Pivalaldehyde: A Deep Dive into a Unique Specialty Aldehyde
The Journey of Pivalaldehyde Through History
Chemistry has a way of surprising everyone, and pivalaldehyde is one of those molecules that’s managed to stick around in the background of organic synthesis for decades. The roots of this compound go back to early 20th-century explorations of branched-chain aldehydes. Researchers set out to modify existing aldehydes, tweaking structures to see how branching impacted reactivity and biological behavior. Out of these early chemical experiments, pivalaldehyde emerged, marking a real shift in how chemists thought about steric effects and structural hindrance. The bulky tert-butyl group attached to the aldehyde carbon carved out a niche for pivalaldehyde, pushing its applications beyond routine carbonyl chemistry. As industries embraced custom synthesis, this quirky aldehyde secured a place in laboratories dedicated to advancing pharmaceuticals, fragrances, and fine chemicals.
What Sets Pivalaldehyde Apart
At first glance, pivalaldehyde doesn't look like much: a small molecule, clear liquid in the bottle, with a sharp, almost stinging aroma. Its chemical name, 2,2-dimethylpropanal, points to its structure, where the central carbon of a tert-butyl group anchors an aldehyde function. This seemingly simple arrangement turns pivalaldehyde into a model for studies on steric effects. That huge tert-butyl group crowds the reactive center, making typical aldehyde transformations harder, or at least different, than with less hindered relatives like acetaldehyde or propionaldehyde. The result? Pivalaldehyde offers a rare mix of stability in air and water paired with a curious reluctance to overreact, which has people coming back to it for specific synthetic challenges.
The Science Behind Pivalaldehyde’s Physical and Chemical Nature
Anyone handling pivalaldehyde quickly notices the volatility: it evaporates readily at room temperature. With a boiling point hovering just above 75 °C, this compound is less persistent than heavier aldehydes. Its density stays low, reflecting the abundance of light hydrogen and carbon atoms in its backbone. Chemically, the fact that the tert-butyl group flanks the carbonyl carbon changes everything about how this aldehyde behaves. The electron-donating bulk increases resistance to nucleophilic attack, setting it apart from aliphatic cousins. This translates into less reactive behavior in simple addition or condensation reactions, but also opens doors for selectivity where one wants limited side reactions. Stability, volatility, and resilience under mild acid or base all feed into its specialty uses, especially when compared to more temperamental aldehydes.
Technical Details and Labeling: More Than Just a Name
Working with chemicals means knowing what’s in the flask—no room for guesswork. Pivalaldehyde usually comes labeled as 2,2-dimethylpropanal, with synonyms like pivalic aldehyde or neopentanal popping up in catalogs. Regulations require a CAS number, and responsible suppliers provide information on purity grades, water content, and method of storage. Typical bottles flag its flammability and the need to avoid open flames or prolonged exposure to air. Labels echo safety standards, including the need for gloves and ventilation, since volatile organics like this tend to sneak past unprepared handlers. Documents like Safety Data Sheets list not just its dangers, but also offer insight into decomposition and incompatibilities, reminding anyone in the lab that a moment’s carelessness can mean exposure to eye or skin irritants.
How Chemists Prepare Pivalaldehyde
Synthesis of pivalaldehyde often relies on the controlled oxidation of the corresponding alcohol, pivalyl alcohol (neopentyl alcohol). Industrial producers lean on oxidants that favor the delicate balance between complete conversion and over-oxidation. Chromium(VI)-based reagents used to be popular, thanks to high yields, but concerns about toxicity and waste have shifted many labs toward safer oxidants like manganese dioxide or catalytic TEMPO with bleach. Another classic approach starts from pivalic acid, which chemists convert through chlorination to the acid chloride, then reduce to the aldehyde with specialized reagents like DIBAL-H. Each method reflects a tug-of-war between efficiency, environmental impact, and worker safety—a real-world example of how green chemistry shapes today’s specialty chemicals industry.
The Reaction Toolkit and Modification Potential
Chemists value pivalaldehyde for the way its crowded structure dictates outcomes. The molecule’s inherent steric bulk makes its reactivity in nucleophilic addition, like those with Grignard reagents or ammonia derivatives, far lower than you'd see with less hindered aldehydes. This doesn’t mean it's useless—just more selective. For example, oxime and hydrazone formation proceeds, but often at slower rates and with higher selectivity for desired products. The tert-butyl group keeps radical or redox pathways honest, limiting over-reactions that sometimes derail syntheses. In the hands of creative researchers, pivalaldehyde can serve as a blocking group or a precursor to bulky building blocks for pharmaceutical intermediates or advanced ligands in catalysis, showing that limitations can spur innovation.
