Pentaerythritol: Foundations, Uses, and the Road Ahead
Historical Development
Pentaerythritol has roots that reach back more than a century. Chemists first synthesized it in the late 19th century, a time marked by the hunger to invent new compounds and unlock potential in industrial chemicals. Over the years, innovators turned a once obscure polyol into an industrial staple, thanks to a unique molecular structure. Through advancements in chemical engineering, manufacturers steadily scaled production and improved purity, setting the stage for broader applications. Anybody who has watched a raw material quietly take on a pivotal role across industries can appreciate how chemistry shapes daily life, sometimes without fanfare. Pentaerythritol’s steady rise from a lab novelty into widespread use shows how curiosity-driven science has lasting ripple effects.
Product Overview
Few folks outside of certain science fields recognize pentaerythritol, yet it turns up in everything from paints to explosives. As a white, crystalline solid with four hydroxyl groups branching off its carbon backbone, this compound works as a building block for many important chemicals. Folks producing synthetic resins, alkyd paints, varnishes, adhesives, and lubricants all rely on its consistent behavior. Its ability to lend stability and durability wherever it goes earns it a critical role, not because it dazzles but because it quietly solves practical problems where reliability matters most. What usually draws industry veterans to pentaerythritol is its balance of chemical functionality and physical predictability.
Physical & Chemical Properties
Anyone who has worked with pentaerythritol knows it by its white, odorless crystals and taste reminiscent of sweetness. It melts at relatively high temperatures, resisting decomposition under heat unless strong acids or bases enter the picture. Its solubility presents a split personality: eagerly dissolving in hot water, yet holding back in cold one; not much for mixing with most organic solvents unless under duress. This stubborn streak brings benefits. Stable, unreactive under normal conditions, it avoids unwanted surprises where consistency is vital, such as formulations for coatings and lubricants. Its tetrafunctional nature means four alcohol groups hang ready for chemical reaction, so chemists can easily modify and craft derivatives that fit specific needs—one of the big reasons it built a solid fanbase among formulators.
Technical Specifications & Labeling
Tracing a pentaerythritol sack from warehouse to storage shouldn’t raise eyebrows if labeling stays clear and honest. Good practice demands listing purity—often above 98 percent for industrial batches. Solid content, moisture, and melting point get their spot in technical sheets. Labels tend to warn that dust can form combustible clouds if handled carelessly. These measures serve more than compliance. They streamline workflow for anyone working with the raw material, reducing unpleasant surprises during blending or transfer, and making routine safety checks smoother. People in the trenches remember that clear technical specs translate into less trouble downstream; details count for those grinding through high-volume processes under time pressure.
Preparation Method
Industry folk generally produce pentaerythritol by reacting acetaldehyde with formaldehyde under alkaline conditions. A batch operation starts with mixing the two feedstocks in a reactor. Alkali comes next, not only to catalyze but to push the reaction forward until the crystals finally separate out. Filtration and purification give the product its characteristic look. This method, refined since the 1930s, strikes a balance between yield, cost, and environmental footprint—never a trivial thing in mass manufacturing. Steps get tweaked depending on desired purity, but the method reflects a tradition of practical refinement—a testament to collective ingenuity, driven by real-world pressures to turn out large quantities dependably without drilling holes in profit margins.
Chemical Reactions & Modifications
Four free hydroxyl groups spell opportunity. Chemists exploit these reactive sites to create esters, ethers, and polymeric compounds. Manufacturers prepare pentaerythritol esters by reacting the base material with fatty acids, leading to derivatives used in lubricant oils or plasticizers. Cross-linking with carboxylic acids produces robust resins for protective coatings. Fireworks and explosives industries leverage its ability to form nitrates, turning it into pentaerythritol tetranitrate (PETN)—a key energetic material. Those who spend years on process improvement see pentaerythritol’s flexibility as a tool that adapts to changing demands, sometimes even rescuing production lines when shifts in raw material prices or regulation force a change of plans.
Synonyms & Product Names
Pentaerythritol goes by several names if you comb through supplier catalogs or technical literature. Synonyms include tetramethylolmethane and 2,2-bis(hydroxymethyl)-1,3-propanediol. In some sectors, folks condense it to PETR, though that can cause confusion with polyethylene terephthalate, so context comes to the rescue. The core identity does not change, and once you spot one of these synonyms on a bag, you learn to connect it to the underlying chemistry that defines its performance and risk profile. Synonyms remind us of the global reach of foundational chemicals—same substance, many tongues.
