Phosphorous Acid: The Overlooked Power Player in Chemistry and Industry
Historical Development
Phosphorous acid often gets overshadowed by its sibling, phosphoric acid, yet its path through chemical history holds real weight. In the early 19th century, the search for ways to make phosphorus-based compounds safer and more reliable pulled phosphorous acid into the spotlight. Chemists learned—sometimes the hard way—that heating moist phosphorus with air could be risky business, but with the right methods, they could bottle a compound with unique properties. I remember reading about Wilhelm Scheele’s work, where he wrangled red phosphorus to produce different acids, setting the foundation for what we use in labs and industry today. Now, phosphorous acid stands as a quiet but steady workhorse in chemical transformations.
Product Overview
Ask folks on the street about acids, and phosphorous acid rarely makes the list. Yet in technical circles, people recognize its value in synthesis, metallurgy, and agriculture. In my experience, researchers and industry specialists appreciate it for its reducing ability and its role in making glyphosate—an herbicide that changed global farming practices. Phosphorous acid doesn’t get flashy displays on hardware store shelves, but its impact trickles through fields, factories, and science classrooms alike.
Physical and Chemical Properties
This acid shows up as a white, crystalline solid that melts without much fuss around 70 °C. If you crack open a bottle and catch a whiff, the odor feels sharp but not overwhelming. Its formula, H₃PO₃, looks simple but packs a punch in terms of chemical dynamics. Because phosphorous acid quickly absorbs water from humid air, chemists don’t leave it out for long. When mixed with water, it forms a solution that can act as both a weak acid and a powerful reducing agent—attributes that make it stand out from the more familiar phosphoric acid. Handling it reveals its corrosive effect: I’ve seen metal stirrers tarnished if left too long in contact.
Technical Specifications and Labeling
Any container of phosphorous acid often carries clear warnings—corrosive, keep dry, keep sealed—echoing those early mistakes with misplaced curiosity and shoddy storage. Labels should spell out concentration, recommended storage conditions, and hazard symbols. Beyond the warnings, specifications focus on purity, usually above 98% for most chemical syntheses. Industrially, manufacturers guarantee tight tolerances on moisture and contaminant metals, because a bit of stray iron or copper can set off unwanted side reactions down the line. It’s one of those compounds where even a tiny bit of impurity can make a big difference, especially when making complex organic compounds or high-purity materials.
Preparation Method
Producing phosphorous acid relies mainly on hydrolyzing phosphorus trichloride with water—an exothermic and fume-filled process. The first step usually combines phosphorus and chlorine to make the trichloride, which then reacts with water or steam. I once watched a demonstration where a few drops of water turned a flask of phosphorus trichloride into a steaming, hissing cauldron, as clouds of hydrogen chloride gas spilled out. That’s the raw chemistry that underlies much more polished industrial runs, which focus on capturing and scrubbing away those byproducts.
Chemical Reactions and Modifications
The true value of phosphorous acid emerges when you hand it over to a skilled chemist or chemical engineer. As a reducing agent, it converts metal ions in solution to lower oxidation states—useful for stripping films, regenerating catalysts, or reducing nitrites. Its reactions with alkalis produce phosphites, which show up in specialty products for agriculture or electronics. In organic synthesis, it protects sensitive molecules from oxidation, shaping complex molecules for pharmaceuticals and agrochemicals. The reduction tricks go beyond theory books; I’ve seen lab projects sink or swim based on how this acid handled specific impurities or functional groups.
Synonyms and Product Names
On inventory sheets or invoices, you might spot phosphorous acid listed under names such as orthophosphorous acid, H₃PO₃, or phosphonic acid. Mislabeling sometimes causes confusion, especially between phosphoric and phosphorous acids. Only a careful reading of structure and oxidation state sorts out the true identity, which matters a lot in industrial settings. Not knowing the difference can wreck an entire production batch or send unwanted gases wafting through a lab’s ventilation system.