What’s in a Name: Synonyms and Product Naming Conventions
Pivalaldehyde goes by plenty of names, depending on whether a chemist values IUPAC formality or classic trade usage. "2,2-dimethylpropanal" spells out the structure, while "neopentanal" refers back to its neopentyl skeleton. Some catalogs keep things simple with just "pivalic aldehyde." Each name reflects different priorities: precision, tradition, or ease of reference. A product’s catalog listing might include all three, and that speaks to the overlapping circles of nomenclature in chemistry, where a single molecule often treads many paths to market.
Safety Standards and Operational Realities
Dealing with pivalaldehyde means paying attention to more than just chemical purity. Volatility and irritant properties make adequate ventilation a non-negotiable part of lab setups. Direct contact can inflame sensitive skin or eyes—protective gear should never gather dust on the shelf. Spills demand quick cleanup, since lingering vapors put everyone at risk. Since aldehydes in general can trigger allergic responses or act as respiratory irritants, labs handling this substance must train staff in proper handling, storage, and emergency protocols. Disposal calls for neutralization or incineration under controlled conditions, steering clear of environmental contamination. Adhering to evolving safety legislation reduces risks and keeps both the workforce and wider community protected.
Pivalaldehyde’s Calling Cards: Application Areas
Real demand for pivalaldehyde often comes from sectors that need both selectivity and stability. In pharmaceutical R&D, the compound becomes a building block, letting chemists add bulk to intermediates or shield reactive centers with its tert-butyl arm. Specialty coatings and resins exploit its controlled reactivity to build polymers with rigid, branched frameworks. Flavors and fragrance industries take a more niche interest—its sharp odor and volatility play a role in complex aroma profiles and scent masking efforts. Meanwhile, academic groups use pivalaldehyde to probe how branching impacts reaction pathways and side product formation, generating fundamental data that feed back into industry process design. Each sector tailors use to its particular demands, leveraging the unique combination of reactivity and steric protection.
Research and Innovation: The State of Play
No specialty chemical stands still, and pivalaldehyde is no exception. Researchers continually test new pathways to cleaner, more cost-effective synthesis. Recent advances in green oxidations, especially electrochemical and biocatalytic methods, hold promise for lower waste production and safer reactions. Studies charting reaction mechanics deepen understanding of how tert-butyl branching impacts nucleophilic attack, while others use pivalaldehyde as a model to train AI systems on chemical prediction and automated synthesis. Work on catalysis and ligand development often circles back to this aldehyde, seeking out ways to leverage its unique bulk. The focus on sustainable chemistry means the future of pivalaldehyde rests as much on cleaner processes as on traditional applications.
Toxicity and Human Health Concerns
Pivalaldehyde, like most low molecular weight aldehydes, carries risks that science takes seriously. Acute exposure to vapors can inflame mucus membranes, trigger headaches, or lead to more serious nervous system effects. Chronic, low-level exposure raises questions about sensitization and immune responses, with animal studies pointing to potential for moderate toxicity if misused. Regulatory agencies keep a close watch on exposure limits, adapting standards as new data emerge. In the workplace, continuous monitoring and staff education remain critical, ensuring early detection of adverse health impacts and quick intervention. Research targets both immediate effects—such as irritation or short-term sickness—and lonq-term impacts, which remain under active study due to the relatively rare, but persistent, use of pivalaldehyde in industry settings.
The Road Ahead for Pivalaldehyde
Looking into what lies ahead, two things drive the conversation: sustainability and shifting demand in advanced manufacturing. As labs race to shrink their environmental footprint, efforts focus on greener syntheses using recyclable catalysts, safer oxidants, and less energy-intensive processes. The evolving landscape of pharmaceutical and polymer manufacturing underscores the need for building blocks like pivalaldehyde that combine resilience with tunable reactivity. Researchers keep chipping away at new applications, exploring hybrid molecules for electronics, specialized adhesives, or medical devices, building on the backbone provided by the tert-butyl-aldehyde structure. As the world moves toward precision chemistry in everything from drug development to new materials, the persistent utility of pivalaldehyde keeps it on the short list for chemists who want control, selectivity, and solid performance without the nasty surprises that come with more volatile or less stable alternatives.
What is Pivalaldehyde used for?
