Phthalic Anhydride: Industry Workhorse, Environmental Question Mark
From Coal Tar to Chemicals: A Century of Phthalic Anhydride
Phthalic anhydride hit the big time long before most of today’s plastics even existed. Chemists found it in the late 1800s, pulling it from coal tar, which sounds primitive now but was cutting-edge at the time. Anyone watching the chemical industry change over decades could see how quickly phthalic anhydride moved from odd laboratory curiosity to key ingredient in building everyday products, from the paints on school walls to the resins in boats and cars. Synthetic routes improved along the way, swapping coal tar for processes that use either naphthalene or o-xylene as starting points. Those changes weren’t just about getting more product out—manufacturers chased better cost control and easier handling. Large-scale production took off, especially once phthalic anhydride set up shop as a basic material in the gigantic alkyd resin and plasticizer industries.
Tangling with the Molecule: What Sets Phthalic Anhydride Apart
Any chemist will notice the sharp look and pungent odor of phthalic anhydride’s flaky white crystals. This isn’t just trivia; working with it makes the senses perk up, because the dust makes throats itch and eyes water. The compound stands out for its readiness to snap back into the acid form with a little water or humidity—eat away at a bag with some damp air, and you find an acrid mess and sore mucous membranes. It melts at just above 130°C, so it can swing between solid and liquid in most processing plants. You get volatility that makes safety goggles a necessity on the floor, and a purity requirement that puts pressure on suppliers. As a chemical, phthalic anhydride’s structure opens it up to all sorts of reactions, thanks to those carbonyl groups crammed onto a small aromatic ring. This accounts for its use nearly everywhere dyes, pigments, and synthetic fibers get made.
Diving into Preparation and Modifications
If you have ever worked in chemical engineering, you know large-scale production relies on more than clever molecular rearrangements—it’s about what’s cheap, reliable, and safe under pressure. Naphthalene oxidation, using a vanadium pentoxide catalyst, ended up kicking off most of the world’s phthalic anhydride for decades. The o-xylene oxidation route offered a valuable alternative, especially in regions with cheap petroleum feedstock. Both methods push a hydrocarbon through hot air across a catalyst bed, snatching off hydrogen, throwing on oxygen, and spinning out a vapor stream that cools down to throw white phthalic anhydride flakes everywhere. Tweaks to these processes mean producers can chase higher yields and lower environmental emissions, but the basic principle hasn’t shifted much since mid-twentieth century.
Synonyms, Standards, and Labeling Realities
Walking the aisles of industrial suppliers, you notice phthalic anhydride hiding behind several aliases: 1,3-isobenzofurandione, o-phthalic anhydride, or simple PA. Official paperwork often switches between these names, sometimes muddying what’s in a drum or a shipment form. Chemical catalogs require sharp eyes, and even the labels reflect a legacy of older naming conventions meshed with modern hazard warnings: bold hazard pictograms to remind you about its irritating properties, alongside older industrial codes now found at the margins. Actual product comes in bags or drums labeled with hazard codes and storage advice, since even a routine spill chews through labels, boxes, and air quality in a ventless room.
Working Safely—Hard-Learned Lessons
People in the chemical industry remember minor incidents because they escalate fast with phthalic anhydride. It flows well as a molten liquid but throws off vapors that attack the nose and throat. It stings skin, especially in the presence of sweat or cuts. Safer operating standards grow out of these incidents: full respiratory protection for transfers, proper gloves, strict temperature controls, and storage far from heat or moisture. Regulatory standards from agencies like OSHA and the European Chemicals Agency wake up plant management to the health side of things, pushing limits for concentration in air and attention to ventilation. These codes reflect both the chemical’s ready reactivity and its long track record of health complaints from workers. It’s not about theoretical risks—older workers carry memories of persistent coughs and heavy chest tightness after too much exposure on lines that left fabric filters unchecked.
