Pivalic Anhydride: Chemical Character and Real-World Perspectives
Historical Development
Asking chemists about the lineage of pivalic anhydride brings up the broader story of the chemical industry’s march through the 20th century. Pivalic anhydride, structurally penned as a dimer of pivalic acid, didn’t become a staple overnight. Early synthetic routes arose from the need for tailored anhydrides in organic synthesis labs, especially around the times when bespoke acids and their derivatives started being drafted into wider chemical transformations. Academic benchwork in the postwar era took the lessons of efficient dehydration and ran with them, driving methods that turned abundant pivalic acid into the more reactive anhydride. Once large chemical manufacturers picked up on its utility, more refined and scalable routes became standard practice. The modern chemist can still see traces of this historical evolution, especially in how process safety and feedstock selection are handled.
Product Overview
Pivalic anhydride doesn’t get the same attention as the big-name reagents, but its applications punch well above its weight. This anhydride delivers a combination of bulkiness and benign leaving group qualities, carving out a niche for itself in pharmaceuticals, esters, and polymer building blocks. The global production may not rival the volumes seen with acetic or phthalic anhydrides, yet the specificity of its applications earns it a dedicated following among organic and process chemists. Smaller batch jobs or custom syntheses often count on it where selectivity and steric factors matter.
Physical and Chemical Properties
A bottle of pivalic anhydride tells a story even before the stopper opens. The substance appears as a colorless liquid, sometimes with an acerbic whiff that warns of its anhydride nature. With a boiling point hovering in the vicinity of typical lab heating mantles and a moderate density, it handles easily under standard chemical room conditions. Its chemical structure, featuring two tert-butyl moieties flanking an anhydride bridge, imparts significant steric hindrance. That means it doesn’t react as violently or indiscriminately as leaner anhydrides do. Water splits it back to pivalic acid, and alcohols or amines convert it to esters and amides, respectively, making it a tool for precise functionalization in organic labs.
Technical Specifications & Labeling Practices
Lab bottles tend to feature common synonyms — trimethylacetic anhydride crops up at times, though “pivalic anhydride” remains the most recognized tag among chemists. Purity levels matter, especially for pharmaceutical intermediates, and most commercial supplies meet or exceed 98%. Impurities in the form of pivalic acid or related tertiary compounds get flagged quickly, as they complicate downstream usage. Safety labels warn of irritant nature, with tight recommendations for gloves and face protection. Proper chemical labeling, up-to-date hazard statements, and manufacturer traceability track industry best practices and meet the expectation laid out in global chemical regulations.
Preparation Method
Classic preparations involve the dehydration of pivalic acid, a method that stands on the shoulders of time-tested laboratory processes. Dehydrating agents like phosphorus pentachloride or oxalyl chloride were once the mainstays. Later, efficiency and environmental goals nudged chemists to opt for more selective conditions, swapping out harsher reagents with less hazardous alternatives wherever possible. Even today, the quest for greener syntheses—fewer chlorinated byproducts, less difficult waste—pushes R&D efforts. Pilot plants now prefer to optimize yields, reduce side reactions, and minimize the environmental footprint, reflecting a broader industry shift toward responsible manufacturing.
Chemical Reactions & Modifications
Workups involving pivalic anhydride rarely feature drama—just steady, predictable transformations. It acylates alcohols and amines to form esters and amides, often used when bulkier groups block unwanted pathways or introduce stability. The tert-butyl group resists further reaction, so modifications stick to the anhydride bond. Its role in introducing pivaloyl protection illustrates how organic chemists rely on subtle tweaks for selectivity in synthesis. Reactions using pivalic anhydride often run at moderate temperatures, with yields that make it worth dealing with the prickly odor and requisite ventilation.
Synonyms & Product Names
Chemists shorthand pivalic anhydride as “Piv2O” in notes and protocols. I’ve also seen "trimethylacetic anhydride" printed on invoices, but most research groups skip secondary names. Suppliers may layer on their own trade codes for cataloging, though these codes rarely make it into the everyday speech of labs or patent filings. I spent years in a synthetic lab where nobody used anything but “pivalic anhydride”—simplicity wins.