Safety & Operational Standards
Safety managers stress that pentaerythritol powder and dust can create fire risks—nothing theoretical, all hard-earned from factory incidents. In large-scale plants, dust collection, ventilation, and spark-free equipment get used as a matter of routine. No one wants to risk ignition, so practices like grounding tools and keeping floors clean move from suggestion to requirement. Health risks track closely with exposure. Skin irritation remains unlikely, though eye contact bothers workers. The biggest concern comes from inhaling dust, which underscores the need for masks and proper housekeeping. Good training and sensible adherence to protocols keep risks low, teaching a hard lesson that safety is an ongoing commitment instead of a box to check at orientation.
Application Area
Pentaerythritol touches many corners of industry. The alkyd resins in everyday paints and coatings count on it for robustness and washability—features that save the cost of premature reapplication. In lubricants, chemists turn to pentaerythritol as a base for high-performance synthetic esters, giving machinery and engines a fighting chance against wear and tear. Its fire retardancy and resistance to heat earn it spots in specialty plastics, often in electrical and construction products. Explosives manufacturers value pentaerythritol as a precursor to PETN, contributing precision and reliability crucial for mining and defense. Even in the pharmaceutical world, certain drugs get stabilized using derivatives. Year after year, I watch it hold ground as a go-to ingredient where function and dependability trump novelty.
Research & Development
In labs and R&D centers, pentaerythritol remains a testbed for creative minds looking for safer, greener, or more efficient pathways. Trends in sustainability push chemists to develop bio-based alternatives or tweak the old synthetic process to cut down on formaldehyde use. Teams continue to research new esters and additives using pentaerythritol, aiming to extend shelf life, reduce toxicity, or enhance biodegradability. Advanced coatings for electronics or medical devices increasingly rely on clever modifications of this molecule. If you sit through conferences or read fresh patent filings, you sense the push to reimagine traditional polyols’ roles in emerging tech fields, just as past generations transformed coal tar derivatives into modern plastics.
Toxicity Research
A lot of ink covers the safety record of pentaerythritol. Toxicologists run tests to nail down its effect on the environment and human health. Results tend to show low oral toxicity in mammals, which helps explain its persistent industrial favor. Regulators in the US, EU, and Japan, after years of scrutiny, generally allow its use with standard precautions. Aquatic toxicity falls below worrisome thresholds, and long-term persistence looks manageable. There’s always room for vigilance. Waste handling and emissions from large plants still demand attention because outdated systems can turn a safe compound into a community irritant. Safety reviews should keep pace with process changes—a lesson learned from watching regulatory misses in related chemicals.
Future Prospects
Pentaerythritol sticks around in large part because of its versatility, but the next decade will likely test how resilient it stays under pressure from new sustainability laws and customer demands. The trend toward bio-sourced chemicals might shift sourcing patterns or open room for modified versions with superior environmental profiles. Process engineers keep looking to reduce energy use and cut greenhouse gas emissions tied to formaldehyde and acetaldehyde precursors. These changes could push clever tweaks in the classic synthetic route or open room for biotechnological production. For all the talk of green chemistry, companies will only switch supply if new solutions meet cost targets and work in existing applications without fuss. The quiet resilience of pentaerythritol makes it a reliable foundation, but continued innovation—whether in greener process chemistry, safer derivatives, or broader applications—will decide if it thrives or fades into the background as upstarts take over.
What is Pentaerythritol used for?
A Building Block for Industry
Pentaerythritol shows up in more parts of our lives than most folks realize. If you look around your house, chances are you’ll spot at least one product where this chemical stepped in behind the scenes. In my years working with contractors and manufacturers, I’ve learned that this white, crystalline substance plays a big role in making materials stronger, safer, and more reliable than older methods allowed.
Resins and Paints
If you care about long-lasting paint finishes on buildings or cars, you’ve got pentaerythritol to thank. It works as a core ingredient in making alkyd resins. These resins help paint dry faster, stand up to rough weather, and resist cracking. The reason manufacturers choose pentaerythritol over other chemicals comes down to the balance it gives between flexibility and toughness. I’ve seen factories switch over to these newer resins and watch their products gain years of life compared to older formulas.
