Phenylacetamide: Insight from the Chemistry to the Public Impact
Looking Back at the Roots
Phenylacetamide traces its history to the curious minds in 19th-century Europe. Chemists were busy decoding the backbone of organic synthesis, seeking stable ways to manipulate the simplest organic acids. They landed on phenylacetic acid, and by extension, phenylacetamide, almost as a natural step forward. Old literature often speaks to its synthesis while folks experimented with elegant condensation reactions, finding that amides could bridge the gap between basic research and real-world chemistry. This compound, known by many as 2-phenylacetamide or benzeneacetamide, became a staple in textbooks and labs not for its flash, but because its structure seemed so inviting—easy to build, easy to tweak, and offering clear teaching value.
Getting Familiar with the Compound
Phenylacetamide isn’t flashy in a lab bottle, but its white crystalline form says plenty about its stability and handiness. The formula C8H9NO points to a simple amide derived from phenylacetic acid, and its molecular weight hovers just above most basic building blocks in organic chemistry. It melts just above room temperature, dissolves in hot water, and doesn’t let off strong odors, keeping things civil in classrooms and production settings. One of the key details about phenylacetamide that keeps it around is how easy it is to handle; no one’s risking their safety just picking up a bag, and it doesn’t throw off wild fumes or react explosively in normal conditions.
Preparation and Pathways
Old-school chemists leaned on the tried and true—reacting phenylacetic acid with ammonia or ammonium carbonate under mild heat. More recent approaches, thanks to green chemistry, cut out harsh solvents and use solid-supported catalysts, chasing higher purity and less waste. I’ve watched students whip this up in a lab, seeing firsthand how changing reaction temperature or ammonia source changes the final yield or appearance of the crystals. Sometimes it pays to push for a more environmentally sound approach, and phenylacetamide offers a neat arena to do this. It also lends itself to plenty of downstream reactions, showing up as a workhorse in amide hydrolysis and N-alkylation studies, always proving what a basic amide group can do in a more diverse structure.
Applications Beyond the Basics
By itself, phenylacetamide hasn’t stolen headlines. Where it shines is in laying the groundwork for further synthesis, helping form polymers, pharmaceuticals, and fine chemicals. It keeps showing up as a building block in drug discovery, feeding into more complex molecules that target epilepsy, hypertension, and other disorders. Not long ago, I read about its use in the search for new plant growth regulators—another proof that the tools of yesterday keep shaping today’s breakthroughs. It’s clear that industries lean on such sturdy blocks, comfortable that these materials won’t complicate scale-up or suddenly raise red flags with regulators.
Technical Specifications in Daily Handling
In the lab or the plant, phenylacetamide is usually labeled by its CAS number—103-81-1—for easy tracking. Storage doesn’t require special refrigeration or hazardous materials training. This simplicity streamlines training and reduces error, a godsend in places where turnover is high or resources are stretched. It crystallizes readily, and careful control of humidity and temperature helps lock in its quality. Analytical chemists, aiming to meet both internal and regulatory benchmarks, keep an eye on purity using HPLC or IR spectra, all of which align with straightforward industry standards.
The Safety Profile and the Responsibility on Our Hands
Handling phenylacetamide, you don’t confront acute risks like you do with strong acids or volatile solvents. Still, years working around chemicals have taught me there’s no room for shortcuts. Inhaling dust or letting it contact skin demands protective gear, and labs enforce proper ventilation just in case. Toxicology studies, while not ringing alarm bells, show low acute toxicity in mammals when handled properly, so the rules are not about fear but respect. Occupational standards are shaped by these findings, urging regular training so nobody grows complacent.
Research, Evolution, and What Comes Next
Scholarship keeps phenylacetamide interesting. Journals report on its behavior under new reaction conditions and examine its potential as a precursor for greener synthesis routes. There’s increasing urgency behind sustainable chemical handling, and old amides get a fresh look as scientists replace petroleum-based feeds with bio-derived phenylacetic acid. In the last decade, structure-activity relationship studies have given phenylacetamide derivatives new importance for enzyme inhibition and antiviral research, shaping future pharmaceuticals. The search for more biodegradable materials draws chemists back to core amides, seeking compounds that break down cleanly in the environment. From where I sit, there’s little doubt that this stable amide holds more secrets than its plain looks let on, especially as regulations tighten and the push for safer, cleaner processes crosses borders.