Safety & Operational Standards
Dealings with this chemical call for attentive handling. The volatile fumes sting mucous membranes, and direct contact irritates skin. Proper fume hoods, goggles, and nitrile gloves form the basic PPE set, while spill cleanup guidelines align with standard protocols for acid anhydrides. Storage stays straightforward—sealed bottles, away from moisture and strong bases. Industrial setups work with containment and air filtration in mind, due to both safety and compliance with workplace exposure limits. Transport containers must be robust, leak-resistant, and clearly labeled, reflecting modern expectations around chemical logistics and hazard communication.
Application Area
The shelf life of pivalic anhydride in research spaces circles back to its role in the synthesis of pharma intermediates and specialty polymers. Chemists lean on it for protecting group strategies or acylation reactions, especially in projects demanding non-reactive, bulky groups. The material’s unique shape precludes over-reactions, earning it a dedicated following among process chemists scaling up medicinal compounds. Polymer labs occasionally turn to it for backbone modification or to put steric brakes on cross-linking, trading its cost for clean, predictable chemistry.
Research & Development
Development efforts target making the production and downstream application of pivalic anhydride both safer and more sustainable. Synthetic chemists have been working toward catalytic or solid-phase methods that produce less waste and avoid hazardous reagents. Analytical chemists dig into purity profiles, detecting trace contaminants using high-performance liquid chromatography or gas chromatography. The pharmaceutical sector keeps a close eye on residual anhydrides in drug candidates, encouraging suppliers to refine purification steps. I’ve watched collaborative projects between academia and industry focus on both new synthetic applications and greener process improvements, signaling a steady commitment to keeping the chemical current, compliant, and competitive.
Toxicity Research
Animal studies and cellular test panels have confirmed what the nose suspects: pivalic anhydride irritates tissues and should stay out of direct contact with skin and eyes. Inhalation of vapors causes respiratory discomfort, and chronic exposure isn’t advisable. Acute toxicity sits in the moderate range compared to other acid anhydrides, though anyone working with it in sizable volumes takes extra precautions to avoid accidental release. Regulatory agencies evaluate new data continually to adjust workplace exposure standards and transport limitations. Researchers catalog and minimize long-term environmental and health impacts using both in vitro and environmental degradation studies.
Future Prospects
Innovation in the world of fine chemicals doesn’t pause, and pivalic anhydride’s future draws from green chemistry, tighter environmental guidelines, and demand in pharmaceuticals. Production routes look to cut out hazardous reagents, support circular economy goals, and meet stricter purity requirements for health applications. Downstream, new synthesis protocols take advantage of pivalic anhydride’s selectivity and steric shielding for complex drug or polymer architectures. If chemists and engineers keep improving both the sustainability of its production and the value it adds in specialty sectors, it won’t disappear from lab shelves anytime soon.
What is Pivalic Anhydride used for?
Understanding Its Place in Real-World Chemistry
Pivalic anhydride isn’t the chemical you hear about at the grocery store, but it plays a quiet role in modern chemistry. I first ran into it while helping a friend with research back in college—chalky, pungent, and kept under lock and key in the lab. While it seems pretty obscure to most people, this compound has stepped into the spotlight for both good and questionable reasons. So, let’s dig in: what makes it worth talking about?
Key Uses and Its Importance
The bulk of pivalic anhydride’s use falls in the chemical industry. Skilled chemists lean on it when they need to craft specialty chemicals, especially pivalates and complex esters. These show up in a slew of places, from flavorings to perfumes and pharmaceutical ingredients. It wouldn’t really catch your attention unless you spend time paging through chemical supply catalogs or mixing things in a lab yourself. Still, as a building block, it’s regularly on the requisition lists at R&D facilities and pharma labs.
Pharmaceuticals count on compounds like this to protect sensitive molecules while building up antibiotics or antiretrovirals. One well-known story is how specific reactions can create the backbone for drugs like some penicillins. I remember hearing from former classmates in pharma jobs that without anhydrides like pivalic, the process could get both costlier and messier. Production efficiency matters here—getting more effective medicine to shelves quickly affects everyone, especially sick patients waiting for relief.