Application Zones: Where Phthalic Anhydride Shows Up
Everyday products weave phthalic anhydride in ways most people don’t realize. If you like tough, glossy finishes on hardware or woodwork, odds are high these properties come from alkyd resins built from this single compound. Walk through a home improvement store, and it’s easy to spot paints and coatings whose colors and textures depend on phthalic-derived intermediates. Car parts—especially lightweight composites—owe much to unsaturated polyester resins, again rooting back to phthalic anhydride. The flexible qualities of vinyl floors or automotive panels often connect to phthalate plasticizers, which track their chemical lineage to production runs that kick out phthalic anhydride as a base. Even medicine bottles, wire insulation, and shoe soles sometimes draw on chemical tricks first figured out using this material. Environmental critics warn about the spread of plasticizer residues in dust and bodies, but the broad reach of the chemical means it will stay in demand until sweeping changes appear across the global plastics economy.
Peering into Research—Both Promise and Limitations
Research on phthalic anhydride never stands still. Organic chemists love exploring novel reactions to build smarter dyes, polymers, or active ingredients in crop protection. There’s ground to cover with green chemistry initiatives, which press for lower-emission synthesis routes, safer catalyst technologies, and recycling pathways for byproducts no longer treated as disposable waste. Academic groups push modifications on the aromatic ring, aiming for cleaner or more selective synthetic transformations. Despite all the progress, industry hesitates before massive changes—redesigning factories and supply chains takes capital and political will.
Toxicity: Old Fears, New Questions
Plenty of long-haul workers in plasticizer or resin plants will tell you that even brief unprotected exposure to phthalic anhydride means a nasty few hours of coughing and sore eyes. Modern toxicology studies back up these lived experiences: this material reliably irritates mucous membranes, eyes, and skin. Animal studies hint at longer-term effects, including possible respiratory sensitization with repeated overexposure. Major concern over phthalate plasticizers, which spring from phthalic anhydride, lands in environmental debates—persistent organic waste, questionable breakdown products, and bioaccumulation worries swirl around the plastics industry. Even with limits in place, the science keeps nudging regulators toward tighter oversight, especially as lab techniques leap forward and detect ever-smaller residues in air, water, and food. The tug of war continues between health research and profit-driven production goals, and too often, people on the line in plants pay the price for shortcuts.
What Comes Next?
Future prospects for phthalic anhydride matter to more than just chemical companies. Anyone invested in safer consumer products, cleaner air, or sustainable manufacturing ought to keep an eye on this industry. The shift to biobased feedstocks might bring alternatives, yet economic pressures still favor this well-understood, high-volume compound. Some researchers pitch closed-loop recycling, new catalysts, or even molecular tweaks to cut out the most controversial plasticizer products entirely. It’s possible to lower emissions and make the handling less hazardous, but achieving major breakthroughs takes slow, coordinated effort across supply chains, production lines, and governments. Communities around chemical plants should press for more transparency and better pollution control. Industry leaders who ignore those calls risk ending up on the wrong side of public trust, no matter how efficiently they churn out raw material.
What is phthalic anhydride used for?
Bridging Chemistry and Everyday Life
Ask anyone about phthalic anhydride and you’ll probably get a puzzled look. On paper, it sounds like a word straight out of a chemistry textbook. Yet it has a hand in making a surprising range of products feel, look, and last the way they do. The world of manufactured goods, especially plastics, paints, and coatings, leans heavily on this compound. My curiosity about “invisible” chemicals began after noticing that the smell of new plastic toys always seemed familiar—thanks to some reading, I learned phthalic anhydride often plays a role there.
From Raw Resin to Flexible Plastics
Phthalic anhydride earns its stripes by acting as a key building block for making plasticizers, the agents that keep plastics soft and bendable. These plasticizers—commonly phthalates like DEHP—turn stiff plastic into flexible consumer goods. Vinyl flooring, shower curtains, and even inflatable pool toys rely on this characteristic. Plastics on supermarket shelves that can handle both rough tossing and repeated bending would fall apart much sooner without the magic touch of plasticizers made from phthalic anhydride.
The story doesn’t stop with plastics. Phthalic anhydride also finds a home in the world of alkyd resins, used for paints and coatings. These resins help protect wood, metal, and even walls from moisture and wear. Doors and window frames covered in glossy enamel owe their shine and resilience to these chemical processes. Many of us take for granted the endurance of high-gloss surfaces in kitchens and bathrooms, forgetting the chemistry that holds them together.
