Nitromethane: More Than a Racing Fuel
Historical Development
Nitromethane brings up images of roaring drag races and clouded starting lines now, but its past stretches back to the curiosity of nineteenth-century chemists. It slipped onto the scene through the work of early synthetic chemistry, with the first accounts of its preparation appearing in the mid-1800s. Research didn’t stay caged in the lab—by the early twentieth century, engineers and manufacturers started exploring it for safer explosives and specialty fuels. The story of nitromethane runs alongside the growth of the chemical age. People learned to harness nitromethane’s unique properties both as a stand-in for more dangerous nitrocompounds and as a tool for building new organic molecules. This compound wasn’t always burning laps around a track; it found its way into ink production, solvents, and—surprisingly—fungicide. A century ago, folks didn’t have the luxury of isolated, pure nitromethane. They often struggled with mixtures and wild side reactions. Today, we have tightly controlled preparation and purification. Seeing this evolution makes it clear why nitromethane keeps showing up in cutting-edge research.
Product Overview
Most people see nitromethane as a racing fuel, but that misses the bigger picture. Chemists rely on its polar structure for reactions that build complex carbon frameworks—from pharmaceuticals to energetic materials. Industrial processes use nitromethane for its knack at dissolving both polar and nonpolar compounds. Painters and ink makers still count on it as a solvent thanks to its fast evaporation and punchy solvency. In labs, it’s a foundation for syntheses that stretch from agricultural chemicals to explosives. If nitromethane vanished overnight, we’d feel it from dragways to factories and research labs.
Physical & Chemical Properties
Explaining nitromethane starts with looking at its appearance and smell: a clear, oily liquid with a bitter almond scent. Its formula, CH3NO2, leaves nothing to chance—it blends high oxygen content with a sturdy methyl group. Water loves mixing with nitromethane, so it doesn’t split away or separate. Its boiling point keeps it liquid in everyday settings, but it lets go of vapor quickly enough to require good ventilation. What’s always struck me is how this small molecule holds enough oxygen in its structure to make it a self-contained powerhouse in combustion. Handling it safely calls for respect; spills mean fumes, and the right mix with air and a spark can spell disaster.
Technical Specifications & Labeling
Bottles and drums of nitromethane follow strict rules—markings communicate more than just the name. Red diamond labels warn of flammability, hazard phrases shout out risks, and even small shipments demand paperwork and regulated packaging. That labeling keeps both warehouse workers and firefighters in the know. Knowing the storage temperature and the risks means fewer surprises for everyone up and down the supply chain. Rules for transport aren’t just for show. They’re there because a leak or spill can cause a mess nobody wants—both for cleanup crews and for the environment. Direct experience with these rules on factory floors goes a long way—those warnings and checklists save lives.
Preparation Method
Early chemists struggled to isolate nitromethane from complex reaction mixtures. These days, manufacturers stick to tried-and-true routes, mostly through the nitration of propane or, in smaller settings, direct treatment of methane with nitric acid under carefully managed pressures and temperatures. The smell isn’t just an annoyance—it’s a clue from the molecule itself, and a sign to double-check ventilation. Today’s plants rely on automation, closed systems, and continuous monitoring to protect workers and minimize emissions. Tight controls—human and technological—keep yields high and waste low. From my perspective, modern process engineering and environmental management have tamed a process that once threatened both workers and downstream communities.
Chemical Reactions & Modifications
Nitromethane draws chemists looking for a reactive functional group that plays nice in synthesis. React it with aldehydes and bases, and you land in Henry reaction territory—perfect for building up nitroalcohols, which show up everywhere from medicine to fragrance chemistry. Throw the right metals into the mix, and you start hydrogenating nitromethane into methylamine, a precursor for drugs, dyes, and countless chemicals. Its single carbon and nitro group mean it slips easily into mechanistic studies and reaction screens. In my own experience, nitromethane offers both challenge and opportunity—a gentle hand avoids unwanted side reactions and makes selective chemistry possible. It truly rewards a careful eye and a steady hand at the bench.
Synonyms & Product Names
Search for nitromethane in a database, and you’ll likely trip across names like Nitrocarbol and mononitromethane. Some old technical bulletins even call it “nitroformic acid.” Industrial suppliers brand it with catalog numbers and trade names, but any sharp-eyed chemist recognizes CH3NO2 by its physical signatures. Knowing those synonyms isn’t just a matter of trivia; they matter for navigating global shipping papers, customs inspections, and multi-lingual safety sheets. Mixing up names leads to confusion—and occasionally to safety mishaps—so good training always stresses keeping aliases straight.
