Looking at Pentachloroethane Through a Modern Lens
The Path Pentachloroethane Has Traveled
Try walking through the story of industrial chemistry in the past hundred years, and pentachloroethane will pop up sooner than many expect. Chemists started working on chlorinated hydrocarbons in earnest during the late nineteenth and early twentieth centuries. Pentachloroethane was never the star of the show, sitting behind more famous cousins like chloroform or trichloroethylene, but it still had more than a cameo. Early industrial research into hydrocarbons usually focused on squeezing out new solvents, intermediates, and in some cases, compounds that could serve as precursors for anesthetics, dry cleaning agents, or insecticides. Pentachloroethane slotted into this growing chemical arsenal. My first few weeks in a chemical archives library revealed as much—old safety manuals from the 1950s listed it along with stern warnings. That history matters because the industrial-scale push for new compounds helped put many people today in the position to weigh risks and rewards associated with compounds like pentachloroethane.
What Pentachloroethane Brings to the Table
Pentachloroethane holds its spot in the chemistry world as a stable, dense, and colorless liquid under ambient conditions. Its molecular formula, C2HCl5, might look uninteresting to non-specialists, yet every extra atom of chlorine packed into the molecule changes its behavior. Dense scent and volatility remind me of laboratory afternoons, with the faint chemical tang lingering at the edge of the room. Boiling near 162°C and with a melting point around -39°C, this substance resists rapid evaporation but doesn’t resist much cold. These physical qualities made it appealing for solvent work decades ago. Tossing it in with organic reactions, it doesn’t explode with reactivity, mostly holding its own and resisting breakdown unless pushed. It’s clear, it’s nonflammable, and, for a while, it checked boxes for cost-cutting industrial applications.
Technical Details and Labels: A Necessary Conversation
Ask anyone with experience in chemical processing, and they’ll say that technical specs aren’t just for pedants. Manufacturers and handlers want to know about purity grades, residual contaminants, and suitable storage containers (glass or lined steel, no room for error). The Global Harmonized System and OSHA rules demand explicit signaling about hazards. Workers, especially those in older facilities, have learned to spot the diamond-shaped warning stickers and color codes without blinking. In the real world, labels need to call out its risks—carcinogenic suspicion, environmental worries, and volatility. These labels rarely slow things down but keep people from making avoidable mistakes that, over decades, can haunt companies and communities.
Making Pentachloroethane: The Chemistry on the Table
Synthesizing pentachloroethane isn’t a kitchen-table affair. Factories rely on careful chlorination of ethylene dichloride or trichloroethylene. Reactions involve bubbling chlorine gas through the parent hydrocarbon, using high temperatures and UV light as the sparks for substitution reactions. Chemists see the challenge: controlling the reaction to avoid producing hexachloroethane or other byproducts. The process usually takes place in closed systems, with off-gases scrubbed and significant monitoring. Early chemists shrugged at the environmental worries, but today, process engineers confront a stricter world. Every step, from raw chlorine handling to final distillation, presents a safety puzzle—environmental controls and leak prevention play an outsized role in plant design and operator training now than they did during the mid-20th century.
Sharing Names and Aliases in Chemistry’s Web
Chemicals earn more names than some rock stars, and pentachloroethane answers to plenty: 1,1,1,2,2-pentachloroethane and pentachloroethyl are common variants. Synonyms can trip up even the experienced—my early years spent poring over research papers made that lesson plain. These names matter when hunting for data, checking legal restrictions, or cross-referencing supply lists. Paper trail clarity seems trivial to outsiders, but too many accidents and regulatory missteps trace back to slips in chemical naming or identification. No matter the nickname, the substance brings its own baggage, and responsible professionals pay attention instead of skating past these details.
Main Chemical Tricks: Reactions and Playing Dress-Up
Chemists don’t just collect chemicals—they transform them. Pentachloroethane joins the game as both a starting material and a bystander. Heat or strong base splits off hydrochloric acid, giving trichloroethylene, which lands with a loud splash in industrial use. Some routes use pentachloroethane as a stepping stone in the march from ethylene to heavier chlorinated products or fluorinated derivatives. I once watched a team struggle with a persistent side-reaction: under alkaline conditions, pentachloroethane stubbornly resisted full conversion, reminding everyone that lab-scale chemistry and plant-scale reality rarely line up perfectly.
