Nitroethane: More Than a Chemistry Sideshow
Looking Back: Tracing the Roots of Nitroethane
Nitroethane isn’t a household word, but it’s found a role in both chemical history and modern labs. Chemistry students usually meet it early on as one of the simplest nitro compounds. Stories of its development stretch back to the dawn of organic chemistry, when visionaries in Europe stumbled onto nitroalkanes while synthesizing explosives and solvents. Recipes often began with ethyl halides reacting with silver nitrite or the classic “Victor Meyer” method, showing just how inventive 19th-century chemists had to be to access new molecules. For over a century, industry stuck to similar prep strategies, first to explore new fuels or solvents, and then to develop practical routes for manufacturing at scale.
Understanding the Stuff: What Sets Nitroethane Apart
Holding a clear, oily liquid in your hand, nitroethane looks plain, but a sniff lets off a strong fruity odor—a hallmark of many nitro compounds. Its lower boiling point, around 114°C, makes it more volatile than similar alcohols. The compound’s secret sauce lies in the nitro group: With a strongly electron-withdrawing effect, it shifts the reactivity of the ethane skeleton. Unlike ethanol, nitroethane won’t mix easily with water, though it dissolves in organic solvents such as ether and acetone. Its density lands a bit heavier than water, a sign that there’s more going on inside the molecule than meets the eye. It lights up chemical reactions, acting as both a substrate and a launching point for more complex transformations thanks to the acidity of the hydrogen next to the nitro group.
The Nitty-Gritty: Technical Considerations & Lab Labels
Any bottle of nitroethane in a lab comes labeled with familiar identifiers like “nitroethane,” “ethyl nitrite,” or sometimes trade names depending on region. The chemical formula, C2H5NO2, sits alongside a UN number for shipping, and hazard pictograms remind users that nitroethane is flammable and needs careful handling. Pressure can build in closed containers, and the compound should be kept away from sparks and heat. While its flash point isn’t as low as some solvents, it demands respect, both in storage and transport, for its fire risk and health hazards.
How Chemists Cook It Up: Paths to Nitroethane
If you’ve ever spent time searching chemical catalogs, genuine nitroethane commands a higher price than its simple composition would suggest. One widespread lab method relies on the nucleophilic substitution of 1-bromoethane by silver nitrite, generating nitroethane and silver bromide. Industrial chemists adapted these old-school approaches, but with bigger reactors and better waste handling. Efficiency matters, so tweaks using cheaper starting materials or optimized catalysts can cut both cost and environmental impact. The chemical industry constantly reviews these methods based on feedstock availability, regulatory trends, and safety, including the ever-present risk of explosive byproducts.
What Nitroethane Can Do in a Reaction Flask
Any organic chemist spots the acid-like hydrogen on the carbon next to the nitro group and thinks of Henry (nitroaldol) reactions, which tie nitroethane to aldehydes and ketones to build beta-nitro alcohols. Few reagents let you add both carbon and nitrogen in one go as well as nitroethane. This trick has earned it a niche in synthesizing pharmaceuticals, dyes, and flavors. Under more aggressive conditions—strong bases or reducing agents—nitroethane can shed its nitro group to make amines or even build up into more elaborate constructions, like heterocycles.
Names on the Label: Synonyms and Shop Talk
Chemists rarely let a good compound go by one name. Besides “nitroethane,” you’ll hear “ethyl nitrite,” though this sometimes causes confusion with entirely different molecules. Some shops and journals refer to it by its systematic name, sometimes by abbreviations like NE. Precision in names matters, especially for regulatory bodies tracking the sale and use of chemicals that appear on watch lists for explosives or narcotics synthesis.
Safety in Practice: Standards and Precautions
Working with nitroethane is all about respect for flammability and moderate toxicity. Labs with any safety culture keep nitroethane away from flames, electrical discharges, and oxidizers. Spill kits, tight-fitting gloves, and solid ventilation come standard. Reports link chronic exposure to neurological and liver symptoms—facts that drive strict limits on permissible exposure. Regulations vary across countries, prompted not just by health but by concerns over illicit use as a precursor.
