Pivaloyl Chloride: Its Past, Current Reach, and the Road Ahead
The Road from Discovery to Industry: A Look Back
Pivaloyl chloride started its journey after early twentieth-century chemists pursued new ways to make acid chlorides more controllable and reactive. Before it entered large-scale use, scientists worked in cramped labs, mixing isobutyric acid with reagents like thionyl chloride. Labs stank of sharp vapors, hands tingled from the corrosive sting, and early research relied heavily on trial, error, and a willingness to risk one’s eyebrows. Over the decades, industrial adoption grew. Specialty chemical firms standardized methods to produce and handle it with greater safety. The sharing of knowledge through patents and scientific journals didn’t just make it easier to manufacture. It allowed smaller labs to use pivaloyl chloride without worrying about basic mistakes that could ruin equipment or harm staff. In a time when chemical safety training wasn't so rigorous, having reliable access to pure pivaloyl chloride meant less risk and faster progress in pharmaceutical and agrochemical synthesis.
The Product: What Sets Pivaloyl Chloride Apart?
With its clear, colorless liquid appearance and strong, pungent odor, pivaloyl chloride never lets anyone forget its reactive nature. It stands apart because it brings a tert-butyl group to the party, not just any acyl group. That tert-butyl shield gives it a unique profile: reactions often run cleaner, yields can be better, and its side products stand out for easy separation. The modern packaging—usually glass or corrosion-resistant steel drums—reflects a direct response to its aggressive nature. Containers feature clear hazard warnings, regulatory symbols, and traceability information. I remember workplace debates about switching vendors because one mislabeled drum could spell disaster during inventory checks. It’s not just an acid chloride; it’s a material that demands respect and constant vigilance.
Unpacking Its Properties & Straight-Talking Technical Details
Pivaloyl chloride boils near 105 °C and loves to attack moisture in the air, forming HCl fumes on contact. Its density runs higher than water, and it dissolves best in organic solvents like dichloromethane and ethyl acetate. Chemists prize it for its chemical bite: teaming up with active methylene groups, forming esters, and capping free amines with a snap. Its reactivity simplifies protection steps in peptide or polymer chemistry, and the tert-butyl group helps block unwanted side reactions. I once compared it to other acid chlorides in a series of syntheses—no other compound matched its sweet spot between speed and selectivity. But you need to keep water far away; even a drop can trigger blistering white smoke and release hydrogen chloride gas. That physical aggression shapes how we handle and store it—never letting up, never relaxing procedures.
How the Chemical World Makes Pivaloyl Chloride
Synthetic routes for pivaloyl chloride usually begin with pivalic acid, derived industrially from isobutyraldehyde oxidation. To turn the acid into the chloride, most facilities react it with thionyl chloride, phosphorus pentachloride, or oxalyl chloride. Some setups stick to thionyl chloride because it produces cleaner byproducts, mainly sulfur dioxide and HCl. In the lab, thionyl chloride gets added in a hood, slow and steady, never poured all at once. Splattering or overheating the mix makes for a memorable fire drill. The reaction is over when vigorous bubbling slows, meaning most of the acid has converted. Then, distillation pulls off pure pivaloyl chloride. Tighter regulations on chlorinating agents and vent-gas treatment have improved worker safety and reduced the environmental footprint compared to mid-century practice, but every batch still needs careful handling and monitoring.
Quick to React, Eager to Transform: The Busy Chemistry of Pivaloyl Chloride
Board-certified as a reagent in almost every organic chemist’s vocabulary, pivaloyl chloride modifies all sorts of molecules. In the hands of an experienced chemist, it converts alcohols to esters, amines to amides, and carboxylic acids to acid anhydrides. The tert-butyl motif proves useful because it resists further changes in many settings, making it an ideal sparring partner in protecting group chemistry. Synthesis of biologically active compounds—think API precursors for antibiotics, heart medications, or pest repellents—often relies on well-behaved acid chlorides. In my own experience, its directness saves purification steps, freeing time and resources for more creative work. The academic literature and patents stretch far back, listing derivatives that owe their existence to one fast attack by pivaloyl chloride in chilly, solvent-rich glassware.