Safety & Operational Standards
Anyone handling nitromethane learns a quick respect for its hazards. It burns fast, and sparks can turn a spill into a blaze. Never store it with bases or amines unless you want trouble—those mixtures can detonate. Every chemist and technician working with nitromethane suits up with gloves, splash goggles, and flame-resistant gear. Ventilation systems matter as much as any other piece of equipment. Leaving a drum cracked open on a shift can make whole buildings uninhabitable. Workers stay sharp by drilling safety standards and running emergency simulations. In the lab or on the shop floor, personal experiences with near-misses keep standards strict. Stories from the past—the ones where someone ignored the rules and paid the price—stick with you more than any formal training ever could.
Application Area
Nitromethane’s territory covers more than most folks realize. The thunderous explosion of Top Fuel drag racing claims the headlines, but the chemistry world finds nitromethane essential. Manufacturers use it to build pharmaceuticals and advanced materials, thanks to its ability to donate both carbon and nitrogen in one shot. It breaks down stubborn coatings and residues in industrial cleaning. Rocket scientists and defense engineers lean on its predictable burn properties for propellants. Even ink manufacturers appreciate its solvency for high-end printing. Out of all the compounds I’ve worked with, nitromethane’s list of users constantly surprises me. It finds jobs in places as different as hospitals, racetracks, and aerospace labs—proof that basic molecular properties open doors everywhere.
Research & Development
Researchers keep pushing nitromethane into new roles, aiming to squeeze more functionality and value from a simple molecule. Green chemistry projects work on less hazardous alternatives and improved recycling methods. Pharmaceutical chemists invent routes that snap nitromethane into position for synthesis of complex, bioactive molecules. Material scientists stretch its use to build specialty coatings and advanced polymers. In academic settings, exploring the selective transformation of nitromethane leads to insights that ripple across fields like catalysis and energy storage. Discussions at recent conferences emphasize digital process monitoring and green-safety engineering—evidence that nitromethane’s future won’t look just like its past. From where I stand, researchers won’t drop nitromethane soon, but they keep hunting for safer, cleaner, and more sustainable approaches.
Toxicity Research
Early reports of nitromethane suggested it might be safer than other nitrocompounds, but experience and modern studies show real concerns. Breathing the vapors can irritate the respiratory system. High doses damage the nervous system, and long-term exposure connects with changes in blood chemistry. The environmental story is just as tricky: spilled nitromethane percolates into soil and groundwater, threatening plant and animal health. Real-world cases forced regulatory agencies to issue strict exposure guidelines and environmental controls. Over the years, scientists tracked metabolites, watched for subtle long-term effects, and refined detection methods for nitromethane in air, water, and tissues. Having worked with it myself, I don’t ignore those early warning signs—protective measures and constant monitoring make the difference between safe chemistry and silent danger.
Future Prospects
Nitromethane stands on the edge of another transformation. Battery scientists eye it for energy storage advances, hoping its chemistry can help improve modern devices. Green energy researchers explore blending it with renewable feedstocks, aiming to step away from fossil-derived chemicals. Racing may remain its most public use, but quieter innovations in medicine, materials, and environmental management could redefine nitromethane’s role in everyday life. As global safety and environmental standards rise, the challenge shifts toward making nitromethane safer, friendlier to the environment, and more compatible with sustainable industry. I’ve watched new generations of chemists embrace both the promise and responsibility that comes with using powerful molecules like nitromethane. The molecule’s story keeps growing—which suggests we haven’t come close to unlocking its full potential yet.
What is Nitromethane commonly used for?
A Fuel with a Punch
Nitromethane grabs attention mainly for the role it plays in motorsports. Dragsters use it because regular gasoline just doesn’t cut it when pushing the limits of speed. Nitromethane carries oxygen within its own structure, so engines running on this fuel can pack in more power with less air. It’s that raw strength that makes it the fuel of choice for top-fuel drag racing. I remember standing at a racetrack, the air thick with the sharp smell of exhaust, feeling the roar of engines shake the grandstands — that power, in a large part, comes from nitromethane.
