Potassium Cyanate: A Closer Look through Science and Industry
Historical Development
The story of potassium cyanate traces all the way back to the early 19th century lab benches. Friedrich Wöhler, known for breaking the vitalism barrier, managed to synthesize urea using ammonium cyanate — a process unthinkable at the time for connecting simple inorganic salts to organic life itself. This work didn’t just open doors for potassium cyanate; it shifted thinking about biological chemistry. Potassium cyanate’s history always extends beyond textbooks, showing how accidents and bold ideas in chemistry can end up laying groundwork for future biotech, pharmaceutical, and polymer advancements. For students and scientists, remembering this helps keep the curiosity alive and puts the reagent in a context that’s bigger than just lab work.
Product Overview
Potassium cyanate has earned a spot as a curious staple in both organic synthesis and industrial efforts. The formula KOCN makes it easy to remember, and most people recognize it as a white, powdery solid. Laboratories using this compound often value its consistent behavior, since it shows up as a reliable nucleophile and a go-to agent for certain substitution reactions. Companies might encounter it in the manufacture of herbicides, pharmaceuticals, and even specialty polymers. Laying hands on a bottle of potassium cyanate means dealing with a product rooted in two centuries of creative experimentation.
Physical & Chemical Properties
Potassium cyanate looks simple enough at first glance, presenting as a white powder that dissolves readily in water. This solubility helps in many lab-scale reactions where uniform distribution in a solution matters. With a melting point over 300 degrees Celsius, it stands up to a fair bit of thermal stress, which is useful in industrial settings where heating can’t be avoided. What stands out about its chemistry is the difference between its cyanate group and more infamous cousins like cyanide. Potassium cyanate acts as a less toxic, more stable alternative, which allows safer handling in many processes. But that doesn’t mean it should be taken lightly, since improper mixing or heating in open air can stir up risk.
Technical Specifications & Labeling
Labels on potassium cyanate bottles reflect careful regulatory standards. Typical packaging lists the CAS number 590-28-3 and identifies the hazard warnings and handling instructions. These resonate with the safety culture in chemical work, and regulatory rules pull no punches — anyone storing or using it needs to meet local and international storage controls. Shipping, receiving, and working with potassium cyanate leaves little room for error, and that’s for the best. Reading these labels becomes second nature for anyone pulling lab reagents from the shelf, and this form of routine vigilance beats complacency every time.
Preparation Method
Most potassium cyanate in circulation comes from direct synthesis involving potassium carbonate and urea. Heating this blend produces the cyanate alongside carbon dioxide and ammonia; those side gases tell a story about reaction pathways and careful balancing acts in the reactor. Labs equipped with the right glass and ventilation find this route straightforward, but it still demands consistent monitoring. The sensitivity of this synthesis to heat and moisture leaves little room for distraction during scale-up, and any deviation might waste resources or create unwanted byproducts. Here, experience plays just as important a role as theory, because every batch teaches a lesson about thermal management and process discipline.
Chemical Reactions & Modifications
Potassium cyanate’s real value shows up in its ability to introduce the carbamoyl group into organic molecules — this property delivers a foundation for synthesizing ureas, semicarbazides, and hydantoins. Pharma chemists especially enjoy exploiting these reactions for drug intermediates. In the lab, dropping potassium cyanate into a mix with ammonium salts or primary amines fires off reactions that convert basic building blocks into more complex ones. I’ve watched research teams coax new behaviors out of this basic salt by pairing it with transition metal catalysts, opening up creative paths for isocyanate chemistry without pulling in nastier alternatives. Any tweaks or attempts to substitute potassium cyanate often run into tradeoffs between safety, scalability, and cost — all points that labs weigh every time a project shifts focus.
Synonyms & Product Names
Potassium cyanate crops up under a scatter of other names. Some call it Cyanic acid, potassium salt. Others refer to it simply as Kocn. Chemical catalogs don’t hide behind brand names for basic reagents, but it pays to double-check, since some older texts or international labels still use synonyms from the past century. That confusion in naming can tangle ordering systems or mislead new lab staff, so consistent training and attention help in keeping procurement tight and inventories correct.