Safety Habits and Working Standards
Few topics draw as much debate in the workforce as chemical safety. Pentachloroethane, though not as infamous as PCBs or dioxins, shares some of their stubborn, persistent hazards. Old industrial settings often skipped detailed ventilation, and stories circulate between seasoned workers about minor spills that sparked headaches and coughs. Modern standards require splash goggles, gloves (chemical-resistant, not the flimsy type from a hardware aisle), and, for most operations, negative-pressure rooms or fume hoods. Local exhausts pull vapors away, and chemical spill kits stand at attention in labs lucky enough to have a budget. Strict storage away from acids, bases, and open flames earns a nod from any seasoned handler. Disposal now faces a labyrinth of hazardous waste rules—incinerators, if equipped to scrub out chlorine half-life, tend to take the bulk. Sloppy handling creates real costs, and the paperwork trail grows longer every time someone skips a protocol or cuts corners.
Where Pentachloroethane Has Been Put to Work
Looking over thirty years of industrial shifts, pentachloroethane hasn’t been a blockbuster, but it’s pulled its weight. It found a home as an intermediate in the manufacture of trichloroethylene and some specialty chlorinated solvents. Dry cleaning once dipped a toe in, but safety and toxicity concerns chased that industry elsewhere long before most people noticed. Research labs sometimes use it as a reagent—particularly in organic syntheses where dense, inert liquids help drive or control certain reactions. What became unmistakable as new regulations came in was that many companies moved away from pentachloroethane, to limit liability and handle changing views on environmental protection. The story matches what happened to many “lesser” chlorinated solvents: old uses fade, while some niche roles persist, especially in specialized chemical syntheses.
R&D and Toxicity: What the Data Shows
Research into pentachloroethane’s behavior in humans and in the environment picked up as the tide turned against “forever chemicals” and chlorinated organics, in general. Rats exposed in chronic toxicity studies developed liver and kidney problems, pointing to real risks. OSHA’s stricter guidelines and international treaties like the Stockholm Convention leaned on such studies. If a chemical can resist breaking down in the wild and bioaccumulate, no matter how minor its mainstream industrial use, regulators watch with a wary eye. As the years pass, environmental chemists dig up evidence showing that pentachloroethane, like related compounds, stands up to degradation, running the risk of lingering in water, soil, and air. Because it’s less widely used today, the public health peril sits below headlines, but smaller spills and improper disposal can still cause real headaches for environmental cleanup teams.
Looking to the Next Page: Where Pentachloroethane Might Go
Thinking about the future for pentachloroethane means thinking about the future of chlorinated solvent chemistry as a whole. Companies ask tough questions about environmental burden and work to identify or invent safer, more biodegradable substitutes. Product stewardship programs encourage drop-in replacements, process redesigns, or even skipping intermediate stages that depend on legacy chemicals like pentachloroethane. Some research groups chase after new uses, including targeted organic synthesis or specialized materials science, but the risks hang heavy. My own conversations with industrial chemists leave me convinced that pentachloroethane will serve mainly as an academic curiosity and occasional synthetic tool. Its fate won’t turn on technical capability alone; it’ll hinge on how societies weigh worker safety, environmental legacy, and the true cost of clinging to old ways.
What is Pentachloroethane used for?
Navigating Industrial Chemistry’s Workhorse
Pentachloroethane rarely gets mentioned outside chemistry circles, but many people rely on its byproducts without ever hearing its name. This chemical puts in the grunt work deep in industrial processes, mostly as an intermediate rather than a main attraction. I remember working in a lab that stored odd drums, their warnings written in a code few outside the field would recognize. Pentachloroethane came up whenever someone talked about legacy chemicals with persistent uses.
Main Uses in the Chemical Industry
One of the main jobs comes from serving as a building block for more widely used chemicals. Large manufacturers use pentachloroethane to produce trichloroethylene and tetrachloroethylene—solvents you find in degreasing applications and old-school dry cleaning. These more common chemicals do most of their work degreasing metal parts, stripping paint, or even cleaning textiles, but they often start from pentachloroethane in the production plant.
It also sees occasional use as a solvent itself, though its toxicity limits how much gets used outside closed chemical systems. Solvents like these strip away grease and oil from industrial parts much faster than soap and water, saving factories both time and money.
Chemical synthesis relies on intermediates that can handle harsh conditions. Pentachloroethane stands out because it doesn't break down easily. This stability makes it a good choice when precision and consistency matter, and some reactions depend on its particular molecular structure.
