Pivalic Acid: The Workhorse You Didn’t Know About
Historical Development
Pivalic acid’s story stretches back to the 19th century, during a time when organic chemistry was rapidly opening up new frontiers in medicine and industry. It’s tough not to appreciate the spark that early chemists must have felt, teasing apart molecules in the hope of stumbling onto something new. Through careful distillation and relentless curiosity, investigators unlocked the secret behind the structure of this seemingly simple molecule — a short, stubby chain with a trimethylated core that gives pivalic acid its toughness and rigidity. As industry sprawled across Europe and later the rest of the world, this compound found a foothold in specialty chemistry thanks to its reliability and functionality, long before most people could spot a carboxylic acid group on a structural diagram.
Product Overview
Pivalic acid stands apart from straight-chain acids due to its branched, shield-shaped carbon skeleton. Its full name, 2,2-dimethylpropanoic acid, hints at its molecular steadfastness. In the grand lineup of carboxylic acids, it resists degradation more than most, owes much to its bulky tert-butyl group, and sits in countless labs as a specialty acid. You might not see glossy advertisements for pivalic acid, yet behind closed doors of fine chemical synthesis, it punches above its weight. Lab workers, process chemists, and research teams have learned to rely on its stability during challenging reactions, especially where other acids might wilt or fail.
Physical & Chemical Properties
This acid brings a distinctive bouquet, landing somewhere between pungency and clinical sharpness. Solid at room temperature, crystalline and white, it has a melting point hovering around 35°C and boils at 164°C. Unlike some of its relatives, pivalic acid shrugs off the urge to lose a proton too easily. It dissolves moderately in water, better in ether, and mingles freely with most organic solvents. A low level of volatility means less evaporation and a safer handling experience, which seasoned chemists remember after too many headaches from acetone or acetic acid fumes. The crowded methyl groups don’t just lend steric bulk. They lock in stability, sheltering the carboxyl group from attack, and that backbone resists easy breakdown, making derivatives stiffer and more persistent in tough reaction conditions.
Technical Specifications & Labeling
For those handling large volumes, reliable identification matters, especially since misuse with volatile acids leads to mistakes. Pivalic acid, with its CAS number 75-98-9 and chemical formula C5H10O2, ships in solid, crystalline form, usually packed in airtight, moisture-resistant containers. Labels carry hazard notations, often warning of irritant characteristics and the need for gloved handling. Industry standards call for 99% purity unless you’re customizing for specific downstream work, where even trace contaminants change outcomes. Respected suppliers run every lot through melting point checks and chromatographic purity scans, ensuring peace of mind for users who recall what a contaminated batch does to a week’s work.
Preparation Method
The traditional preparation borrows from the enduring tactics of organic synthesis. Pivalic acid comes from the hydrolysis of neopentyl chloride, or in modern plants, from the oxidation of pinacolone — a byproduct that’s more available than it used to be thanks to the scale of global petrochemicals. In labs, the classic haloform reaction steps up, with researchers controlling pH and temperature to encourage selectivity and avoid byproduct headaches. Acylation of tert-butyl chloride with carbon monoxide (under acid catalysis) finds use as plants scale up production. These process developments always circle back to the appeal of making a pure, branched acid in clean, reproducible runs, sometimes serving as the ideal teaching example for up-and-coming chemists eager to see industrial chemistry in action.
Chemical Reactions & Modifications
Pivalic acid stands as a resolute starting point in many synthetic schemes. Chemists value its role as a building block for esters, amides, and as a protective group in multi-step syntheses. Its robust structure limits side reactions, a trait friends in pharmaceutical labs regularly appreciate, especially during peptide or complex small-molecule construction. Pivalates appear in polymer chemistry, too, giving materials improved resistance to heat and hydrolysis. Basic reactions—esterification with alcohols, amidation with amines—stay straightforward even on scale, which boosts yield and predictability. Thanks to the shield-like tert-butyl portion, derivatives keep their structure under situations that trash less-branched acids, so it’s unsurprising how often pivalic acid sneaks into the source materials of modern performance chemicals.