Controversy and Scrutiny
Not every use of pivalic anhydride seems beneficial. Authorities watched the market closely, especially as headlines in some parts of the globe linked it to illegal drug manufacturing. Sophisticated groups once harnessed chemicals like this to make synthetic drugs. This led governments such as the ones in the US and EU to place tighter controls, flagging it as a substance with potential for illicit use. Picture a legitimate scientist frustrated at delays due to red tape, all because of misuse by criminal groups. During a past lab internship, I saw researchers jump through hoops to order such chemicals—one more form, one more proof of the project’s legitimacy, and another phone call from compliance teams.
Balancing Act: Progress and Responsibility
We rely on chemicals like pivalic anhydride for continued progress. You feel its absence in pharma, flavor science, materials, and even agricultural labs. At the same time, it’s only right to keep a close eye on its distribution and use. Public safety always comes first. Those controls might slow down the workflow, but they make a lot of sense after watching the harm that loose chemical markets can cause. There’s no perfect answer, but stronger vetting procedures and real-time tracking can help good science keep moving while stopping shady business in its tracks.
So next time someone tosses out complicated names like pivalic anhydride, remember its journey isn’t just about white coats and bubbling flasks. It’s about innovation but also about safeguards—both matter if we care about keeping progress steady and our communities safer.
What are the safety precautions when handling Pivalic Anhydride?
Everyday Decisions Matter
Pivalic anhydride rarely turns up in conversations outside the lab, but those who handle it know a single mistake can carry lifelong consequences. This compound, used for advanced chemical synthesis and pharmaceutical research, brings real risks along. A sharp, unpleasant odor usually signals its presence. Inhaling those vapors burns more than just nostrils—it irritates the throat and lungs, and more serious exposures cause breathing trouble. Even a seasoned chemist feels respect when they break the seal on a new bottle.
Direct Contact Bites Back
Spills on bare skin sting instantly because pivalic anhydride reacts with moisture. Unprotected eyes water, eyelids clench, and vision might blur. The danger ramps up if contact lasts more than a few seconds. Stories circulate of accidental splashes in crowded fume hoods. Managing one of those rare incidents taught me to triple-check for gloves and face shields. No shortcut repays a trip to the emergency eye-wash station. Surfaces and containers touched by the liquid keep the threat alive even after the main clean-up.
Air Quality: A Frontline Shield
Every time I worked with this chemical, I relied heavily on a certified fume hood. It moves the vapors away before you can breathe them in. Sealed goggles, splash-resistant lab coats, and double-nitrile gloves form layers of defense. One lab mate learned too late that thin vinyl gloves dissolve quickly around this stuff. Strong ventilation doesn’t just keep the air clear. It signals respect for the person working next to you—their lungs deserve protection, too.
Handling and Storage Lessons
Pivalic anhydride reacts with water and alcohols—sometimes with a pop, sometimes with dangerous acid byproducts. Soaked lab tissues and leftover solution bottles need disposal as hazardous waste. Keeping containers tightly sealed limits air contact. Dry storage, well marked and away from flammable materials, lowers the chance of a chain reaction after a spill. In one lab, failing to read that label cost someone half a day lost to clean-up and a stern safety lecture. Training made sure it wouldn’t happen to others.
Emergency Steps: No Excuses
Quick reactions keep injuries from becoming tragedies. That means knowing the nearest shower, eyewash, and exit. Safety data sheets lay out exact procedures, but real-world practice drills also count. Skin contact? Strip gloves, rinse under running water, seek medical attention. Splash in the eyes? Lean close to an eyewash for fifteen minutes, even if it feels fine after five. Fainting or choking calls for emergency responders—never tough it out or leave someone alone.
The Human Factor
People like to cut corners after years of routine, but shortcuts with pivalic anhydride write stories no one wants told. Consistent safety habits protect your hands, eyes, breath, and reputation as a reliable team player. Sharing experience and mistakes lowers risk for the whole lab, so speak up about near-misses and system flaws. The small effort to double-check equipment and procedures pays off every single day.