Explosion Safety and Fire Resistance
Most people don’t connect everyday chemicals to life-saving materials, but pentaerythritol does heavy lifting in making nitrocellulose and explosives more stable. The same building block that helps your paint last on the wall also keeps explosives steady until they’re needed for mining or military use. More importantly for families, this chemical helps create flame retardants and fire-resistant plastics. Fire marshals I’ve worked with have stressed how advances in these chemicals made homes safer and gave firefighters more time to respond during emergencies.
Lubricants and Plastics
Factories rely on machines with moving parts, and keeping those parts running often comes down to good synthetic lubricants. Pentaerythritol lets oil makers create stable, reliable lubricants that resist breaking down at high temperatures. Engine oils and industrial greases would break down much faster under pressure without this ingredient, leading to more breakdowns, higher repair bills, and even safety risks. On the plastic side, pentaerythritol makes products like PVC tougher and less likely to degrade from sunlight or chemicals.
An Eco-Friendlier Option
Concerns about toxic byproducts and pollution come up a lot in my conversations with environmental engineers. Pentaerythritol has gained ground as a greener option—its production process creates fewer dangerous side products than some older chemicals. For people working to reduce industrial pollution, this choice marks a step in the right direction. Studies reported in journals such as Environmental Science & Technology show less persistent organic pollution coming out of plants using pentaerythritol-based processes compared to alternatives heavy with heavy metals or halogens.
Solutions and the Way Forward
Even a workhorse like pentaerythritol faces tough questions. World demand keeps climbing, and raw material supply sometimes feels tight. Chemical engineers I know are already working on routes that waste less and get a higher yield per batch. They’ve talked about using renewable raw materials like plant starches to make the process more sustainable. For communities living near manufacturing plants, stronger regulations and better air filters could cut down on dust and vapor release.
As every generation puts new demands on materials—be it safer electronics, sturdier bridges, or cleaner production—pentaerythritol continues to adapt. Its continued popularity among manufacturers shows that practical chemistry, quietly working in the background, still shapes the world for the better.
Is Pentaerythritol safe to handle?
A Closer Look at Pentaerythritol
Pentaerythritol shows up in industries that reach from paint formulation to explosives. People who haven’t worked in labs or industrial plants might have never heard about it, but for chemists and manufacturers, this crystalline powder can seem pretty familiar. The main question that comes up—especially for anybody who needs to handle it regularly—is whether this substance can be considered safe to use, and what kinds of precautions keep the risk low.
Understanding the Real Risks
I’ve spent time in chemical storage rooms, and it’s always clear that “non-toxic” never means “no risk.” Pentaerythritol doesn’t act like heavy metal salts or the more notorious reagents, yet it doesn’t belong in your coffee, either. According to safety data and the European Chemicals Agency, pentaerythritol has low toxicity via skin or breathing exposure in normal industrial settings. People who touch it with bare hands usually don’t notice any immediate reactions. Skin issues only crop up after repeated or prolonged exposure, and even then, most folks see mild irritation or redness at worst.
Breathing dust from any fine powder can lead to coughing or a sore throat. The same risks exist with flour or talcum powder. The real problem starts if you ignore protective equipment, forget about ventilation, or don’t handle spills right away. Breathing in a huge amount of dust, by accident or neglect, can irritate the airways. That lesson holds for nearly every powdered material in the lab or the factory.
Why Handling Practices Matter
Early in my career, I saw a technician try to weigh out a batch of pentaerythritol without a mask or gloves. White dust clouded in the air, and the tech started to sneeze, looking thoroughly annoyed. He joked about “harmless dust” but went straight for eye drops after rubbing his face. This short episode stuck with me. It proved that even safe substances can turn uncomfortable—or risky—if you don’t show them respect.
Studies from the National Institute for Occupational Safety and Health back this up, showing no cancer risk or bioaccumulation in the body from pentaerythritol. Most problems involve temporary irritation. Eco-toxicity studies also put it in the “low concern” category. So, if you handle it like you’d handle flour or cement—gloves, dust mask, and ventilation—you set yourself up for a routine, uneventful day.