Everyday Paints, Perfumes, and Beyond
Many households keep a tin or two of varnish for touch-ups, but fewer people talk about what makes the varnish spread smoothly and dry without yellowing too soon. That’s another quiet contribution by phthalic anhydride’s derivatives. It helps paints glide on more evenly and dry to a tough finish, which in turn lowers the frequency of repainting drives and touch-ups. That saves both time and money in the long run.
Industries use phthalic anhydride to make dyes, pigments, and certain perfumes as well. The vibrant reds and blues in synthetic textiles rely on pigments crafted from this compound. Some synthetic fragrances in soaps and detergents also trace their roots back to phthalic anhydride. Even though we barely notice it, life without these colorful or fragrant products might feel a lot duller.
Balancing Progress and Safety
All this convenience doesn’t come without challenges. Research has shown concerns over the health effects of phthalates—especially for children. Regulatory bodies, including the US Environmental Protection Agency and the European Chemicals Agency, have tightened the reins on how these substances get used. As someone who has handled everything from dollar-store toys to high-end paint, it feels reassuring knowing there’s oversight, but the responsibility doesn’t end there.
Safe handling and continued research matter. Switching to alternatives like bio-based plasticizers looks promising. Some industries have invested in non-phthalate options where possible. The average person can also pay attention to labeling, stick with trusted brands, and stay informed about material choices in products brought into the home.
Phthalic anhydride stands out in the world of chemistry. Its uses touch more lives than most know. Understanding how each product’s journey begins in a lab can give us all a better grip on the choices we make and the products we trust.
Is phthalic anhydride hazardous to health?
Breaking Down the Real Concerns
Phthalic anhydride shows up in plastics, paints, and even some automotive parts. Factories work with it every day. People sometimes ask if this chemical poses risks. Based on my own days working near manufacturing lines, I’ve seen how easy it is for vapor or fine particles to get into the air. Most folks outside that world don’t realize how chemicals drift and how even a small exposure piles up over time.
One thing that stands out: phthalic anhydride acts as an irritant. Inhaling dust or fumes can hit your throat and nose. Sometimes mucus gets thick, your nose burns, or eyes water like you’re cutting onions. A study by the National Institute for Occupational Safety and Health found that workers often complain about these symptoms. That isn’t much of a surprise. When I worked back-of-house in an old warehouse, just a little chemical dust in the air brought out coughs—ten-fold for those without a good mask.
With longer exposure, sensitization gets real. Once your immune system reacts, allergic asthma steps into play. Some workers have left jobs because of breathing troubles. The American Conference of Governmental Industrial Hygienists classifies phthalic anhydride as a respiratory sensitizer. Even at levels considered “safe,” there have been reports of chest tightness and breathlessness. After years on the job, a few folks I knew never fully stopped wheezing.
There’s another layer. Research points to mild risk of skin problems too. Coming in direct contact, folks have dealt with rashes and sores. In my experience, gloves cracked and even a small leak could bring out a red, itchy patch by the day’s end. Sometimes contact happened just from leaning against a surface. Most routines fix this by swapping gloves often and keeping washing stations close. Cutting corners has always backfired for those who got lazy about hand protection.
Fact-Checking Cancer and Other Risks
Cancer risk tends to worry people. So far, researchers haven’t placed phthalic anhydride in the high-alert zone. According to the International Agency for Research on Cancer, there’s no solid proof linking it directly to human cancer. Still, anyone careless with protective equipment tends to put themselves in the line of fire for other chemicals, many of which do have stronger links to serious illness.
Some look at birth defects or hormonal shifts as possibilities. Science doesn’t show strong evidence that phthalic anhydride causes these issues, but caution rules the day. The bigger problem comes from being around other related chemicals at the same time, where effects may add up.
Solutions From Real Life
I’ve learned that the most important fixes aren’t fancy. Factories use proper ventilation. Simple extractor fans do most of the heavy lifting. Respirators and gloves hold equal importance—no worker should walk in unprotected. Regular breaks for washing up matter more than most folks realize. Most hang safety signs for show, but real change comes from supervisors enforcing mask rules and checking in when anyone shows symptoms.
For people worried about surrounding neighborhoods, local air monitoring offers answers. Community groups have pressed companies to share test results. It keeps them honest. Workers deserve the same transparency. Company health checks catch new cases of allergies or asthma before they get worse, and this proactive approach saves both livelihoods and lives.