Protection Stems From Experience
Pivalic anhydride doesn’t take mercy on the unprepared. Rely on the facts—irritant, corrosive, triggers runaway reactions with water—and then remember the quick lessons every senior technician learns over time. Use the tools, trust your training, and treat every bottle with the gravity it deserves. Every lab owes its safety record to the people inside, putting good habits into practice every shift.
What is the chemical formula and structure of Pivalic Anhydride?
Why Get Personal About Pivalic Anhydride?
Some compounds make you think twice about the chemistry behind everyday reactions, and pivalic anhydride caught my eye early in my studies. I remember the smell—a sharp, sour quality—that comes with working with acid anhydrides. This particular one pulls double duty in both organic synthesis and the creation of specialty chemicals. Understanding what pivalic anhydride brings to the table helps anyone tinker with or scale up organic reactions.
Chemical Formula of Pivalic Anhydride
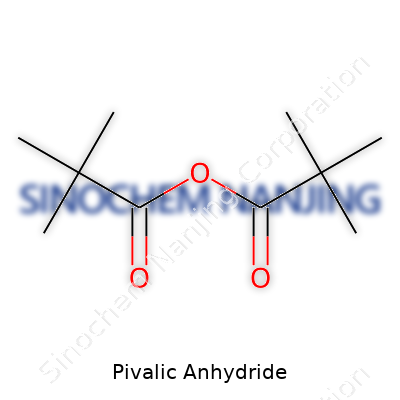
The chemical formula comes down to C10H18O3. Each molecule fuses two pivalic acid (trimethylacetic acid) units through a central oxygen atom, squeezing out a water molecule along the way. Simple as a reaction, but easy to mess up if expectations for yield and reactivity aren’t realistic.
Molecular Structure Matters
Visualize two bulky t-butyl groups (basically, a carbon with three methyl groups hanging on) leashed to a central oxygen “bridge.” Drawing it out, you’ll spot each t-butyl (C(CH3)3) cluster attached to a carbonyl (C=O), then both carbonyls fasten onto a single oxygen atom. The layout looks like this:
(CH3)3C–CO–O–CO–C(CH3)3
Compared to plain anhydrides, the tert-butyl groups make the structure stand out. The anhydride bond sits right in the middle. Those bulky groups around it keep it from getting knocked around too easily in side reactions. For me, working in the lab usually meant the reagents stayed where they belonged—no random water vapor sneaking in to ruin everything.
Role in Synthesis and Challenges
Pivalic anhydride is not just a simple derivative—people use it as an acylating agent. It reacts sharply with alcohols and amines, an important step to protect sensitive functional groups during complex organic syntheses. The presence of those big t-butyl groups slows things down compared to something like acetic anhydride. Sometimes, this comes in handy: it helps control the reaction’s pace, letting chemists carefully tune outcomes and avoid over-reaction.
On practical terms, storage and handling matter. Contact with moisture means game over—the compound splits and gives back pivalic acid. This calls for tight handling routines, especially on humid days or in busy teaching labs where containers don’t always get capped fast. Safety goggles weren’t just for show. The sharp, almost peppery odor serves as a quick reminder not to stick your nose too close. According to the Global Harmonization System (GHS), it can irritate skin, eyes, and airways, so fume hoods become a must.
Applications—Why Bother?
Pivalic anhydride plays a big part in prepping esters that resist hydrolysis, especially in the pharmaceutical world. Pivalic esters hold tight to their structure, helping drugs cruise through the body until they need to let loose the active component. In fragrances and specialty coatings, this stability matters just as much. I saw teams use it to build blocks for polymers, too, giving end products a toughness they wouldn’t get from generic acylating agents.
Ideas For Safer and Smarter Handling
Switching to automated pipetting or gloveboxes takes a lot of stress out of working with noxious reagents. Training and clear labeling make sure that junior staff don’t grab the wrong bottle—or leave it open. Tech advances let teams track inventory and temperature in real time, slashing the risk of spoiled batches. Pivalic anhydride rewards good prep work with smooth reactions and, usually, minimal waste.
How should Pivalic Anhydride be stored?