Responsible Solutions for Safe Handling
Accidents don’t ask if their cause topped a safety chart. That’s why workers should keep personal protective equipment close. Companies have a duty to remind staff about goggles and properly labeled containers. Good ventilation, regular safety drills, and well-written procedures keep incidents from becoming stories people retell for years. Spills need to be cleaned with a damp method—never swept dry—so dust never gets the chance to drift into the air. These habits never get old, and they do more to build a culture of safety than any wall chart.
Pentaerythritol keeps showing up in modern manufacturing for a reason—it gets the job done without bringing major health baggage along. But every workplace owes its workers simple respect for risk, clear instructions, and easy access to safety gear. Treat it with care and solid information from credible sources, and you can keep your day just as safe as another stack of office paperwork—all with a lot less risk of a cut or a burn.
What is the chemical formula of Pentaerythritol?
The Chemical Makeup of Pentaerythritol
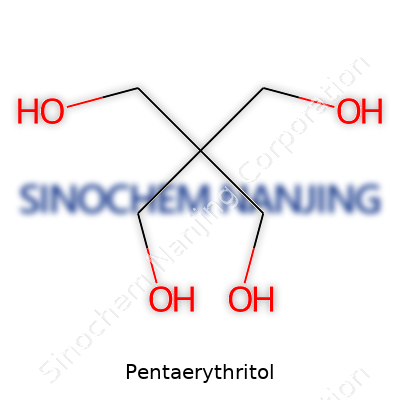
Pentaerythritol carries the chemical formula C5H12O4. This compound has four hydroxyl groups bonded to a single carbon backbone. Imagine a compact carbon "hub" where each arm grabs onto an -OH group. That makes it a type of polyol, which translates to a molecule loaded with multiple alcohol groups.
Why the Formula C5H12O4 Matters in Industry
Many might overlook ordinary chemical names, but Pentaerythritol’s structure drives its real-world uses. The four hydroxyl groups lead to serious reactivity, letting it form tight chemical networks. Manufacturers lean on it for stable synthetic lubricants, paints, polyester resins, and especially alkyd resins. This core structure stands out in explosives manufacturing too, shining in the form of pentaerythritol tetranitrate (PETN). Explosives need compounds that pack lots of energy and burst reliability; Pentaerythritol’s compact backbone checks those boxes.
Bringing Chemistry and Safety Together
I remember a chemistry professor holding up a bottle of white Pentaerythritol crystals and stressing how tiny adjustments to a molecule’s makeup yield strikingly different properties. The difference between a paint resin and an explosive comes down to how the same base molecule gets processed and reacted. A chemical formula isn’t just numbers and letters—it’s a blueprint that controls results in an industrial blender or a lab reactor. That’s why workers in chemical plants keep careful track of even minor changes in reactants and ratios.
Where Precision Pays Off
Take alkyd paints, a staple in everything from bridges to boats. Here Pentaerythritol strengthens resins and helps paints stand up to moisture and weather. Moving away from lead-based stabilizers was a big step for environmental health. Pentaerythritol’s structure let manufacturers switch to safer, longer-lasting formulas without sacrificing quality. In lubricants and synthetic oils, its formula provides backbone flexibility and high viscosity—all without breaking down fast under heat. Long engine life owes a favor to these chemical roots.
Addressing Environmental Impact
Chemicals like Pentaerythritol hit the news whenever environmental debates heat up. Polyols can present challenges if they’re mismanaged in waste streams. Supporting greener practices means careful control of byproducts during manufacturing and developing recycling strategies for end-of-life resins or products. Chemical makers have started using renewable raw materials as part of a broader shift to sustainability. Some European producers now base the entire synthetic route off forest-based feedstocks, skipping over petroleum where possible. Clear labeling, government standards, and third-party certification all help convince downstream users their paints and lubricants don’t leave a heavy footprint.
Shaping Tomorrow’s Choices
People often see formulas like C5H12O4 as just trivia. But for families that want safer homes, industries craving reliable products, or workers whose health matters at every step, understanding the chemicals in daily life makes a real difference. Pentaerythritol’s story reflects the bigger push in science to balance efficiency with safety. More research into plant-based production, improved recycling, and safer handling will keep these molecules working for—not against—communities worldwide.
How is Pentaerythritol stored?
What Is Pentaerythritol and Why Does Storage Matter?