Health and Environmental Risks
Factories that make or process pentachloroethane can't ignore the health side of things. If someone inhales its vapors or gets it on the skin, the liver and nervous system face real threats. Working for a company that paid close attention to chemical hygiene, I saw safety procedures spelled out in exhausting detail for chemicals less risky than pentachloroethane. Wearing gloves and ventilation masks forms the first line of defense, but leaks still happen.
This chemical doesn’t disappear quickly in the environment. Once it escapes, it hangs around in soil and water, and over time breaks into compounds that hit drinking water sources across the country. The EPA listed pentachloroethane and its relatives as hazardous, linking prolonged exposure to cancer and other chronic illnesses.
Looking for Safer Alternatives
Regulators across multiple countries put pressure on industry to find replacements. Regulations, tighter limits, and expensive cleanup costs force companies to keep searching for less hazardous chemicals. For companies still relying on pentachloroethane, switching to other solvents or greener intermediates brings down risk and stays ahead of tougher laws. The European Union has already pushed some sectors to overhaul their entire chemical supply chains, nudging the United States to do the same.
Modern green chemistry looks beyond making chemical reactions more efficient. It aims to design chemicals that break down into harmless stuff after use. The rise of solvents made from renewable resources shows that industry can adopt better options without losing performance.
Balancing Industry, Safety, and Progress
Pentachloroethane’s story gives a real lesson: what worked for industry in the past may become a headache in the future. Most of us rarely see this chemical up close—but without a push for safer, smarter chemistry, old hazards can linger long after the workers go home. Strong oversight, smarter design, and plenty of investment in safer options can keep workers and communities healthy while meeting the world’s demand for high-performance materials.
Is Pentachloroethane hazardous to health?
A Closer Look at Pentachloroethane
Pentachloroethane might sound like nothing more than a chemical from a science textbook, but it plays a part in the real world. Factories once used it in refrigerants and as an intermediate for making other chemicals like trichloroethylene. As time passed, experts learned more about the risks—and started to ring alarm bells.
Direct Exposure and Its Effects
People working in industrial settings face direct contact with pentachloroethane. Even without years behind a lab bench, I’ve heard stories from chemical workers who noticed irritation in their eyes or throats after a few hours. Breathing in vapors can bring headaches, dizziness, nausea, or lightheadedness. Prolonged workplace exposure adds risk for liver and kidney issues. The U.S. National Institute for Occupational Safety and Health (NIOSH) considers this chemical a serious substance to handle with care. High concentrations can knock a person unconscious. Chloroform-like solvents, including pentachloroethane, prove toxic in small spaces—especially when safety equipment gets ignored. Acute exposure never gives the body much time to fight back.
Risks Beyond the Factory Floor
Pentachloroethane’s dangers aren’t limited to plant workers. Improper disposal or leaks cause contamination in water or soil. Once, a community near an old facility saw strange tastes in their water. Health teams came in, ran tests, and found traces of pentachloroethane. Even low-level contact causes worry, as this chemical tends to stick around in ground and drinking water. Studies say long-term exposure may contribute to cancer or harm reproductive health. More research continues, but health agencies like the U.S. Environmental Protection Agency (EPA) have labeled it a probable carcinogen. They recommend quick action if pentachloroethane shows up in public resources.
Why This Matters
Chemicals from past decades still linger in many environments. I grew up by a river known for upstream dumping. Neighbors always wondered why some kids developed strange rashes or asthma. Nobody blamed one substance, but stories stuck. Pentachloroethane isn’t the only hazardous pollutant, but it stands out as one that’s both persistent and potent. Health is more than staying free from aches. Breathing clean air and drinking clean water mean everything to a community. When pentachloroethane enters the picture, people want straight answers—not spin from industry spokespeople.
Finding Solutions and Moving Forward
Industries can’t easily ignore chemical hazards anymore. Regulatory pressure mounts with every news story about groundwater pollution. Companies can adopt safer alternatives or redesign processes to phase out pentachloroethane. For communities, staying informed and asking questions make a difference. The EPA, local health departments, and watchdog groups provide resources for home testing and safety protocols. Engineers and chemists continue to develop products with fewer health risks, showing it’s possible to keep progress without sacrificing safety. Regular monitoring around plants and stronger cleanup standards serve as the best ways to protect health going forward.
Pentachloroethane sits in that group of chemicals where science, law, and common sense must work together. The risks are real. Choosing safety and demanding transparency lead to better answers for everyone.What are the storage requirements for Pentachloroethane?