Why Safe Storage Can’t Be Ignored
Pivalic anhydride carries a bit of a punch. It reacts strongly with a lot of everyday lab substances, so unsafe storage can ruin supplies, damage expensive equipment, and even lead to fires or toxic fumes. Inexperienced chemists might think it just needs to stay off the floor or away from sunlight. That’s a recipe for trouble. I’ve worked in facilities where a leaky container put a sharp, biting odor in the air. It isn’t just a risk to experiments—it’s a risk to everyone in the building.
Control the Temperature and Humidity
Keeping the temperature steady matters a lot. Most labs stick to cool, dry locations, because heat or moisture can turn pivalic anhydride into something you don’t want in the air or on your skin. Moisture spells disaster because the chemical breaks down, releasing acid fumes and heat. From the labs I’ve worked in, desiccators or climate-controlled rooms help, especially in summer humidity spikes. Never even think about storing the stuff near a hot water heater or a sunlit window: those temperature swings just call for trouble.
The Right Kind of Container Makes All the Difference
Just grabbing any bottle off the shelf is asking for leaks and corrosion. Glass bottles with tight, Teflon-lined caps have saved many from emergency cleanups. Never use containers with rubber stoppers, as anhydrides chew through those fast. Double containment—placing your main container in a sealed, acid-resistant bin—is the routine in chemistry departments that take safety seriously. This saves you from sleepless nights worrying about slow, invisible leaks. In one lab, a careless student stored pivalic anhydride above other chemicals, and the drip landed in an open base. Quick reactions—yelling included—stopped things from getting worse, but it shouldn’t have happened in the first place.
Keep It Separate from Incompatibles
Pivalic anhydride will not stay quiet if someone stores it near alcohols, water-based reagents, or open packs of amines. Combining them can mean fire, smoke, and ruined chemistry. Safety officers I’ve met hammer home that flammables and strong acids belong in different cabinets, with anhydrides in their own spot. Dedicated secondary cabinets with chemical-resistant trays or liners control spills. These small steps keep neighboring reagents safe, which means fewer expensive cleanups and no frightening evacuation alarms.
Label Everything and Mind the Rules
The habit of slapping on a half-ripped tape label just opens the door for mistakes. Write out full names, hazard pictograms, and dates—my own mistakes have shown me that clear, bold labels save trouble when half the bottles look the same. Let colleagues know what’s inside; never count on memory to keep them safe. Local regulations and institutional SOPs should shape your storage system, but in practice, the care shown in real labs goes beyond rules printed on posters.
Training Brings It All Together
Book knowledge alone isn’t enough. Periodic hands-on safety refreshers make the difference, especially for new hires or those learning the ropes in a research setting. I’ve watched newer coworkers get comfortable once they practiced opening and closing containers safely and actually walked through what to do if something spills. Real, lived experience in the lab proves that preparation outpaces just having the right shelves or bottles. Building that culture of care keeps everyone working, experimenting, and going home healthy at the end of the day.
Is Pivalic Anhydride hazardous or toxic?
Understanding What We Face
Pivalic anhydride does not show up in daily routines, or on grocery shelves, but it plays a role in the chemistry world. Used in labs and for certain chemical reactions, it’s a strong and reactive substance. This stuff matters because we value workplace safety, healthy communities, and smart manufacturing. I’ve seen firsthand how overlooked chemicals, even in small research settings, unsettle teams. A bottle hits the floor, a cloud forms, or a fume hoods stops drawing, and everybody’s priorities shift in seconds.
Safety Risks and Health Concerns
Some folks only worry if a substance causes cancer, but danger rarely waits for chronic exposure. Pivalic anhydride carries real risks on contact, not only years down the line. Breathing in its fumes can cause major irritation in the nose and lungs, leading to coughing, trouble breathing, and even a burning throat. If it lands on the skin or in the eyes, you get pain and redness—sometimes blistering burns. Eyes suffer the most. Permanent injury can follow from one bad splash without quick action.
OSHA and NIOSH both stress the hazards of strong acylating agents—pivalic anhydride falls solidly in this camp. The compound reacts strongly with water and releases pivalic acid. Water is everywhere—in air, on skin, in the eyes. That reaction doesn’t wait for a high-school chemistry lab; it starts the second the liquid meets any moisture. Smell is no warning either since pivalic anhydride’s scent can be too faint to notice, even at dangerous concentrations.