Safety and Operational Standards
Working with phosphorous acid comes with clear, non-negotiable ground rules—eye protection, gloves, and good ventilation are a given. If splashed on skin, it burns, and fumes irritate the respiratory tract. Forgetting safe practices leads to injuries that linger. Regulatory agencies, including OSHA and the European Chemicals Agency, place strict demand on labeling, exposure limits, and disposal. I’ve seen company safety officers drill new hires on keeping containers closed and preventing accidental mixing with bases or strong oxidizers. Cleaning up spills requires neutralizing with sodium bicarbonate and using non-reactive absorbents. Long experience shows that small lapses—the temptation to work quickly or skip a ventilation step—can bring big trouble.
Application Area
Agriculture takes a big share of phosphorous acid production. Plants benefit from fertilizers based on phosphites, which are less likely to lock into soil minerals—making nutrients more available over time. Fungicide sprays gain an extra layer of disease resistance when farmers include phosphonate salts. The electronics sector taps into its reducing power during etching or cleaning steps, and metal finishers rely on it to prepare surfaces and remove corrosion scales. In other cases, water treatment facilities add controlled amounts to adjust water chemistry, helping to manage lead or copper leaching in municipal pipes. I’ve known greenhouses that swear by foliar sprays made with phosphite derivatives, citing better plant health and reduced losses.
Research and Development
Breakthroughs in phosphorous acid chemistry typically come from small tweaks—a catalyst here, an energy-saving method there. Universities and private labs look for ways to make its production greener, since chlorine handling still leaves a heavy environmental footprint. Some studies chase new organic reactions that could speed up pharmaceutical manufacturing and cut costs. I watched graduate students spend months searching for catalysts that let phosphorous acid perform reductions at lower temperatures or with fewer nasty byproducts. Even in well-established sectors, a clever improvement can unlock big value or meet stricter regulatory rules.
Toxicity Research
Scientists have dug into the acute and chronic toxicity of phosphorous acid, with most studies showing that it’s less hazardous than some related compounds but clearly not benign. Acute exposure eats away at tissue and causes strong irritation. Over time, repeated low doses can damage the liver and kidneys—making secure handling and disposal a top priority. Regulatory bodies use animal studies to set workplace exposure limits and guide emergency response procedures. Still, rare cases of environmental contamination make it clear that prevention matters more than reaction. Streamlining safety training and upgrading protective equipment remain the best tools.
Future Prospects
The future for phosphorous acid looks closely tied to changes in environmental regulations and the growing appetite for precision agriculture. Farmers want safer, more effective ways to boost yields with fewer environmental risks—a need that brings attention to phosphite-based products. In tech sectors, demand for ever-purer chemicals for semiconductors or specialty ceramics keeps pressure on suppliers to refine production and cut contaminants. Researchers look for alternative synthesis pathways that eliminate hazardous intermediates and lower carbon footprints. As renewable energy and electrification gather steam, new uses for reducing agents like phosphorous acid could emerge in battery manufacturing or recycling. Each new regulation or technological advance pushes the industry to adapt, and my experience tells me that companies finding safer, cleaner ways to make and use phosphorous acid will gain a real edge.
What is Phosphorous Acid used for?
Phosphorous Acid in Farming
Walk into any agricultural supply store, and you will find products based on phosphorous acid. Farmers and gardeners often look for ways to protect their crops from disease, boost yields, and deal with tough environmental conditions. This chemical delivers in a big way—especially for fruit growers. Grapes, citrus, and avocados suffer from fungal diseases like downy mildew and root rot. Sprays or drenches with phosphorous acid help keep these destructive pathogens under control. My father’s vineyard managed a nasty outbreak of root rot one summer, and after switching to phosphorous acid treatments, we watched sickly vines green up and blossom again. The immediate impact on crop health put the science into real-life perspective.
Boosting Plant Nutrition
Farmers use this acid because it supplies phosphorus—a vital nutrient for plant growth—more directly than some older fertilizers. Phosphorous acid does not stick around in the same form as standard phosphate fertilizers. Plants transform it and use the resulting phosphorus to build sturdy roots, strengthen cell walls, and get that deep, healthy green in their leaves. High-value crops like fruit trees and vegetables, which often reward even small nutritional tweaks, show stronger yields with phosphorous acid in the mix. My neighbor added it to his tomato field during a particularly rainy season, and his plants stayed upright and disease-free, even as more traditional fertilizer users in the area struggled.