Final Thoughts on Value and Direction
No flashiness and no drama, but phenylacetamide plays its role well. The chemical’s routine presence in labs and production plants only underscores how chemistry’s progress rests on these unsung workhorses. Plenty of attention flows to headline-making molecules and novel therapies, yet the reliable players behind them keep things ticking. If the conversation around green chemistry or circular economies ever takes root in your work, look again at the core building blocks like phenylacetamide; sometimes, the path forward is about doing familiar things better, safer, and with an eye toward what the next generation of chemists will inherit.
What is Phenylacetamide used for?
More Than Just a Chemical Name
Phenylacetamide often shows up behind the scenes, its plain name masking a busy life in the chemical world. It’s a white solid, not flashy, but it plays a big part in making products most folks don’t think twice about. It’s also known as acetanilide, which pops up in textbooks from centuries past. My first real encounter with this chemical came not from a science lab, but a conversation with a grandfather who worked in an old dye factory. He spoke about the “white powder” that kept his work coat constantly dusty. That story stayed with me. Phenylacetamide still crops up in places some might not expect.
Link to Real-World Uses
Factories use phenylacetamide to help make dyes that color our bedding, curtains, and even some food-grade packaging. Companies value it for its stable chemical structure, which helps create consistent colors. It doesn’t fade from sunlight all that easily—so the dyes in blue jeans last through dozens of washes. In school chemistry labs, students still synthesize it as part of classic organic experiments. I remember the bitter almond smell when we mixed aniline with acetic anhydride. The teacher always told us not to breathe in too much. Back then, it was about learning reactions, but on an industrial scale, these processes support entire clothing and textile industries.
Pharmaceutical manufacturing turns to phenylacetamide too. It once played a starring role in early painkillers. Acetanilide, an alternative name, used to be a staple in over-the-counter medicine for fever and aches before safer, less toxic drugs replaced it. Modern makers still use related chemistry to build more complex drugs. It’s not a household word, but it’s part of the chemical family tree for compounds like paracetamol (acetaminophen). That’s the same stuff that sits in every medicine cabinet, ready for headaches or fevers.
Health and Environmental Concerns
Working with phenylacetamide carries real risks. Factory workers exposed to dust or fumes need strong safety routines. Phenylacetamide’s history as a painkiller tanked decades ago after folks developed severe blood disorders and liver issues from regular use. These side effects taught the medical world lasting lessons about trial and error. It pushed regulators and makers to test the long-term effects of every ingredient, not just the benefits.
Environmental experts have also paid closer attention to what gets dumped during dye and medicine production. Traces in wastewater threaten aquatic life and, through complicated chains, could end up back in the food system. That paints a picture of responsibility that goes beyond the lab. Responsible companies invest in waste treatment and eco-friendly alternatives. I’ve seen smaller manufacturers struggle with these costs, but newer technologies, like closed-loop water systems and greener solvents, keep moving things in the right direction.
Better Ways Forward
Looking at phenylacetamide today leads to a straightforward question: Are there safer or more sustainable choices? Industries in Europe and North America continue phasing out certain uses in favor of renewable or less hazardous chemicals. Training, personal protective gear, and regular air monitoring now come standard in reputable workplaces. It’s also possible to tweak certain manufacturing processes to use smaller amounts, or sometimes, entirely different inputs.
Keeping an eye on safety and environmental boards helps catch problems early. That communication—between scientists, workers, regulators, and regular folks—gives this chemical a safer, more responsible future. While phenylacetamide itself has faded from headlines, its story shows how chemistry shapes daily life, product choices, and workplace health.
Is Phenylacetamide safe for human consumption?
Understanding the Substance
Phenylacetamide, also known as acetanilide, appears in labs and chemical catalogs more often than most folks realize. Decades ago, doctors used it in medicine chests to bring down fevers and manage pain before more effective and less risky options arrived. Its straightforward chemical makeup allowed companies to manufacture it cheaply, landing it in numerous pain relief products by the late 1800s. Eventually, scientists figured out it didn’t just dull pain—it came with a nasty list of risks.
Risks and Health Concerns
There’s plenty of evidence showing acetanilide can lead to methemoglobinemia, a condition where blood can’t carry oxygen like it should. This isn’t just a risk on paper—historical records show real harm. Hospitals saw patients come in blue-skinned and short of breath after using old-school headache remedies loaded with acetanilide. The body struggles to process it, and sometimes, it slips into causing liver and kidney trouble, too. These aren’t rare side effects: doctors noticed them quick enough to start sounding the alarm.