Synonyms & Product Names
Chemists hear pivalic acid but may jot down 2,2-dimethylpropanoic acid, trimethylacetic acid, or even NEHA. Language varies across continents and schools, though the molecule stays the same. Specialty chemical suppliers often stock it under various labels rooted in its IUPAC or classical names. This can trip up students checking reference literature or workers tracking down MSDS sheets, making cross-referencing part of the daily grind for anyone dealing with more than a handful of acids.
Safety & Operational Standards
Repeated exposure calls for respect. Skin contact irritates, inhalation causes discomfort, so proper gloves, goggles, and a working fume hood stay non-negotiable, especially since crystalline pivalic acid stings in open wounds. Most researchers learn protocol the hard way—after a careless afternoon leaves red knuckles or a burning throat. Storage in steel or glass, away from bases and reactive agents, sidesteps most mishaps. Clear labeling wards off accidents, which long-time lab staff realize protects both newcomer and expert. Disposal routines reflect the growing demand for environmental accountability. Standard practice includes neutralization and organized waste streams, as unchecked carboxylic acids sometimes sneak by into water systems, drawing scrutiny from regulatory bodies. Compliance rests on knowledge, habit, and a healthy fear of repeating mistakes of the past.
Application Area
Pivalic acid’s reach stretches far. Its esters and salts play vital roles in pharmaceuticals, perfumery, and agrochemistry. Pharmaceuticals rely on it for active compound modification, delaying metabolism or improving bioavailability. Crop protectants and growth regulators feature pivalic acid derivatives that boost environmental stability, extending shelf life and field potency. Even high-performance plastics and engineered resins owe part of their toughness to this bulky acid’s contributions. In flavor and fragrance work, pivalate esters add subtlety and persistence to formulations where evaporation rates matter. Its sheerness and tenacity stand as a testament to how a simple, short-chained acid supports industries that rely on precision and predictability in every batch.
Research & Development
Ongoing projects push pivalic acid into new corners of science. Medicinal chemists continually tweak pivaloyl derivatives to improve the delivery and action of novel drugs. Polymer research values the acid group’s ability to resist breakdown, especially under harsh conditions like automotive or aerospace environments. Researchers chase more sustainable production methods, hunting for routes that cut down on raw materials, waste, and emissions, especially now that supply chain concerns push efficiency back into the spotlight. At universities, trainers use pivalic acid to demonstrate the real-world utility of functionalized acids, often paralleling broader lessons in green chemistry and career-long habits of careful reagent selection.
Toxicity Research
Toxicologists say pivalic acid isn’t the worst offender among carboxylic acids, though they don’t dismiss its hazards. Inhalation, ingestion, and skin exposure yield discomfort, sometimes enough to draw blood test abnormalities, with repeated or heavy exposure leading to liver and kidney stress in animal studies. Agencies reviewing environmental impacts watch for long-term persistence, since the branched structure can resist natural degradation pathways. Responsible labs commit to tightly controlled usage, swift cleanup cycles, and regular refresher training, so that bad habits don’t spiral into routine violations or community risk.
Future Prospects
Looking forward, the push toward greener chemistry means pivalic acid’s supporters face pressure to tighten up synthesis and downstream handling. Trends lean toward biosynthetic alternatives, efficient recycling of process solvents, and switchovers to continuous-flow manufacturing. New derivatives entering clinical trials each year keep demand healthy, while materials science drives the exploration of pivalate-based polymers for demanding, high-temperature settings. The story of pivalic acid showcases how overlooked compounds still have plenty of stories to tell. Its journey isn’t just about what’s on the label, but how its resilience, accessibility, and adaptability shape the products and experiences that define daily life for many, often without recognition, but always with quiet impact.
What is pivalic acid used for?
Not Just Another Industrial Chemical
You probably won’t see pivalic acid sitting on a grocery shelf. Yet for a lot of products people use daily, this small molecule works behind the scenes. Speaking as someone who enjoys tinkering with chemistry experiments and pays attention to product safety, I know certain chemicals find their way into both industrial and everyday life. Pivalic acid is one of those unassuming but useful compounds. Looking like a clear, sometimes faintly yellow liquid, it has a distinct scent and packs a punch in the right settings.