Other Names, Same Substance
Chemists can be an inventive bunch, especially when it comes to naming compounds. Pivaloyl chloride has picked up monikers like trimethylacetyl chloride and 2,2-dimethylpropanoyl chloride. The various names can confuse newcomers or those jumping between academic and industrial texts. In multinational labs, shipments sometimes carry trade names, code strings, or local language equivalents. Accurate labeling prevents mix-ups, and I’ve seen projects grind to a halt because someone mistook pivalyl for propionyl. In a world where one slip can cost weeks of work, clarity in naming remains a daily concern.
Staying Safe and Setting Standards
With pivaloyl chloride’s toxic and corrosive bite, safety training is non-negotiable. Good ventilation, fume hoods, gloves resistant to acids, and goggles stand as basic requirements with this chemical. I remember the dull, aching headache that came after a minor breach of protocol—the gas can sting the eyes and lungs within seconds. Strict labeling, double-sealed containers, and written procedures exist for good reason. Regulatory standards now require detailed risk statements, not just pictograms or generic hazards. Spill containment gear sits ready near workstations, and storage follows the rules learned from painful accidents a generation ago. Every bottle in our lab archives came with material safety data, and we relied on incident reports to keep improving our habits. Complacency fades fast when a single mishap fills the room with acrid fumes that linger for hours.
Shaping Science and Industry: Application and R&D
Across the chemical industry, pivaloyl chloride wrestles with a tough workload. It finds regular employment making pharmaceuticals, especially where a pivaloylated intermediate buffers a sensitive synthetic step. Agrochemicals rely on it for countless herbicides and fungicide precursors, and in flavor and fragrance chemistry, its role is hard to replace. Research teams often test new catalytic cycles, trying to lower waste or make reactions run at milder temperatures. In academia, the hunt for greener production methods never stops. Some works published over the years showcase enzymatic alternatives, but the main route remains relentlessly chemical. Electric reactors and continuous-flow systems show promise but require serious investment, both in money and talent. No easy solutions exist; innovation soaks up time and expertise far faster than most people expect.
The Other Side: Health Risks and Toxicity Research
Even seasoned chemists walk with extra caution around pivaloyl chloride. The acid vapor can burn skin and lungs, causing lasting health problems after exposure. Reports document case studies of accidental splashes or chronic inhalation where victims needed more than basic first aid. Toxicity testing found that exposure damages mucous membranes and can provoke acute reactions in the airways. Regulatory authorities class this family of reagents as dangerous to people and the environment, requiring strict disposal and monitoring. Newer research looks at low-level, long-term exposure in manufacturing workers. The studies dig into subtle symptoms—dermatitis, respiratory issues, or increased sensitivities—not just dramatic accidents. This sustained attention forces companies to rethink old habits, update their training, and adopt more responsive protective equipment. It costs money up front but reduces lawsuits and health costs down the line.
What the Road Ahead Might Bring for Pivaloyl Chloride
Demand for safe, efficient synthetic reagents keeps the spotlight on pivaloyl chloride. The industry faces pressure to cut environmental emissions, curb hazardous waste, and find alternatives that do not compromise reactivity. Some startups work on solid or supported forms to limit direct exposure. Recycling spent reagents and capturing HCl gas for reuse stands as a promising direction, but cost and reliability hold things back. Digital tracking, real-time monitoring for leaks, and new training protocols push the entire sector forward. Policymakers look for ways to reward companies that develop safer, “greener” synthetic routes, but changes roll out slowly. In research settings, scientists keep probing the core chemistry—trying to lower activation energies, reduce toxic byproducts, or find biocatalytic shortcuts. Success in these areas would ripple across pharmaceuticals, plastics, agrichemicals, and beyond. Until then, pivaloyl chloride remains a chemical that rewards careful handling and creative problem-solving. Lessons learned in the lab bind one generation of chemists to the next, and every bottle on the shelf carries echoes of older experiments, triumphs, and — sometimes — hard-earned scars.
What is Pivaloyl Chloride used for?
What Role Does Pivaloyl Chloride Play?
Pivaloyl chloride sounds like a tongue-twister, but behind that name, there’s a lot of action. This chemical lands its punch in the world of pharmaceuticals, agrochemicals, and in making special ingredients for plastics. I remember my first time hearing about it in a college organic chemistry class. The professor showed a diagram and all I could think was, “People really build medicines with that?” Turns out, they do—and much more.