Safety & Operational Standards
No responsible chemist shrugs off the risks tied to potassium cyanate. Even though it’s less toxic than potassium cyanide, its potential for releasing cyanide under acidic or heated conditions calls for gloves, safety glasses, and a properly vented workspace. Inhalation, skin contact, and accidental releases have consequences — not just for people but for lab environments and waste streams. Procedures go beyond simple reminders, pushing for regular safety drills and more effective chemical hygiene protocols. I’ve seen that even veterans in the lab take another minute to read Safety Data Sheets. Spills, leaks, and fires tied to potassium compounds often involve first responders and environmental crews, so good labeling and emergency training pay off every day.
Application Area
Potassium cyanate serves several industries. In pharmaceuticals, it sits at the root of urea-based drug intermediate syntheses. Agrochemical manufacturing leans on its behavior for certain herbicidal pathways, where efficiency and stability shape pricing and availability. Polymer chemists reach for potassium cyanate or related salts to produce specialty materials with tailored physical properties. In academic settings, it remains a teaching aid — perfect for illustrating concepts from nucleophilicity to basic polymer chemistry. Seeing the compound in so many sectors reflects both its utility and the constant drive in chemistry to simplify connections from bench to product.
Research & Development
Researchers keep poking at potassium cyanate, seeking new reactions and safer uses. Green chemistry circles have targeted its preparation, looking for ways to cut out harsh conditions and lower waste. Some teams focus on catalytic improvements, searching for cheaper, easier routes for introducing carbamoyl moieties without generating excess salts. Analytical chemists investigate sensory methods and detection limits, since tighter monitoring means safer workplaces and better environmental controls. As a research substrate, potassium cyanate remains appealing because it’s both stable enough to store and reactive enough for a range of creative tests.
Toxicity Research
Toxicologists know potassium cyanate requires respect. Acute toxicity sits below that of cyanides, but chronic exposure can’t be ignored. Animal studies draw lines between doses and nervous system impacts, while larger studies look for reproductive effects and subtle biochemical shifts from repeated low-level exposure. Wastewater engineers pay close attention to degradation products, since incomplete disposal opens doors for cyanide release. I’ve seen debates at conferences about how much regulation should focus on immediate handling versus downstream risk, and the changes in guidance from one country to another often spark more research. Regular review of published data supports both safer handling and more thoughtful environmental policies.
Future Prospects
Potassium cyanate may look old-fashioned, but there’s plenty of life left in its chemistry. Labs hungry for safer isocyanate alternatives keep running experiments on more benign synthesis pathways. Recycling and reclaiming cyanate salts shows promise for slashing process costs in big chemical plants. As the push for sustainable chemistry grows, companies search for tighter controls on cyanate formation and decomposition. Advances in catalysis and reaction engineering stand ready to unlock new ways of using potassium cyanate, from custom pharmaceuticals to greener crop solutions. This simple salt reminds us that even established compounds have room for innovation if research keeps driving forward.
What is Potassium Cyanate used for?
Why Potassium Cyanate Matters in Chemistry
I remember seeing potassium cyanate for the first time in a university chemistry storeroom, tucked on a shelf with other slightly intimidating chemicals. Most people don’t bump into this stuff unless they work in a lab or a factory. Chemists and researchers reach for potassium cyanate because it plays a key role in some important chemical reactions, especially in making other compounds that fuel bigger discoveries, medicines, and even crop protection.
Crafting Medications and Exploring Science
Drug research teams often use potassium cyanate to build useful molecules. In pharmaceutics, it’s known for helping synthesize compounds called ureas and semi-carbazides. These chemicals show up in some medicines and also help researchers test how drugs behave in the body. Hospitals and clinics want better, safer treatments—this is one small building block that often sits at the start of a new medicine’s journey.