Building Better Polymers and Plastics
Anyone who's ever tried to understand what makes a durable plastic water bottle tough will run into chemicals like pivalic acid. Manufacturers often use it as a building block for specialty polymers. Some plastics need specific characteristics, such as resisting high temperatures or staying rigid under pressure. Pivalic acid helps create these properties. Modern electronics, car parts, and even home goods benefit from tweaks in polymer chemistry, much of the time using this acid as a starting point.
Supporting the Pharmaceutical Field
The story shifts in medicine. Pharmaceuticals need specialized chemicals in their creation. Pivalic acid acts as a raw material in the synthesis of various drugs. Take prodrugs, for example: these get broken down inside the body to release active medicine. Chemists often add pivalate groups to a molecule for better absorption or longer shelf stability. This strategy boosts how medicine works once taken. Antibiotics and anti-inflammatory medications in particular lean on pivalic acid’s properties to work efficiently.
More Than Just a Solvent
Lab settings sometimes demand a tough, stable solvent. Pivalic acid steps in firmly here, making it easier to separate chemicals or drive certain reactions forward. Its resistance to oxidation and heat gives it an edge over some traditional options. During college research, I’ve seen chemists reach for pivalic acid, knowing they can push experiments without worrying about quick breakdowns or spoiling delicate reactions.
Safety: A Big Piece of the Puzzle
Working with pivalic acid comes with responsibility. It’s not something to splash around carelessly. Inhaling its fumes or getting any on your skin could lead to irritation. Companies and labs must follow strict guidelines for handling, disposal, and storage. It’s worth pointing out that its use, especially in pharmaceuticals, comes under tight regulation. Organizations such as the FDA and EPA watch chemicals like this closely to ensure human health and protect the environment.
Looking for Better Options
Chemical industries keep searching for safer, greener materials. Pivalic acid, while useful, falls under scrutiny because of possible environmental impacts if mismanaged. Green chemistry pushes scientists to design molecules that break down easily, pose less risk to water sources, or require less energy to produce. Some research groups try replacing pivalic acid in certain processes with more sustainable alternatives, or even look for ways to recycle it efficiently from waste streams. These changes, although slow, aim for both performance and safety.
Paying Attention to Tomorrow’s Chemistry
One lesson from following the story of pivalic acid: chemistry shapes everyday life in ways most people don’t notice. From plastics to pills, small molecules play big roles. Keeping an eye on how industries use chemicals, and how they manage waste, helps push innovation toward healthier, safer products.
Is pivalic acid safe to handle?
The Real Risks of Working with Pivalic Acid
Pivalic acid pops up in plenty of chemical labs, especially where people deal with organic synthesis. Anyone working with chemicals knows that even everyday substances can pack hidden dangers, so it makes sense to look at pivalic acid with a careful eye.
Pivalic acid doesn’t grab headlines the way stronger acids do, but its hazards deserve notice. The chemical, known for its sharp, creamy odor, feels like a basic organic acid to many, but its skin and respiratory risks deserve just as much respect as more aggressive compounds. Even small splashes irritate the skin and eyes, sometimes causing redness or burning that takes time to fade. I’ve seen folks in research circles who skip gloves on “routine days” and regret it after a careless touch.
The Material Safety Data Sheet (MSDS) for pivalic acid calls out its acute toxicity when inhaled or ingested. Vapors from heating or accidental spills cause coughing, shortness of breath, and for some, triggering of asthma symptoms. In my experience, unfamiliarity invites shortcuts—people get lazy, thinking "it's just an acid," and skip using fume hoods. Even brief exposure to its dust or vapor can leave a scratchy throat or itching eyes.
Safe Practices Pay Off
Good ventilation stays key during any handling. Fume hoods never sit unused on chemical benches for a reason—local exhaust capture delivers the best protection in a real lab. Open windows or basic fans can't replace that, no matter how small the job looks. Goggles and lightweight gloves become standard gear because prevention beats scrambling for eye wash. The acid’s solid crystals sting far quicker than folks expect.
Accidents sometimes happen because of improper storage. Pivalic acid reacts strongly with oxidizers and bases. I once watched a shelf get rearranged without thinking, and incompatible reagents ended up within inches. Storing acids away from anything reactive or moisture-sensitive eliminates one more source of accidents. It works best to keep it in a tight container in a cool, dry spot, never near food or open drinking cups. Even trace contamination on shared surfaces can linger and create headaches for others later.