Professional drag racers mix nitromethane with methanol to get the right combination of power and engine stability. A typical dragster running on this concoction can jump from zero to 330 miles an hour in just a few seconds. Regular gasoline doesn’t offer this thrill, no matter how you blend it.
An Essential Tool in Research and Industry
Beyond racing, nitromethane gets a lot of use in chemical labs and manufacturing. Chemists like it for making explosives, pharmaceuticals, and solvents. It acts as a starting point in producing certain antibiotics and painkillers, including medications used to manage epilepsy and other disorders. Safety comes into play here. Companies that handle nitromethane spend resources on worker education and storage protocols, since accidents with this compound can end in disaster. A mistake on a site using nitromethane might cost lives or shut down a plant.
In the lab, nitromethane’s value lies in its reaction with other compounds. It works well when making synthetic products, especially where researchers want a short, direct path to their goal. Data from chemical safety boards in several countries highlight nitromethane’s strong performance as a solvent and its ability to speed up key reactions.
Model Engines and Remote-Control Hobbies
People see nitromethane at work in smaller settings, too. Hobbyists who build and race remote-controlled cars and planes use nitromethane in their model engines. These engines run faster and smoother, giving a more exciting experience. Stores that sell model kits stock premixed fuels for these machines, with nitromethane as a selling point. Enthusiasts often compare notes about which mixtures offer better speed or longer engine life. My neighbor’s backyard would fill with the sound of model cars zipping through obstacle courses, all powered by this punchy fuel.
Safety, Regulation, and the Path Forward
Not everything about nitromethane is rosy. Law enforcement and regulatory bodies track its sale due to its explosive properties. It’s on watch lists because people have tried using it for illegal purposes. Governments respond with licensing, background checks, and tighter controls for buyers. Genuine users sometimes face delays and paperwork, but these steps save lives. I’ve seen stories of accidents that might have turned tragic if regulations hadn’t been strict.
Going forward, it makes sense to keep public education front and center. Most people buy nitromethane for legitimate goals, but simple mistakes and misinformation have led to accidents. Training, clear labeling, and industry partnerships can help keep nitromethane both useful and safe. People who rely on it — racers, chemists, hobbyists — benefit from understanding exactly what’s in their hands. The more we respect what nitromethane can do, the better we handle the risks that come with it.
Is Nitromethane dangerous to handle or store?
Getting to Know Nitromethane
Nitromethane shows up at the drag strip, roaring inside the engines of top-fuel cars. It's an energetic, pale liquid, packing more oxygen in every molecule than gasoline. This means bigger combustion, louder races, and much faster cars. Besides motorsport, factories sometimes use nitromethane in chemical manufacturing, pharmaceuticals, and explosives.
Fueling Risk
Most people outside professional racing and industry rarely encounter nitromethane. At first glance it looks harmless, a clear liquid without much odor. But forget that. Storing and handling nitromethane brings a long list of risks that regular folks can’t ignore.
Drop a spark or let a canister warm up next to a hot pipe and things can get ugly in seconds. Nitromethane burns hot and fast, but given the right conditions, it also explodes. The Oklahoma City bombing in 1995 stands as a tragic reminder. Nitromethane fed the fireball that ripped through those federal offices. Government regulators, including OSHA and the DEA, keep a close eye on its movement for that reason.
Why Handling Gets Tricky
Pouring gas into a lawnmower comes with some risk, but filling a dragster with nitromethane demands much more care. Nitromethane absorbs into skin easily, so even a spill across hands can lead to headaches, nausea, and sometimes worse. When mixed into the air, its vapors irritate eyes and lungs; long exposure just multiplies the risk.
Improper containers lead to leaks, and vapor can ignite from static charges or a forgotten spark. Blunt trauma like dropping a metal barrel on a hard surface brings another risk—nitromethane responds poorly to physical shock.
Storage: More Than Just a Locked Door
A locked shed at the backyard isn’t enough. Nitro should stay away from sunlight, heat, any flame, and anything that could bang against it. Industry best practice calls for specialized, explosion-proof storage: double-walled steel containers, plenty of venting, and strict temperature controls. Only trained professionals ought to move or mix this stuff. Even then, regular training and emergency drills stay mandatory.