Learning from decades in and out of manufacturing, common sense rules above everything. Workers and nearby families stay safer with clear routines, simple equipment, and managers willing to listen. Facts only help if folks put them into practice each day.
What are the storage requirements for phthalic anhydride?
The Real Reason Storage Standards Matter
Phthalic anhydride finds a regular spot in the chemical industry, mainly because it forms the backbone of things like plasticizers, resins, pigments, and dyes. Anyone who’s spent time in a facility with these materials knows storage isn’t a detail you get to skim over. Chemistry in real life doesn’t match the tidy diagrams from textbooks. We’re not just talking about keeping stock orderly—the storage method matters because phthalic anhydride reacts sharply with water and opens the door to hazards if mistakes slip in.
Keeping It Dry—Moisture’s a Problem
I remember my first encounter with phthalic anhydride. I expected the usual bagged powder. Instead, I watched lumps form inside storage bins, all thanks to humid air sneaking past a bad seal. This chemical reacts quickly with moisture to form phthalic acid, which clogs lines and triggers maintenance nightmares. So it makes sense that every container needs to be air-tight and watertight; compromised bags or loose lids cost more in labor and lost product than many realize. Facilities that invest in sealed drums or glass-lined bins sidestep headaches. Even warehouse flooring should be dry—once that powder cakes together, it’s rarely usable again.
Temperature: Not Just About “Room Temp”
Some folks glaze over the label saying, “store at room temperature.” But heat sources like boilers or afternoon sun on a loading bay—these turn storage zones risky. If this solid hits about 131°C (268°F), it melts and gives off fumes that sting the eyes and nose. OSHA calls out phthalic anhydride for its potential to irritate or trigger asthma symptoms. Facilities aiming to reduce sick days or keep staff on the floor take this threat seriously; they keep storage at steady, moderate temperatures, away from radiators or machinery exhaust. Simple shading and insulation protect not just the material but the people around it.
No Shortcuts with Safety—Personal Experience Counts
Having worked in labs and warehouses, the worst accidents show up when teams take shortcuts. Spilled phthalic anhydride doesn’t stay in one place—just opening a container the wrong way can send dust everywhere, kicking off allergic reactions and persistent coughs. That’s why chemical-safe gloves, goggles, and masks earn their reputation. A single slip-up, like failing to isolate storage from other reactive chemicals—especially acids, alkalis, or open water sources—creates a recipe for emergencies.
Following Regulations and Building Good Habits
From the start, regulatory groups like OSHA and the EPA have outlined storage procedures for a reason. Rules call for labeling every bin and separating incompatibles by secure barriers. Facilities use metal or plastic containers lined to resist corrosion, with clearly marked warning signs in place. Regular inspections and quick cleanups after spills go a long way. It helps to run real drills on response; even experienced teams benefit from hands-on training.
Smart Solutions for Modern Storage
As industry leans into automation, options like humidity-controlled storage, electronic leak sensors, and inventory systems cut down on risk. Investing in sealed vessels and quality packaging, rather than improvising with basic bins, adds years to both product shelf life and employee health. It isn’t just about checking boxes; it’s about respect for everyone who handles these materials each day.
Good storage of phthalic anhydride isn’t just policy—it’s respect in action for co-workers, the environment, and your own bottom line.
How is phthalic anhydride produced?
The Main Route: Oxidation of O-xylene and Naphthalene
Phthalic anhydride shows up in everyday objects like plastic toys, paints, and the insulation in wires. What gets it there is a specific industrial process that hasn’t changed much in decades. Most factories produce it by oxidizing either o-xylene or, much less often nowadays, naphthalene. In both cases, air and a metal catalyst step in to drive a reaction that transforms the starting chemical into phthalic anhydride vapor.
The o-xylene process wins out in regions with ready petroleum supplies because o-xylene comes from oil refining. Inside the reactor, temperatures reach between 350 and 400°C, and a vanadium pentoxide catalyst helps crack apart the o-xylene. Air forces the oxidation, yielding a mix of gases that cool and condense into the solid white flakes recognized as phthalic anhydride. Factories usually recover leftover heat from this reaction to save energy, which helps keep costs a bit more manageable.