Pentaerythritol shows up a lot in manufacturing paints, explosives, plastics, and lubricants. It comes as a white, odorless powder that doesn’t shout danger, but safe storage really matters. Mishandling this chemical runs up costs fast—spilled product, ruined batches, even serious accidents.
Staying Dry and Secure
From experience in industrial settings, humidity ruins chemicals faster than almost anything else. Pentaerythritol pulls in moisture from the air like a sponge, so dry storage space is non-negotiable. Most facilities use sealed drums or heavy-duty plastic bags inside sturdy containers. Why so much effort? Moisture changes chemical properties, causing clumps or caking, which might slow down production lines. Also, nobody wants a lab surprise, so keeping spill-proof containers locked up goes beyond the rulebook—it keeps workers safe.
Temperature Is No Joke
Storage rooms for pentaerythritol don’t need to double as freezers, but stable, moderate temperature keeps the compound sound. Direct sunlight leads to uneven warming, which never ends well with powders. So, closed rooms with good airflow—never near heat sources—work best. Out-of-the-way areas keep foot traffic low. People have a habit of stacking random boxes, which leads to tears in packaging and leaks.
Label Everything You Store
In a rush, it’s tempting to toss extra pails of powder in any available spot and scribble a nickname on the label. That’s risky. Clear, permanent labeling with the full chemical name, date, and handling instructions prevents mix-ups down the road. I’ve watched a shipment go back to the sender because of poor labeling. Mistakes cost a company more than just dollars—reputation is tough to rebuild once clients lose trust.
Keep Away from Acids and Bases
Pentaerythritol looks innocent, but hazardous reactions can spark off if acids, bases, or strong oxidizers stand too close. Segregation matters. People stack drums of cleaning supplies next to specialty chemicals all the time, and I've seen near misses because folks forgot a little spill can travel fast. Keeping incompatible chemicals several aisles apart or on different shelves is a practice worth following, no matter the pressure in a busy workday.
Emergency Plans and Regular Checks
Things get overlooked. Someone bumps a drum into a wall, dents it, and pushes it to the back. That drum could leak before anyone notices. Scheduled inspections help catch cracked lids and faded labels before an accident. Employees understand that if you trust them with information and training—like proper use of personal protective gear and a clear response plan—they spot problems early.
Forward-Thinking Solutions
Technology offers new options for tracking conditions inside storage spaces. Smart sensors find leaks quickly, track humidity and temperature, and alert managers. Investing in good pallets and racks prevents container breakage. Rotating your stock to keep older lots in use cuts down on long-term degradation risk.
People First—Always
Nothing replaces good training and a safety culture. Anyone working around chemicals like pentaerythritol needs practical knowledge, not just a binder full of rules. In my time on factory floors, teams who looked out for each other made fewer mistakes. Safe storage means fewer health scares, less environmental mess, and a stronger, more reliable business.
What industries commonly use Pentaerythritol?
How Paints and Coatings Rely on Pentaerythritol
Walk into any hardware store, and you’ll see gallon after gallon of paint promising long-lasting color and weather protection. A lot of that performance starts in the chemistry lab, where pentaerythritol earns its spot in the recipe for alkyd resins. These resins form the backbone of paints and coatings, bringing hardness and durability. I remember painting my first apartment and watching rain bounce right off the trim. That toughness owes something to this unassuming polyol because it helps resins cross-link into sturdy films, keeping wood and metal safer for years. Rapid fading and peeling are much less common thanks, in part, to how pentaerythritol shapes modern coatings.
Role in Lubricant Additives
Cars, bicycles, factory machines—pretty much anything with moving metal parts—benefit from synthetic lubricants. Pentaerythritol steps up here as a building block for esters found in top-tier lubricants. These improve flow at low temperatures and stick around at high ones. Mechanics know grease that won’t break down under pressure can save on repair bills and downtime. The use of pentaerythritol-based esters shows up in the transmission fluids that keep an engine running smooth during summer and dead winter alike. It’s good to see chemistry having your back under the hood.