Getting to Know Pivalaldehyde
Pivalaldehyde might not sound like something most folks talk about at the dinner table, but its place in science is bigger than you might expect. This compound stands out due to its unique structure, with its chunky tert-butyl group right next to an aldehyde function. In plain English, it’s a versatile building block, an ingredient that chemists reach for when ordinary chemicals just won’t cut it.
Why Chemists Rely on It
I've seen pivalaldehyde mentioned in chemistry labs, especially in research that needs something both reactive and sterically hindered. This compound lets scientists make more complex molecules by blocking certain reactions and steering the chemistry in the right direction. Think of it as giving a recipe a special seasoning that changes the whole dish for the better.
Aldehydes in general can spark a lot of reactions, but pivalaldehyde does more: its bulky side helps shape the results. It often shows up in the early steps of pharmaceutical drug development. The world rarely sees the direct impact because the real payoff surfaces only after a string of precise chemical tweaks. Some advanced medications—cancer treatments, antibiotics—trace roots back to starting materials like pivalaldehyde. This matters, because what you start with often decides how safe or effective the final drug will be.
Behind the Scenes in Industry
Outside the pharmaceutical world, pivalaldehyde has made appearances in specialty material science. Engineers use similar aldehyde compounds to kick off polymer reactions or synthesize advanced plastics. Durable resins, coatings, and adhesives benefit from building blocks just like this one, chosen for their ability to handle heat or chemical exposure.
Some technologies demand pure final products—electronics, medical tools, and aerospace parts spring to mind. For this, even the minor roles played by starting chemicals take on real importance. I remember seeing case studies where swapping one aldehyde for another improved stability or changed the way surfaces reacted under stress. These minor tweaks can end up delivering longer-lasting products or safer outcomes for consumers.
Safety and Ethical Concerns
Pivalaldehyde, like many chemicals, asks for respect. Exposure can irritate skin and lungs—chemists rely on gloves, goggles, and proper ventilation. This isn’t just for lab safety: it shapes how companies transport and dispose of chemicals, raising essential environmental questions.
For those of us who care about safe workplaces and clean water, it’s a reminder that every obscure ingredient comes with responsibility. Regulations step in, but they don’t cover every scenario. As science pushes forward, industry faces a challenge: keep innovation moving without cutting corners on health or the environment. I’ve watched debates over chemical regulation play out—balancing progress against risk can get heated, especially where potent substances are involved.
Innovation and Alternatives
Green chemistry now encourages researchers to look for starting materials that break down easily and reduce toxic waste. For pivalaldehyde, some newer labs test renewable feedstocks or recycle parts of the molecule after use. These shifts aim to cut pollution and protect communities around chemical plants.
Curiosity about lesser-known compounds often pushes science forward. Each specialty ingredient—pivalaldehyde included—reminds us that every new breakthrough carries a story of risk, reward, and the search for responsible solutions.
What are the physical and chemical properties of Pivalaldehyde?
What Makes Pivalaldehyde Stand Out
Pivalaldehyde, with its more technical name 2,2-dimethylpropanal, often catches the eye in a lab setting thanks to its unique smell and structure. The molecule carries a bulky tert-butyl group, which makes it different from most simple aldehydes that tend to have smaller side chains. Anyone who's spent much time handling organic chemicals might recognize its distinct sharp aroma, reminding one of apple peels layered with something slightly medicinal. This smell alone marks it as a chemical you don't soon forget.
Getting Down to Physical Details
You won't find this stuff solid at room temperature. Pivalaldehyde turns up as a clear, colorless liquid. Its boiling point lands around 74°C (165°F), a solid marker that it evaporates quicker than water. Pour a little on your glove (don't, really), and it'll start vanishing in the air fast. Its melting point sits far below zero, about -65°C, which means storing it in a general cold room won't freeze it solid.
The substance has a higher density than water, around 0.8 grams per cubic centimeter, so a drop floating on water might just struggle to dissolve rather than sink. For context, that’s lighter than chloroform but heavier than ether — a sweet spot for many organic solvents. Its vapor pressure doesn't let it linger quietly; it evaporates with enthusiasm if left open to air. In most college labs, it’s kept tightly capped because those fumes sneak out with a vengeance.
Chemical Personality That Shapes Its Use
Pivalaldehyde leans on the same kind of carbonyl chemistry that makes aldehydes reactive. It takes on nucleophiles, likes to join condensation reactions, and won’t shy away from oxidation or reduction. Its notable feature rests in that tert-butyl (C(CH3)3) tail, which acts like a shield. That bulk keeps a lot of reactive species at bay and slows down changes at the aldehyde carbon.