Environmental Threats
Every chemical mishap eventually affects more than just the people in the room. A small spill swirling down the drain won’t vanish, as wastewater treatment rarely neutralizes chemicals this reactive. Pivalic anhydride in waterways turns into pivalic acid, which can disrupt living things in rivers and lakes. Persistent pollution likes to slip past early detection in lab audits. Once, at a field project in graduate school, we flagged unknown residues in a marsh after a minor lab leak upstream—tests found traces that kept showing up for weeks.
How We Tackle the Issue
Nobody should hope for perfect memory, so labeling and real training make the difference. The most effective labs keep tight records, label every container, and require goggles, gloves, and full coverage when handling anything like pivalic anhydride. Scrambling for a safety sheet after a spill never works as well as a posted sign or a practiced drill. I've seen old-timers in the lab shrug at protective gear until one mishap puts them in an eyewash station yelling for help. Equipment isn’t a suggestion—it’s the line between a close call and an ER visit.
Community awareness comes next. Local fire departments and hazardous materials teams need info on the substances in their region. Even municipalities not known for heavy industry hold stocks of lab chemicals in schools and small factories. Tracking production, usage, and disposal keeps the chain of responsibility clear. Big gains in safety come from regular audits and an open-door policy for workers to report unsafe practices.
Enforcing What Matters
Rules around hazardous substances don’t work unless everyone—from high school teachers to research chemists to janitorial staff—feels free to ask questions. The best safety record comes from this culture of respect, not just strict rules. I’ve seen the difference after a team meeting where everyone could share lessons from mishaps. Pivalic anhydride, like a lot of strong chemicals, teaches quick respect. Handling it with care and attention protects not only those in the room, but neighbors and future generations as well.
| Names | |
| Preferred IUPAC name | 2,2-Dimethylpropanoic anhydride |
| Other names |
Trimethylacetic anhydride Pivalic acid anhydride 2,2-Dimethylpropanoic anhydride |
| Pronunciation | /paɪˈvælɪk ænˈhaɪdraɪd/ |
| Identifiers | |
| CAS Number | ['1112-86-7'] |
| 3D model (JSmol) | `C1(C(C)(C)C(=O)OC(=O)C(C)(C)C)` |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:39058 |
| ChEMBL | CHEBI:39060 |
| ChemSpider | 12440 |
| DrugBank | DB11262 |
| ECHA InfoCard | 100.013.293 |
| EC Number | EC 211-481-1 |
| Gmelin Reference | 85477 |
| KEGG | C14321 |
| MeSH | D010901 |
| PubChem CID | 9172 |
| RTECS number | AT8225000 |
| UNII | 6QK513393A |
| UN number | UN2522 |
| Properties | |
| Chemical formula | C10H18O3 |
| Molar mass | 186.22 g/mol |
| Appearance | Colorless liquid |
| Odor | pungent |
| Density | 0.932 g/cm3 |
| Solubility in water | Decomposes in water |
| log P | 0.47 |
| Vapor pressure | 0.14 mmHg (25 °C) |
| Acidity (pKa) | 8.4 |
| Basicity (pKb) | 14.17 |
| Magnetic susceptibility (χ) | -7.39 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.371 |
| Viscosity | 1.32 mPa·s (20 °C) |
| Dipole moment | 1.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -833.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –1812.6 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H302, H314, H410 |
| Precautionary statements | P280, P261, P305+P351+P338, P308+P313, P310 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 99 °C |
| Autoignition temperature | 221 °C (430 °F; 494 K) |
| Lethal dose or concentration | LD50 oral rat 763 mg/kg |
| LD50 (median dose) | LD50 (median dose): 316 mg/kg (oral, rat) |
| NIOSH | SD9175000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Pivalic Anhydride: "No OSHA PEL established |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 250 ppm |
| Related compounds | |
| Related compounds |
Trimethylacetic acid Isobutyric anhydride Acetic anhydride Propionic anhydride |