Insurance companies treat nitromethane as a high-risk chemical. Regulations require logging all shipments and reporting theft or loss instantly to federal authorities. Those policies exist for a reason—unattended nitromethane in an accident zone or during a warehouse fire puts surrounding buildings and workers in extreme danger.
Safer Racing, Safer Industry
Racers wear gloves and respirators, and work with anti-static tools. Labs use chemical hoods and only open containers on grounded metal plates. Getting permission to use nitromethane takes paperwork and regular audits. That keeps most accidents rare, but everyone who touches this liquid knows what could go wrong.
Public safety demands vigilance. Clear labeling, locked access, and no shortcuts. Ignoring even one safety step has ended in ruined careers, burned buildings, and lost lives. Nitromethane might let dragsters blast down the strip, but it gives zero leeway to anyone who underestimates what it can do outside a controlled environment.
What are the safety precautions when using Nitromethane?
Understanding the Real Risks
Nitromethane draws a crowd in motorsports, but its risks stretch beyond the racetrack. Just a whiff at the racetrack will tell you how different this stuff is compared to regular fuels. Nitromethane carries more oxygen within its structure, which helps engines run hotter and faster, but it also means the hazards multiply. This chemical easily ignites, releases potent fumes, and can even explode under certain conditions. Even people without a chemistry background tend to know that anything used to make powerful model engines roar isn’t just another household item.
Personal Experiences Are Wake-Up Calls
I remember walking into a garage where nitromethane had spilled near a battery charger. The harsh, sharp scent felt like a slap, and my gut instantly knew to get fresh air. Luck played a role that day — just a single spark could have led to real disaster. This wake-up call led me to read up on the details. I learned that nitromethane vapors linger low to the ground. Spark-producing tools, even the humble screwdriver, become much more dangerous around it. Stories like this stick with you and underscore why following basic rules isn’t just ticking boxes.
Simple Steps, Big Impact
Keeping nitromethane safe starts with treating it seriously, never like ordinary fuel. It always stays in tightly sealed containers, away from sunlight and heat. Steel drums are the go-to — plastic can break down, releasing fumes or leaking. Handling means gloves on, goggles tight, and no distractions. Even a distracted moment while pouring can splash nitro onto your clothes, and that’s not just a laundry problem. It’s skin-irritant territory.
Ventilation isn’t just a nice idea. Those fumes carry health risks. They irritate the lungs and eyes. Continued exposure can lead to headaches, dizziness, and worse. A powered exhaust fan keeps things safer in closed garages, and open doors let the air pull chemical odors away fast. Anyone using nitromethane indoors should consider an air quality monitor — a small investment for the peace of mind it brings.
Fire and Explosion Hazards
Nitromethane deserves respect for its flash point. Any spark, even static from synthetic clothing, can trigger a fire. Static wristbands, non-sparking tools, and keeping cell phones away from the work area seriously lower the odds of disaster. I always kept a Class B fire extinguisher handy. The wrong type just spreads the fire or does nothing. Water is not the answer for nitromethane fires — foam or dry chemical agents work best.
Storage and Transportation Rules
Just moving nitromethane should give pause. It needs labeling, and nobody loads it in the back seat of a family car. Enclosed, non-vented vehicles trap fumes, turning minor leaks into major risks. Transport in small, approved quantities, with clear signage, brings risk down to an acceptable level. Always check local regulations, as some cities have extra rules for even small amounts.
Seeing the Bigger Picture
Nitromethane provides thrilling performance, but the flip-side can’t be ignored. Awareness, protective gear, and preparation are non-negotiable. Accidents may be rare with good habits, but complacency is what leads to problems people regret. Anyone handling nitromethane, enthusiast or pro, owes it to themselves (and anyone nearby) to take every step possible to reduce risk.
Can Nitromethane be shipped internationally?
Why Shipping Nitromethane Is Not Straightforward
Nitromethane draws plenty of attention from regulators, chemists, racers, and businesses. For anyone who’s ever cracked open a model airplane engine or has an interest in drag racing, nitromethane lights up the imagination. Yet this chemical comes with difficulties—especially when someone tries to ship it across the globe. Strict regulations remain in place, and anyone unaware of those sometimes faces large fines or even an unexpected visit from law enforcement.