Potassium cyanate also helps scientists unravel the secrets of enzymes and proteins. Protein chemists employ it to turn proteins into their “carbamylated” forms. Watching how proteins behave after carbamylation can shed light on diseases like kidney failure, where this exact modification takes place in the body. This opens the door to better tests and more accurate understanding of what goes wrong in diseases.
Workhorse in Manufacturing
Factories lean on potassium cyanate for several down-to-earth tasks. Metal treatment plants use it to toughen and harden steel, so car parts, tools, and heavy-duty machinery last longer. Cyanate salts change the chemical structure of steel at the surface, leading to equipment that runs smoother and breaks down less. One machine breaking mid-shift can halt an entire line. Fewer breakdowns means more steady paychecks for workers and fewer delays for buyers.
Potassium cyanate also shows up in the production lines for herbicides. Crop yields rise when weeds stay down. Modern agriculture depends on chemicals that stop weeds yet let corn, wheat, and soybeans thrive. The base materials for some popular weed killers start with this very compound.
Safety and Handling Concerns
The risks with potassium cyanate can't be ignored. Like other cyanate or cyanide-related materials, it deserves careful handling. Touching or inhaling it by mistake can harm people, so factory floors and labs require training, gloves, and goggles. I’ve seen strict protocols put in place: spill kits near every workstation and emergency response steps posted on walls. Shops selling chemicals have to track who buys and how much leaves the shelf.
People often ask if potassium cyanate poisons waterways or the environment. Unlike potassium cyanide, which raises alarm bells immediately, potassium cyanate breaks down faster and causes fewer long-term problems—yet spills or poor disposal can do harm. Local governments and international agencies watch over chemical handling closely. The right laws and careful workers keep both people and soil safe.
Looking Toward Alternatives and Better Practices
Over the years, chemists and engineers have aimed for cleaner, safer alternatives when possible. Some factories now use less hazardous chemicals for hardening steel or formulating medicines. Researchers keep experimenting with greener routes, like shifting to less dangerous reagents or inventing processes that generate less waste. Anyone who’s worked in a lab knows the power of curiosity and caution working together. Continuous improvements, effective training, and strong oversight all help balance productivity with safety and environmental health.
Is Potassium Cyanate dangerous or toxic?
Understanding the Substance
Potassium cyanate sometimes pops up in unexpected places. I’ve seen it listed in textbooks, talked about in classrooms, and even used in certain specialized industries. People who spend time in chemistry labs might recognize it as a white, odorless salt. Most folks don’t come across it during their everyday lives, but the name alone is enough to spark questions. I'm not shy to say that seeing “cyanate” on a label makes my own radar go off. Even if handled safely in science, few want these chemicals near their communities or homes.
How Potassium Cyanate Behaves in the Body
This substance can spell trouble if swallowed, spilled on the skin, or mixed the wrong way in the lab. It reacts with water, releasing cyanate ions. Given the “cyan-” family reputation, with cyanide notorious for its acute toxicity, a lot of concern focuses on health effects. Once it's in the body, potassium cyanate can break down into cyanide under acidic conditions. Several studies show exposure irritates the eyes, skin, and respiratory tract. Swallowing larger amounts creates headaches, nausea, confusion, and muscle weakness — and could hit much harder, depending on dose.
Risks in Workplaces and Labs
Anyone handling potassium cyanate in factories, laboratories, or universities needs to stay sharp and work under solid protocols. I remember friends in research who always wore gloves, goggles, and worked under fume hoods. Gloves aren’t just for show. Skin contact leads to redness and irritation. Inhaling dust shouldn't happen working with proper ventilation, but accidents happen in labs every year across the country. According to the U.S. National Institute for Occupational Safety and Health, even moderate contact can pose hazards.
Long-term, I’ve read evidence connecting heavy, repeated exposure to changes in metabolism and issues for the nervous system. Those handling it daily are encouraged to watch their health and avoid casual handling. Companies keep detailed safety sheets on site and train staff to contain spills right away. Hospital emergency personnel keep up-to-date with antidotes, should accidental ingestion or exposure occur.