Naphthalene-based production runs on a similar principle but nowadays is more common in countries with a strong coal industry since naphthalene comes from coal tar. It’s less favored for efficiency and supply reasons. Modern producers often build their plants close to oil refineries or facilities making things like PVC and plasticizers, cutting down on shipping and logistical headaches.
Impact on People and the Environment
Factories that make phthalic anhydride use large volumes of energy and give off gases like carbon monoxide and carbon dioxide. Phthalic anhydride itself is irritating to eyes and lungs; long-term exposure to dust in the air raises real health concerns for plant workers. Factories try to clamp down on emissions by installing scrubbers to catch phthalic anhydride particles before they escape the plant. Several countries set workplace air standards that require air monitoring and regular health checks for workers.
Waste heat recovery, as well as water treatment and waste gas filtration, reduce damage to the environment. I remember touring a chemical plant near the Gulf Coast. The big sign reading “Zero Discharge Goal” sounded ambitious but represented a real shift in mindset over two decades. Managers spent years retrofitting their plant with energy recapture systems. Local waterways, once streaked green and red from chemical runoff, now look clear again. That transparency matters to surrounding towns relying on fishing and clean water.
Paths Toward Safer, Cleaner Production
Efficiency and safety sit front-and-center for anyone running a chemical plant today. More companies keep pushing for better catalysts—less waste, stronger yields, longer lifespans. Some researchers look into bio-based starting materials, hoping to one day replace o-xylene with compounds from agricultural waste, though scaling up remains tricky and expensive. Even a small bump in catalyst efficiency can cut down on both waste and greenhouse gases, given the millions of tons produced annually.
Phthalic anhydride sticks with the world because products using it—for example, flexible PVC plastics and polyester resins—stay in high demand. Yet there's growing public pressure to put environmental and worker health front-of-mind. My own view: Anything that helps plants run cleaner without risking jobs or safety is a step in the right direction. Strict monitoring, ongoing research into better catalysts, and investment in new technologies will define how this basic chemical fits into a future aiming for sustainability.
What safety precautions should be taken when handling phthalic anhydride?
Why Responsibility Matters in Industrial Settings
I’ve spent years around industrial production sites, and I’ve seen safety become more than just a rulebook—it’s a way people watch out for each other. Phthalic anhydride doesn’t make headlines unless there’s trouble: spills, chemical burns, or sudden coughing fits that clear a whole work area. The powdery, crystalline material looks harmless, but after seeing colleagues react badly when a cloud escapes during a transfer, I stopped taking it lightly. Hidden danger lurks in that dust.
Personal Stories Shape Approaches
A lot of common sense comes from what people have witnessed. One veteran operator shared how years of not taking proper care with respiratory gear led to chronic respiratory problems. He used to dismiss the warnings, thinking a quick job without a mask couldn’t hurt. After a few trips to the occupational doctor, he changed his tune. Hearing those types of stories, the risks of this compound become very real.
Risks and Health Concerns
Phthalic anhydride irritates the skin, eyes, and airways. Direct exposure means red, watery eyes and skin rashes. Breathing in the dust often brings on coughing fits and, for some, can trigger asthma. The American Conference of Governmental Industrial Hygienists set a pretty low threshold for airborne phthalic anhydride—just 1 part per million over an eight-hour shift. Continued overexposure can lead to chronic bronchitis. NIOSH and OSHA also flag it as a hazardous chemical. Seasoned safety personnel know that relying on luck is no way to handle it.
Real-World Safety Steps
PPE is the starting point. Anyone handling this stuff puts on chemical goggles or full-face shields, gloves rated for corrosive chemicals (like nitrile or butyl rubber), and proper work clothing. Serious facilities insist on respirators—especially during transfers or cleanups. Relying on just a dust mask falls short. Cartridge-style respirators rated for organic vapors and acidic gases keep exposure down, and using them isn’t negotiable.
Engineering controls make just as big a difference. In one production plant, we built a closed transfer system to move phthalic anhydride from the bag to the reactor. That cut down fugitive dust and made a visible difference in air quality. Local exhaust hoods right at the mixing station catch stray particles. Ventilation has to be up to snuff—no shortcuts waiting for the “big upgrade.” Even after improvements, monitors and alarms keep tabs on air quality, sounding alerts if things get dicey. People trust clean air only with proof.