High Risk Chemical, Tougher Rules
Nitromethane is flammable, creates toxic fumes, and has earned a reputation for being more than just a fuel additive. Incidents related to its improper handling or misuse have led to tragedies and prompted heavier oversight. In the United States, nitromethane gets the label “hazardous material.” The Department of Transportation (DOT) and the Bureau of Alcohol, Tobacco, Firearms, and Explosives (ATF) both watch it closely, sometimes treating it almost like an explosive. Europe includes it in the REACH framework, enforcing rules on production, sales, and shipment.
Freight carriers treat nitromethane shipments like handling radioactive materials. Some shipping giants—think FedEx or DHL—just steer clear, refusing to touch the stuff. If they agree to move it, the price skyrockets and paperwork stacks up. That includes detailed manifests, international safety data sheets, and compliance with the International Air Transport Association’s dangerous goods regulations.
International Differences Make It Complicated
Here’s where things go sideways. Every country has its own rules. The U.S. will allow shipments under some conditions: the vessel and packaging have, by law, to meet Department of Transportation rules. Mexico restricts imports—same for Australia, especially for private buyers. India lists it as a chemical of concern. In the European Union, nitromethane made headlines after illicit use, and now buyers need special permits. Even places with loose domestic controls often clamp down at the border.
Personal experience with customs forms tells me officials rarely show flexibility. There was a time someone I knew needed nitromethane for race tuning. The package got stuck in customs for weeks in Germany because the importer overlooked one detail. Freight forwarding companies explained how unlisted substances delay everything. Once they flagged the shipment, local police visited, just to do a routine check. This isn’t rare; chemistry hobbyists, researchers, and engine tuners all share similar horror stories.
Public Safety and Industry Needs: Walking the Tightrope
Looking at risk, these tough regulations make sense. Public safety carries more weight than a racing win or a research project. Bomb threats, arson, and the growing awareness of chemicals in terrorism all fed the strict approach. Yet, industry and legitimate hobbyists need a balance that doesn’t bring all legitimate commerce to a halt.
Solutions won’t come from relaxing standards, but smart policy can smooth legitimate trade. Industry groups often lobby for clear labeling and standardized paperwork. Chemists and buyers push for better training that helps border agents tell the difference between the engine builder and the bad actor. Traceable licensing, transparent end-user declarations, and digital tracking could make shipments less risky while keeping loopholes closed.
Staying Informed and Safe
Anyone seeking to bring nitromethane across borders has to read the fine print. Governments worldwide offer information portals, and consulting regulators ahead of time avoids disaster. Experts in international law and chemical logistics can bridge gaps left by patchwork global rules.
If shipping nitromethane sounds tough, it’s because every step involves scrutiny. That keeps people safer, but it challenges those with real industrial or scientific purposes. Staying honest, working with the right partners, and handling everything by the book can keep the process moving.
What is the shelf life of Nitromethane?
Understanding How Nitromethane Ages
Nitromethane doesn’t show its age like milk in the fridge. This chemical looks about the same year after year if stored right, but that doesn’t mean it stays fresh forever. Over time, nitromethane faces threats from heat, sunlight, air, and sometimes just the passage of months sitting on a shelf. Left open to humidity or stored in a room that sees big temperature swings, what’s inside that drum can slowly change. Most manufacturers say nitromethane keeps for at least two years if you follow their storage tips—sealed tight, away from light, nowhere near heat sources or sparks. I’ve seen good product last longer when people don’t cut corners, but those extra months aren’t something you should bet safety on.
Why Shelf Life Isn’t Just a Label Issue
If someone keeps nitromethane too long, the risks go up. The liquid might still fuel an engine or serve its purpose as a solvent, but invisible changes could be brewing: breakdown products can form, sometimes turning it acidic. Acidity eats at the metal tanks or containers, and in rare cases, this jumpstarts an unhealthy chain of reactions that can trigger leaks, corrosion, or even incidents you don’t want happening in any workshop or garage. I’ve spoken with people who got away with using old nitromethane but others lost equipment or had near-misses that left them shaken.
Regulators take a careful look at old nitromethane. Safety codes like those from the National Fire Protection Association don’t set a hard-stop date, but they point out that safe storage is as much about checks and rotation as it is about the label. Big racing teams and industrial users track drums with dates and keep regular inspection logs—not for paperwork’s sake, but because surprises cost lives and dollars. Small-time users sometimes don’t have that discipline, which leads to forgotten stock and dangerous surprises.