Fire Safety Products
Ask anyone who’s worked in building or wiring about concerns over fire, and you’ll hear real stories of near-misses and the difference fire retardants can make. Pentaerythritol finds its way into flame retardants for plastics, textiles, and electronic components. Manufacturers mix it into insulation, circuit boards, and fabrics. The key: it helps finished products resist burning and slow the spread of flames if something sparks. Safer appliances and electronics help give peace of mind at home and in public spaces. As fire codes keep getting stricter, this material remains in demand from safety engineers who don’t want to roll the dice.
Pharmaceuticals and Healthcare Applications
Folks in the pharmaceutical industry depend on consistency when making pills, ointments, and medical creams. Pentaerythritol makes its way into creams and emulsions as a stabilizer and base material. It gives products a texture that spreads well on the skin and helps control the release of active ingredients. If you’ve ever used an ointment that soothed pain and stuck around without making a mess, chances are good you’ve seen the results of some clever chemistry. Reliability matters here—people trust their medication to work right, day in and day out.
Plasticizers and Adhesives
Most folks use products glued or bonded together every day. The things that keep shoes from coming apart or wires firmly sealed inside flexible cables often use plasticizers made from pentaerythritol. These softening agents turn rigid compounds into something bendable and strong. In adhesives, this means glue won’t suddenly turn crumbly in cold weather or ooze in a heatwave. Reliable bonds keep products functioning and safe, which matters from construction to everyday consumer items.
Moving Toward Safer, Greener Chemistry
It’s hard to ignore concerns about safety and sustainability in modern manufacturing. The search for less hazardous raw materials and reduced waste is ongoing. Pentaerythritol, by supporting products that resist fire and weather, helps reduce waste, repair needs, and property loss. The chemical industry keeps looking for renewable sources and safer processes to make essential compounds like this one, pushing for advances that balance utility with long-term safety.
| Names | |
| Preferred IUPAC name | 2,2-Bis(hydroxymethyl)propane-1,3-diol |
| Other names |
Pentaerythrite Monopentaerythritol 2,2-Bis(hydroxymethyl)-1,3-propanediol Tetramethylolmethane |
| Pronunciation | /ˌpɛn.tə.ɪˈrɪθ.rɪ.tɒl/ |
| Identifiers | |
| CAS Number | 115-77-5 |
| 3D model (JSmol) | `load =3pel` |
| Beilstein Reference | 1101003 |
| ChEBI | CHEBI:18009 |
| ChEMBL | CHEMBL140209 |
| ChemSpider | 10983 |
| DrugBank | DB14426 |
| ECHA InfoCard | 19e3b6b2-02f4-4364-9e94-2fe53f1d47eb |
| EC Number | 201-334-6 |
| Gmelin Reference | 88149 |
| KEGG | C01083 |
| MeSH | D010400 |
| PubChem CID | 8530 |
| RTECS number | SD7320000 |
| UNII | 6Z70NJ5I87 |
| UN number | 1347 |
| CompTox Dashboard (EPA) | DTXSID1025472 |
| Properties | |
| Chemical formula | C5H12O4 |
| Molar mass | 136.15 g/mol |
| Appearance | white crystalline powder |
| Odor | Odorless |
| Density | 1.39 g/cm³ |
| Solubility in water | Moderate (1.2 g/100 mL at 25 °C) |
| log P | -0.97 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 13.0 |
| Basicity (pKb) | 8.24 |
| Magnetic susceptibility (χ) | -7.2e-6 cm³/mol |
| Refractive index (nD) | 1.462 |
| Dipole moment | 1.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -538.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6785 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | C01EB15 |
| Hazards | |
| GHS labelling | GHS classification: Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008. No GHS pictogram, signal word, hazard statements, or precautionary statements are required for pentaerythritol. |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: Not a hazardous substance or mixture according to the Globally Harmonized System (GHS). |
| Precautionary statements | P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | > 385°C |
| Autoignition temperature | 285 °C |
| Explosive limits | Explosive limits: Dust may form explosive mixture with air |
| Lethal dose or concentration | LD50 (oral, rat): 10,000 mg/kg |
| LD50 (median dose) | 6,600 mg/kg (rat, oral) |
| NIOSH | WI7750000 |
| PEL (Permissible) | PEL for pentaerythritol: 15 mg/m3 (total dust), 5 mg/m3 (respirable fraction) |
| REL (Recommended) | 320 |
| Related compounds | |
| Related compounds |
Methenamine Trimethylolethane Trimethylolpropane Neopentyl glycol |