Where Nitroethane Winds Up: Main Fields of Use
You’ll spot nitroethane anywhere people need a polar, relatively non-reactive solvent—such as in adhesives and paint removers. Older rocket hobbyists remember it fondly as a fuel, valued for both its energy content and ignition properties, though modern regulations pinch this use. Synthetic chemists use it to bridge the gap between small molecules and more complex drugs or agrochemicals, making it a popular starting material for pharmaceutical research and commercial manufacturing.
Research Keeps Pushing the Story Forward
The chemistry isn’t standing still. Academic and industrial teams keep searching for greener methods to make and use nitroethane, nudged by tougher environmental regulations and sustainability targets. Research dives into the electronic structure, refining selective reactions, and even using it in flow chemistry setups to boost safety and yield. As more applications surface, especially in pharmaceutical innovation or specialty materials, demand for high-purity nitroethane with fewer byproducts sets new hurdles for chemists to clear.
Toxicity: Know the Hazards
Toxicity studies show that nitroethane doesn't cause acute poisoning easily, but repeated exposure increases risks of central nervous system depression, liver stress, and blood disorders. Reports from industry and health agencies warn against inhalation and skin contact since long-term effects can compound silently. Animal studies highlight changes in red blood cell counts and enzyme levels, though exact data for humans in occupational settings can be tricky to pin down. These concerns drive adoption of better engineering controls, airtight reactors, and personal protective equipment.
Looking Ahead: The Future of Nitroethane
Nitroethane’s story keeps evolving as new tools and challenges shake up the chemical world. Pressures to make synthesis more sustainable and less hazardous shape the next chapter. As regulation tightens around chemical precursors, the industry explores alternative routes, and new uses appear in electronics, advanced polymers, and as specialized intermediates. There's promise in catalysis, process optimization, and even biotechnological strategies for safer, cleaner production. Researchers with an eye on greener chemistry already dream up new catalysts and pathways that use renewable raw materials, aiming to steer nitroethane chemistry away from its reputational snags and toward more responsible innovation.
What is Nitroethane used for?
Understanding Nitroethane’s Place in the Lab
Chemistry textbooks list nitroethane alongside common reagents, though most folks outside science circles never come across it. This colorless, oily liquid means little on its own, but inside laboratories it opens up a world of possibilities. Nitroethane offers more than one use. Its biggest spotlight falls on its role in synthetic chemistry, acting as a building block in the creation of other compounds.
Practical Applications in Industry
Nitroethane steps up as a solvent for extractions, cleaning solutions, and specialty coatings. Its knack for dissolving greasy residues makes it useful in formulating certain cleaners and removing adhesives. Racing enthusiasts have known for decades that adding nitroethane to fuel mixtures gives a power boost. This use lines up with small-scale racing—think model engines and remote-controlled cars—rather than full-sized automobiles.
Pharmaceutical companies keep nitroethane close by for early-stage development of medicines. Chemists reach for it during the preparation of intermediates, especially in drugs where the nitro group becomes important later in the process. While such steps might seem minor, the direction a reaction takes sometimes depends on having a reagent like nitroethane on hand.
A Double Edge: Risks and Restrictions
Not every use of nitroethane draws praise. Its chemical properties allow easy transformation into compounds which can veer into illegal territory. Government agencies around the world watch it closely, especially after noticing its place in the synthesis of certain controlled substances. Because of this, regulations have tightened. Buyers often face strict documentation checks and must explain how they plan to use it.
Safety deserves its own attention here. Nitroethane brings flammability concerns, and inhaling its vapors causes headaches or worse if left unchecked. I remember my first experience storing some for a side project at the university. The safety officer checked everything twice: proper labeling, leak-proof containers, and a reminder about good ventilation. It wasn’t about scaring anyone; the risks were just real and needed respect.