Environmental Concerns and Safe Disposal
Down the drain isn't an option. Cyanate salts, in high quantities, hurt aquatic life and build up in the environment. Researchers at Cornell University outline how improper disposal can contaminate water. For communities near research centers or plants, environmental monitoring and transparency matter most. Scientists can neutralize the substance in controlled ways, using basic solutions to make it safer before disposal. Ordinary folks don’t have those setups at home.
What Makes Potassium Cyanate Worth the Precautions
Some might wonder why chemists keep potassium cyanate around, given the risks. As someone fascinated by new medicines, it’s clear that chemists use it to create treatments or research new compounds. It plays a part in making organic chemicals, herbicides, and a few specialty drugs. While the benefits are real, responsible oversight can't get overlooked. Too often, the news carries stories of accidental poisonings by improper storage or unsupervised use.
Solutions and Good Practices
Workers and students need hands-on safety training. Laboratories require working eye washes, gloves, and exactly labeled containers. Community education plays a part too. Every school should teach the basics of hazardous materials and what to do if an accident occurs. Local authorities must regularly inspect industrial sites and share inspection records publicly. Disposal companies need clear rules, verified by local governments, to avoid shortcutting the process. Manufacturers must keep up routine reviews of procedures. Many disasters start with tiny missteps: loose lids or confusion about labeling.
Potassium cyanate won’t become less dangerous with time, but common sense, science-based regulations, and transparent communication go a long way in keeping people safe.
How should Potassium Cyanate be stored?
Getting Real About Chemical Safety
Most people never need to think about potassium cyanate. Those working in chemistry labs or handling specialty chemicals know all too well that common sense must meet science. Potassium cyanate offers a unique set of risks that deserve proper respect. Storage routines for this compound directly impact both workplace safety and public health. I learned this lesson early while volunteering in a university research lab, watching a careless colleague spill a small sample. The resulting panic—alarm, hasty mop-up, and a round of stern warnings—showed how quickly things can turn dangerous if safety gets ignored.
Storing Away from Trouble
Keeping potassium cyanate in simple containers puts too many people at risk. Airtight, labeled bottles crafted from glass or high-grade plastic give a sturdy line of defense. Metal containers invite trouble, since cyanate can corrode certain metals, releasing compounds you don’t want to breathe or touch. I’ve watched safe practices save labs from headaches many times—clear labeling and segregation of chemicals go a long way. Always keep potassium cyanate far from acids. Even trace moisture paired with acid can produce toxic cyanide gas. Cross-contamination caused more than one incident in my days at a shared school stockroom; clear signage and dedicated shelves ended the emergency calls.
Environmental Conditions Matter
Potassium cyanate wants a cool, dry, stable environment. Direct sunlight or humidity can break it down, increasing the risk of disastrous chemical reactions. Do not trust a stuffy closet or makeshift cabinet. Professional-grade chemical storage cabinets with ventilation and spill protection cut risk. Even at home, basic awareness helps—looking at temperature, humidity, and air flow, to minimize accidents.
Securing What Matters
No chemical should sit where unauthorized hands can find it. I once heard about a janitor accidentally moving bottles around because they looked just like cleaning products. Small lapses like that rapidly become emergencies. Locks, access logs, and employee training stop mishaps before they start. In research settings, routine inventory checks keep things from going missing or being stored too long.
Tackling Emergencies—Preparation Shows Respect
Some folks think certain chemicals like potassium cyanate can simply be left alone, but unpredictability becomes a problem if no one prepares for the worst. Emergency eyewash stations, ventilated storage areas, spill kits, and clear plans matter as much as the original safe storage. I’ve seen busy labs skip regular safety drills—until an accident forced everyone to scramble. Practicing and running down checklists must become habit.
Standard Guidance—Why Rules Exist
Organizations like OSHA, NIOSH, and the CDC lay out strict guidelines for a reason. Following these doesn’t earn style points, but I’ve seen their wisdom in real life. Sharp labeling, careful segregation, strong ventilation, and a practical approach to access combine to keep both newcomers and seasoned chemists safe. No amount of experience replaces consistent applications of these standards. A smart storage plan means fewer accidents and healthier lives for everyone working close to hazardous chemicals.