The Backbone in Medicine-Making
Whenever you take an antibiotic or pick up a prescription with a complicated name, there’s a fair chance pivaloyl chloride played a role in putting it together. It helps protect certain parts of molecules during chemical reactions. Chemists call this a “protecting group.” It behaves almost like a security guard, watching over delicate sections of a molecule so they survive the rough parts of drug synthesis. Once the hard work is done, this “guard” gets dismissed, leaving behind the target molecule, ready for the next stage. Pivaloyl chloride’s knack for providing this protection gives experts the freedom to design some of the most effective drugs known to science.
From Pesticides to Plastics
I once met a researcher who worked in crop science. She told stories about battling invasive pests, and I soon learned that pivaloyl chloride helps create many modern crop-protection chemicals. These pesticides make their way onto farmlands across the globe, boosting plant yields and helping feed billions.
Pivaloyl chloride also slips into the world of plastic additives. Some industries add it while producing materials used for car interiors or phone cases. The result? More durable plastics, better able to withstand heat and stress—qualities that keep products working even with everyday wear and tear.
Risks Lurking Behind the Laboratory Door
The trouble starts when pivaloyl chloride gets used carelessly. I’ve read stories about mishaps in labs, with serious burns or health complications all because someone treated this compound like a kitchen ingredient. If inhaled or spilled, it can cause everything from skin wounds to breathing trouble. Not long ago, strict regulations forced companies to upgrade their storage and handling processes. Now, workers suit up with face shields and thick gloves, learning to treat this material with respect.
Many countries keep tight controls on its transport, mainly because pivaloyl chloride can produce dangerous byproducts if handled wrong. Accident records have pushed chemical plants to invest in better training and emergency gear.
Building a Safer Future
Chemists are always hunting for safer alternatives. Some have started using new processes that cut back on dangerous intermediates like pivaloyl chloride. In my time talking with green-chemistry advocates, the message is clear: safer chemicals mean fewer accidents and less environmental worry.
Education plays a huge role. Younger scientists train with strict safety routines, and companies share best practices around the globe. Technology now helps monitor air quality in facilities where pivaloyl chloride gets used, cutting down exposure to unsafe levels.
Even with its dangers, pivaloyl chloride has carved out a spot in modern chemistry. The work continues—making every dosage of medicine safer and every harvest healthier, while still pushing for practices that protect workers and the wider environment.
What are the safety precautions when handling Pivaloyl Chloride?
Getting to Know the Risks
Pivaloyl chloride belongs to a group of chemicals with a short fuse. In my work around research labs, I’ve seen how even seasoned hands give this compound special respect. Splashing a bit on your skin or catching a whiff of its fumes doesn’t play out like a TV drama — it hurts. This chemical can burn on contact, cause painful eye injuries, and it can hit the lungs pretty hard if inhaled. One wrong move, a break in concentration, and you might end up in the medical wing, wishing you paid more attention during safety briefings.
The Role of Solid Protection
Relying on thin gloves or rolling up the sleeves won’t cut it. Thick nitrile or neoprene gloves, safety goggles, a face shield, sturdy lab coat, and long pants play a big role in keeping the stuff off your skin. I worked alongside a chemist who once tried to pour a few milliliters without full gear under the excuse of “just a quick transfer.” She regretted it fast — even a drop caused stinging and left a red mark. Missing this layer of physical protection guarantees a bad time with pivaloyl chloride.
Ventilation Isn’t Optional
Foul-smelling fumes rise from pivaloyl chloride before you’ve even cracked the bottle. Breathing it in has sent lab workers home with coughing, sore throats, and headaches. Cracking a window doesn’t make the grade. This is a job for a chemical fume hood. If you don’t have one, don’t open the bottle. Simple rule. At research universities, strict policies keep this stuff behind a sash and under exhaust fans. Feeling short of breath or dizzy at your bench? Walk away and let someone know. Long-term outcomes from repeated inhalation don’t make for happy reading.