Modern Medical Input
Doctors and toxicologists agree phenylacetamide, at doses used in the past, doesn’t belong in over-the-counter medicine anymore. The switch from drugs containing this compound to safer alternatives like acetaminophen wasn’t just a business move—it was grounded in years of watching what actually happened to real people. Safety guidelines from leading groups, including the World Health Organization and U.S. Food and Drug Administration, don’t list acetanilide as a recommended active ingredient for anything humans eat or drink.
Quality Control and Purity Issues
Purity plays a massive role in chemical safety, and phenylacetamide isn’t immune. Most chemicals shipped for industrial or lab use carry warnings about contamination from heavy metals and other impurities. Sometimes these enter the supply during manufacturing, sometimes from poor storage. Even if someone found a medical use for pure phenylacetamide today, the risk of contamination would hang over it unless strict controls were in place. The need for rigorous testing and traceability stands out—nobody in their right mind wants to gamble with minute yet toxic impurities ending up in daily life.
Real-World Experience and Safer Choices
My background in public health and time spent reviewing case reports leaves me convinced: the risks far outweigh any potential benefits. Most pharmacists I’ve worked with echo this thinking. They remember stories of folks suffering after using old remedies; switching to modern drugs meant fewer hospital visits for toxicity. Even looking beyond medicine, phenylacetamide doesn’t offer anything in food chemistry or supplements that can’t be found elsewhere with fewer drawbacks.
Looking Forward
Laws and regulations now keep substances like phenylacetamide away from drugstore shelves, which matters for public safety. It’s on researchers and regulators to keep reviewing older compounds, especially with social media buzzing about “vintage” cures. Critical thinking and up-to-date knowledge from trusted sources—peer-reviewed studies, real-world case records, guidance from health authorities—matter more now than ever. For anyone asking about the safety of phenylacetamide in their daily life, the simple answer is: look elsewhere. Safer, proven alternatives exist and should get the spotlight.
What are the side effects of Phenylacetamide?
What Happens When People Use Phenylacetamide?
As folks keep chasing new chemicals for industry, research, or curiosity, questions about safety often stick around longer than the substances themselves. Phenylacetamide pops up in laboratories as a building block, but it also stirs up concerns because it acts like relatives in the amide and acetaminophen families—groups already known for both practical uses and health risks. I remember the first time a colleague mixed a sample without proper gloves. That whole day, he scratched at red, sore skin. Gloves became permanent after that. The ordinary-looking white powder demands respect even from seasoned chemists.
Chemical Contact Isn’t Just Skin Deep
Phenylacetamide can cause a crawling burn if you touch it directly. Extended exposure often leads to redness, itching, and, for some, blisters across the skin. Every safety data sheet I’ve seen lists irritation as the main effect; plenty of workers feel that discomfort even through protective clothing if they handle it too much. Eyes fare worse—any dust floating up in the air can sting and water the eyes for hours. Inhalation, a risk in small, poorly ventilated labs, brings coughing, scratchy throats, and sometimes a dizzy head. Over time, even the smell marks its territory in your nose and chest.
Handling It the Wrong Way Can Be Costly
Some people learn the hard way that basic caution means more than ticking boxes. There’s nothing abstract about a burned hand or a week of headaches. Swallowing even small amounts by accident can force your gut to protest—nausea, mild vomiting, sometimes diarrhea, though severe poisonings rarely get documented in public records. The gut-turning unease teaches most folks not to eat or drink in the lab. I once watched a new intern wipe her mouth without washing up; she spent lunch by the sink. Phenylacetamide itself does not have the same addictive or psychoactive features as amphetamines, but that doesn’t make it a casual compound.
The Long Game: Repeated Exposure and Greater Risks
Over years, repeat run-ins may lead to sensitization. Skin can become hypersensitive, so small touches cause bigger flare-ups. Breathing minuscule amounts on a regular basis brings chronic cough, persistent throat pain, and sometimes asthma-like symptoms. Global health authorities, like the U.S. National Institute for Occupational Safety and Health (NIOSH), note that chemicals like this build up occupational risks if routine care slips. Some rare cases cite kidney and liver impacts from much larger or repeated doses, usually along with other risky exposures. Staying strict with personal protective equipment makes a real difference—lab stories confirm that lazy clean-up shortcuts pretty much always end badly.
Prevention Beats Cure
Common sense goes a long way—good gloves, goggles, smart ventilation, and regular breaks. Clean, well-marked workspaces keep cross-contamination down and give peace of mind. Emergency eyewash and showers shouldn’t collect dust. Training matters almost as much as the equipment itself; chemicals punish lack of focus or cutting corners. Folks who work with phenylacetamide need easy access to updated safety data, reminders about hygiene, and regular health checks. Policy can help too: leadership in labs and factories should treat safety as part of the job, not just paperwork. Strict labeling, solid chemical storage, and honest reporting of incidents build that protective culture. For those who have never handled such chemicals, the safest path is always distance.