Balancing Innovation and Oversight
Modern chemistry moves fast, but calls for accountability grow just as quickly. Factories now rely on narrower supply chains, making it easier to follow the journey of each liter. Responsible sourcing, careful storage, and transparent reporting address two important needs—keeping industry moving forward and making sure chemicals land only in trustworthy hands.
Some analysts in public health suggest that education helps most. Informing users not only about potential benefits but also dangers steers both industry and the curious hobbyist away from risky behavior. Companies can lean on credible suppliers, ask for certificates, and train staff on hazardous materials management. Research groups and regulators have already started working together, agreeing that trust grows from openness and vigilance at every step.
Looking Forward
Nitroethane proves valuable, but the science community treats it with the respect it deserves. Honest conversation between regulators, scientists, and suppliers will decide its legacy in the coming years. A practical chemical, yes, but its story reminds us that progress asks for both excitement and caution, in equal measure.
Is Nitroethane hazardous or toxic?
Nitroethane in Daily Context
Nitroethane isn’t something most folks bump into at the grocery store. It shows up in labs, in certain industrial cleaning products, racing fuels, and even in some nail polish removers. Out of curiosity, years ago I checked the label of an old can of hobby fuel, saw the word “nitroethane,” and reached for my phone. I remember furrowing my brow over some of the warnings. The chemical sounded kind of exotic, but the label’s fine print spelled out some plain dangers.
Health Risks and Why They Matter
People who spend time around nitroethane should know a few clear facts. Inhalation, skin absorption, or swallowing this substance can make you pretty sick. Inhaling its vapors causes dizziness, headaches, breathing trouble, and nausea. Spend a while around concentrated fumes, and it can knock your nervous system sideways. Some case reports mention long-term issues with regular, unprotected exposure, ranging from memory trouble to numb fingers. Nitroethane isn’t listed as a proven carcinogen, but the short-term risks can’t be shrugged off.
Direct skin or eye contact stings and brings redness and irritation. After a friend accidentally spilled a tiny amount on his arm, he ended up with a red patch for days. Swallowing nitroethane—something children or curious pets might do—is absolutely more concerning. Swallow just a bit, and symptoms can ratchet up to confusion, tremors, and trouble breathing.
Fire Hazards
Nitroethane is highly flammable. A leak near a heater or open flame turns a bad day into an emergency. Combustion can also release toxic fumes. Most safety data sheets urge folks to store nitroethane away from ignition sources. As someone who has seen ignitable materials mishandled, these warnings deserve respect. It’s not just about property damage—lives can change in minutes because of one overlooked spark.
Environmental Concerns and Legal Controls
This chemical, if not handled properly, seeps into soil or drains and lingers. Some evidence signals potential harm to aquatic life. Though nitroethane breaks down eventually, spills and poor storage habits can leave a mark on waterways and plant roots. Laws keep a close watch on the purchase and use of nitroethane. In some places, regulations treat it almost like controlled substances, mostly to reduce misuse in making illicit drugs or explosives. Background checks and permits reduce the chances of dangerous incidents.
Smart Handling Practices
When dealing with substances like nitroethane, respect and caution go a long way. Wear gloves and goggles. Only open containers in well-ventilated spaces, ideally with a fume hood on hand. Never eat, drink, or smoke where the chemical gets handled. If an accident happens, know where to find the local emergency number. Training matters—a quick refresher course can help workers avoid both the rare and the likely disasters.
Institutions and companies that use nitroethane owe it to their teams to provide solid safety training. Occupational exposure limits, established by agencies like OSHA, set hard rules for how much vapor is too much. Regular air quality tests and personal monitors catch problems before they cross the line. If storage happens on-site, it should stay in approved containers away from heat and locked up tight.