Water and Pivaloyl Chloride: A Bad Mix
Spilling the chemical is already bad news. Letting it mix with water turns the situation into a small disaster. Water causes it to break down and release hydrogen chloride gas. The cloud smells sharp, bites the airways, and irritates every bit of skin it touches. I remember a minor splash on a wet bench causing an emergency evacuation. Everyone felt the sting even down the hall. Dry workspaces and chemical-absorbing pads are the norm in labs using this reagent. If someone hasn’t caught on to this risk, they aren’t ready to handle this job yet.
Storage and Waste — Cut No Corners
Pivaloyl chloride doesn’t “get along” with many other chemicals. It eats through cork or rubber stoppers and corrodes metal. I always keep it under a chemical-ventilated cabinet, tightly sealed, and as far away from bases, water, or alcohols as possible. Never store leftovers in an unlabeled container. And don’t dump waste down the drain. Waste collection and disposal must follow the rules — otherwise, the next person can get hurt even if they behave.
Being Ready for Emergencies
Even experienced people slip up and accidents catch everyone off guard. Emergency showers, eyewash stations, and clear exit routes cut down the damage. Quick response makes the difference. I’ve seen someone use the eyewash after a droplet flicked from a pipette — she walked out shaken, but she kept her sight. Never try to muscle through a spill or splash on your own. Call for help. Stay safe by knowing exactly what to do, before the bottle ever comes off the shelf.
What is the chemical formula of Pivaloyl Chloride?
Looking at the Structure and Formula
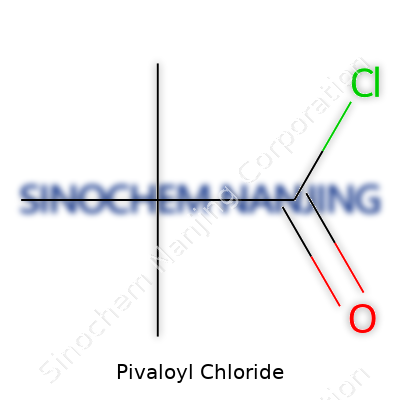
Pivaloyl chloride goes by the molecular formula C5H9ClO. Breaking it down, this comes from a pivalic acid backbone, where the carboxylic group links to three methyl branches, and the acid’s hydroxyl swaps for a chlorine atom. This small change gives chemists a reactive acyl chloride that’s a favorite for organic synthesis, especially when quick acylation is needed on a bulky substrate.
Real Uses in the Lab
I’ve handled pivaloyl chloride in research projects focused on drug precursors. It reacts fast, carries its signature smell, and always calls for careful handling. People in pharmaceutical chemistry use it to build tough or complex molecules—sometimes protecting sensitive groups or introducing a shield that resists unwanted reactions. For instance, I used pivaloyl chloride to install large, sturdy blocking groups on a fragile base. The ability to steer a synthetic route around unwanted branches really matters when deadlines loom.
Health and Environmental Considerations
Pivaloyl chloride produces hydrogen chloride fumes on contact with water, so opening the bottle means releasing sharp, irritating vapor. Exposure stings the eyes, nose, and throat—and spills burn. The Environmental Protection Agency warns about runoff since these chlorinated compounds break down slowly in soil and water, and fume control in the lab matters just as much as glove and goggle use. Accidental release could lead to toxic acid build-up, especially if local rules for chemical disposal aren’t respected.
Why Molecules Like This Deserve Attention
Molecules like pivaloyl chloride show up behind the scenes in cancer treatments, antibiotics, even high-performance plastics. Despite its promise, the real-world risks are not just theoretical. In the lab, I have seen new graduate students cough from improper handling, rushing to open pivaloyl chloride bottles or skipping the fume hood. This chemical’s sharp reactivity makes it an asset, but it tests our habits, too. Every bottle carries an edge. That sharpness can teach caution and good lab practice faster than hours of lecture.
Improving Safety and Sustainability
Labs and production facilities catch on to these lessons by switching to closed systems, better training, and digitized monitoring. A fresh bottle means a new safety review among colleagues, not a silent self-check. Chemical manufacturers keep searching for alternatives—acylating agents that deliver the same power but with less hazard and wastewater impact. Green chemistry teams aim to replace old-school acyl chlorides with less hazardous, more biodegradable choices. These efforts keep the best synthetic routes open without turning a blind eye to long-term health or environmental fallout.
