. With rising pressure to run cleaner operations, possible solutions should hit several marks—safety, recycling, and long-term cost. Customers keep moving toward options that solve more than just the immediate task. By adapting production strategies or updating procurement policies, businesses in these sectors can shape a safer future for their workers and the communities around them. Governments and buyers who fund these industries carry the conversation even further, pushing for choices that encourage both progress and stewardship.
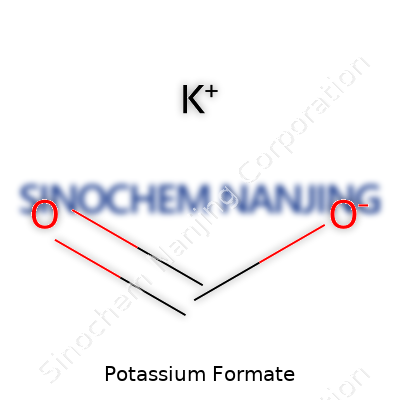
| Names | |
| Preferred IUPAC name | Potassium methanoate |
| Other names |
Potassium methanoate Formic acid potassium salt Potassium salt of formic acid |
| Pronunciation | /pəˈtæsiəm ˈfɔːr.meɪt/ |
| Identifiers | |
| CAS Number | 590-29-4 |
| Beilstein Reference | 3561416 |
| ChEBI | CHEBI:63315 |
| ChEMBL | CHEMBL1201550 |
| ChemSpider | 71397 |
| DrugBank | DB14511 |
| ECHA InfoCard | EC 209-677-9 |
| EC Number | '209-677-9' |
| Gmelin Reference | Gmelin Reference: 1768 |
| KEGG | C18697 |
| MeSH | D019277 |
| PubChem CID | 23674749 |
| RTECS number | RR0350000 |
| UNII | 6G8W6S4L8E |
| UN number | UN 3474 |
| Properties | |
| Chemical formula | KHCO2 |
| Molar mass | 84.115 g/mol |
| Appearance | White crystalline solid or colorless liquid |
| Odor | Odorless |
| Density | 1.56 g/cm³ |
| Solubility in water | Miscible |
| log P | -3.6 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.75 |
| Basicity (pKb) | 8.7 |
| Magnetic susceptibility (χ) | `-30.6·10⁻⁶ cm³/mol` |
| Refractive index (nD) | 1.370 |
| Viscosity | 1.9 cP (20°C, 52% solution) |
| Dipole moment | 1.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 95.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -657.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -567 kJ/mol |
| Pharmacology | |
| ATC code | B05XA17 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | Keep only in original packaging. Wash hands thoroughly after handling. Avoid release to the environment. |
| NFPA 704 (fire diamond) | 1-0-0 |
| Lethal dose or concentration | LD50 (oral, rat): 5500 mg/kg |
| LD50 (median dose) | LD50 (median dose) for Potassium Formate: Oral-rat LD50: 5500 mg/kg |
| NIOSH | SWG |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Potassium Formate: Not established |
| REL (Recommended) | 400 mg/L |
| Related compounds | |
| Related compounds |
Formic acid Potassium acetate Sodium formate Calcium formate Potassium carbonate |