The Human Factor
Complacency leads to problems, not the pivalic acid itself. At home or in industry, clear protocols and well-placed warnings make all the difference. Up-to-date safety training does more than tick a compliance box. It empowers lab techs and students to recognize symptoms, respond to spills fast, and skip the hush-hush attitude that sometimes turns a small issue into a medical call.
Pivalic acid has seen its use in pharmaceuticals, esters, and specialty materials. It has a role in chemistry, but that doesn’t mean it’s completely benign. People handling it should read the label, review the MSDS, and never cut corners. Those steps align with best practices recommended by both OSHA and the American Chemical Society.
Looking Ahead
Safer workspaces start with honest talk about risk, regular drills, and enough personal protective equipment within arm’s reach. Spreading real experiences—close calls and minor burns included—helps new chemists watch their step. Pivalic acid embodies the gray area between casual and dangerous status. Careful respect, not fear or indifference, keeps everybody safe.
What is the chemical formula of pivalic acid?
A Look at Pivalic Acid’s Structure
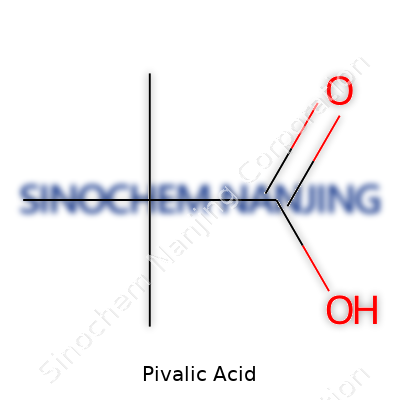
Ask a chemist about pivalic acid and you’ll see recognition in their eyes. The chemical formula stands as C5H10O2. This simple set of letters and numbers packs a punch when it comes to understanding just how carbon-based compounds get used, studied, and even regulated. The backbone of pivalic acid comes from three methyl groups arranged around a central carbon. Top it with a carboxylic acid group, and it shapes up as a small, sturdy molecule. Researchers call it also 2,2-dimethylpropanoic acid, but most in the lab stick with "pivalic acid."
Why the Structure Matters
The shape of pivalic acid matters to more than just chemists bent over lab benches. If you’ve spent any time around plastics or pharmaceuticals, you might have seen pivalic acid turning up as a building block. The structure—three methyls on a single carbon—makes the molecule resistant to breakdown by many common chemicals and microbes. Industries see value here because chemical stability means fewer headaches in storage and processing. No one likes surprises when it comes to shelf-life.
In practice, I have watched colleagues debate over using pivalic acid in organic synthesis. The bulky groups often protect a reaction site, ensuring nothing sneaks in and messes up an experiment. That protectiveness pays off in drug research, where enzymes often break down smaller acids. Pivalic acid doesn’t give up so easily, making it a favorite for chemists developing stable drug formulations or exploring new polymer blends.
Health, Safety, and Regulation
No conversation about chemicals in daily use skips concerns over safety. Pivalic acid’s stability brings two sides to the table. On one hand, it doesn’t easily react with skin or tissue, so casual contact rarely causes harm. Spill some on a glove, and soapy water usually does the trick. On the other hand, its usefulness has pushed researchers to ask larger questions about persistence in the environment. What feels stable in the lab sometimes lingers in water or soil, leading to calls for responsible handling and disposal practices.
I’ve seen safety data sheets that remind everyone to wear goggles, avoid inhaling dust, and keep pivalic acid away from heat sources since it can irritate the eyes and, at high concentrations, the lungs. These reminders get repeated in safety meetings, not just out of formality, but because small slip-ups can cascade into much bigger messes.
Looking at Solutions
Researchers keep working on ways to harness pivalic acid’s good qualities while minimizing the downsides. Newer production techniques aim to cut waste and limit emissions during manufacture. In the lab, we use sealed containers and track inventory closely, both for economic reasons and to keep the environment clean. Some start-ups talk about offering greener substitutes for uses where extreme chemical stability isn’t crucial.