Industrial and Chemical Roles
The influence of phosphorous acid extends beyond the farm. The chemical industry relies on it to make certain phosphonates—helpful for water treatment and cleaning applications. Phosphonates from phosphorous acid keep water pipes and boiler systems from clogging up with mineral scale. This might not sound exciting, but anyone who’s dealt with burst pipes or inefficient heating knows how quickly a plumbing disaster can drain your wallet. I helped a small food factory in my town address repeated scale-ups in their equipment by switching to cleaners using these specific chemicals, and they cut their downtime dramatically.
Facing Environmental and Health Questions
No solution is perfect. Some folks worry about runoff from fertilized fields and its impact on lakes and rivers. Too much phosphorus can feed algae blooms, which choke out fish and wreck water quality for nearby towns. The key is applying only what crops need, with proper timing before rains. I grew up next to a lake that suffered one bad bloom after another, and it took coordinated action between farmers and local officials to get things back under control—part of which meant updating how and when phosphorous acid-based fertilizers went onto fields.
Shopping for Safe Choices
On the home gardening front, people often pick up plant foods without reading the label. Phosphorous acid is perfectly safe in small, recommended doses, but concentrated solutions can irritate skin or damage plants if overused. I once splashed some on my arm and got an unpleasant rash that took a week to settle. Careful handling and storage solve most of these issues. Reading labels does not take long, and it keeps gardens healthy without surprising side effects.
Finding a Path Forward
Phosphorous acid serves plenty of useful purposes, from saving crops to keeping industry humming. Using this tool responsibly means staying aware of how it moves through our soils and waterways. Farmers, gardeners, and industry workers need up-to-date training and clear information. Investing in new soil tests and smart application systems pays off for everyone—better crops, safer water, and fewer regrets down the line.
Is Phosphorous Acid safe to handle?
Looking at Phosphorous Acid as a Chemical Worker
Stepping into a space where chemicals play a central role, you develop a different kind of respect for all the tools at your disposal. Phosphorous acid isn’t the most common compound on the shelf, but those handling it should treat it with the care it demands. As someone who’s spent years around warehouses and labs, I’ve seen how easy-going routines can turn risky if folks ignore hazards tied to chemicals like this one.
Health and Environmental Risks
This acid does more than just help in the making of products like water treatment solutions and plant nutrition formulas. If you breathe in its fumes or touch it without gloves, your body will protest fast—irritated eyes, coughing, maybe even some burns. The chemical won’t do permanent damage at a glance, but it knows how to bite skin and lungs. Inhaling the dust or mist for a long stretch can lead to some tough respiratory issues. I remember a colleague, rushed out of a mixing room, visibly shaken and hacking after a valve broke loose and sprayed a fine mist. He learned quickly that just “being careful” isn’t enough; real safety means respecting boundaries and rules, every time.
Importance of Education and Training
Training changes the game when it comes to chemical safety. Clear instructions save limbs and lives. Simple steps like wearing the right gloves, goggles, and a respirator go a long way. I’ve seen some teams rely on hastily printed labels or word-of-mouth warnings, which haven’t stopped careless slip-ups. Those mistakes can hurt not only the handler but also anyone else who walks by. Investing time in regular instruction—showing new hires how to clean up spills and why ventilation fans matter—becomes more than a checklist. It turns into a culture of accountability.
Lessons from Worksite Blunders
No system can entirely replace human attention. Spill kits look sturdy on a supply rack, but only if people use them. Once, in a greenhouse supply shed, an employee reached for a leaky jug with bare hands thinking it wasn’t serious. The burns on his palms reminded everyone on the team how easily routines slip when folks overlook minor leaks or don’t double-check storage. Every incident like this also pushes managers to keep better logs and tighten up physical controls.
Safe Handling is a Team Effort
My personal experience tells me that safety flourishes where workers share responsibility. Supervisors set the tone, but co-workers watch out for each other. Unsafe handling can lead to legal trouble, environmental messes, ruined crops—problems that ripple out from a single mistake. Storing chemicals in labeled, undamaged containers, tracking inventory tightly, and sticking to written procedures aren’t up for debate. Nobody enjoys paperwork, but careful records ensure answers when leaks happen and help trace contamination before it grows.