Room for Improvement
Replacing nitroethane with safer alternatives exists in some fields. Industries that prioritize greener practices look for solvents and additives less likely to harm people or the environment. Educating everyone—from lab techs to warehouse staff—strengthens the safety net. Waste treatment systems designed specifically for nitroethane keep rivers, groundwater, and wildlife safer. Every preventable spill or mishap avoided is one less story about regret and one more about common sense saving the day.
What safety precautions should be taken when handling Nitroethane?
Why Safety Takes Priority with Nitroethane
If you’ve ever opened a bottle of nitroethane in a lab, the first thing you’ll notice is the sharp, sweet odor. The stuff seems almost innocent, but years in chemical handling have taught me to respect it. Nitroethane shows up in solvents, fuel additives, and even as an intermediate for pharmaceutical synthesis. Though it’s not an everyday material for most people, mishandling it can quickly turn into a trip to the emergency room or bring in a visit from the fire department.
Risks and Hazards: Clear and Present
Let’s skip the jargon. Nitroethane burns easily. Vapors catch fire far faster than water boils, and any static spark or open flame nearby could set off an accident. Breathing the fumes gets rough on the lungs, and the liquid stings the skin. Prolonged exposure sometimes leads to headaches, confusion, or even a kind of chemical drunkenness that’s no joke. It absorbs pretty well through skin, so gloves aren’t optional trust me, I’ve seen enough red, irritated hands to learn that lesson for life.
Good Habits: Small Steps, Big Difference
Before even opening a container, grab the right gloves—nitrile works best—and a pair of goggles. Keep a lab coat handy and buttoned, and make sure sleeves aren’t loose. Earthing or grounding the work area helps prevent static, which can spell disaster with a flammable liquid like this. Store it far from oxidizers like nitric acid and out of reach of any open flames or heat sources. I picked up the habit of using real chemical-rated splash goggles, not just safety glasses, after a close call with a volatile splash. Sometimes it’s tempting to rush, but patience saves skin and eyes.
Only work with nitroethane where you’ve got plenty of ventilation. I like to double-check that the fume hood actually works if I plan to open the bottle. If there’s a spill, getting to fresh air comes first; don’t try to muscle through it. Rags and towels used for cleanup go straight to a flame-proof waste can. Leaving them lying around is asking for trouble, and fire codes exist for a reason.
Emergency Plans: Prepare, Don’t Guess
Chemical burns and inhalation risks aren’t things anyone wants to deal with on the fly. Emergency showers and eye-wash stations become your best friends in a spill. I’ve seen folks freeze up in an accident, not remembering where to run. Taking a minute to locate gear ahead of time, even if it means being “that person” on your first day, pays off down the line. Every lab I’ve been in posts the number for the local poison control center right above the chemicals. That’s not just for show. If anyone inhales too much vapor or gets splashed, authorities want details on what happened and what’s been done so far. Time counts.
The Bigger Picture: Trust the System, Trust Yourself
Shelves full of labeled flasks and clean counters don’t guarantee safety. It’s easy to get careless, especially on long days or during repetitive tasks. Watching colleagues take shortcuts has taught me just how fast a quiet afternoon can turn hectic. Regulations and training aren’t red tape—they really keep people out of the hospital. Professional organizations, OSHA guidelines, and institutional safety protocols all draw from hard-earned experience. Even so, personal responsibility means checking the safety data sheet, asking questions if anything’s unclear, and never assuming yesterday’s batch went fine so today will be the same.
Can Nitroethane be shipped internationally?
A Chemical with Multiple Sides
Nitroethane gives off a sharp scent, familiar if you’ve been around flavor labs or certain industrial sites. On paper, it acts as a solvent, finds uses in chemical syntheses, and pops up in a handful of specialty products. The problem? Nitroethane walks a tightrope between legitimate industry roles and darker corners involving controlled substances.