Pivalic acid highlights the give-and-take that runs through much of industrial chemistry. Balancing performance, cost, and safety calls for details. Instead of cutting corners, keeping updated with current safety practices and recycling programs lowers risks on shop floors and in the wider world. The chemical formula C5H10O2 means more than just atoms—it’s a reminder that even small molecules make a broad impact when handled thoughtfully.
How should pivalic acid be stored?
Experience Teaches the Value of Diligence
Storing chemicals in the lab looks easy, until someone forgets a detail. I’ve watched colleagues scramble after a mislabeled bottle turned an afternoon into a cleanup nightmare. Pivalic acid stands out for its strong odor and sharp, almost vinegary quality – it’s unmistakable once you’ve worked with it. Leaving a bottle uncapped, or shoving it among incompatible materials, risks ruining expensive equipment and could put people in harm’s way.
Risks Can Multiply under Improper Storage
Pivalic acid is flammable. It gives off fumes that can irritate eyes and the respiratory tract. Its melting point sits lower than room temperature, so it doesn’t always look the same – one day it’s a solid, by the next afternoon it’s half-melted in a warm lab. From what I’ve witnessed, this unpredictability tricks new staff into lowering their guard. Vapor exposure happens before anyone realizes a spill occurred. In a chemistry department, I once watched a technician rush to neutralize a small puddle after poor storage led to condensation around the bottle seal. That same week, lingering odors tipped off staff to improper ventilation. These moments stick with me. It only takes one oversight to impact an entire workspace.
Best Practices Build a Culture of Safety
I’ve learned the value of reading safety data alongside customer testimonials. Pivalic acid belongs away from open flame, heat, strong oxidizers, or bases – it’s not wise to pop it onto a random shelf near caustic agents. Cool, dry storerooms with plenty of ventilation make a difference. A single, well-sealed glass container cuts down the risk of leaks and keeps odors contained. I always double-check the lid and label, since faded writing leads to confusion later.
Gloves, goggles, and a lab coat form the most basic layer of protection. If a spill does happen, access to spill absorbents and a working fume hood make cleanup safer. From what I’ve practiced, minimal handling means fewer opportunities for accidents. In my experience, keeping the bottle within reach but in a labeled, protected cabinet adds a layer of security. The best labs use safety training to reinforce these habits, not just file paperwork. Teams discuss past mistakes, boosting everyone’s focus and caution.
Supporting Safe Handling with Facts and Vigilance
Pivalic acid finds use in pharmaceuticals and polymers, but its hazards extend to amateur makers and students. Standard safety data indicates storage temperatures between 15°C and 25°C, away from sunlight and humidity. Strong acids or bases nearby speed up decomposition, so physical separation matters. Every serious storage space relies on a chemical inventory sheet updated regularly. Not long ago I watched an audit reveal several containers bought long ago, now past expiration. The supervisor seized the moment to launch a cleanup project, which prevented possible exposure to degraded chemicals.
The lesson here: it’s not just about following rules. Personal stakes run high with chemicals. Comfort grows with practice and attention, and a single shortcut brings regret. Treating pivalic acid with respect means a safer, lighter workload for everyone. That’s a standard I want to see everywhere chemicals get handled.
What are the physical properties of pivalic acid?
Getting to Know Pivalic Acid
Anyone who has stepped into a proper laboratory will recognize the sharp, penetrating odor of certain organic acids. Pivalic acid, also known as trimethylacetic acid, finds its way into many labs and chemical syntheses, but people often underestimate its straightforward yet unique physical character. If you pour a small sample into a beaker, the first thing you’ll notice is that it looks like water — a clear, colorless liquid or sometimes a soft, crystalline solid, especially if the room runs cool. Lab coats and old textbooks describe the melting point sitting around 35–36°C, so it sometimes appears solid even at room temperature.
The odor stands out: strong and sour, almost hard to miss, a bit like everyday vinegar but with a more industrial punch. This makes all the difference during work, both a warning and a navigational cue, especially if you have a nose trained to pick up toxic vapors. I remember carefully uncapping a bottle for the first time: the hiss, the aroma, the instant understanding this wasn’t something to leave unsealed overnight.