Building a Better Routine
Solutions don’t need to be fancy. Working with phosphorous acid requires common-sense rules: good gloves, proper goggles, regular cleanup checks, and real attention to storage. Immediate cleanup of spills with the right neutralizers and supplies stops minor issues from becoming major crises. Reviewing and updating safety protocols as materials and uses change keeps people ahead of potential trouble. Managers should encourage every team member to ask questions and report concerns. That encourages trust and helps catch mistakes before they hurt anyone.
Final Thoughts
Phosphorous acid belongs in the category of chemicals that demand respect. Safe handling calls for a strong routine, reliable teamwork, and ongoing learning. Every person who steps into a workspace stocked with hazardous materials needs to stand up for the whole team’s safety, using experience and education to keep health and career intact.
What is the chemical formula of Phosphorous Acid?
Phosphorous Acid: Not Just Another Laboratory Reagent
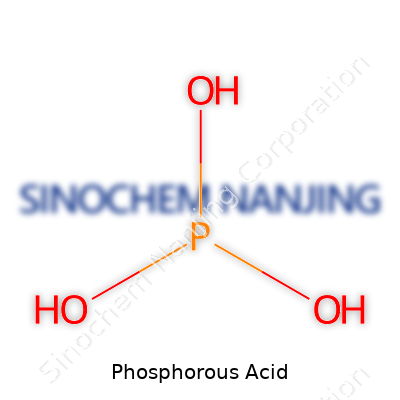
Phosphorous acid, with the chemical formula H3PO3, plays a bigger role than most people realize. This compound pops up not only in chemistry textbooks but also across various fields like agriculture and industry. It's easy to overlook the specifics of substances until you catch a reminder of how deeply linked they are to daily necessities and practices.
Breaking Down the Formula
Looking at the formula, you notice three hydrogen atoms, one phosphorus atom, and three oxygen atoms. This ratio creates properties quite different from phosphoric acid, which comes with the formula H3PO4. That single oxygen atom less in phosphorous acid changes its behavior and value. I've often found that understanding the difference in formulas gives insight into how certain nutrients interact with plants or why one compound delivers different results compared to its close relatives.
Application Rooted in Agriculture and Industry
Phosphorous acid stands out in the field of crop protection. Farmers rely on its salts—commonly called phosphites—as fungicides. These salts trigger defense mechanisms within plants, increasing resistance to problems like downy mildew or late blight. The application isn't magic; the science rests on the way phosphites travel through plant tissues and activate protective responses. With crop diseases threatening farmer incomes and food supplies, this seemingly simple chemical keeps significant harvest losses at bay.
In manufacturing, phosphorous acid serves as a reducing agent or acts in the preparation of phosphite salts. I've seen technical processes for water treatment or the synthesis of certain organophosphorus compounds that would falter without a reliable source of H3PO3. It's not about novelty—it's about reliability and proven action. The knowledge of its formula leads chemists to predict reactivity and choose the right material for each job.
Potential Environmental and Health Concerns
Every chemical offers benefits, but nobody gets a free pass. Overuse or mishandling of any phosphorus-based product brings the risk of environmental runoff, which may contribute to waterway pollution and algal blooms. Applying phosphorous acid beyond recommended levels won't boost returns and likely causes more harm. While some may want faster results in the field, patience and measured approaches protect soils and waterways. Educators and extension agents keep reminding folks about stewardship over quick wins.
Building Safer and Smarter Practices
Knowledge of the chemical formula means more than memorization. It allows users to calculate proper dosages and anticipate how the compound reacts in mixtures or under different conditions. Following label instructions and guidelines from reliable agencies reduces environmental risks while achieving the desired outcome in crops or products. Schooling in basic chemical literacy proves valuable—not just to future chemists, but to any grower or professional working with these compounds.