Trying to oxidize it straight to an acid calls for patience or a strong hand with the reagents. The molecule doesn’t roll over easily. At the same time, the steric crowding from the tert-butyl group makes it tough for bases and nucleophiles to crowd around the carbonyl site for simple addition reactions. Chemists looking to build something big often lean on this property, using pivalaldehyde as a building block that resists unwanted side reactions.
Handling with Respect: Safety and Reactivity
Breathing in vapors can irritate your nose and throat pretty quickly. It’s best to work with it under a fume hood with gloves and goggles on. Skin contact rarely causes a reaction, but it leaves behind that smell for hours. Spills evaporate fast, making cleanup about catching vapors before they drift up your nose.
Storing pivalaldehyde doesn’t require cold temperatures, but direct sunlight speeds up unwanted reactions. Over time, it forms peroxides — that’s a big red flag in any lab. Old bottles should get tested and handled carefully. Disposal means mixing with an oxidizer or incinerating it under controlled conditions to avoid harm to your colleagues or community.
Why It Matters
Labs need substances that let them run reactions without unexpected jumps. Pivalaldehyde fills a niche where reactivity gets tuned down, not revved up. Researchers use it to build complex molecules, with the tert-butyl group acting like armor in multi-step syntheses. Anyone designing safe and efficient routes to pharmaceuticals probably passes this aldehyde on their journey. Understanding its physical quirks and chemical habits keeps the work honest and the risks clear.
How should Pivalaldehyde be stored?
The Risks You Face with Pivalaldehyde
Pivalaldehyde isn’t your average chemical. With a strong, pungent scent and a reputation for being flammable and reactive, it commands respect and vigilance inside any laboratory where it shows up. I’ve seen researchers almost shrug off storage advice with less risky chemicals, but you can’t cut corners with this one. Let a bottle stay open or bring it near heat, you’re not just risking an accident—you could set off a fire.
Tight Lids, Cool Spaces, and Ventilation
Picture yourself coming into a lab after a weekend, only to catch a whiff of something sharp and biting in the nose. That’s often the sign of a bottle that wasn’t sealed right or ended up too warm. Pivalaldehyde likes to escape into the air, and nobody wants its fumes in their workspace. The right way to keep this chemical involves airtight containers—glass with tight, chemical-resistant lids, never plastic that might degrade or leak fumes.
Forget about storing it on some old shelf at room temperature. Refrigerators designed for flammable materials provide a stable, low-temperature environment, which limits evaporation and reduces fire risk. Any ordinary fridge or freezer can spark or leak air back into the lab, but explosion-proof models protect the room and anyone walking by.
Light and Air: Enemies of Stability
Some folks ignore just how much light speeds up decomposition or the way oxygen sparks reactivity. I’ve watched bottles left under laboratory lights turn yellow and start building up gas pressure inside after only a few months. Opaque wrapping or amber glass keeps unwanted ultraviolet rays out, and regular checks catch signs of degradation.
The best scientists in my experience develop habits: double-checking labels, replacing cracked lids, and keeping chemicals in secondary containers. Accidents usually trace back to someone rushing, thinking, “It’ll only be for a few days.” With volatile aldehydes, those shortcuts never pay off.
Preventing Spills and Unplanned Reactions
Bad storage behavior often leads to bigger disasters. Once, I saw a technician shove a fresh delivery onto a crowded shelf, setting Pivalaldehyde beside a strong acid. Separate storage keeps reactive chemicals from mixing and turning a minor spill into a toxic event. Floors that slope toward a chemical hood, with proper spill trays below, provide extra protection. I remember a minor spill that could’ve sent fumes across half the building if it weren’t for well-placed trays and the habit of opening containers inside the hood.
Labeling bottles with clear, legible details makes it easier to spot mistakes before they happen. In tightly-run labs, I always see logs tracking who opened what and when, because good information stops small issues from turning into full-blown emergencies.
Regular Training and Emergency Planning
No storage system replaces a trained team. I’ve watched newcomers get lax with personal protective equipment, sometimes leaving gloves off for just a second. Proper training—reinforced every few months—reminds everyone why protection matters. Emergency protocols, displayed right on entry doors and drilled into memory, set the foundation for safe chemical storage.