Regulatory Hurdles—A Tangled Web
Shipping nitroethane across borders introduces a mess of paperwork and suspicion. I’ve watched colleagues try to import lab chemicals, only to face governments that treat every package as a potential threat. For nitroethane, regulators tighten their grip because some buyers use it for illicit drug manufacturing. In the United States, the DEA keeps a close watch. In Europe, REACH and various customs offices raise barriers. Many Asian countries follow suit. No shipper wants a customs agent cracking open a drum and finding errors on the manifest, or worse—a shipment seized for “security concerns.”
The Demand and the Risks
Industries want nitroethane because it does the job that other solvents struggle with. Paint strippers, fuel additives, and specialty glass cleaners rely on it. Chemists inside research labs and on factory floors tell me nitroethane holds a niche spot—too versatile to ban outright, too risky to ignore. Every order brings up paperwork, background checks, and proof of end use. One mistake can land a company on a government watchlist, cut off suppliers, or block import rights. Not exactly something any business wants.
Practical Realities of Shipping
Most chemical suppliers won’t even offer international shipping without reams of supporting documents. Standard requirement: end-user declarations. Sometimes, it also means site visits or video verification calls just to confirm legitimate use. Freight companies often say no to nitroethane orders outright because insurance premiums spike on anything tied to drug precursors.
Rules change fast. Customs agencies can demand new licenses overnight, block ships from docking, or retroactively penalize brokers who mishandled forms. Personally, even ordering common solvents for lab use brought the kind of questioning that felt more like a criminal investigation than a transaction.
Making Compliance Work
Finding a reliable supplier and a trustworthy freight partner is the first step. Vet every order: double-check import codes, substance lists, and country-specific rules. Use exporters with proven track records in hazardous materials and controlled chemicals—that background matters more than ever. On the import side, know the customs office’s guidelines cold. A single missing stamp can mean months of delays or the loss of an entire container load.
Advocacy matters, too. Industry groups push governments to improve clarity, offer secure tracking tools, and reduce unnecessary hold-ups for legitimate buyers. Publicizing the value of nitroethane in specialty manufacturing keeps policies rooted in reality, not fear. Training logistics teams to handle documentation builds trust with authorities, speeds up shipping, and slashes waste.
Safe Path Forward
Every leg of nitroethane’s journey needs careful handling. Companies prepared for documentation, traceability, and transparency lower risk, cut costs, and keep valuable chemicals flowing. Meanwhile, efforts to keep dangerous misuse in check remain critical for public safety, which demands vigilance from every link in the chain.
What is the purity level of the Nitroethane product?
What Defines Nitroethane Purity?
Nitroethane offers promise across several industries, but performance always ties back to purity. In laboratories and manufacturing, specs for nitroethane purity can run anywhere from 95% to a benchmark of 99.5% or even greater. That extra half-percent may not mean much at first glance, but trace impurities sometimes create bigger headaches than people expect. Even a little water or an unexpected byproduct left over from synthesis can throw off key chemical reactions.
Real-World Impacts of Purity
Many chemists don’t cut corners because inconsistent quality leads to failed experiments or product recalls. Impurities don’t always show up right away—sometimes you only spot them later as odd color changes, or worse, as dangerous byproducts, especially during scale-up from bench-top to industrial runs. I’ve seen manufacturers waste weeks tracing a problem back to a “pure” bottle that missed out on proper final checks.
Take pharmaceuticals or flavors. These industries stake their reputation on tight tolerances. Regulatory watchdogs keep lab techs alert, as just one percentage point of contamination could shift a formula’s toxicity or throw off an entire sensory profile. The wrong impurity can carry health risks, create new hazards, or violate safety codes, leading to serious fines or lawsuits.
Testing and Verifying Purity
Companies serious about delivering trustworthy nitroethane rely on gas chromatography, mass spectrometry, moisture analysis, and other rigorous tests. Customers deserve full reports. In practice, nobody accepts just a generic grade—chemists and engineers ask suppliers for certificates of analysis with every lot. Anything less, and buyers start looking elsewhere.