Real-World Responsibility
The science classroom provides the foundation, but practical experience brings that knowledge to life. Each time I saw someone disregard the importance of the correct formula, the result was waste or an unplanned mess to clean up. Having a clear understanding of H3PO3 keeps people grounded in fact-based decisions. Phosphorous acid isn’t a mystery; with sound information and a respect for science, its use delivers value safely and sustainably.
How should Phosphorous Acid be stored?
Understanding the Real Risks
Phosphorous acid rolls into warehouses and labs with a reputation for being a workhorse, not a troublemaker. Still, toss a careless storage routine into the mix and trouble finds a way in. Anyone who's spent time handling chemical supplies has seen labels fade, containers corrode, and a once trusted barrel become a potential hazard. Phosphorous acid won’t explode or catch fire out of the blue, but water, air, and metal spell disaster for this material. Combine moisture and phosphorous acid and the result is a slow, corrosive creep on storage shelves and even after a few months, you get sticky, dangerous residue where a safe drum used to sit.
Solid Choices for Storage Containers
Polyethylene or polypropylene containers stand up to acid’s strength, holding up for years without giving in to that silent corrosion. Many labs and warehouses go cheap with container choice and land in hot water after a leak, so cutting corners doesn’t make sense. Metal drums, especially steel, break down bit by bit with acid. It takes experience to spot a nearly-invisible pinhole or slow leak in a drum—and sometimes you figure it out only after you see a puddle. Stick with containers specifically marketed for storing acidic materials, and don’t mix them with other chemicals or recycled barrels that come from an unknown source.
Keeping Out Moisture and Air
Moisture is the classic silent thief here. Leave a cap loose or store phosphorous acid in a humid spot, and sooner or later you’re dealing with diluted acid and unstable residue. Tighten every lid and use air-tight containers. That goes double for any room with wash sinks, steamy vents, or high humidity. Even a little water makes phosphorous acid decompose faster, releasing tricky vapors that make a routine job dangerous.
Temperature Counts
Phosphorous acid likes steady temperatures around room level. Too much heat and you’re looking at slow breakdown, possible pressure build-up, and container warping. Direct sunlight cooks containers and, over time, weakens their structure. Very cold conditions can cause precipitation or make the acid hard to handle if you need to measure or transfer it. Warehouses that rely only on outside air see big swings in temperature, and storage gets risky. Staff should check storage closets, storage sheds, and indoor shelves regularly—especially through changing seasons.
Label It Right—Every Time
A clear label with basic details including date of receipt, batch number, and a warning about corrosion might sound overkill, but experience tells a different story. More than once I’ve come across mystery drums with only a faded barcode left and workers guessing what’s inside. Emergency teams can’t make safe calls with guesswork, and even routine daily use gets messy without a clear label.
Training and Housekeeping
Warehouse routines should cover daily checks for leaks or corrosion—no exceptions. Staff need to know the real consequences of a casual approach to acid storage. Eyes on the shelving, containers, and floor can catch problems long before they turn serious. Written procedures outlining safe transfer, handling, and clean-up turn words and warnings into habits.
Big Picture: Prevention Beats Clean-Up
Responsible storage isn’t about paranoia, it’s about consistent routines and paying attention, every time. Chemical burns, ruined floors, or emergency calls on a Friday night—these happen less often in places where everyone takes a moment to follow basic rules. The stakes are real, but the steps are simple—choose the right containers, keep lids sealed, watch the room conditions, and double-check those labels. Sometimes, it’s the little routines that keep disaster off the map.
What are the differences between Phosphorous Acid and Phosphoric Acid?
A Tale of Two Acids
I’ve spent my fair share of time around both phosphorous acid and phosphoric acid. Most people run into these acids in agriculture, cleaning products, or even food additives without giving much thought to what separates them. The two sound almost identical, but their chemistry and uses couldn’t be more different, and mixing them up can spell real trouble—especially on a farm or in a factory.
Chemical Structure Cuts to the Core
Let’s not get lost in jargon. Phosphoric acid, often written as H3PO4, delivers three hydrogen atoms willing to hop off and interact. This flexibility makes it so useful in fertilizers and soft drinks. Phosphorous acid (H3PO3), on the other hand, only lets two hydrogen atoms detach. This small change shifts the way it behaves completely, both in the lab and in real-world applications.