Understanding the Risks
Anyone dealing with pentachloroethane faces more than a simple inconvenience. This chemical’s hazards can’t be taken lightly. Repeated studies highlight its volatile and potentially toxic behavior, so sloppy handling puts both workers and the environment in real danger. I’ve learned in my own experience working near solvent storage tanks that mistakes with chlorinated solvents, even for a short time, can lead to headaches or worse. These are not old-wives’ tales—they’re direct lessons from folks affected by vapor accumulation and poor ventilation.
Solid Containment Makes a Difference
A storage plan begins with a dedicated area. Pentachloroethane demands a sealed, corrosion-resistant drum or tank. Both carbon steel with a strong internal coating and high-grade polyethylene storage containers have proven themselves under demanding conditions. Not every general chemical drum fits the bill. A slight leak turns a minor spill into a costly cleanup, and you'll never forget the first time you smell those chlorinated vapors getting loose in a warehouse.
Temperature and Ventilation Control
Direct sunlight and heat sources challenge the integrity of pentachloroethane containers. Over the years, I’ve seen warehouse teams cut corners by stacking drums near heater units, only to end up with expansion that warped container lids. Storage in a cool, dry spot keeps danger in check. Mechanical ventilation—robust, not just an open window—plays a huge role, because vapor build-up leads to explosions or illness. Industrial fans and well-planned vent pathways don’t just meet regulatory lists—they bring real peace of mind during long hours around the storage site.
Fire Prevention and Emergency Planning
Flammable and reactive materials spell out the need for fire separation. Storing pentachloroethane next to oxidizers or acids is asking for an emergency. I’ve worked with safety experts who drill the message that every stored drum must have a buffer—sometimes a full containment bay with non-sparking tools in close reach. Fire alarms and chemical spill kits belong within arm’s reach, not buried in some distant storeroom. Workers who know the routes to safety showers and eyewash stations feel more confident facing a worst-case scenario.
Labeling, Training, and Inspection
Regulatory advice from the EPA and OSHA always comes back to clear labeling. Drums without clear hazard markers turn a risky situation into a guessing game for new employees. Assigning someone to check labels and log container conditions each week highlights worn seals or corrosion early, saving both product and headache. Proper training means more than a signed sheet—it means practical drills and real talk about the risks. Companies with a culture of reporting near-misses and maintenance issues build trust and improve standards over time.
Paths Forward: Solutions and Investments
Technical fixes exist—automated leak detection, updated fire suppression units, and online monitoring for temperature and pressure shifts. These come with upfront costs, but disasters cost far more in fines, lawsuits, and clean-up. Working with local hazardous materials teams when planning storage layouts helps the company and first responders. Insurance rates for chemical storage reflect accident records, so investing in safety tweaks pays back in more ways than one.
How should Pentachloroethane be handled safely?
Understanding What We're Dealing With
Pentachloroethane isn’t the sort of stuff you use around the house, but it does have a place in certain industrial settings. I remember walking through a chemical plant years ago and seeing barrels of complex-sounding solvents stored in ventilated sheds. The sharp, distinctive odor of some chloro-organics sticks in your mind, reminding you that these substances require attention every step of the way. Pentachloroethane isn’t any different. It’s known to mess with the nervous system, irritate the lungs, and bring heavy risks to people who don’t take protective measures seriously.
Taking Personal Protection Seriously
Folks working with pentachloroethane need to suit up. A simple pair of gloves doesn’t cut it. Proper gear -- chemical-resistant gloves, safety goggles, and full-coverage clothing -- shields skin and eyes from splashes or accidental contact. I’ve seen workers double-check their gear, run their hands along the seams of their gloves, and make sure respirators were properly fitted. No shortcuts. Inhalation risks demand a well-fitted mask with cartridges designed for volatile organics. Even brief exposure can lead to dizziness, headaches, or worse.
Work Area Conditions Matter
Every story I’ve heard about chemical incidents usually comes back to poor ventilation or carelessness in handling. Doors wide open, fans on, exhaust systems working—ventilation can save someone’s health. I recall a technician who insisted on working near a window, even on cold days, because he knew a whiff of something nasty could ruin more than his shift. Good ventilation helps keep vapor concentrations low, giving people an extra buffer against mistakes.