Storing Pivalaldehyde calls for respect, good habits, and a clear mind. You can build safety into daily routines, not because rules demand it, but because it keeps everyone healthy. Even today, the smartest chemists I know call out problems early, fix them fast, and stay humble around chemicals with dangerous tempers.
Is Pivalaldehyde hazardous and what safety precautions are needed?
Pivalaldehyde: Small Molecule, Big Responsibilities
Pivalaldehyde, a colorless liquid, shows up in laboratories more often than you'd expect. I’ve worked in organic synthesis and noticed that folks sometimes treat less flashy chemicals with less respect. This one deserves genuine attention. It’s got a sharp, penetrating odor that signals right away it isn’t just some harmless solvent. This stuff packs a punch if mishandled.
Hazard Profile: Why Caution Matters
Based on material safety data and first-hand experience, pivalaldehyde acts as a strong irritant. Vapors cause burning sensations to eyes and throat, and that’s not something you want to push off as a minor nuisance. On contact with skin, it can trigger redness, itching, and even blistering if contact lasts longer than just a few seconds. One spill on my lab sleeve taught me to double up gloves every time.
It’s not just a skin or eye irritant. The chemical tends to form peroxides, which become shock-sensitive over time. That means storage in open air or in clear glass, especially near windows or heat, runs the risk of the compound breaking down and causing explosions. Colleagues from other labs have had near-misses due to this very reason.
Health agencies draw strict exposure limits for good reason. Inhalation at high concentrations leads to coughing and dizziness. Chronic exposure can lead to headaches and, based on animal studies, the risk of damaging liver or kidney function grows with repeated exposure. Each fact pushes home the point: this is not a pantry staple — it’s a lab hazard.
Steps for Safe Handling
Pivalaldehyde belongs in fume hoods, not on open benches. Airflow removes vapors before they hit your lungs. Goggles are not optional here. Face shields work even better, especially during bulk transfers. Short gloves with thin nitrile often tear open, so thicker, longer gloves provide genuine protection. Wearing a lab coat keeps skin covered, and shoe covers handle splash risks on the floor.
Flammability makes things trickier. Heating belts or hot plates shouldn’t come close unless you’re equipped for high-flashpoint solvents. I know a chemist who lost months of work after a container ignited next to a faulty plug; the clean-up cost thousands and halved their reputation in the group. Store pivalaldehyde under nitrogen in dark, sealed bottles, preferably behind secured cabinets. Adding stabilizers slows peroxide buildup.
Disposal and Emergency Responses
Spill control kits should always be within reach where pivalaldehyde gets opened. Neutralize small spills with inert absorbents, and never shove contaminated rags into open trash bins. Hazardous waste streams handle all unused or expired material — pouring it down a drain damages more than just the pipes.
Accidents still happen, even with checks and balances. Quick rinsing under water helps after skin exposure, and eyewash stations save vision if splashes strike the face. Health and safety officers train folks for a reason: following their protocols isn’t about red tape, it’s about going home healthy.
Building a Culture of Safety
It’s easy to think of only the big explosive molecules as dangerous, but pivalaldehyde proves otherwise. Running a lab goes beyond just getting results; it’s about responsibility for everyone who steps through its doors. If someone looks at a label and shrugs off the risks, that signals the need for stronger safety discussions and peer accountability. Knowing the hazards, respecting them, and acting with care makes all the difference.
What is the CAS number and molecular formula of Pivalaldehyde?
Understanding the Basics
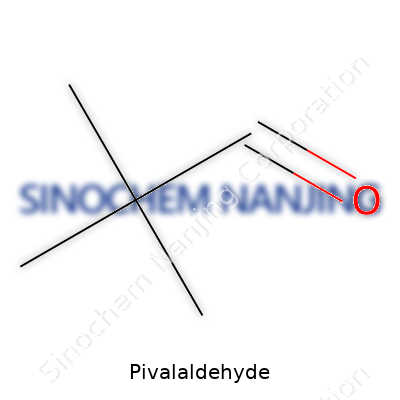
Pivalaldehyde highlights how organic chemistry connects laboratory science to real-world impact. Its CAS number is 630-19-3, a straightforward label any chemist can use to order or research this compound. The molecular formula reads C5H10O. On paper, this may look simple, but it signals a molecule with five carbon atoms, ten hydrogen atoms, and one oxygen atom. Anyone familiar with aldehydes will recognize the formyl group attached to a bulky tert-butyl structure. This structure affects how it reacts and handles in a chemical setting.