Real-World Uses—Why One Doesn’t Replace the Other
I spoke with a friend running a vineyard, and he’ll tell you that phosphorous acid earns its keep in plant disease management. Farmers trust it for fighting off oomycete pathogens like Phytophthora, which can wipe out crops in a season. They don’t grab phosphoric acid for that job, because it doesn’t have the same disease-fighting properties. Instead, phosphoric acid usually finds its way into fertilizer blends to boost phosphorus content for root development and growth. You’ll also find it acidifying soda beverages and rust-removal products.
In manufacturing, using the wrong acid creates headaches. Mixing up these acids leads to wasted batches, failed products, or even equipment damage. Phosphorous acid’s reducing nature can interact with metals in unpredictable ways, while phosphoric acid works better for controlled pH settings or phosphate supply.
Handling and Health
Neither substance belongs in untrained hands. Phosphoric acid creates issues with skin and eyes during handling, but large-scale exposure to phosphorous acid vapors brings extra risk. Fumes irritate airways and can harm tissues over time. Knowing the distinction between these acids means safer storage and use, especially since labeling mistakes aren’t unheard of.
Cost and Environmental Impacts
Manufacturing both acids leaves its mark on the environment. Phosphoric acid usually comes from phosphate rock, a resource that’s getting harder to mine sustainably. Excessive use, especially in fertilizer, fuels phosphorus runoff, which pollutes rivers and promotes algae growth—choking aquatic life in the process.
Phosphorous acid demands tighter controls in production and transport, mainly because its chemical properties make it more reactive and volatile. The agriculture sector needs to keep a close eye on application rates, since overuse offers no nutritional benefit to crops and only raises environmental risks.
What’s the Path Forward?
Anyone working around these acids—farmers, food techs, industrial engineers—should push for better training and clearer product labeling. Switch to digital tracking or use barcodes to avoid mix-ups in storage. Schools and training programs can do more to teach responsible handling and spill management.
On a bigger scale, industry and research should work together to limit environmental impacts tied to both acids. Researchers at public universities already look for alternatives and ways to recycle phosphorus from waste. Supporting these efforts and choosing products from transparent suppliers matters if you want to make a difference in sustainability and safety.
| Names | |
| Preferred IUPAC name | phosphonic acid |
| Other names |
Phosphonic acid Orthophosphorous acid Phosphorous(III) acid |
| Pronunciation | /ˈfɒs.fər.əs ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 10294-56-1 |
| Beilstein Reference | Beilstein Reference: 1207173 |
| ChEBI | CHEBI:29805 |
| ChEMBL | CHEMBL116431 |
| ChemSpider | 61460 |
| DrugBank | DB09449 |
| ECHA InfoCard | ECHA InfoCard: 100.030.045 |
| EC Number | 015-012-00-0 |
| Gmelin Reference | 527 |
| KEGG | C01108 |
| MeSH | D010768 |
| PubChem CID | 23986 |
| RTECS number | TH4900000 |
| UNII | 7J9327U17H |
| UN number | UN1803 |
| CompTox Dashboard (EPA) | DTXSID6024269 |
| Properties | |
| Chemical formula | H3PO3 |
| Molar mass | 81.996 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.651 g/cm³ |
| Solubility in water | Very soluble |
| log P | -0.77 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.3 |
| Basicity (pKb) | 1.3 |
| Magnetic susceptibility (χ) | -52.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.384 |
| Viscosity | Low viscosity |
| Dipole moment | 1.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1100 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1281 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V07AB |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, harmful if swallowed or inhaled. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H290, H314 |
| Precautionary statements | P261, P280, P301+P330+P331, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-0-1-A |
| Autoignition temperature | 300°C |
| Lethal dose or concentration | LD50 (oral, rat): 1895 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 1895 mg/kg |
| NIOSH | SC8925000 |
| REL (Recommended) | 30 mg/L |
| IDLH (Immediate danger) | Not listed. |
| Related compounds | |
| Related compounds |
Phosphoric acid Hypophosphorous acid Phosphine Phosphates Phosphite Phosphorus pentachloride |