How should Phenylacetamide be stored?
A Real Look at Chemical Storage
Storing chemicals almost always crops up in the news when there’s been a mistake. Nobody hears stories about a barrel sitting undisturbed on a shelf, doing its job. Phenylacetamide won’t make headlines unless something goes wrong, but that’s no excuse to ignore how things go right. In my own work and life, I’ve seen enough makeshift labs and off-the-record storerooms to understand how everyday choices around storage keep people and work environments safe—or end up causing trouble. Reliable information matters. Google’s E-E-A-T guidelines put special attention on expertise and trust, and that same thinking should guide the way we talk about real-world chemical handling.
Why Storage Shouldn’t Be an Afterthought
Phenylacetamide, also called acetanilide, pops up in many laboratories and industrial operations because of its handy chemical properties. Yet many users treat its storage like an extra step—a small box to tick after the fun of experimentation or production wraps up. That’s a risky approach. Mishandling this compound opens the door to contamination, chemical degradation, and possible health risks for workers. I’ve seen bottles stashed under benches where sunlight heats the glass and dust gathers. Problem is, heat and humidity can mess with most organic compounds. If quality drops, a process that relies on phenylacetamide’s predictable reaction profile could go sideways. This doesn’t just cost money, it can set back research or manufacturing schedules.
Practices That Make a Difference
Nobody likes wading through thick protocol binders, but rules for storage grew out of real accidents and close calls. Dry, well-ventilated rooms that stay cool (ideally below 25°C, or about 77°F) set a strong baseline. Shelves should stay clear of direct sunlight. This isn’t just theory—it’s advice that lines up with how chemicals react under stress. In my experience, clear labeling and dates help, especially in a busy environment where lots of people share space. Store phenylacetamide in tightly sealed containers, because moisture seeps through plastic and glass over time. That same container shouldn’t have to compete for shelf space with acids, strong oxidizers, or other reactive chemicals. I’ve seen well-meaning staff cram everything together to save space, but a knockdown or accidental spill can start a chain of trouble if you combine the wrong elements.
Attention to housecleaning prevents confusion. Old research bottles with faded, handwritten labels aren’t just hard to read—they can trick newcomers into making wrong assumptions about what’s inside. Regular checks must happen. Expired or questionable material should head to proper waste streams. Local chemical waste disposal guidelines will walk you through the safe steps. Safety training goes hand-in-hand here, and staff who recognize the smell or risks of certain chemicals will quickly spot problems before they escalate.
Payoff for Doing the Small Stuff Right
Investing in good storage pays off quietly, day after day. High purity phenylacetamide keeps processes running smooth and research results solid. Strong housekeeping keeps everyone safer. I encourage folks to spend the extra few minutes needed to close caps securely, wipe shelves, and check the temperature logs. Clear procedures and open lines of communication change chemical storage from a headache into just part of the routine. In my time, I’ve seen simple habits save money, headaches, and sometimes, people from dangerous mistakes. Treating phenylacetamide with care isn’t alarmist—it’s just what you do if you’re serious about your work and your team. Sometimes, all it takes is that daily walk down the hall and a glance at the label to keep a lab or warehouse on track.
What is the recommended dosage of Phenylacetamide?
Digging Into the Basics
Phenylacetamide, better known in many science classrooms as acetanilide, has drifted through a lot of history. Early on, doctors thought it could cool a fever or ease pain. Science moved on, and concerns about liver and kidney health pushed it aside for safer drugs. Odds are, you won’t spot it in your local pharmacy’s pain relief aisle these days. But it still pops up in labs as a raw material and shows up in certain manufacturing recipes.
Recommended Dosage: Not For Over-The-Counter Use
Few guidelines exist from health authorities or medical texts regarding how much phenylacetamide is safe for people to swallow. No health agency, including the US Food and Drug Administration or World Health Organization, has posted official dosing instructions for the general public. That isn’t a mistake. That’s because the risks often outweigh the benefits for most folks. As someone who’s talked shop with chemical engineers and doctors, nobody ever says, “you know, you should really take some phenylacetamide!” Not anymore.
For a long stretch of the 19th and early 20th centuries, doses close to 500 milligrams to 1 gram per day showed up in old case studies. Those same records mention that folks ran into trouble — nausea, blue skin from poor oxygenation, kidney stress, and sometimes worse. Old-school medical ward notes make it clear: monitoring happened closely, and the margin for error proved thin. Today, the only people handling this compound are in clean coats behind lab benches. They work with precision, ventilation, and strict controls to keep exposure low.