Learning from Experience
Every chemist has a pivaloyl chloride story—some funny, some cautionary, all useful. Watching experienced chemists treat it with respect, double-checking PPE and venting setups, drove home the idea that fast, powerful molecules bring both progress and responsibility. The chemical formula C5H9ClO won’t appear on most medicine labels or consumer products, but the methods it unlocks leave fingerprints everywhere. In chemistry, the smallest formula can carry big weight. I know mine—do you?
How should Pivaloyl Chloride be stored?
No-Nonsense Chemical Storage
Anyone who’s handled pivaloyl chloride knows that it tests the responsibility of a lab. It’s not a chemical you want lying around on the bench or getting too comfortable in a storeroom corner. Pivaloyl chloride, useful in so many industrial reactions—from pharmaceuticals to agrochemicals—has a side to it that can ruin more than just your day if ignored. It chews up skin, irritates your lungs, and violently reacts with water. A few splashes can send sharp fumes into the air. I’ve seen glassware etch itself within minutes due to mishandling. The stuff demands strict boundaries.
Rules That Matter for Everyone
You keep pivaloyl chloride in a place built for corrosives, well away from moisture and reactive chemicals. A strong, airtight bottle—usually glass or a special kind of plastic—stands up to its nature much better than metal containers, which pivaloyl chloride can eat through. I always double-check for secure lids before I walk away, and I swear by desiccators or cabinets lined with inert secondary containment, as spills burn and spread fast.
If you’ve ever rushed to find a leaking bottle of this chemical, you’ll get why shelving material counts. Cheap wood warps and absorbs. Stainless steel and chemical-resistant plastic trays solve the problem. Keep it cool, too. You want to store it below 25°C; the higher the temperature, the quicker it decomposes, releasing hydrochloric acid or worse. I’ve worked in labs where cheap AC setups saved our shelves from sticky, hazardous residues. Even small, regular investments in monitoring temperature and humidity pay off when the alternative could mean shutting the lab for days.
Training and Lab Culture
No policy or safety sheet protects people like repetition and example. I’ve seen organizations lose hard drives and notebooks to pivaloyl chloride that wandered beyond its storage cabinet. People respect the chemical only after real stories circulate—stories with real costs. Good labs walk new folks through the rules each season, not just during hiring. Inevitably, someone asks why it can’t sit near the acids shelf. All it takes is a bump or a drip from a bottle of water-based acid, and you get clouds of gas or a puddle that won’t quit reacting. That’s why separation inside chemical storage is never up for negotiation.
Real Consequences, Real Solutions
Labs investing in clear labels for every corrosive bottle and secondary trays for every shelf reduce confusion. Emergency showers and eyewash stations—occasionally tested, not just installed—keep everyone aware of the game. The worst situations crop up in under-ventilated store rooms. Installing exhaust fans, even simple ones, keeps vapors out of people’s lungs. Sensor-based leak alarms or humidity sensors now come cheaper than many people expect.
Local regulations get strict for good reason. I always recommend reviewing Safety Data Sheets before accepting a shipment of pivaloyl chloride. Chemical companies don’t exaggerate the risks. Smart storage means using all their guidance, not cutting corners. Invest in organization and good containers, reinforce with regular checks, and keep every staff member in the loop. Taking shortcuts earns nothing but downtime and danger.
What are the common applications of Pivaloyl Chloride in industry?
Chemicals That Shape Modern Manufacturing
Pivaloyl chloride shows up in far more places than most people expect. I first noticed its impact working in a small chemical plant. Every day, trucks would come and go, hauling away barrels filled with clear, strong-smelling liquid—a reminder that chemistry drives plenty of the things we touch and use. Pivaloyl chloride, in particular, finds its way into processes that power much of the pharmaceutical and agrochemical fields, and I quickly learned how deep its reach goes.
Pharmaceutical Synthesis: The Unsung Helper
Most major drug manufacturers rely on pivaloyl chloride to make medicines safer and more effective. It steps in as an acylating agent, which means it helps attach protective “caps” onto different building blocks of drugs. These caps shield parts of a molecule during complex assembly, then come off when no longer needed. This process lets chemists piece together life-saving medicines with much better precision. Finished antibiotics, painkillers, and antiviral drugs can all trace part of their existence back to a handful of ingredients like pivaloyl chloride making the work possible. According to industry reports, about 70% of all pivaloyl chloride ends up in pharmaceutical use.