Employee Training Sets the Tone
Handouts and posters don’t stick unless supervisors demonstrate the routines themselves—showing how to check a respirator seal or secure a barrel. The best-run shifts I’ve seen always start with a few words about any lingering phthalic anhydride leftovers or odd readings on the air monitor. No one assumes that a new hire knows how to wash off a chemical burn—everyone gets thorough training and refresher courses. This helps workers know not just the “what,” but the “why.”
Waste Handling and Decontamination
Waste material waits in labeled, sealed drums—not left in open tubs where dust can drift. Team members clean up spills immediately with wet wiping or HEPA vacuums instead of sweeping, since dry sweeping stirs up more danger. Showers and eye wash stations sit close by and get checked often. No one waits to report a small incident—from habit, workers speak up when something doesn’t look right.
Practical Solutions Pay Off
Handling phthalic anhydride doesn’t just demand care; it insists on teamwork and vigilance. People who take personal responsibility, speak up about problems, and actually use the safety tools available make the work environment safer for everybody. There’s no substitute for looking out for one another and trusting real-world experience.
| Names | |
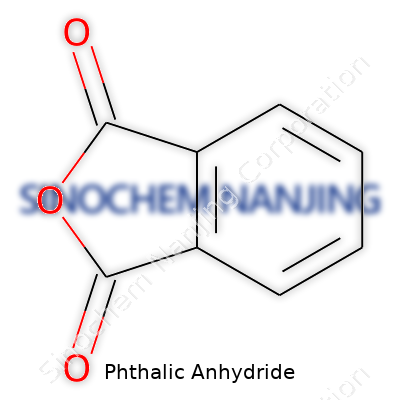
| Preferred IUPAC name | 3,4-Benzenedicarboxylic anhydride |
| Other names |
Phthalic acid anhydride 1,3-Isobenzofurandione Phthalic anhydride flakes PA NSC 407593 |
| Pronunciation | /ˈθælɪk ænˈhaɪdraɪd/ |
| Identifiers | |
| CAS Number | 85-44-9 |
| 3D model (JSmol) | `3D model (JSmol) string` for **Phthalic Anhydride**: ``` C1=CC2=C(C=C1)C(=O)OC2=O ``` |
| Beilstein Reference | 'Beilstein Reference: 1909611' |
| ChEBI | CHEBI:17618 |
| ChEMBL | CHEMBL1401 |
| ChemSpider | 1579 |
| DrugBank | DB03126 |
| ECHA InfoCard | 03d82c07-6e5b-4577-aa8a-ed6faf3b41d1 |
| EC Number | 204-873-0 |
| Gmelin Reference | 786 |
| KEGG | C01483 |
| MeSH | D010681 |
| PubChem CID | 1004 |
| RTECS number | TH9625000 |
| UNII | 97C5T2UQ7J |
| UN number | UN2214 |
| Properties | |
| Chemical formula | C8H4O3 |
| Molar mass | 148.12 g/mol |
| Appearance | White crystalline solid |
| Odor | Slightly aromatic |
| Density | 1.53 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.60 |
| Vapor pressure | 0.02 mmHg (20°C) |
| Acidity (pKa) | 1.3 |
| Basicity (pKb) | 1.60 |
| Magnetic susceptibility (χ) | -46.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.607 |
| Viscosity | 1.7 cp |
| Dipole moment | 2.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 325.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -531.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –3163 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if inhaled or swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H317, H319, H334, H335 |
| Precautionary statements | P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P363, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-2-0-W |
| Flash point | 129°C |
| Autoignition temperature | 432°C |
| Explosive limits | 1.7–10.4% |
| Lethal dose or concentration | LD50 oral rat 1530 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 1530 mg/kg |
| NIOSH | TC4025000 |
| PEL (Permissible) | 5 ppm (mg/m³) |
| REL (Recommended) | 6 mg/m3 |
| IDLH (Immediate danger) | 60 ppm |
| Related compounds | |
| Related compounds |
Phthalic acid Terephthalic acid Isophthalic acid Phthalimide Naphthalene Maleic anhydride |