Why Its Identification Matters
Chemists rarely choose a compound without checking its CAS number. This numeric code helps avoid mix-ups, with each unique chemical having its own identifier. If a researcher needs pivalaldehyde for a synthesis, mistaking it for another aldehyde would ruin the process, cost time, and possibly create safety hazards. I’ve seen confusion from misidentified chemicals tank weeks of lab work, all because a number was off by one digit. CAS numbers don’t just belong to databases; they keep the wheels turning in both research and industry.
Real-World Connections and Research
Pivalaldehyde isn’t the flashiest molecule, but it fills important roles. In organic synthesis, its tertiary structure keeps reactions selective. It often shows up as a building block for pharmaceuticals or specialized polymers. Not every chemist gets excited about aldehyde synthesis, but a tough route to a drug precursor gets a whole lot easier with a reliable starting material.
Questions about purity or supply also come up a lot. When buying chemicals, a wrong lot number means the difference between a smooth reaction and a failed experiment. The confidence comes from recognition — you’re getting C5H10O, not something close enough to pass for it in a quick glance. Speaking from direct work in an academic laboratory, I learned the importance of cross-referencing CAS numbers — it forms the backbone of chemical inventory management. Errors slow everyone down and can jeopardize results.
Safety and Handling
Pivalaldehyde carries the usual aldehyde risks. The compound is volatile, with an odor that both trained and untrained noses notice right away. Aldehydes, including this one, can irritate skin and mucous membranes. In my own lab, proper ventilation and protective gloves were non-negotiable. Even small spills demand careful cleanup, using plenty of water and avoiding contact. Information from safety data sheets and chemical supplier guidelines provides the best protection against unexpected exposure. Safety protocols prevent health issues and keep research on track.
Improving Accountability and Traceability
Better digital record-keeping could help the industry track every drop of pivalaldehyde. Linking the CAS number directly to safety data and batch information makes traceability clearer. If something goes wrong, investigators can quickly trace it to the source — whether it’s an impurity or a mislabeled container. This approach creates a feedback loop, tightening controls on quality and guides better regulatory compliance. Research teams, manufacturers, and regulators all benefit from clear, consistent data at every stage.
Looking at the Bigger Picture
Something as simple as a CAS number and a formula prevents costly mistakes and boosts safety. Training newcomers in lab work should always include lessons about chemical identification, going past the basics to show what a few digits and a handful of symbols really mean. No matter the career stage, everyone can use reminders about the value of precision and due diligence in handling chemicals like pivalaldehyde.
| Names | |
| Preferred IUPAC name | 2,2-Dimethylpropanal |
| Other names |
2,2-Dimethylpropanal Pivalic aldehyde Neopentanal |
| Pronunciation | /paɪˈvæl.æl.dɪ.haɪd/ |
| Identifiers | |
| CAS Number | 630-19-3 |
| 3D model (JSmol) | `Pivalaldehyde|CID_12219` |
| Beilstein Reference | 1718737 |
| ChEBI | CHEBI:36010 |
| ChEMBL | CHEMBL16268 |
| ChemSpider | 68213 |
| DrugBank | DB04116 |
| ECHA InfoCard | 100.003.950 |
| EC Number | 204-594-3 |
| Gmelin Reference | 83217 |
| KEGG | C06106 |
| MeSH | D010993 |
| PubChem CID | 12318 |
| RTECS number | **UV3150000** |
| UNII | 2O8F1NFS8K |
| UN number | UN2377 |
| CompTox Dashboard (EPA) | DTXSID2039246 |
| Properties | |
| Chemical formula | C5H10O |
| Molar mass | 100.159 g/mol |
| Appearance | Colorless liquid |
| Odor | pungent |
| Density | 0.780 g/mL |
| Solubility in water | slightly soluble |
| log P | 1.51 |
| Vapor pressure | 2.8 mmHg (20°C) |
| Acidity (pKa) | 17.0 |
| Basicity (pKb) | 13.45 |
| Magnetic susceptibility (χ) | -41.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.349 |
| Viscosity | 1.61 mPa·s (20 °C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 321.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -232.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2335 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 6 °C (43 °F; 279 K) |
| Autoignition temperature | 215 °C (419 °F; 488 K) |
| Explosive limits | Lower: 1.2%, Upper: 7.2% |
| Lethal dose or concentration | LD50 (oral, rat): 650 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 830 mg/kg |
| NIOSH | PIV000 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Pivalic acid Pivalamide Pinacolone Neopentyl alcohol Neopentyl chloride |