Boiling Point and Handling
Pivalic acid boils up at about 164°C. This seems middle-of-the-road among similar-sized organic acids, yet it shifts how it behaves under heat. Set a reaction flask over a Bunsen burner and before long, the room fills with that unmistakable vapor. If you work in a lab with limited ventilation, you’ll quickly learn the wisdom of safety protocols and a working fume hood — no one wants lingering acid fumes.
Water solubility is pretty low for pivalic acid. Drop it into a glass of water, and most stays apart, forming oily beads or a floating layer. This comes from its bulky shape, thanks to its three methyl groups crowded together. As a chemist, it’s clear: if you want to dissolve pivalic acid, you turn to organic solvents such as ether or chloroform rather than water. The lower solubility can present issues for any waste disposal procedure, making it important to follow local chemical safety guidance to protect groundwater and sewer systems.
Density and Storage Concerns
The density lands around 0.93 grams per cubic centimeter — a touch lighter than water. Pouring it from one flask to another, the difference won’t jump out at you; it still flows with a familiar viscosity. The key to storage lies in its reactivity and volatility. Leaving the cap loose means you’re losing product every hour, and the liquid can slowly eat into certain plastics or metals. Lab practice — and life experience — always points toward glass containers with tight stoppers stored in cool cupboards.
Applications and Safety
In the real world, the physical properties of pivalic acid guide every application. Most of its industrial use draws on its toughness and ability to resist breakdown. It’s used to make useful esters and as a protecting group in organic synthesis because it doesn’t fall apart easily. This property means fewer unwanted byproducts, greater yield, and better control during complex chemical reactions. Those characteristics come straight from its physical form — low water solubility, stability, a high melting point for its size.
Respect for its odor, volatility, and reactivity shapes daily lab life. Gloves and eye protection remain standard. Leaving containers open or working without proper airflow leads to unnecessary exposure, and the physical experience teaches care and attention far better than a safety manual ever did. The unique combination of physical properties — from the powerful smell to the low solubility — ensures pivalic acid finds steady use where reliability and predictable behavior count. In chemistry, knowing how a substance looks, smells, and moves sets the stage for safer and more effective work.
| Names | |
| Preferred IUPAC name | 2,2-Dimethylpropanoic acid |
| Other names |
Trimethylacetic acid 2,2-Dimethylpropanoic acid Pivalate |
| Pronunciation | /paɪˈvælɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 75-98-9 |
| Beilstein Reference | 1209229 |
| ChEBI | CHEBI:38536 |
| ChEMBL | CHEMBL418444 |
| ChemSpider | 6769 |
| DrugBank | DB03912 |
| ECHA InfoCard | 100.007.963 |
| EC Number | 204-689-0 |
| Gmelin Reference | 8188 |
| KEGG | C00956 |
| MeSH | D010875 |
| PubChem CID | 7908 |
| RTECS number | **OV1925000** |
| UNII | 8TS35J9S8W |
| UN number | UN2529 |
| Properties | |
| Chemical formula | C5H10O2 |
| Molar mass | 102.13 g/mol |
| Appearance | White crystalline powder |
| Odor | pungent |
| Density | 0.934 g/cm³ |
| Solubility in water | 5.1 g/L (20 °C) |
| log P | 1.51 |
| Vapor pressure | <0.1 hPa (20 °C) |
| Acidity (pKa) | 5.03 |
| Basicity (pKb) | 10.10 |
| Magnetic susceptibility (χ) | χ = -54.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 1.15 mPa·s (25 °C) |
| Dipole moment | 1.59 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -486.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2886.6 kJ/mol |
| Pharmacology | |
| ATC code | A16AA08 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05, GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P280, P261, P305+P351+P338, P304+P340, P310, P312 |
| NFPA 704 (fire diamond) | 2-3-0-W |
| Flash point | 161 °C (322 °F; 434 K) |
| Autoignition temperature | 385 °C (725 °F; 658 K) |
| Explosive limits | Upper: 2.6%, Lower: 1.1% |
| Lethal dose or concentration | LD50 oral rat 1400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1700 mg/kg |
| NIOSH | SN4025000 |
| PEL (Permissible) | No PEL established. |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Trimethylacetic anhydride Neopentyl alcohol Neopentyl bromide Pivaloyl chloride Pivalamide |