Agrochemical Manufacturing: Crops and More
Walk through a crop field, and it’s easy to forget that “simple” fertilizers or pesticides have big stories behind them. Pivaloyl chloride steps up in several key reactions to make new-generation herbicides, fungicides, and insecticides. Without ingredients like this, a lot of field treatments would spoil during production, or never make it to market in the first place. A 2023 review in the “Journal of Agricultural and Food Chemistry” pointed out how pivaloyl chloride reduced waste and improved yields for common active ingredients. Farmers may never hear the name, but their harvest depends on its reliability.
Custom Chemicals and Fine Fragrances
Not every use of pivaloyl chloride deals with massive machines. High-end fragrance makers turn to it for synthesizing rare perfumery ingredients—especially those with complex, delicate notes. In a small workshop setting, chemists combine pivaloyl chloride with specialty alcohols or amines to build exotic scents that stand apart from cheap imitations. Food flavorings, too, often start with similar reactions. Even though these batches are tiny compared to pharmaceuticals, the purity and consistency required make pivaloyl chloride indispensable.
Industrial Challenges and Safer Solutions
Pivaloyl chloride demands respect—its sharp fumes can burn eyes and skin, and even brief exposure can send workers running for fresh air. This pushes labs and factories to invest in improved ventilation, sealed containers, and better training for everyone on site. The chemical’s reactivity means even small leaks can escalate into safety incidents. I’ve seen experienced teams scramble to contain spills because, as one safety manager always said, “This stuff won’t give you a second warning.” Regulatory agencies have started increasing oversight, encouraging the use of personal protective equipment and routine health monitoring for chemical workers.
Some research teams in the European Union and Asia experiment with greener alternatives, hoping to cut down hazards and environmental impact. So far, pivaloyl chloride remains the standard because its efficiency outpaces most substitutes. Still, investments in new reaction pathways could eventually yield safer tools for both medicine and agriculture. Until then, skill and experience keep things running smoothly, showing how modern industry adapts to keep risk in check.
| Names | |
| Preferred IUPAC name | 2,2-dimethylpropanoyl chloride |
| Other names |
Pivaloyl chloride Trimethylacetyl chloride 2,2-Dimethylpropanoyl chloride Neopentanoyl chloride |
| Pronunciation | /paɪˈvælɔɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | “3282-30-2” |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:82232 |
| ChEMBL | CHEMBL135888 |
| ChemSpider | 68214 |
| DrugBank | DB11360 |
| ECHA InfoCard | 100.008.535 |
| EC Number | 204-188-7 |
| Gmelin Reference | 74123 |
| KEGG | C14243 |
| MeSH | D011669 |
| PubChem CID | 8773 |
| RTECS number | KN0450000 |
| UNII | Z776010087 |
| UN number | UN1780 |
| CompTox Dashboard (EPA) | DTXSID9086523 |
| Properties | |
| Chemical formula | C5H9ClO |
| Molar mass | 112.55 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 0.973 g/mL at 25 °C |
| Solubility in water | Reacts |
| log P | 2.52 |
| Vapor pressure | 8 mmHg (20°C) |
| Acidity (pKa) | 1.0 |
| Basicity (pKb) | Basicity (pKb): 10.33 |
| Magnetic susceptibility (χ) | -5.37 × 10⁻⁹ cm³/mol |
| Refractive index (nD) | nD 1.398 |
| Viscosity | 0.685 cP (20°C) |
| Dipole moment | 2.28 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 324.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -322.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -685.9 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02, GHS05, GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H335 |
| Precautionary statements | P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P311, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-2-W |
| Flash point | 46 °C |
| Autoignition temperature | 410 °C |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 370 mg/kg |
| NIOSH | SN2100000 |
| REL (Recommended) | 0.5-2.5 ppm |
| IDLH (Immediate danger) | IDLH: 25 ppm |
| Related compounds | |
| Related compounds |
Pivalic acid Pivalamide Pivaloyl fluoride Trimethylacetyl chloride Neopentanoyl chloride |