Storage: Out of the Way, Out of Trouble
Nobody wants a drum leaking in a crowded storeroom. I’ve seen storerooms labeled clear as day, with pentachloroethane barrels kept away from heat, sunlight, and anything even remotely flammable. Cool, dry rooms, strong shelving, and proper labeling remove guesswork and reduce disaster risk. Leak-proof containers and secondary containment trays are everyday basics; a spill on bare ground is hard to clean and can get into groundwater fast.
Dealing with Spills and Waste
A chemical like this doesn’t belong in the sink or in regular trash. Training makes a difference here—everyone should know where absorbent pads and neutralizing materials are kept. I’ve practiced spill drills, racing co-workers to put on gear and cordon off an area without panicking. All contaminated materials go straight to hazardous waste bins, then picked up by certified carriers. Water sources always stay protected.
Accurate Information, Reliable Resources
Knowledge makes a workplace safer. Labels, safety data sheets, and honest conversations about symptoms or close calls can keep others out of harm’s way. The facts are clear—chronic exposure brings cancer risks and heavy environmental damage. No one should feel shy about asking questions or flagging unsafe practices.
Room for Improvement
Chemicals like pentachloroethane call for thoughtful habits that stick, not just half-hearted compliance. Industry leaders can set expectations by rewarding safe behaviors, funding better protective equipment, and encouraging a culture where safety isn’t just a box to check. Newer, safer alternatives keep hitting the market, and switching over means fewer people get hurt in the long run.
What is the chemical formula of Pentachloroethane?
Understanding Pentachloroethane
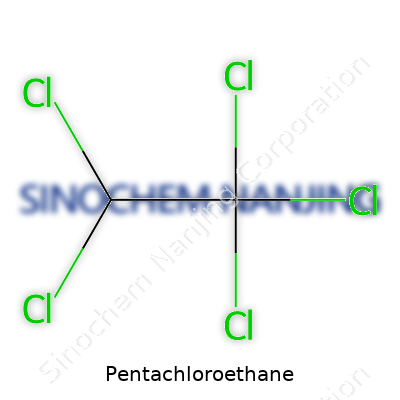
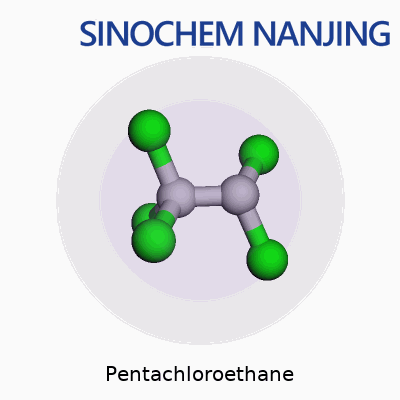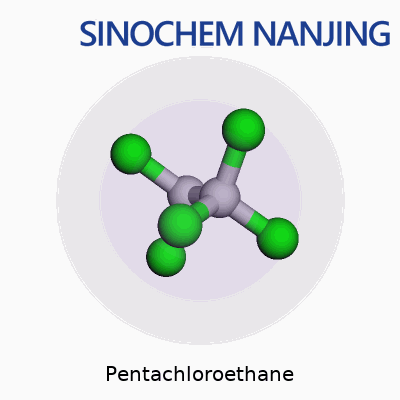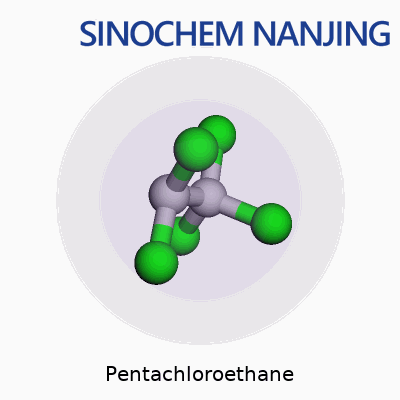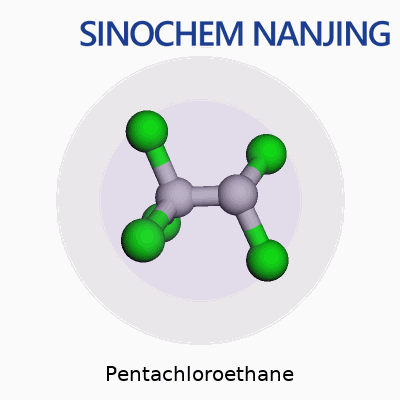
Pentachloroethane, as its name hints, is a chemical compound made from ethane, where five of the hydrogen atoms have been replaced by chlorine atoms. Its formula is C2HCl5. The structure contains two carbon atoms—just like simple ethane—but the heavy swap for chlorine makes it a very different substance from household hydrocarbons like propane or butane.