Real Risks With Stale Stock
The biggest headache—stability. Fresh nitromethane stays fairly predictable under normal conditions. Once it starts breaking down, the risk of spontaneous ignition or violent reactions chills the mood in any shop handling it. I worked with machinists and racers who felt confident grabbing any old can from storage. Stories float around of bottles that “clinked” when poured—sediment at the bottom giving away that breakdown had come calling. That’s the sort of red flag nobody should ignore.
Misusing old nitromethane isn’t only a hazard for people handling it. Waste services raise their prices or refuse containers if they see problems or smell something off. Cleanup grows more complicated, as disposal rules get stricter each year—especially now, with enforcement tighter around chemical hazards.
Smarter Storage and Disposal Fix a Lot
To stretch shelf life, keep nitromethane in steel drums with solid seals, in a place where temperatures don’t spike and sunlight can’t sneak in. Regularly check containers for rust or leaks. Rotate stock—use older containers first, same as people do with food. Anything showing cloudiness, odd smells, or sediment belongs in hazardous waste. For disposal, follow local guidelines exactly; don’t cut costs by skipping steps. Tools like chemical inventory management apps, now common at even small shops, help track age and condition. I’ve seen a few reminders pay for themselves with one avoided incident.
Nitromethane won’t alert you loudly when it’s expired. Staying on top of storage, inspection, and disposal is the clearest way to keep it safe, keep people healthy, and sidestep the expensive mess old chemical stockpiles bring.
| Names | |
| Preferred IUPAC name | Nitromethane |
| Other names |
Nitrocarbol Methylnitron NITROMETAN NITROMETHANE NITRO-METHANE |
| Pronunciation | /ˌnaɪ.trəʊˈmiː.θeɪn/ |
| Identifiers | |
| CAS Number | 75-52-5 |
| Beilstein Reference | 1460119 |
| ChEBI | CHEBI:48107 |
| ChEMBL | CHEMBL137481 |
| ChemSpider | 9143 |
| DrugBank | DB02770 |
| ECHA InfoCard | 100.003.360 |
| EC Number | 603-025-00-9 |
| Gmelin Reference | 828 |
| KEGG | C08336 |
| MeSH | D009600 |
| PubChem CID | 6387 |
| RTECS number | PA9800000 |
| UNII | K4J9Q9N41A |
| UN number | UN1261 |
| CompTox Dashboard (EPA) | DTXSID1020698 |
| Properties | |
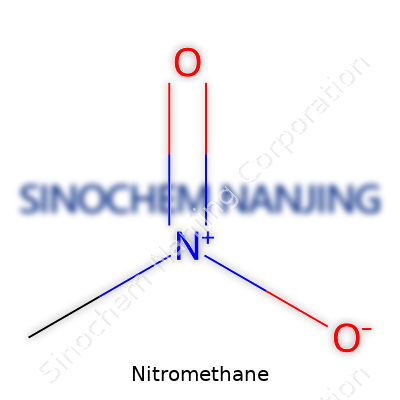
| Chemical formula | CH3NO2 |
| Molar mass | 61.04 g/mol |
| Appearance | Colorless, oily liquid |
| Odor | Disagreeable, fruity odor |
| Density | 1.13 g/cm³ |
| Solubility in water | Fully miscible |
| log P | -0.57 |
| Vapor pressure | 35 mmHg (20°C) |
| Acidity (pKa) | 10.2 |
| Basicity (pKb) | 10.2 |
| Magnetic susceptibility (χ) | -23.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.381 |
| Viscosity | 0.61 mPa·s (at 25 °C) |
| Dipole moment | 3.46 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 104.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -80.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –709.2 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB24 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H302, H319, H335 |
| Precautionary statements | P210, P261, P280, P304+P340, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-1-OX |
| Flash point | 35 °C |
| Autoignition temperature | 373 °C |
| Explosive limits | 7.3–63% (in air) |
| Lethal dose or concentration | LD50 (oral, rat): 940 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 940 mg/kg |
| NIOSH | NIOSH: SN 1610000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Nitromethane: "100 ppm (245 mg/m³) as TWA (OSHA) |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 160 ppm |
| Related compounds | |
| Related compounds |
Nitroethane Nitropropane Nitroethanol Methanol Acetonitrile Formamide |