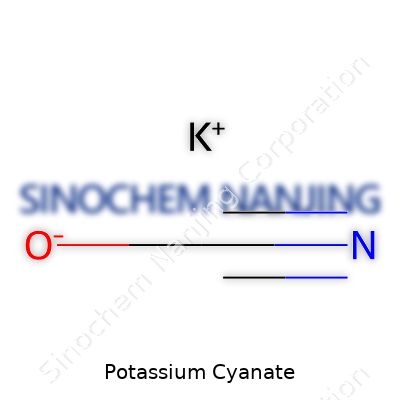
What is the chemical formula of Potassium Cyanate?
Breaking Down the Formula
Potassium cyanate shows up in labs and classrooms as KOCN. This simple string of letters hides a complex story. The K stands for potassium, O is for oxygen, and CN points to the cyanate group. That’s it—three elements, each taking a distinct role. Scientists memorize this formula early, but for most people, it looks like alphabet soup. Once you see how those atoms work together, the bigger picture comes into view.
Where Potassium Cyanate Fits in Real Life
Besides popping up on chemistry exams, KOCN supports several industries. Pharmacies rely on it for making medicines. Factories use it to treat metals and to build chemicals that protect crops. I worked a summer job in a plant that handled substances like this. A lot of our safety routines came down to knowing formulas. Sometimes, just getting a label right kept disasters from happening. That gave me real respect for the details.
Why the Formula Deserves Attention
One reason potassium cyanate turns heads is the confusion around its name. It sounds like potassium cyanide—a whole different beast. People hear “cyanate” and worry about poison. KOCN isn’t as dangerous as cyanide, but it isn’t sugar water either. Safety data shows it can irritate skin and lungs. Knowing its formula means you can look up how to store, handle, and neutralize it. Mistaking one compound for the other could end up causing harm, especially at schools or small labs.
What Science Teaches Us About Simplicity
KOCN’s formula reminds me how small changes create huge differences. One extra atom, and you have a chemical with whole new properties. Education works the same way. Deep understanding grows from simple facts. In high school, my science teacher would drill these formulas, repeating until they stuck. Years later, I see how they stick around not just as trivia, but as tools for safety and discovery.
Trust Through Clear Information
Sharing solid information supports trust. Manufacturers list the chemical formula so workers know what they’re dealing with. Emergency responders check formulas when handling spills. The more accurate and accessible this info stays, the safer communities can be. Potassium cyanate may seem obscure, but honest labeling and good science communication protect lives and the environment. That’s a public service that matters, whether you blend chemicals or just hope your water stays clean.
Better Solutions for Safety and Learning
Access to trustworthy data helps everyone, not just experts. Labels should state KOCN and give warnings that match the real risks. Students and workers need training that goes beyond memorizing symbols. Hands-on demonstrations show why these formulas deserve careful attention. Open channels between scientists, educators, and communities make for more informed decisions. Sometimes safety guidelines lag behind new discoveries, so feedback from people working on the ground speeds up updates that keep us protected.
Moving Toward Safer Chemistry
It all starts with a few letters. KOCN shows how chemistry ties together classrooms, labs, agriculture, and industries. My own work experience made me realize one missed letter can end up on the evening news. So take the formula seriously, keep asking questions, and don’t hesitate to double-check before mixing anything unfamiliar. Informed choices save both time and lives.
What are the safety precautions when handling Potassium Cyanate?
The Real Risks at Hand
Anyone who has ever watched a drop of potassium cyanate hit water knows that things go from calm to questionable pretty fast. This chemical doesn’t have bright warning colors, but it brings a hefty danger if people treat it like baking soda. Inhaling dust, letting it touch skin, even handling the bag wrong – all of it opens the door to poisoning. Cyanate compounds break down and release cyanide, a toxin that stops cells from using oxygen. If someone starts coughing, showing dizziness, or gets short of breath after exposure, the link should pop up right away.