These certificates don’t only matter for top-tier labs. Even basic industrial syntheses, such as those in adhesives or coatings, face unexpected downtime and waste when the solvent or reagent holds unlisted impurities. Clean nitroethane streamlines downstream processing—saving on filtration costs, cutting delays, and making outcomes more predictable. Time lost to troubleshooting easily dwarfs the small savings from unvetted product.
Addressing the Core Challenge
Tighter controls start at the source. Reliable suppliers invest in sealed production equipment, monitor each batch, and isolate the finished product from air and light as they bottle it. That detail pays off: water uptake or air contamination spoils purity even after distillation. Packaging in amber glass or high-density polyethylene reduces leaching and keeps off-the-shelf quality up to spec.
Sourcing nitroethane often raises questions about trust. That trust grows through relationships with suppliers who stand by their processes and offer documentation. From my own stints in research, I learned the hard way that “high purity” can mean different things depending on region, vendor, or application. Don’t just trust the label—back it up with your own spot checks, or have a lab verify the lot during big projects.
Bigger buyers hold leverage by requesting audits and visiting production sites. That transparency lifts everyone’s standards, forcing more corners into the light. It builds a culture where nitroethane truly meets its labeled purity and where companies take pride in consistency, safety, and reliability—not just in filling orders.
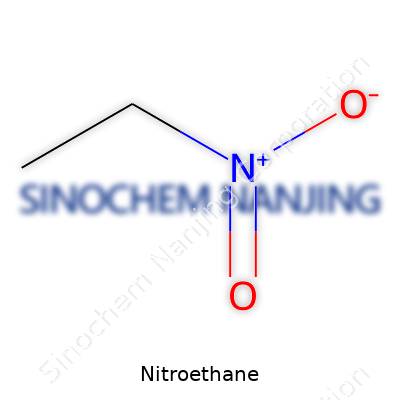
| Names | |
| Preferred IUPAC name | Nitroethane |
| Other names |
Nitroethane Ethyl nitrite Nitrethane |
| Pronunciation | /ˌnaɪ.trəʊˈiː.θeɪn/ |
| Identifiers | |
| CAS Number | 79-24-3 |
| Beilstein Reference | 1908228 |
| ChEBI | CHEBI:50075 |
| ChEMBL | CHEMBL16852 |
| ChemSpider | 6733 |
| DrugBank | DB14019 |
| ECHA InfoCard | ECHA InfoCard 000029-849-7 |
| EC Number | 208-740-4 |
| Gmelin Reference | Gmelin Reference: 843 |
| KEGG | C03261 |
| MeSH | D009586 |
| PubChem CID | 6569 |
| RTECS number | SN0700000 |
| UNII | 789U1901DI |
| UN number | UN2842 |
| Properties | |
| Chemical formula | C2H5NO2 |
| Molar mass | 75.07 g/mol |
| Appearance | Colorless oily liquid |
| Odor | fruity odor |
| Density | 0.991 g/cm³ |
| Solubility in water | Moderate |
| log P | -0.17 |
| Vapor pressure | 3.5 kPa (20 °C) |
| Acidity (pKa) | 8.6 |
| Basicity (pKb) | pKb = 10.2 |
| Magnetic susceptibility (χ) | -8.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.378 |
| Viscosity | 1.07 mPa·s (at 25 °C) |
| Dipole moment | 3.50 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 199.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -162.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −1419 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N02CC07 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H315, H319, H335 |
| Precautionary statements | H225, H315, H319, H335 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 29 °C |
| Autoignition temperature | 488 °C |
| Explosive limits | 2.7–63% |
| Lethal dose or concentration | LD50 oral rat 1100 mg/kg |
| LD50 (median dose) | 1100 mg/kg (rat, oral) |
| NIOSH | NIOSH: SZ7875000 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 100-500 mg |
| IDLH (Immediate danger) | 900 ppm |
| Related compounds | |
| Related compounds |
Nitromethane 1-Nitropropane 2-Nitropropane Nitroethanol Ethyl nitrite |