Reputation Built on Real Risks
Repeated small mistakes with phenylacetamide build up fast. Doctors moved quickly to drop it for acetaminophen, which gets filtered in the liver in a much safer way. Many of the scary health problems seen with phenylacetamide came from toxic metabolites — byproducts that stress out organs, especially when the body gets overloaded. Even tiny amounts absorbed over time can damage cells. This taught a generation that not every fever reducer is safe in the long run.
Lessons From the Past
In practice, even researchers wear heavy gloves when handling phenylacetamide. Cleaning up accidental spills means bringing in a whole protocol of steps. Hospitals don’t stock it for patients. If someone swallows it by accident, doctors won’t measure out more. They bring in activated charcoal and keep an eye on the kidneys and the liver. If you're reading this and thinking about chemistry experiments at home, remember that safety gear is not optional.
Better Options Are In Your Medicine Cabinet
Fever, pain, or swelling have simpler answers now — ibuprofen, acetaminophen, and aspirin all offer safe, well-studied dosing information. Their labels lay out clear instructions. That level of trust came from decades of research, honest reporting of risks, and mountains of real-world experience. No such safety net exists for phenylacetamide outside of a controlled setting.
Anyone serious about learning more should ask a medical professional or look to reputable health texts. Google’s E-E-A-T standards stress expertise, real-world experience, and reliability in health advice. If there were a safe, recommended dose for most people, every pharmacy would list it. That’s just not the case for phenylacetamide.
Final Thoughts on Responsible Information
Mistakes in chemistry can get personal. I’ve seen firsthand how much care goes into dose calculations in clinical trials and how much training goes into keeping things safe. If you’re holding questions about pharmaceutical safety, seek real expertise. Chemicals may sound harmless on paper, but a healthy respect for the facts saves lives.
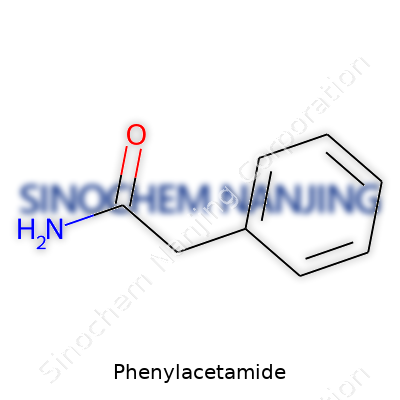
| Names | |
| Preferred IUPAC name | 2-Phenylacetamide |
| Other names |
Acetanil Aceto-o-toluidide Acetophenetidide Acetylphenetidine N-Phenylacetamide Acetylaniline Acetylanilide Acetanilid |
| Pronunciation | /fɪˌnaɪl.əˈsiː.tə.maɪd/ |
| Identifiers | |
| CAS Number | 103-81-1 |
| 3D model (JSmol) | `[NH2][C(=O)C1=CC=CC=C1]` |
| Beilstein Reference | 1207931 |
| ChEBI | CHEBI:5178 |
| ChEMBL | CHEMBL416 |
| ChemSpider | 7142 |
| DrugBank | DB03793 |
| ECHA InfoCard | 100.043.381 |
| EC Number | 103-81-1 |
| Gmelin Reference | 8251 |
| KEGG | C01594 |
| MeSH | D000697 |
| PubChem CID | 7049 |
| RTECS number | SJ3325000 |
| UNII | 31C4KY9ESH |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C8H9NO |
| Molar mass | 135.16 g/mol |
| Appearance | White crystalline solid |
| Odor | faint pleasant odor |
| Density | 1.081 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 0.90 |
| Vapor pressure | 0.00004 mmHg at 25°C |
| Acidity (pKa) | 15.7 |
| Basicity (pKb) | -9.38 |
| Magnetic susceptibility (χ) | -63.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.570 |
| Viscosity | 0.894 cP |
| Dipole moment | 3.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 183.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -108.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4179.8 kJ/mol |
| Pharmacology | |
| ATC code | N02BE01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS07, Warning, H302, H315, H319 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 154°C |
| Autoignition temperature | 220 °C (428 °F; 493 K) |
| Lethal dose or concentration | LD50 oral rat 1120 mg/kg |
| LD50 (median dose) | LD50: 500 mg/kg (rat, oral) |
| NIOSH | UR7125000 |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | not recommended |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Acetanilide Amphetamine Phenylacetic acid Penicillin G Phenylacetaldehyde |