Why Gloves and Goggles Aren’t Just for Show
Stories of lab accidents stick around for a reason. Back in graduate school, a friend forgot to grab gloves for a “quick weigh out.” He only realized the trouble later – itchy skin, that creeping sense he’d made a mistake. Wearing chemical-resistant gloves, splash goggles, and a proper lab coat isn’t just some bureaucratic rule; it’s about keeping sharp with a chemical that doesn’t care about shortcuts. Nitrile gloves stop direct contact, and goggles shield eyes from splash or dust. Without them, it’s open season for skin rashes or even far worse problems.
Ventilation Isn’t Optional
Open windows don’t cut it. Good laboratory practice demands a chemical fume hood for potassium cyanate. The dust acts like it wants to sneak up your nose or get into your lungs, and a bench in the open won’t stop it. A fume hood pulls the vapors and dust away, sending them into a filtration system. A once-over for fume hood airflow before uncapping anything pays off far more than worrying about exposure after the fact.
What Happens If Exposure Occurs
It’s easy to freeze—no one likes to admit they messed up. Still, running straight for an eyewash or safety shower trumps embarrassment. If powder touches skin, a full fifteen-minute rinse with water is the only play. Swallowing potassium cyanate or breathing in a cloud needs urgent hospital care, no exceptions. My own rule in the lab: Always keep emergency contacts taped to the wall, and don’t work alone. People work faster when they’re sure someone has backup and knows the drill.
Handling, Storage, and Disposal: A Non-Negotiable Routine
Potassium cyanate thrives on carelessness. Keep containers sealed, dry, and away from acids that could create hydrogen cyanide gas. Think of every label double-check as insurance against a ruined day. Working on a dedicated bench prevents accidental mixing or cross-contamination – labeling and color-coding storage containers makes confusion rare. Disposal isn’t a “toss it and forget it” thing: hazardous chemical protocols demand collecting even small waste amounts in a labeled bottle. Waiting for a chemical waste pickup beats a spill every time.
A Safer Path Forward
Recent tragedies show why safety training and updated material safety data sheets matter. The best labs run recurring workshops to keep new team members sharp. Staying up to date on OSHA and local chemical rules helps sidestep surprise inspections or fines, and, more importantly, keeps everyone healthier. Taking care around potassium cyanate doesn’t need high drama – just real habits, smart teamwork, and respect for hard-won safety lessons handed down over years.
| Names | |
| Preferred IUPAC name | potassium cyanate |
| Other names |
Cyanic acid potassium salt Potassium cyanate(V) Cyanate of potassium |
| Pronunciation | /poʊˌtæsiəm saɪˈaneɪt/ |
| Identifiers | |
| CAS Number | 590-28-3 |
| Beilstein Reference | 1908220 |
| ChEBI | CHEBI:4866 |
| ChEMBL | CHEMBL1231427 |
| ChemSpider | 62396 |
| DrugBank | DB11309 |
| ECHA InfoCard | 18b9a8c2-0fc1-409c-98eb-c4ca46e2b2f7 |
| EC Number | 209-782-3 |
| Gmelin Reference | Gmelin Reference: 14219 |
| KEGG | C14125 |
| MeSH | D011075 |
| PubChem CID | 10135 |
| RTECS number | SC0650000 |
| UNII | G9E09941I4 |
| UN number | UN2476 |
| Properties | |
| Chemical formula | KOCN |
| Molar mass | 81.115 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.056 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.77 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.7 |
| Basicity (pKb) | pKb = 11.5 |
| Magnetic susceptibility (χ) | +10.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.452 |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.0 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -226.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -234 kJ/mol |
| Pharmacology | |
| ATC code | V03AB31 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause irritation to eyes, respiratory system, and skin. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 3-1-2-W |
| Autoignition temperature | 390°C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 640 mg/kg |
| NIOSH | KN122 |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Sodium cyanate Potassium cyanide Potassium isocyanate |