Why the Formula C2HCl5 Is Important
Knowing the formula might seem like trivial chemistry. For people in chemical manufacturing, waste management, or environmental science, it gives important clues. Chlorinated ethanes, including pentachloroethane, have a way of sticking around in air, water, and soil. All those chlorine atoms influence how pentachloroethane behaves and how tough it can be to get rid of if spilled or leaked. That formula—C2HCl5—tells you it’s loaded with chlorine, which means extra care during use or disposal.
Health and Environmental Impact
Pentachloroethane doesn’t appear in consumer products and rarely comes up in everyday conversation. As someone who spent years researching hazardous waste, I’ve seen the aftermath of improperly managed chlorinated solvents firsthand. Not only do they pose health risks—prolonged exposure may affect the liver and nervous system—but they also resist breakdown in the environment.
The formula makes it less flammable, which might seem like an advantage in industry, but that low flammability comes at a cost. The molecule breaks down slowly in sunlight and doesn’t easily biodegrade, which means it can migrate into groundwater. It relates back to all those chlorine atoms holding the molecule tightly together—nature has a tough time snapping them apart.
How It’s Used, Where It’s Found
Chemists use pentachloroethane as a starting material for other chemicals, including substances used in firefighting foams and refrigerants. It rarely stands alone in manufacturing, often showing up as a byproduct. It helps to remember that a lot of our environmental mishaps have roots in chemical byproducts just like this one. The formula gives a hint about stability, persistence, and the handle-with-care attitude people need around it.
Looking Forward: Practical Solutions
Modern regulations put stricter limits on the handling of chlorinated solvents like pentachloroethane. In my experience, companies now follow tighter reporting and containment measures. Some are switching to less persistent alternatives when possible. The foundation for safer management always comes back to education. Workers who know the significance of those five chlorines—who understand why C2HCl5 matters—tend to play it safe with storage, use, and disposal.
Cleanup once these compounds spill can drag on for years without proper know-how. Community engagement helps, too, because those living near chemical plants often spot problems first. Sharing the basics—like a chemical’s formula and what it means—empowers people to ask sharp questions and demand safer practices. Sometimes basic chemical literacy delivers more than lab coats ever could.
| Names | |
| Preferred IUPAC name | 1,1,1,2,2-Pentachloroethane |
| Other names |
Ethane, pentachloro- Perchloroethane PCE 1,1,1,2,2-Pentachloroethane |
| Pronunciation | /ˌpɛntəˌklɔːroʊˈɛθeɪn/ |
| Identifiers | |
| CAS Number | 76-01-7 |
| Beilstein Reference | 1204853 |
| ChEBI | CHEBI:35821 |
| ChEMBL | CHEMBL135565 |
| ChemSpider | 8842 |
| DrugBank | DB01849 |
| ECHA InfoCard | DTXSID7030695 |
| EC Number | 602-035-00-6 |
| Gmelin Reference | 784 |
| KEGG | C19685 |
| MeSH | D010421 |
| PubChem CID | 8055 |
| RTECS number | KJ5075000 |
| UNII | 36B82AMQ35 |
| UN number | UN1891 |
| Properties | |
| Chemical formula | C2Cl5H |
| Molar mass | 202.28 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic odor |
| Density | 1.68 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.96 |
| Vapor pressure | 23 mmHg (20°C) |
| Acidity (pKa) | 5.55 |
| Basicity (pKb) | 11.38 |
| Magnetic susceptibility (χ) | -0.00025 |
| Refractive index (nD) | 1.531 |
| Viscosity | 0.87 cP (20°C) |
| Dipole moment | 2.16 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 290.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –44.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -886.8 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, suspected of causing cancer, toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335, H351, H373 |
| Precautionary statements | P210, P261, P280, P301+P310, P303+P361+P353, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Autoignition temperature | 215°C |
| Explosive limits | Lower 6.2%, Upper 15.6% |
| Lethal dose or concentration | LD50 oral rat 2500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 2500 mg/kg |
| NIOSH | SN4550000 |
| PEL (Permissible) | 1 ppm (7 mg/m3) |
| REL (Recommended) | 1 ppm (7 mg/m³) |
| IDLH (Immediate danger) | **50 ppm** |
| Related compounds | |
| Related compounds |
Tetrachloroethane Hexachloroethane Trichloroethane Chloroethane |

